Salt Tolerance of Hydrangea Plants Varied among Species and Cultivar within a Species
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Treatments
2.3. Data Collection
2.4. Leaf Mineral Analysis
2.5. Greenhouse Location and Environment
2.6. Experimental Design and Statistical Analysis
3. Results and Discussion
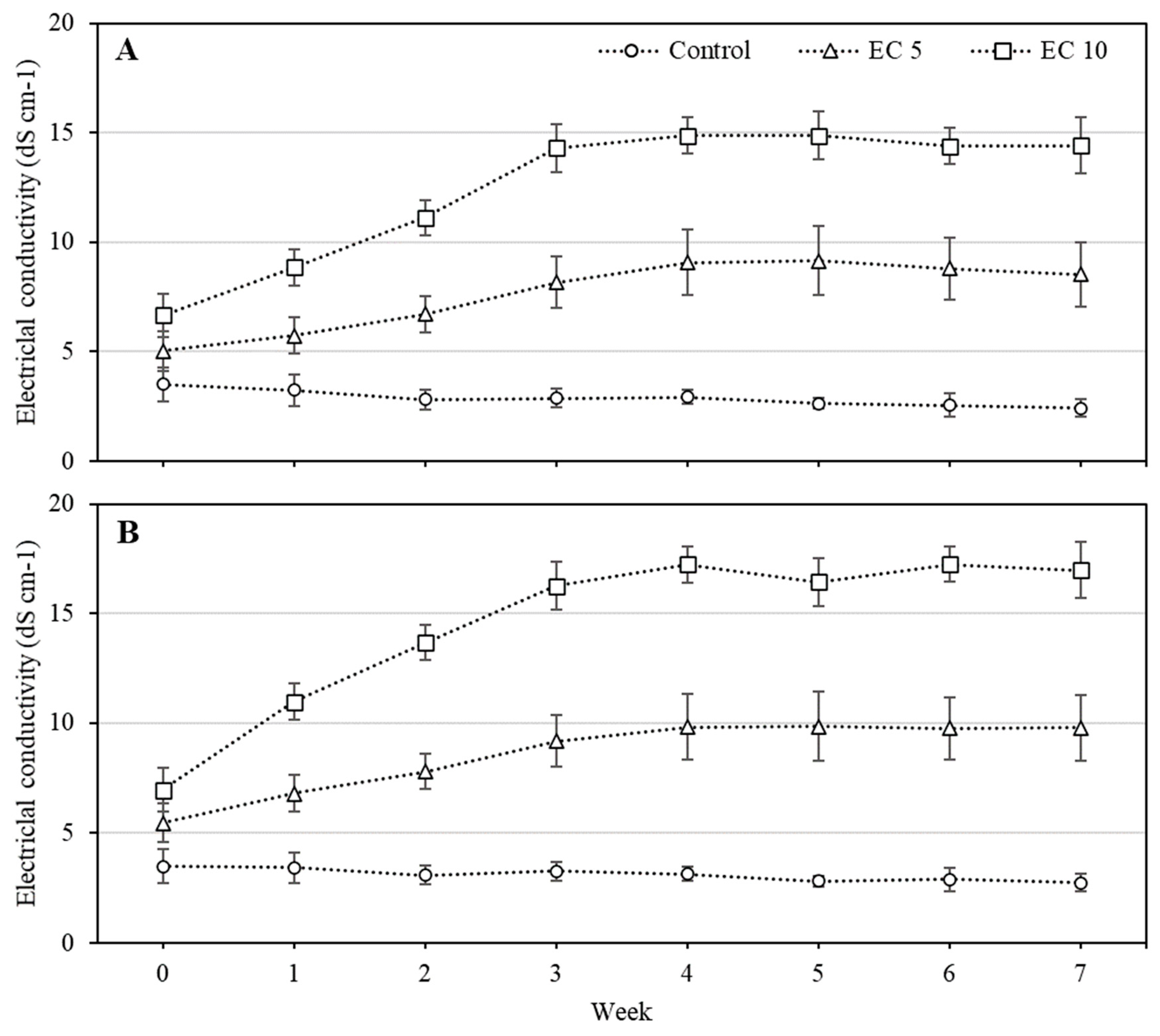
3.1. Leachate EC
3.2. Visual Quality
3.3. Plant Growth
3.4. Mineral Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Garcia-Caparros, P.; Llanderal, A.; Pestana, M.; Correia, P.J.; Lao, M.T. Tolerance mechanisms of three potted ornamental plants grown under moderate salinity. Sci. Hortic. 2016, 201, 84–91. [Google Scholar] [CrossRef]
- Miralles, J.; Valdes, R.; Franco, J.A.; Banon, S.; Sanchez-Blanco, M.J. Irrigation of Hydrangea with saline reclaimed wastewater: Effects of freshwater flushing. Acta Hortic. 2013, 1000, 229–236. [Google Scholar] [CrossRef]
- Niu, G.; Cabrera, R.I. Growth and physiological responses of landscape plants to saline water irrigation—A review. HortScience 2010, 45, 1605–1609. [Google Scholar] [CrossRef]
- Tanji, K.; Grattan, S.; Grieve, C.; Harivandi, A.; Rollins, L.; Shaw, D.; Sheikh, B.; Wu, L. A Comprehensive Literature Review on Salt Management Guide for Landscape Irrigation with Recycled Water in Coastal Southern California. Available online: http://www.salinitymanagement.org (accessed on 10 July 2020).
- Hancioglu, N.E.; Kurunc, A.; Tontul, I.; Topuz, A. Irrigation water salinity effects on oregano (Origanum onites L.) water use, yield and quality parameters. Sci. Hortic. 2019, 247, 327–334. [Google Scholar] [CrossRef]
- Shannon, M.C.; Grieve, C.M. Tolerance of vegetable crops to salinity. Sci. Hortic. 1999, 78, 5–38. [Google Scholar] [CrossRef]
- Fulcher, A.; Owen, J.; Lebude, A. Hydrangea Production: Species-specific Production Guide. Available online: https://extension.tennessee.edu/publications/Documents/PB1840-B.pdf (accessed on 11 July 2020).
- Alexander, L.W. Optimizing pollen germination and pollen viability estimates for Hydrangea macrophylla, Dichroa febrifuga, and their hybrids. Sci. Hortic. 2019, 246, 244–250. [Google Scholar] [CrossRef]
- Khaing, M.T.; Jung, H.J.; Han, T.H. Trend of hydrangea cultivar development. Trends Agric. Life Sci. 2016, 53, 63–68. [Google Scholar]
- Reed, S.M.; Jones, K.D.; Rinehart, T.A. Production and characterization of intergeneric hybrids between Dichroa febrifuga and Hydrangea macrophylla. J. Am. Soc. Hortic. Sci. 2008, 133, 84–91. [Google Scholar] [CrossRef]
- Nordli, E.; Strom, M.; Torre, S. Temperature and photoperiod control of morphology and flowering time in two greenhouse-grown Hydrangea macrophylla cultivars. Sci. Hortic. 2011, 127, 372–377. [Google Scholar] [CrossRef]
- Wu, L.; Dodge, L. A special report for the Elvenia J. Slosson Endowment Fund, Landscape salt tolerance selection guide for recycled water irrigation. University of California, Davis, CA. Available online: http://slosson.ucdavis.edu/files/215300.pdf (accessed on 10 July 2020).
- Liu, Q.; Sun, Y.; Niu, G.; Altland, J.; Chen, L.; Jiang, L. Morphological and physiological responses of ten ornamental species to saline water irrigation. HortScience 2017, 52, 1816–1822. [Google Scholar] [CrossRef]
- Conolly, N.B.; Bassuk, N.L.; MacRae, P.F. Response of five hydrangea species to foliar salt spray. J. Environ. Hortic. 2010, 2, 125–128. [Google Scholar] [CrossRef]
- Wu, S.; Sun, Y.; Niu, G.; Altland, J.; Cabrera, R. Response of 10 aster species to saline water irrigation. HortScience 2016, 51, 197–201. [Google Scholar] [CrossRef]
- Cai, X.; Sun, Y.; Starman, T.; Hall, C.; Niu, G. Response of 18 Earth-Kind® rose cultivars to salt stress. HortScience 2014, 49, 544–549. [Google Scholar] [CrossRef]
- Cavins, T.J.; Whipker, B.E.; Fonteno, W.C. Pourthru: A method for monitoring nutrition in the greenhouse. Acta Hortic. 2008, 779, 289–297. [Google Scholar] [CrossRef]
- Gavlak, R.G.; Horneck, D.A.; Miller, R.O. Plant, Soil, and Water Reference Methods for the Western Region; Western Regional Extension Publication (WREP), 125: Corvallis, OR, USA, 1994. [Google Scholar]
- Havlin, J.L.; Soltanpour, P.N. A nitric acid and plant digest method for use with inductively coupled plasma spectrometry. Commun. Soil Sci. Plant Anal. 1989, 14, 969–980. [Google Scholar] [CrossRef]
- Isaac, R.A.; Johnson, W.C. Collaborative study of wet and dry ashing techniques for the elemental analysis of plant tissue by atomic absorption spectrophotometry. J. Assoc. Off. Anal. Chem. 1975, 58, 436–440. [Google Scholar] [CrossRef]
- Chen, L.; Sun, Y.; Niu, G.; Liu, Q.; Altland, J. Relative salt tolerance of eight Japanese barberry cultivars. HortScience 2017, 52, 1810–1815. [Google Scholar] [CrossRef]
- De Oliveira, E.V.; de Lacerda, C.F.; Neves, A.L.R.; Gheyi, H.R.; Oliveira, D.R.; de Oliveira, F.I.F.; de Araujo Viana, T.V. A new method to evaluate salt tolerance of ornamental plants. Theor. Exp. Plant Physiol. 2018, 30, 173–180. [Google Scholar] [CrossRef]
- Niu, G.; Starman, T.; Byrne, D. Responses of growth and mineral nutrition of garden roses to saline water irrigation. HortScience 2013, 48, 756–761. [Google Scholar] [CrossRef]
- Niu, G.; Davis, T.D.; Masabni, J. A review of salinity tolerance research in horticultural crops. J. Arid Land Stud. 2019, 29, 53–59. [Google Scholar]
- Niu, G.; Rodriguez, D.; McKenney, S. Response of selected wildflower species to saline water irrigation. HortScience 2012, 47, 1351–1355. [Google Scholar] [CrossRef]
- Knight, H.; Trewavas, A.J.; Knight, M.R. Calcium signalling in Arabidopsis thaliana responding to drought and salinity. Plant J. 1997, 12, 1067–1078. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.X.; Li, P.M.; Gao, H.Y. Alleviation of photoinhibition by calcium supplement in salt-treated Rumex leaves. Physiol. Plant. 2007, 129, 386–396. [Google Scholar] [CrossRef]
- Epstein, E. How calcium enhances plant salt tolerance. Science 1998, 280, 1906–1907. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.L.; Flowers, T.J.; Wang, S.M. Mechanisms of sodium uptake by roots of higher plants. Plant Soil 2010, 326, 45. [Google Scholar] [CrossRef]
- Flowers, T.J.; Colmer, T.D. Salinity tolerance in halophytes. New Phytol. 2008, 179, 945–963. [Google Scholar] [CrossRef]
- Epstein, E.; Bloom, A.J. Mineral Nutrition of Plants: Principles and Perspectives, 2nd ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2005; p. 400. [Google Scholar]
- Flowers, T.J.; Làuchli, A. Sodium versus potassium: Substitution and compartmentation. In Encyclopedia of Plant Physiology; Làuchli, A., Bieleski, R.L., Eds.; Inorganic Plant Nutrition 15B; Springer: New York, NY, USA, 1983; Volume 15, pp. 651–681. [Google Scholar]
- White, P.J.; Broadley, M.R. Chloride in soils and its uptake and movement within the plant: A review. Ann. Bot. 2001, 88, 967–988. [Google Scholar] [CrossRef]
- Green, T.G.A.; Sancho, L.G.; Pintado, A.; Saco, D.; Martin, S.; Arroniz-Crespo, M.; Casermeiro, M.A.; de la Cruz Caravaca, M.T.; Cameron, S.; Rozzi, R. Sodium chloride accumulation in glycophyte plants with cyanobacterial symbionts. AoB Plants 2017, 9, plx053. [Google Scholar] [CrossRef]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef]
- Cabrera, R.I. Revisiting the salinity tolerance of crapemyrtles (Lagerstroemia spp.). Arboric. Urban For. 2009, 35, 129–134. [Google Scholar]
- Niu, G.; Xu, W.; Rodriguez, D.; Sun, Y. Growth and physiological responses of maize and sorghum genotypes to salt stress. ISRN Agron. 2012. [Google Scholar] [CrossRef]
- Niu, G.; Sun, Y. Salt tolerance in roses—A review. Acta Hortic. 2019, 1232, 143–150. [Google Scholar] [CrossRef]
| Species | Cultivar | Common Name |
|---|---|---|
| Hydrangea macrophylla | ‘Ayesha’ | Bigleaf hydrangea |
| H. macrophylla | ‘Emotion’ | Bigleaf hydrangea |
| H. macrophylla | ‘Mathilda Gutges’ | Bigleaf hydrangea |
| H. macrophylla | ‘Merritt’s Supreme’ | Bigleaf hydrangea |
| H. macrophylla | ‘Passion’ | YouMe® Bigleaf hydrangea |
| H. paniculata | ‘Interhydia’ | Pink Diamond hardy hydrangea |
| H. paniculata | ‘Bulk’ | Quickfire® panicle hydrangea |
| H. quercifolia | ‘Snowflake’ | Oakleaf hydrangea |
| H. serrata | ‘Preciosa’ | Mountain hydrangea |
| H. serrata × macrophylla | ‘Sabrina’ | Hybrid hydrangea |
| H. serrata × macrophylla | ‘Selina’ | Hybrid hydrangea |
| Cultivar | Visual Score z | Leaf Area (cm2) | Shoot DW (g) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | EC 5 | EC 10 | Control | EC 5 | EC 10 | Control | EC 5 | EC 10 | |
| Experiment 1 | |||||||||
| Ayesha | 4.7 Aa y | 3.9 Ab | 3.8 ABb | 3129 a | 1635 b | 1315 b | 26.5 Aa | 15.9 Ab | 13.9 Ab |
| Emotion | 4.0 Ba | 3.6 Ab | 2.1 Dc | 1675 a | 1166 b | 466 c | 15.6 BCa | 11.6 ABCa | 5.4 Bb |
| Mathilda Gutges | 4.5 ABa | 3.9 Ab | 2.8 Cc | 1674 a | 1158 b | 575 c | 16.7 BCa | 11.4 ABCb | 6.3 Bc |
| Merritt’s Supreme | 4.3 ABa | 3.7 Ab | 3.2 BCb | 1602 a | 978 b | 652 c | 13.7 BCDEa | 9.2 BCDb | 6.9 Bb |
| Passion | 4.1 ABa | 2.8 BCb | 0.8 Ec | 1838 a | 1080 b | 206 c | 16.8 BCa | 13.7 ABa | 4.7 Bb |
| Interhydia | 4.5 ABa | 3.6 Ab | 0.6 Ec | 1228 a | 795 b | - x | 15.2 BCDa | 9.2 BCDb | 6.0 Bb |
| Bulk | 3.0 Ca | 1.1 Db | 0.5 Ec | 507 a | 234 b | - | 6.7 Ea | 4.2 Da | 4.0 Ba |
| Snowflake | 4.4 ABa | 4.1 Aa | 0.6 Eb | 2211 a | 934 b | 814 b | 17.9 Ba | 7.8 CDb | 5.5 Bb |
| Preciosa | 3.1 Ca | 2.4 Cb | 0.7 Ec | 984 a | 514 b | 150 c | 8.0 DEa | 4.8 Db | 3.6 Bb |
| Sabrina | 3.9 Ba | 3.8 Aa | 4.0 Aa | 2257 a | 1528 b | 1205 b | 20.1 ABa | 14.6 CDb | 11.5 Ab |
| Selina | 3.2 Ca | 3.4 ABa | 3.5 ABa | 1250 a | 1003 ab | 726 b | 8.7 CDEa | 8.5 CDa | 7.0 Ba |
| Treatment (T) | *** | *** | *** | ||||||
| Cultivar (C) | *** | *** | *** | ||||||
| T × C | *** | ** | *** | ||||||
| Experiment 2 | |||||||||
| Ayesha | 4.0 ABCa y | 4.0 Aa | 3.3 Ab | 2604 a | 1996 b | 1333 c | 23.1 ABa | 21.7 Aa | 14.9 Ab |
| Emotion | 3.5 BCDa | 2.5 DEb | 1.7 CDc | 1353 a | 947 a | 187 b | 14.8 CDa | 11.7 BCa | 10.6 ABCa |
| Mathilda Gutges | 4.0 ABCa | 3.5 ABCa | 2.3 BCb | 1305 a | 1064 a | 531 b | 14.8 CDa | 10.2 BCDb | 7.2 CDc |
| Merritt’s Supreme | 3.4 BCDa | 3.2 ABCDa | 2.6 ABCa | 1429 a | 843 b | 724 b | 12.8 CDEa | 8.9 BCDEa | 10.3 ABCa |
| Passion | 3.3 CDa | 2.0 Eb | 1.0 DEFc | 1462 a | 934 b | - x | 15.4 BCDa | 13.1 BCb | 4.4 Dc |
| Interhydia | 4.2 ABa | 2.7 CDEb | 0.5 Fc | 1182 a | 573 b | - | 13.8 CDa | 7.9 CDEab | 6.9 CDb |
| Bulk | 3.0 Da | 0.8 Fb | 0.5 EFb | 315 | - | - | 5.8 Ea | 4.4 DEa | 4.1 CDa |
| Snowflake | 4.7 Aa | 3.7 ABa | 1.8 CDb | 2513 a | 1598 ab | 905 b | 24.0 Aa | 14.7 Bab | 9.1 BCDb |
| Preciosa | 3.0 Da | 2.2 Eb | 1.4 DEc | 831 a | 352 b | 209 b | 8.4 DEa | 4.0 Eb | 4.3 Db |
| Sabrina | 3.0 Db | 3.7 ABa | 3.0 ABb | 1966 a | 1496 b | 1136 c | 16.9 ABCa | 14.0 Bab | 12.3 ABb |
| Selina | 3.5 BCDa | 2.8 BCDEa | 2.8 ABa | 1166 a | 824 b | 549 b | 10.6 CDEa | 7.9 CDEb | 6.4 CDb |
| Treatment (T) | *** | *** | *** | ||||||
| Cultivar (C) | *** | *** | *** | ||||||
| T × C | *** | *** | *** | ||||||
| Cultivar | Na+ (mg·g−1) | Cl− (mg·g−1) | Ca2+ (mg·g−1) | K+ (mg·g−1) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | EC 5 | EC 10 | Control | EC 5 | EC 10 | Control | EC 5 | EC 10 | Control | EC 5 | EC 10 | |
| Ayesha | 0.8 Cc z | 6.2 CDb | 9.7 CDa | 3.3 Dc | 30.3 CDb | 44.2 CDEa | 11.6 Eb | 19.8 CDa | 22.0 Ca | 39.5 BCa | 39.4 Ba | 40.0 ABCDa |
| Emotion | 1.5 Ac | 12.8 Bb | 31.6 Aa | 4.4 Dc | 46.2 ABb | 92.1 Aa | 18.7 CDc | 28.7 CDb | 37.8 ABa | 33.4 DEFb | 34.1 BCDab | 37.3 BCDa |
| Mathilda Gutges | 0.9 Cb | 5.6 CDb | 14.8 BCa | 5.2 CDc | 32.9 BCDb | 65.2 Ba | 16.8 CDEc | 23.6 CDb | 31.4 BCa | 39.7 BCa | 40.3 Ba | 43.8 ABa |
| Merritt’s Supreme | 1.5 ABc | 6.0 CDb | 10.5 BCDa | 6.9 BCc | 31.1 CDb | 49.2 BCDEa | 16.8 CDEb | 21.2 CDb | 29.9 BCa | 37.2 BCDb | 38.2 BCb | 42.1 ABCa |
| Passion | 1.7 Ac | 19.0 Ab | 35.8 Aa | 4.6 Dc | 54.8 Ab | 96.7 Aa | 21.2 BCb | 31.6 BCa | 33.0 BCa | 38.3 BCDa | 35.9 BCDab | 33.2 Db |
| Interhydia | 0.2 Db | 3.3 Db | 16.9 BCa | 0.6 Ec | 36.2 BCb | 61.4 BCa | 26.6 Bc | 41.9 ABb | 47.6 Aa | 29.0 EFb | 29.2 Dab | 35.1 CDa |
| Bulk | 0.4 CDa | 2.0 Da | 5.6 Da | 1.1 Eb | 28.2 CDa | 31.2 Ea | 34.0 Aa | 45.0 Aa | 38.1 ABa | 28.6 Fa | 34.5 BCDa | 33.8 CDa |
| Snowflake | 0.1 Db | 2.3 Db | 11.6 BCDa | 0.9 Ec | 20.8 Db | 55.2 BCDa | 15.5 CDEc | 28.1 CDb | 34.2 BCa | 34.5 CDEab | 31.5 CDb | 39.2 BCDa |
| Preciosa | 1.0 BCb | 2.9 Db | 16.8 BCa | 10.3 Ac | 28.9 CDb | 66.4 Ba | 18.1 CDc | 22.8 CDb | 29.8 BCa | 51.0 Aa | 51.2 Aa | 48.4 Aa |
| Sabrina | 1.8 Ac | 9.7 BCb | 18.8 Ba | 8.1 Bc | 32.6 BCDb | 60.5 BCDa | 14.6 DEb | 16.8 Db | 22.8 Ca | 40.9 Ba | 36.9 BCa | 37.3 BCDa |
| Selina | 1.7 Ac | 9.7 BCb | 17.4 BCa | 4.0 Dc | 21.7 CDb | 42.3 DEa | 13.2 DEb | 16.6 Dab | 21.7 Ca | 49.2 Aa | 40.4 Bb | 39.1 BCDb |
| Treatment (T) | *** | *** | *** | * | ||||||||
| Cultivar (C) | *** | *** | *** | *** | ||||||||
| T × C | *** | *** | * | *** | ||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niu, G.; Sun, Y.; Hooks, T.; Altland, J.; Dou, H.; Perez, C. Salt Tolerance of Hydrangea Plants Varied among Species and Cultivar within a Species. Horticulturae 2020, 6, 54. https://doi.org/10.3390/horticulturae6030054
Niu G, Sun Y, Hooks T, Altland J, Dou H, Perez C. Salt Tolerance of Hydrangea Plants Varied among Species and Cultivar within a Species. Horticulturae. 2020; 6(3):54. https://doi.org/10.3390/horticulturae6030054
Chicago/Turabian StyleNiu, Genhua, Youping Sun, Triston Hooks, James Altland, Haijie Dou, and Christina Perez. 2020. "Salt Tolerance of Hydrangea Plants Varied among Species and Cultivar within a Species" Horticulturae 6, no. 3: 54. https://doi.org/10.3390/horticulturae6030054
APA StyleNiu, G., Sun, Y., Hooks, T., Altland, J., Dou, H., & Perez, C. (2020). Salt Tolerance of Hydrangea Plants Varied among Species and Cultivar within a Species. Horticulturae, 6(3), 54. https://doi.org/10.3390/horticulturae6030054








