1. Introduction
Blueberry is a woody perennial species in the family Ericaceae and genus
Vaccinium. The fruit is a true berry with many seeds, with color ranging from light blue to black and a waxy cuticle layer [
1]. Blueberry has been gaining great importance in fruit production, especially because of its recognized taste properties and its nutraceutical qualities as an anti-inflammatory and anti-oxidant, being a health-promoting food [
2]. Blueberry fruits are rich in polyphenols [
3]. These blueberry polyphenols show anti-inflammation activity, related to the balances in pro-inflammatory cytokines, and they could be used as an anti-inflammatory medicine [
4]. Among the phenolic compounds that appear at high levels in blueberries are anthocyanins [
5], flavonols, and phenolic acids [
6]. The anthocyanin found in high amounts in blueberries contributes to preventing several chronic diseases, such as neurodegenerative diseases, cardiovascular disorders, cancer, and diabetes [
7].
Much research has been developed related to the propagation of blueberries. Traditionally, blueberry is propagated by softwood, semi-hardwood, and hardwood cuttings [
8] or even rhizome cuttings of selected clones [
6]. Some challenges in this production are a very low rooting percentage in many genotypes, the amount of time required to propagate and commercialize newly-released cultivars for mass propagation [
8,
9], and phytosanitary problems. In vitro culture (micropropagation) can overcome the limitations of traditional cuttings, presenting an alternative for faster growth [
10] throughout the year (with no seasonal effects) without pathogens [
11]. There are some studies on the in vitro propagation of
Vaccinium species, but only some of these have been done with rabbiteye (
V. virgatum Ait. (syn.
V. ashei Reade)), specifically for the ‘Delite’ rabbiteye cultivar that is suitable for and adapted to regions of southern Brazil. For this specific cultivar, some research concerning in vitro protocol is still required to give more information on the optimal conditions for the development of this technique.
One crucial point in tissue culture techniques is the appropriate use, type, and concentration of growth regulators and the combination of culture medium salts that allows fast, efficient development of the initial explants. Understanding the interference of factors can lead to the development of further regeneration protocols that could be useful for either micropropagation or developing regeneration techniques necessary for plant recovery after cell transformation. There is some research showing that the lack of new shoot growth can make initiation the limiting step in establishing
Vaccinium cultures in vitro [
12]. Studies also show that new growth in vitro is difficult to achieve in
Vaccinium, especially when using plant material from the field [
13].
For the initial phase of in vitro culture, a combination of cytokinins can usually be used. In the initial in vitro culture in one study using nodal segments from softwood cuttings of ‘Ozarkblue’ highbush blueberry (
V. corymbosum), zeatin (ZEA) and 6-(γ-γ-dimethylallylamino)-purine (2Ip) were tested in the initial culture medium in different combinations (18 µM ZEA, 25 µM 2-iP, and 9.1 µM ZEA combined with 25 µM 2iP) using WPM as the basal medium. On medium with ZEA present, shoots developed with green and red leaves. However, on medium containing only 2iP, shoots had light red leaves and callus at the base with stunted growth [
9].
In lowbush blueberry (
V. angustifolium Ait.) cultivated in the initiation phase medium containing 5 µM ZEA or 10 µM 2iP, explants produced elongated shoots with both growth regulators. However, ZEA treatments showed a higher percentage of new shoot growth compared to 2iP for three cultivars [
6].
Wild bilberry (
V. myrtillus L.) and lingonberry (
V. vitis-idaea L.) were tested using buds and shoot tips on a modified MS medium supplemented with 2iP variations from 9.8 to 78.4 µM. For bilberry and lingonberry, the best results were obtained with 49.2 µM and 24.6 µM, respectively. Brownish explants were observed with an increasing 2iP concentration [
13]. For ‘Berkeley’, ‘Bluecrop’ and ‘Earliblue’, highbush blueberries, and ‘O’Neal’ southern highbush blueberry, a medium containing 20 µM ZEA was used in the initiation of cultures [
14].
Concerning the type of basal culture medium, many studies have used WPM as the basic medium for blueberry [
14]. However, some authors have tried to optimize this medium by making some modifications, such as combining MS and WPM media, creating an MW medium [
14], or proposing some changes in the components [
15], leading to a modified WPM. A well-balanced medium is important to prevent stunted growth and physiological disorders [
16]. Some authors have discussed the importance of the balance between nitrogen forms used in tissue culture (NO
3- and NH
4+) as much as the total amount of nitrogen in the culture medium [
17].
The objective of this work was to determine an efficient growth regulator and balance of nitrogen salts for the establishment of ‘Delite’ microcuttings in in vitro culture.
2. Materials and Methods
In this work, three experiments in initial in vitro culture were designed. In the first one, four different cytokinins (ZEA, 2iP, 6-benzylaminopurine (BAP), and kinetin (KIN)) were tested at eight different concentrations. The second experiment tested nine different combinations of the nitrogen salts (NH
4)
2SO
4, KNO
3, and Ca(NO
3)
2·4H
2O, using the modified WPM [
15] as the basic medium. The third experiment tested nine different combinations of two nitrogen salts, NH
4NO
3 and Ca(NO
3)
2·4H
2O, using the original WPM [
18] as the basic medium and compared them with treatment 10 (modified Woody Plant Medium [
15]).
2.1. Plant Material
One-year-old hardwood cuttings were collected during winter from field-grown rabbiteye blueberry ‘Delite’ mother plants at the Experimental Station of Universidade Federal do Paraná, Pinhais/PR. They were treated with an immersion in fungicide solution for 5 min (Cercobin® 0.2%) and stored at a 4 °C temperature at the Micropropagation Laboratory, UFPR, Curitiba/PR for one to two months in plastic bags. Cuttings were placed in glass containers with water in the culture room at 25 °C ± 2 °C under cool day light at 40 μmol m−2 s−1 with a 16-h photoperiod. Newly formed shoots were collected and used as explants for the establishment of cultures.
Two-node segments (0.8–2 cm in length, discarding the apical portion of the donor-explant) were collected and surface sterilized with 70% (v/v) ethanol for 30 s, followed by immersion in 0.5% sodium hypochlorite solution containing 0.1% (v/v) Tween 20 for 5 min. They were washed with sterile deionized water three times inside the laminar flow chamber.
2.2. Culture Medium and Growing Conditions
Explants were isolated in culture tubes (150 × 30 mm), with each containing 6 mL of modified culture medium, differing in each of the three experiments. In all experiments, the medium was supplemented with Murashige and Skoog (MS) [
19] vitamins, 30 g L
−1 sucrose, 0.1 g L
−1 myo-inositol, and 6 g L
−1 agar (Vetec®). The pH of all media was adjusted to 5.2 before autoclaving at 120 °C and 1.5 atm.
2.2.1. Experiment 1: Cytokinins
Microcuttings were isolated in the modified WPM [
2] (
Table 1), supplemented as described above. Eight different concentrations (0, 2.5, 5, 10, 20, 30, 40, and 50 µM) of each of the four cytokinin growth regulators, ZEA, 2iP, BAP, and KIN, were tested, for a total of 32 treatments. ZEA and 2iP, when used, were sterilized through 0.22 µm filters and added to the cooled media. BAP and KIN were added to media before autoclaving.
2.2.2. Experiment 2: Combinations of (NH4)2SO4, KNO3, and Ca(NO3)2·4H2O Using the Modified WPM [15] as the Basic Medium
Explants were isolated using nine different treatments as described in
Table 2, with different amounts (1×, 0.5× or 1.5×) of (NH
4)
2SO
4, KNO
3, and Ca(NO
3)
2·4H
2O (
Table 2), using the modified WPM [
2] (
Table 1) as the basic medium. Media were supplemented as described above, with the addition of cytokinin ZEA (5 µM).
2.2.3. Experiment 3: Combinations of NH4NO3 and Ca(NO3)2·4H2O Using the Original WPM [1] as the Basic Medium
In this third experiment, explants were isolated in 10 different treatments described in
Table 3. Nine treatments were used with different amounts (1×, 0.5× or 1.5×) of NH
4NO
3 and Ca(NO
3)
2·4H
2O, using the original WPM [
18] as the basic medium, and one treatment used the modified WPM [
15] (
Table 1). Media were supplemented as described above, with the addition of cytokinin ZEA (5 µM).
2.3. Growing Conditions
After isolation, cultures were transferred to a culture room and grown at 25 °C ± 2 °C in the dark for eight initial days and then transferred to a 16-h photoperiod with a light intensity of 40 μmol m−2 s−1 provided by cool-day fluorescent lamps.
2.4. Experimental Design, Data Collection, and Statistical Analysis
The experiments were conducted in a completely randomized design. In experiment 1, a two-factor experiment (4 × 8) design was used, with four different cytokinins (ZEA, 2iP, BAP, and KIN) in eight different concentrations (0, 2.5, 5, 10, 20, 30, 40, and 50 µM). There were 32 treatments in total. Each treatment had four replicates of 10 tubes each (one plant per tube), e.g., 40 plants per treatment, resulting in a total of 1280 plants.
In experiment 2, a completely randomized design was used, with nine treatments, according to
Table 2. Each treatment had three replicates of seven tubes each (one plant per tube), e.g., 21 plants per treatment, resulting in 189 plants.
In experiment 3, a completely randomized design was used, with 10 treatments (
Table 3). Each treatment had four replicates of 10 tubes each (one plant per tube), e.g., 40 plants per treatment, resulting in a total of 400 plants.
Plants were evaluated based on many aspects two months (Experiment 1) or three months (Experiments 2 and 3) after initial culture. Contaminated cultures were discarded and not included in the data analysis. Contamination rates ranged from 0 to 7.5% in experiment 1. The final number of explants evaluated is presented in
Table S1. In experiment 2, contamination rates ranged from 0 to 14%; and were 0 to 35% in experiment 3. Survival rate (%) and new shoot growth (%) were recorded. The number of new shoots formed per explant was counted (nº), the length of the longest shoot (millimeters from base to shoot tip) was measured, and the number of leaves of the longest shoot was counted (nº). All the plants were evaluated and had the mean estimated from the plants in each replication, and subsequently, the mean of the three or four replications in each treatment.
In experiment 1, ANOVA, Tukey, and regression analyses did not include values for the zero concentration treatments, since it was clear that a zero concentration did not have any influence on the explant development and it is not a concentration that labs would apply in practice. In the zero concentrations, there was no shoot formation in any of the explants evaluated. Since there was no shoot formation, there was no valid evaluation of number of shoots formed, length of shoot, or number of leaves per shoot. Hence, 28 treatments were statistically analyzed using a two-factor experiment (4 × 7), with four different cytokinins (ZEA, 2iP, BAP, and KIN) at seven different concentrations (2.5, 5, 10, 20, 30, 40, and 50 µM). The results were first transformed to the square root scale and then two-way ANOVA was performed (
Table S2) to detect any interaction between the two factors and to check for any statistically significant difference between treatments at levels 1 and 5%. In the case of interaction between factors, in the variable analyzed, two tests were performed. First, Tukey’s test (
P < 0.05) was performed for each of the cytokinins with each of the concentrations. For factor 2 (different concentrations), regression analysis was performed for each cytokinin with the original data. The best-fitting regression model was obtained and the R
2 value was recorded. In experiments 2 and 3, original data were used, and one-way ANOVA was performed to check for any statistically significant difference between treatments (
P < 0.01). Then, Scott-Knott’s test (
P < 0.05) was performed. For these analyses, the software Assistat® was used.
4. Discussion
In vitro establishment is an important step in tissue culture. It is a critical point where explants come from a different environment and have to adapt to in vitro conditions. One of the key steps in this process is the use of adequate growth regulators and a balance of mineral salts in a suitable concentration. Our results showed a screening comparison of four different cytokinins in eight different concentrations and varying balances of nitrogen salts in ‘Delite’ rabbiteye blueberry, presenting an efficient technique for in vitro plant propagation of this species.
The species and cultivars of the Vaccinium genus show natural variation in in vitro responses. There is high genetic variation in growth regulator responses/needs. Our results, based on linear and quadratic polynomial regression analyses, displayed the effects of cytokinin concentrations and their great impact on the survival of explants, new shoot formation, number of new shoots formed, length of shoots formed, and number of leaves in the shoots.
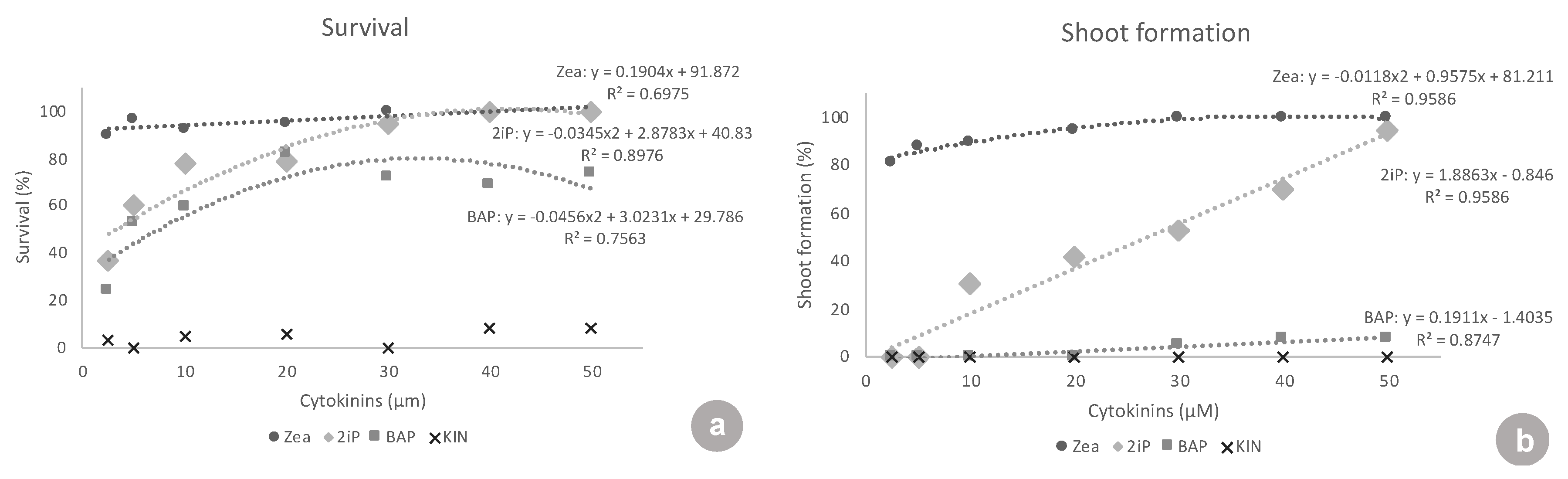
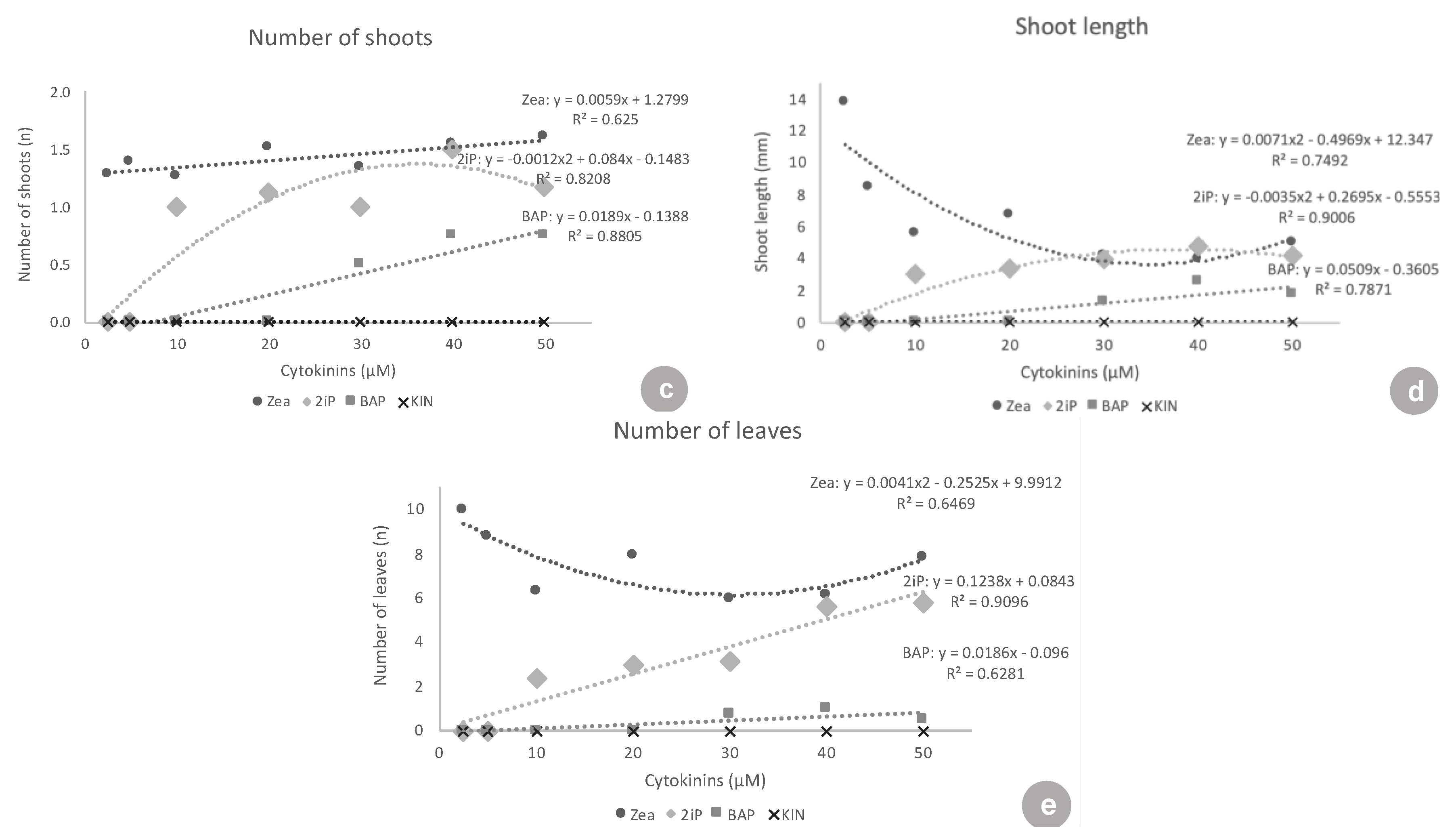
ZEA and 2iP resulted in better responses to in vitro establishment. At the lowest concentrations tested, 2.5 and 5 μM, ZEA was superior to all of the other cytokinins tested, in all the variables analyzed, presenting values of: 89.7 and 96.4% explant survival, 81.3 and 88.2% of explants forming new shoots, 1.3 and 1.4 new shoots formed, 13.8 and 8.4 mm of shoot length, and 10.0 and 8.8 leaves per shoot, respectively. Similar results were observed with highbush blueberry ‘Polaris’ and half-high blueberry ‘St. Cloud’, where ZEA was used at a concentration of 9.1 μM in the shoot establishment in vitro. ZEA was also efficient in inducing shoot proliferation in a liquid medium at 4.6 µM [
10], instead of at higher concentrations. For
V. corymbosum ‘Oskar’,
V. angustifolium ‘Emil’ and ‘Putte’, and
V. corymbosum ×
V. angustifolium ‘Northblue’ establishment, 2 mg L
−1 (9.12 µM) ZEA was used [
20]. In highbush blueberry ‘Duke’ propagation, ZEA at 2 mg L
−1 (9.12 µM) was superior to 2iP or TDZ [
21].
For in vitro shoot proliferation in cranberry (
V. macrocarpon Ait.) cultivars, ZEA at very low concentrations (2–4 µM) showed good results [
22]. In
V. ashei at the multiplication stage, ZEA increased shoot formation compared to 2iP. However, 2iP showed longer shoots with a higher number of nodes [
23]. For initial culture of highbush blueberry, 1 mg L
−1 (2.85 µM) zeatin riboside was used [
24]. In lowbush blueberry, the authors tested 0, 2.3, 4.6, or 9.10 µM ZEA on the elongation of shoots, and concentrations of 2.3 and 4.6 µM gave the best response [
25].
Another important aspect is the growth habit of the ‘Delite’ rabbiteye blueberry cultivar in this study. In particular, in the presence of ZEA and 2iP, it showed a low number of new shoots per explant, but longer shoots, which means that a new subculture could be performed using the nodal segments of the long shoot instead of using new axillary or adventitious shoots formed.
At the lowest concentrations (2.5 and 5 μM), there were no responses to 2iP. Treatments with 2iP started to form shoots only at the concentrations of 10, 20, 30, 40, and 50 μM. Concerning the percentage of explants forming new shoots, 2iP was inferior to ZEA in all of the concentrations, except 50 μM, where both had the same shoot formation rate. This showed that ZEA triggered a response in the explants, even at inferior concentrations (2.5 and 5 μM), and that 2iP was able to lead to some shoot formation only at higher concentrations (10 μM and above). Concerning shoot length, at the concentrations where 2iP started showing new shoots (10–50 μM), the shoots formed were equivalent in length to the shoots formed with ZEA. At concentrations of 10, 20, 30, and 50 μM, both were superior to BAP and KIN. However, when analyzing the number of leaves, ZEA was superior to 2iP at almost all concentrations, except 40 and 50 μM, again demonstrating the need for higher concentrations of 2iP to produce a higher number of leaves. In ‘Brightwell’ blueberry, the authors found that different concentrations of 2iP (5, 10, 15, or 20 mg·L
−1) and TDZ were inferior to 2 mg L
−1 (9.12 µM) ZEA in shoot proliferation [
26]. ZEA at 4 mg·L
−1 (18.24 µM) was more successful than 2iP at 10 or 15 mg·L
−1 (49.2 or 73.8 µM) in establishing
V. corymbosum blueberry cultivars [
12].
BAP did not show any response at the lowest concentrations of 2.5, 5, 10, and 20 μM. BAP only started showing a low response to 30, 40, and 50 μM (5.0–7.8% shoot formation). BAP was always highly inferior to ZEA at all concentrations tested, in all of the variables analyzed, except for the shoot length at 40 μM. Additionally, BAP was inferior to 2iP from 10–50 μM concerning shoot formation, number of shoots, shoot length, and number of leaves. In the same way, in ‘Bluejay’ and ‘Pink Lemonade’ blueberry, the authors found that BAP induced fewer axillary shoots than ZEA, as well as smaller shoots [
27].
Kinetin showed no response concerning shoot formation and had almost no surviving explants (maximum of 8.3% survival), clearly showing that it was not suitable for ‘Delite’ rabbiteye blueberry initiation culture.
In this study, different balances of nitrogen salts were tested. Using the modified WPM medium, no differences were observed among all combinations of nitrogen salts: (NH4)2SO4, KNO3, and Ca(NO3)2·4H2O. ‘Delite’ blueberry showed lower survival (55.7%), shoot formation (55.7%), and shoot length (15.7 mm) in the modified WPM compared with the original WPM (79.4%, 79.4%, and 33.3 mm, respectively).
Using the original WPM, it was observed that treatments containing higher amounts of NH
4NO
3 (1× or 1.5×, instead of 0.5×), as well as the treatment with a higher amount of Ca(NO
3)
2 (1.5×), even with a lower amount of NH
4NO
3 (0.5×), showed the same performance as in WPM without modification. Similarly, in red raspberries, it was found that combinations of intermediate to high NO
3- and intermediate to high NH
4+ developed the most growth in most cultivars [
28].
However, changing the ranges of Ca(NO3)2, in addition to increasing or decreasing the total amount of nitrogen and its nitrate form, would also change the Ca+2 ion. Therefore, the result seen in the treatment Ca(NO3)2 (1.5×) could be related to either nitrogen or calcium in higher amounts, or even both.
This study in a rabbiteye blueberry cultivar represents a basic framework that can be used to understand initial in vitro establishment. It can be useful to describe this process in other Vaccinium cultivars regarding the adjustments necessary to adapt the process to different genotypes.
5. Conclusions
This research showed the effects of different cytokinins at different concentrations and different nitrogen salt ranges on ‘Delite’ rabbiteye blueberry during in vitro establishment, and it provides basic knowledge for further experiments in rabbiteye blueberry tissue culture.
In conclusion, focusing on an efficient strategy for in vitro establishment of ‘Delite’ rabbiteye blueberry, we recommend the lowest concentration tested, 2.5 μM ZEA, which promoted a high survival rate (89.7%), as well as a good response on explants forming new shoots (81.3%). This concentration yielded a number of 1.3 new shoots, a high shoot length (13.8 mm), and 10.0 leaves per shoot. Concerning salt composition, we recommend the original WPM. An increase or decrease in the NH4NO3 and Ca(NO3)2 concentration did not promote better growth than the original medium.
This work is of interest for evaluating different cytokinin and salt compositions in the culture medium for in vitro establishment of rabbiteye blueberry, and it can contribute to developing a deeper knowledge of large-scale propagation, germplasm conservation, and development of other biotechnology techniques in other research fields, such as morphology, plant breeding, and physiology.
Future studies could be developed beyond the research presented here, focusing on fine-tuning the salts composition and concentrations of the growth regulator needed for an efficient response, as well as combining the two most successful cytokinins tested, ZEA and 2iP.