Fresh and Dry Weight Relations Are Predictors of Cycas micronesica Seed Age
Abstract
1. Introduction
2. Materials and Methods
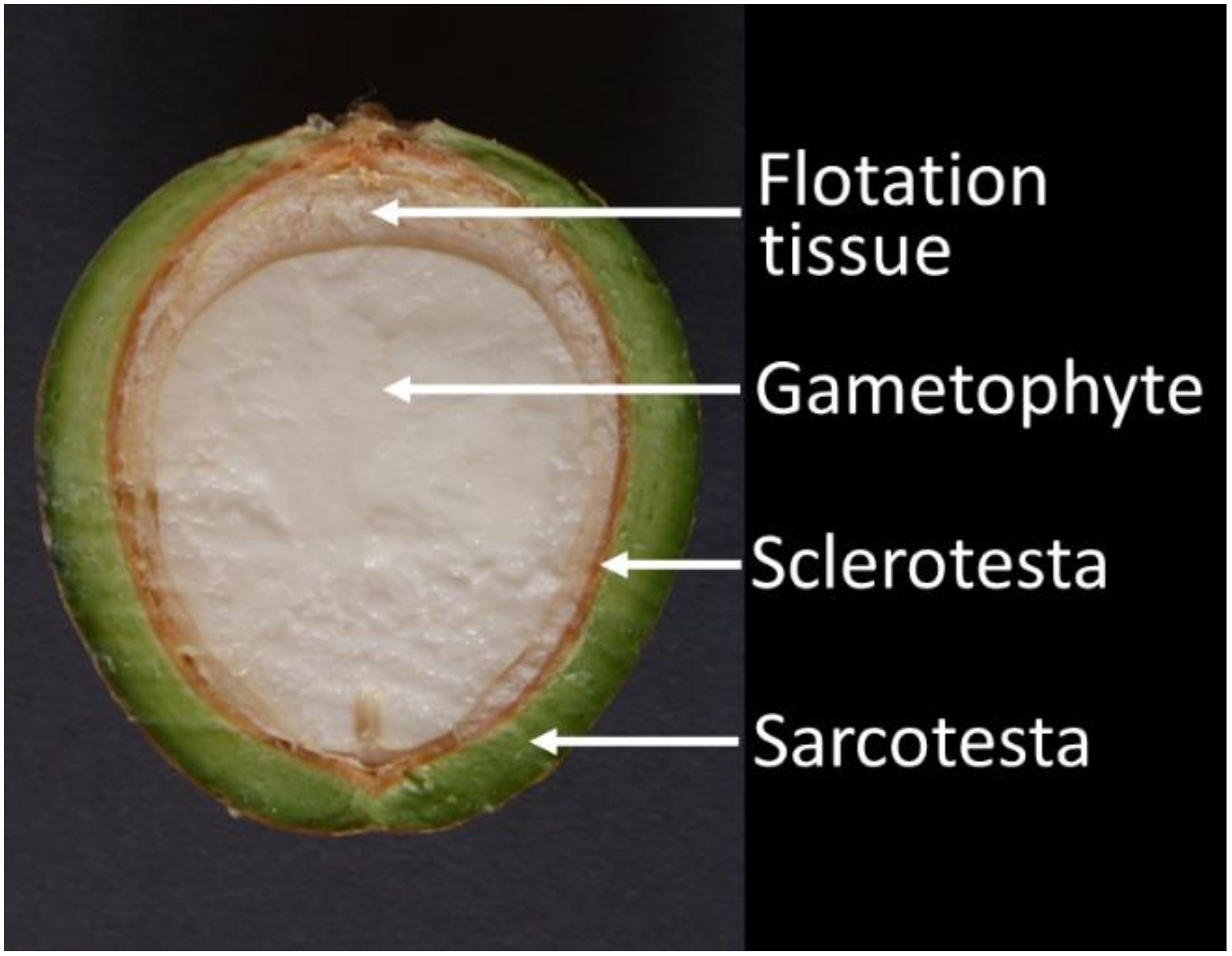
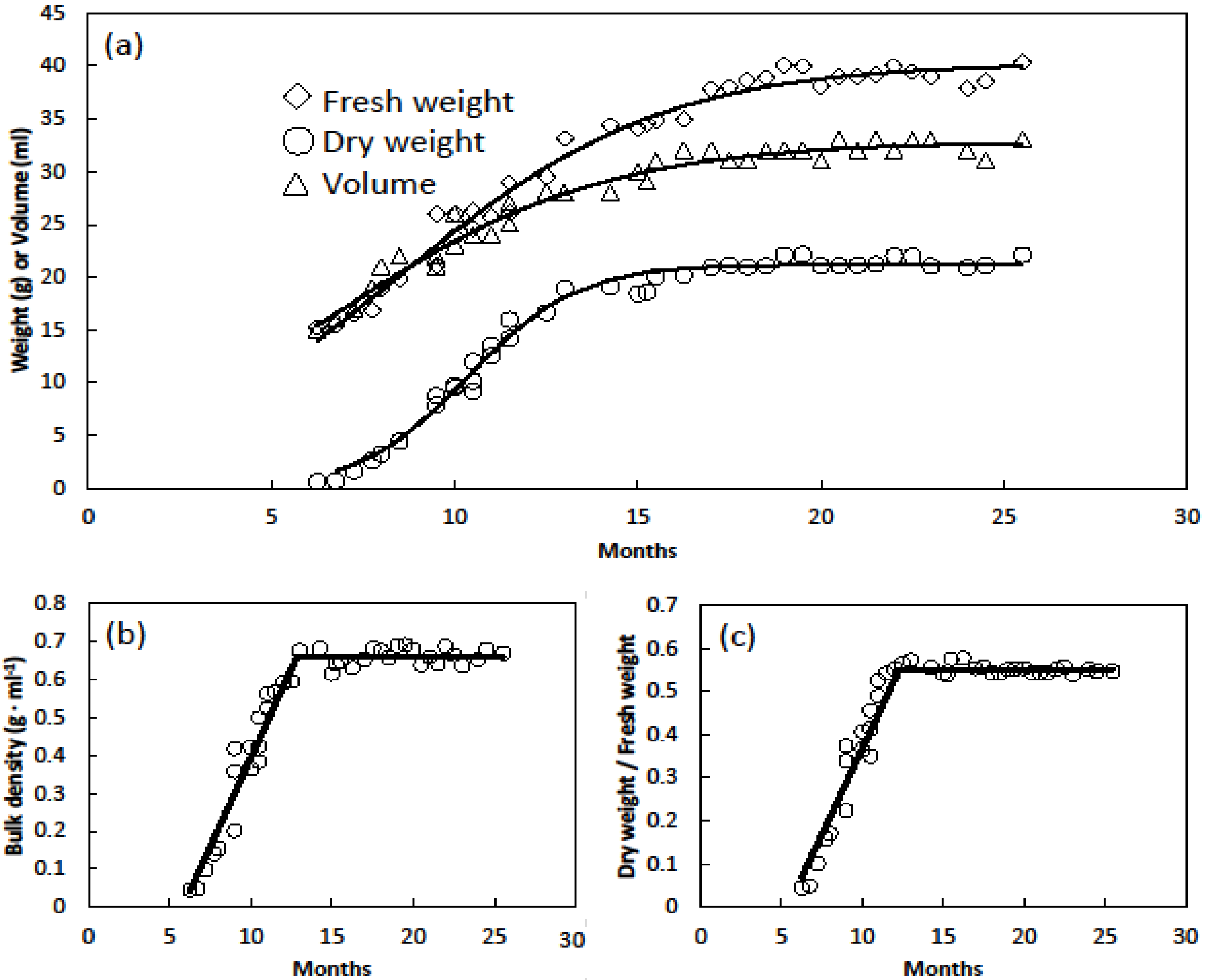
2.1. Gametophyte Study
2.2. Whole Seed Studies
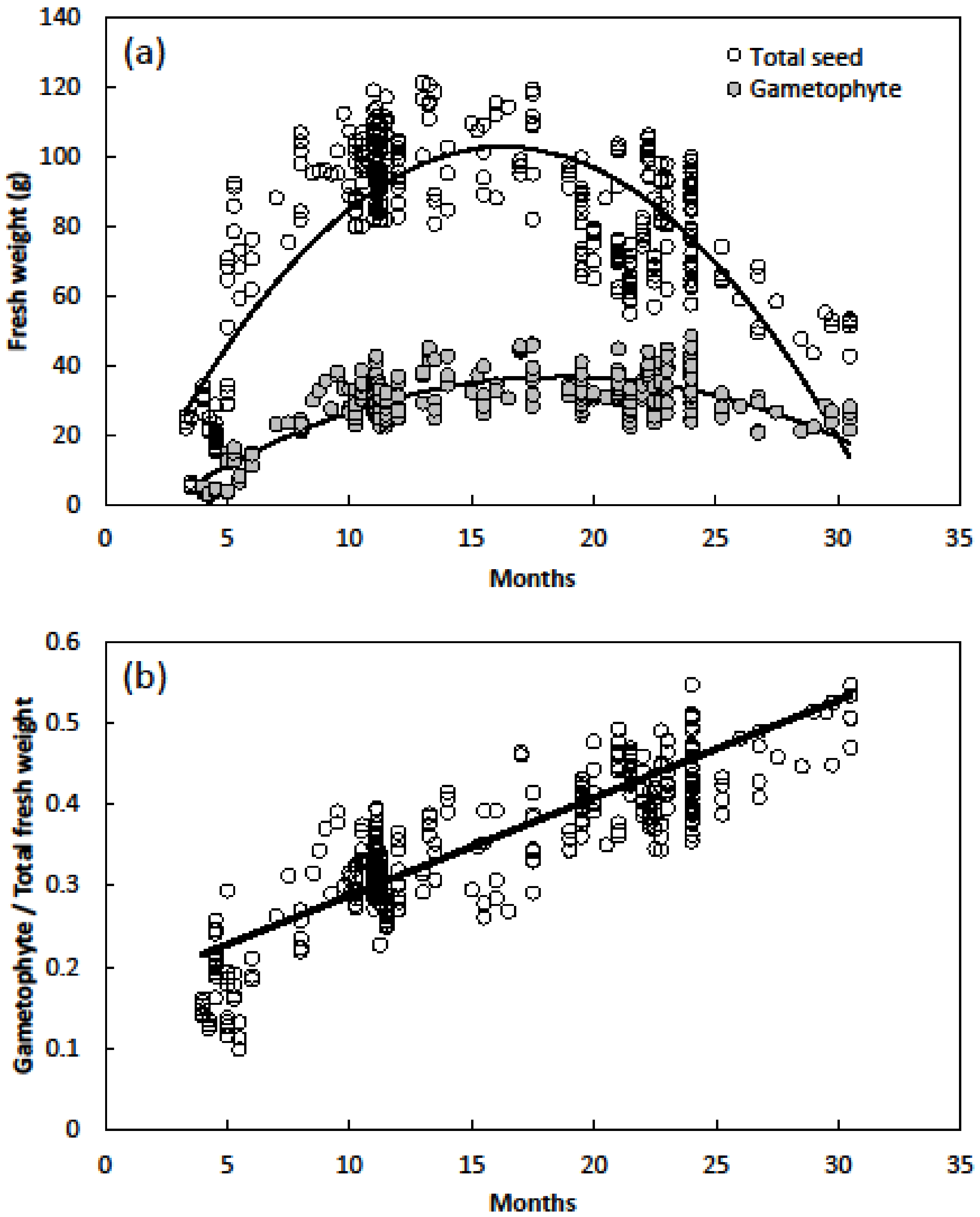
- Fresh weight relations of seeds were determined by comparing gametophyte weight with whole seed weight. Seed age ranged from 3 to 30 months and included 394 seeds.
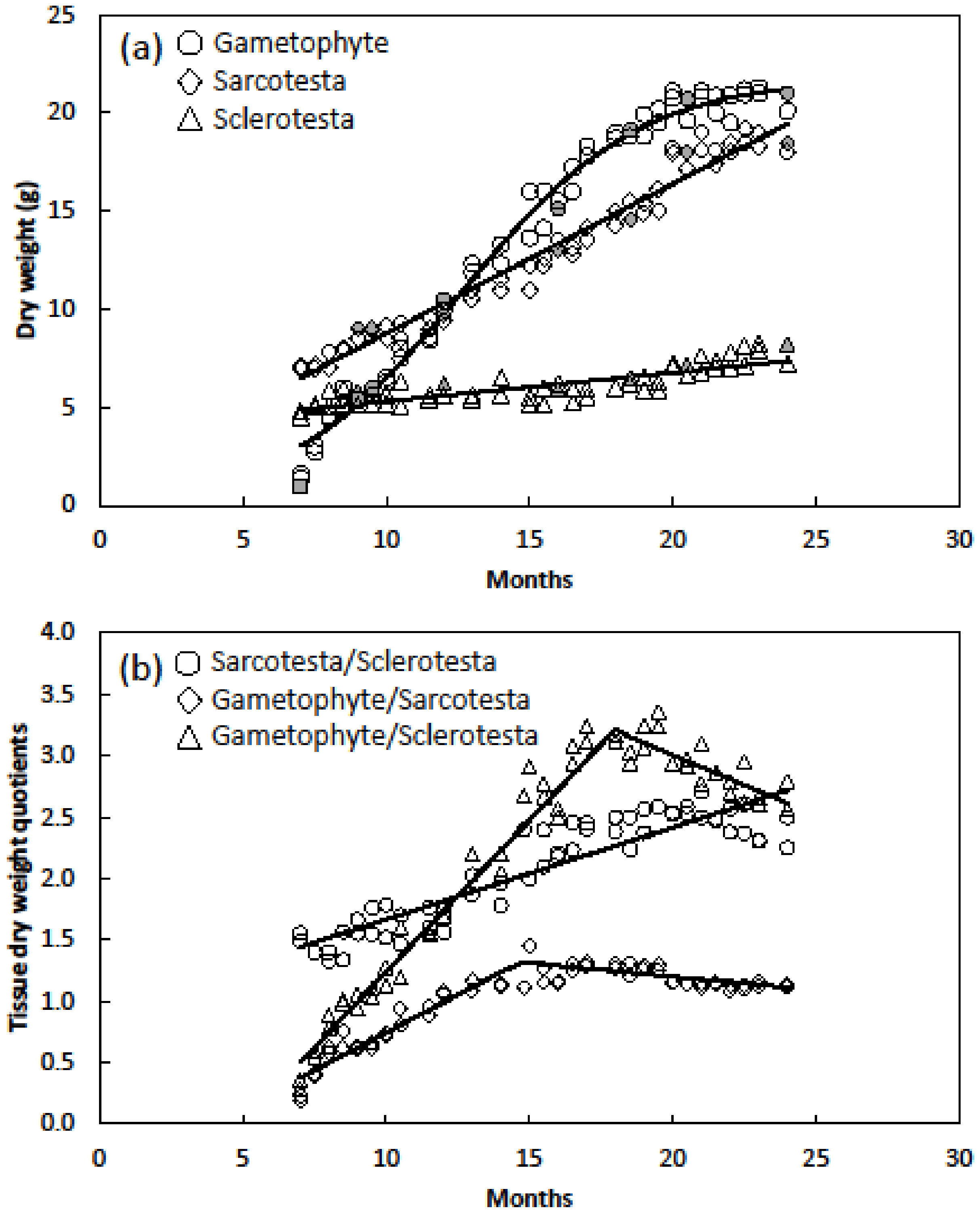
- Dry weight relations of seeds were determined by separating seeds into sarcotesta, sclerotesta and gametophyte tissue. The flotation tissue was included with the sclerotesta category. For older seeds, the embryo was included with the gametophyte category. Seed age ranged from 7 to 24 months and included 60 seeds. To provide a check on the influence of sequentially harvesting seeds of more than one age from the same trees, a population of eight trees were set aside to supply a single seed age.
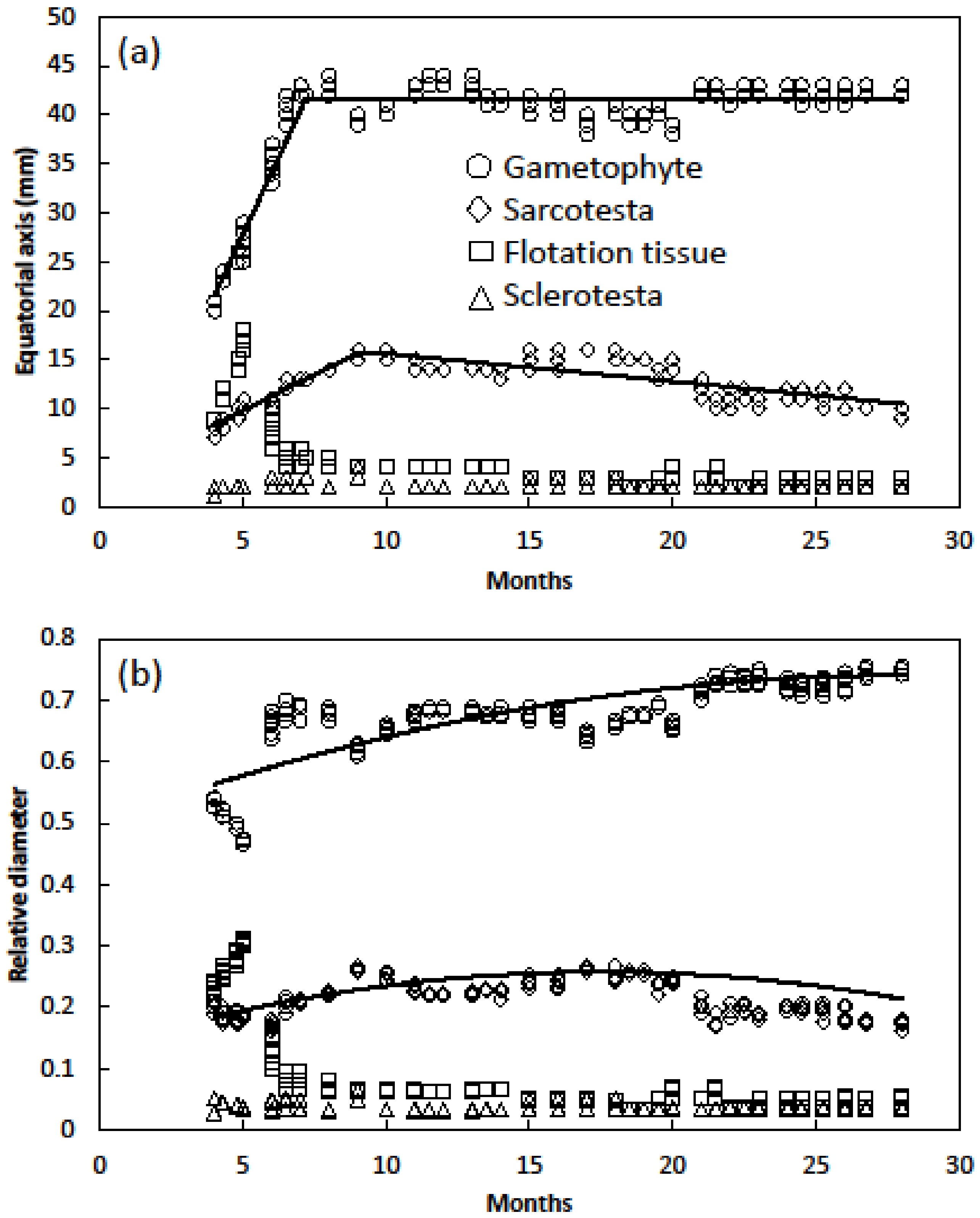
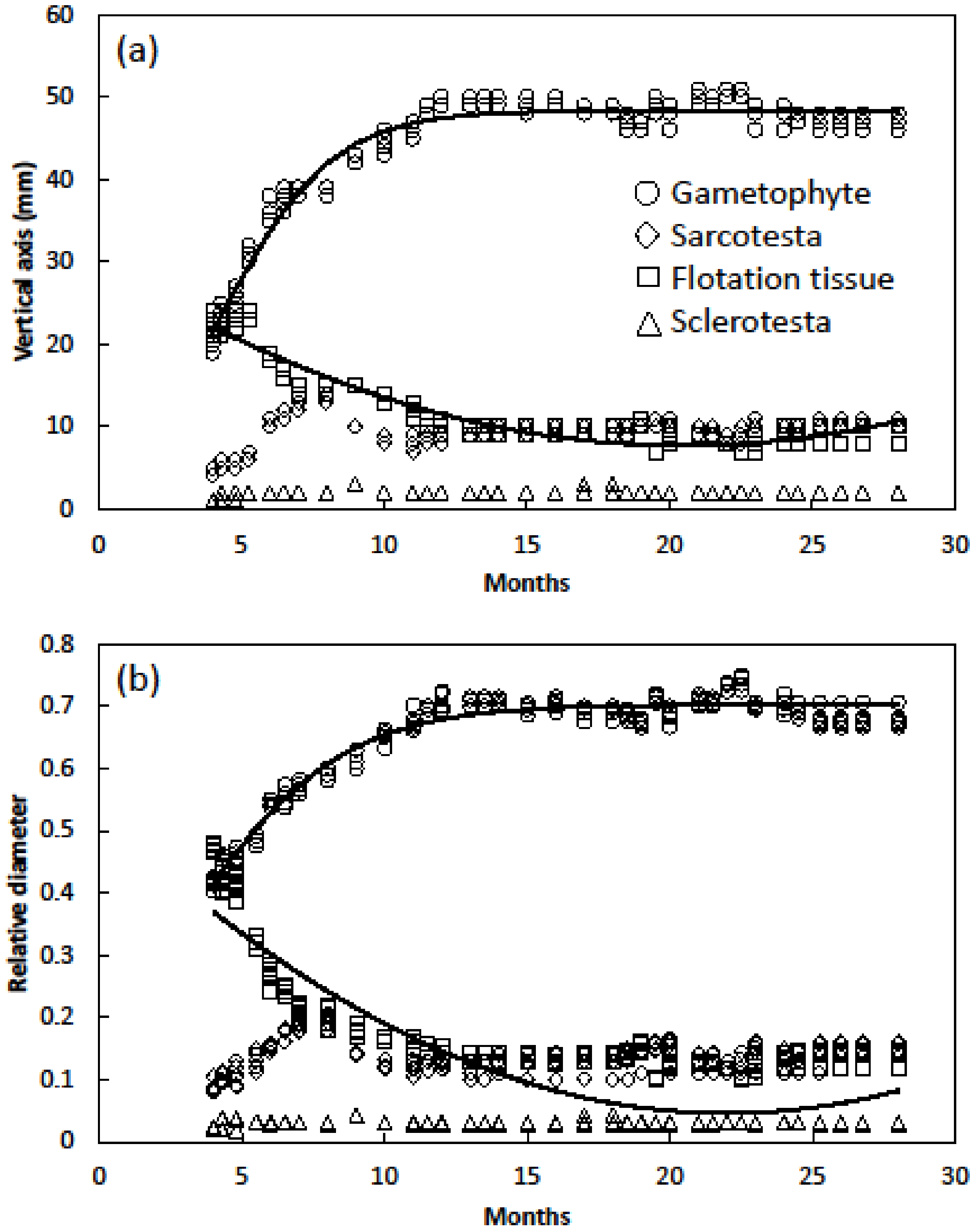
- Seed equatorial dimension relationships were determined by cutting each seed in half then measuring the diameter of sarcotesta, sclerotesta, flotation tissue and gametophyte tissue. Total diameter of each category was determined by adding the diameters of each side. Seed age ranged from 4 to 28 months and included 210 seeds.
- Seed longitudinal dimension relationships were determined with the same seeds by measuring the diameter of sarcotesta, sclerotesta, flotation tissue and gametophyte. Seed age ranged from 4 to 28 months and included 210 seeds.
2.3. Analyses
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rieger, M. Introduction to Fruit Crops; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Prasad, K.; Jacob, S.; Siddiqui, M.W. Fruit maturity, harvesting, and quality standards. In Fruit Maturity, Harvesting, and Quality Standards; Siddiqui, M.W., Ed.; Academic Press: Waltham, MA, USA, 2018; pp. 41–69. [Google Scholar]
- Cox, J. From Vines to Wines; Storey Books: North Adams, MA, USA, 1999. [Google Scholar]
- Gasic, K.; Reighard, G.L.; Windham, J.; Ognjanov, M. Relationships between fruit maturity at harvest and fruit quality in peach. Acta Hortic. 2015, 1084, 643–648. [Google Scholar] [CrossRef]
- Vinson, E.L.; Woods, F.M.; Kemble, J.M.; Perkins-Veazie, P.; Davis, A.; Kessler, J. Use of external indicators to predict maturity of mini-watermelon fruit. HortScience 2010, 45, 1034–1037. [Google Scholar] [CrossRef]
- Norstog, K.J.; Nicholls, T.J. The Biology of the Cycads; Cornell University Press: Ithaca, NY, USA, 1997. [Google Scholar]
- Hill, K.D. The Cycas rumphii complex (Cycadaceae) in New Guinea and the western Pacific. Aust. Syst. Bot. 1994, 7, 543–567. [Google Scholar] [CrossRef]
- Marler, T.E.; Cruz, G.N. Source and sink relations mediate depletion of intrinsic cycad seed carbohydrates by Aulacaspis yasumatsui infestation. HortScience 2019, 54, 1712–1717. [Google Scholar] [CrossRef]
- Marler, T.E.; Lee, V.; Shaw, C.A. Spatial variation of steryl glucosides in Cycas micronesica plants: Within- and among-plant sampling procedures. HortScience 2005, 40, 1607–1611. [Google Scholar] [CrossRef]
- Marler, T.E.; Lee, V.; Chung, J.; Shaw, C.A. Steryl glucoside concentration declines with Cycas micronesica seed age. Funct. Plant Biol. 2006, 33, 857–862. [Google Scholar] [CrossRef]
- Marler, T.E.; Shaw, C.A. Free and glycosylated sterol bioaccumulation in developing Cycas micronesica seeds. Food Chem. 2009, 115, 615–619. [Google Scholar] [CrossRef] [PubMed]
- Marler, T.E.; Lindström, A.J. Scouting the Philippines for Cycas. Mem. N. Y. Bot. Gard. 2018, 117, 519–528. [Google Scholar]
- Marler, T.E.; Lee, V.; Shaw, C.A. Cycad toxins and neurological diseases in Guam: Defining theoretical and experimental standards for correlating human disease with environmental toxins. HortScience 2005, 40, 1598–1606. [Google Scholar] [CrossRef]
- Whitelock, L.M. The Cycads; Timber Press: Portland, OR, USA, 2002. [Google Scholar]
- Jones, D.L. Cycads of the World; Smithsonian Institution Press: Washington, DC, USA, 1993. [Google Scholar]
- Kurland, L.T. Amyotrophic lateral sclerosis and Parkinson’s disease complex on Guam linked to an environmental toxin. Trends Neurosci. 1988, 11, 51–53. [Google Scholar] [CrossRef]
- Mabry, T.J. Selected topics from forty years of natural products research: Betalains to flavonoids, antiviral proteins, and neurotoxic nonprotein amino acids. J. Nat. Prod. 2001, 64, 1596–1604. [Google Scholar] [CrossRef] [PubMed]
- Marler, T.; Haynes, J.; Lindstrom, A. Cycas micronesica. In The IUCN Red List of Threatened Species; IUCN: Gland, Switzerland, 2010. [Google Scholar] [CrossRef]
- Cibrian-Jaramillo, A.; Daly, A.C.; Brenner, E.; DeSalle, R.; Marler, T.E. When North and South don’t mix: Genetic connectivity of a recently endangered oceanic cycad, Cycas micronesica, in Guam using EST-microsatellites. Mol. Ecol. 2010, 19, 2364–2379. [Google Scholar] [CrossRef]
- Marler, T.E.; Dongol, N. Models to describe Cycas micronesica leaf and strobili development. HortScience 2011, 46, 1333–1337. [Google Scholar] [CrossRef]
- Marler, T.E.; Niklas, K. Reproductive effort and success of Cycas micronesica K.D. Hill are affected by habitat. Int. J. Plant Sci. 2011, 172, 700–706. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marler, T.E.; Shaw, C.A. Fresh and Dry Weight Relations Are Predictors of Cycas micronesica Seed Age. Horticulturae 2020, 6, 29. https://doi.org/10.3390/horticulturae6020029
Marler TE, Shaw CA. Fresh and Dry Weight Relations Are Predictors of Cycas micronesica Seed Age. Horticulturae. 2020; 6(2):29. https://doi.org/10.3390/horticulturae6020029
Chicago/Turabian StyleMarler, Thomas E., and Christopher A. Shaw. 2020. "Fresh and Dry Weight Relations Are Predictors of Cycas micronesica Seed Age" Horticulturae 6, no. 2: 29. https://doi.org/10.3390/horticulturae6020029
APA StyleMarler, T. E., & Shaw, C. A. (2020). Fresh and Dry Weight Relations Are Predictors of Cycas micronesica Seed Age. Horticulturae, 6(2), 29. https://doi.org/10.3390/horticulturae6020029





