The Effect of Environment and Nutrients on Hydroponic Lettuce Yield, Quality, and Phytonutrients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Culture and Harvest
2.2. Flavonoid Analysis
2.3. Mineral Composition
2.4. Statistical Analysis
3. Results
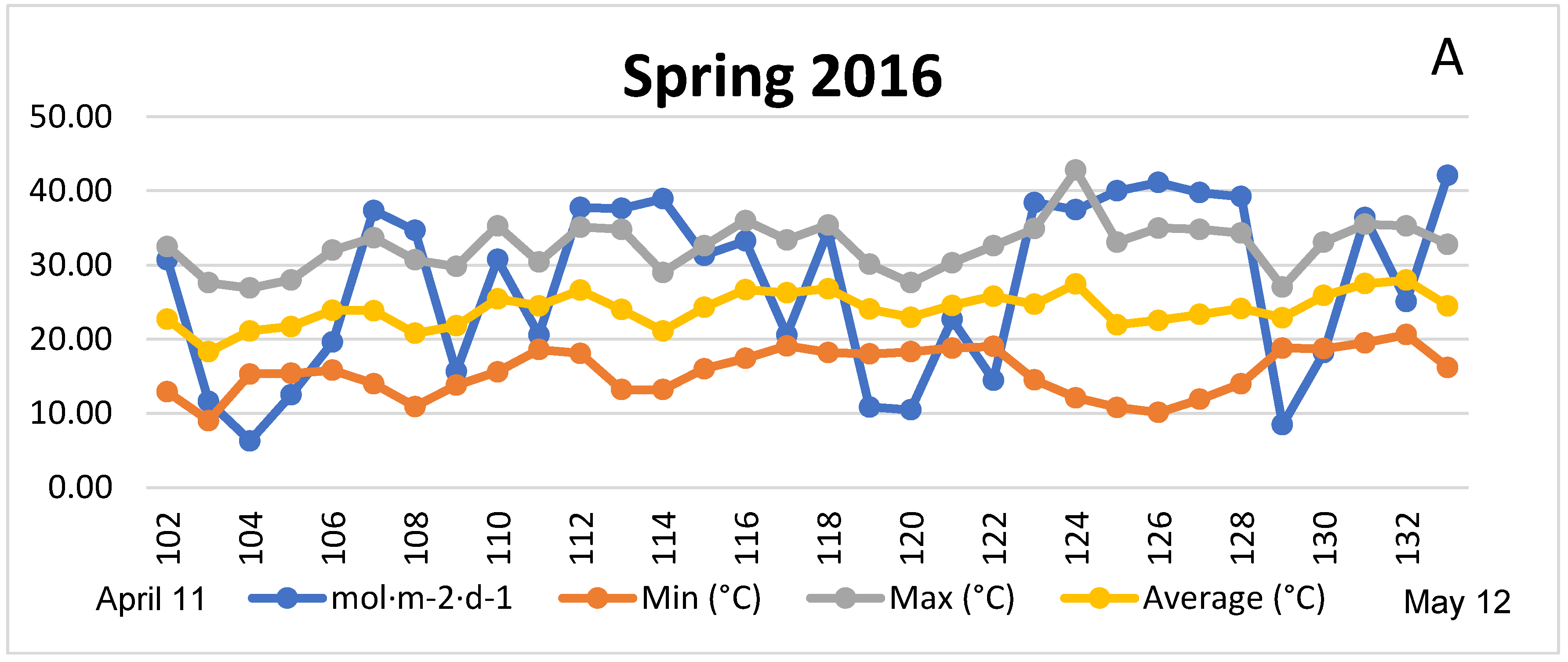
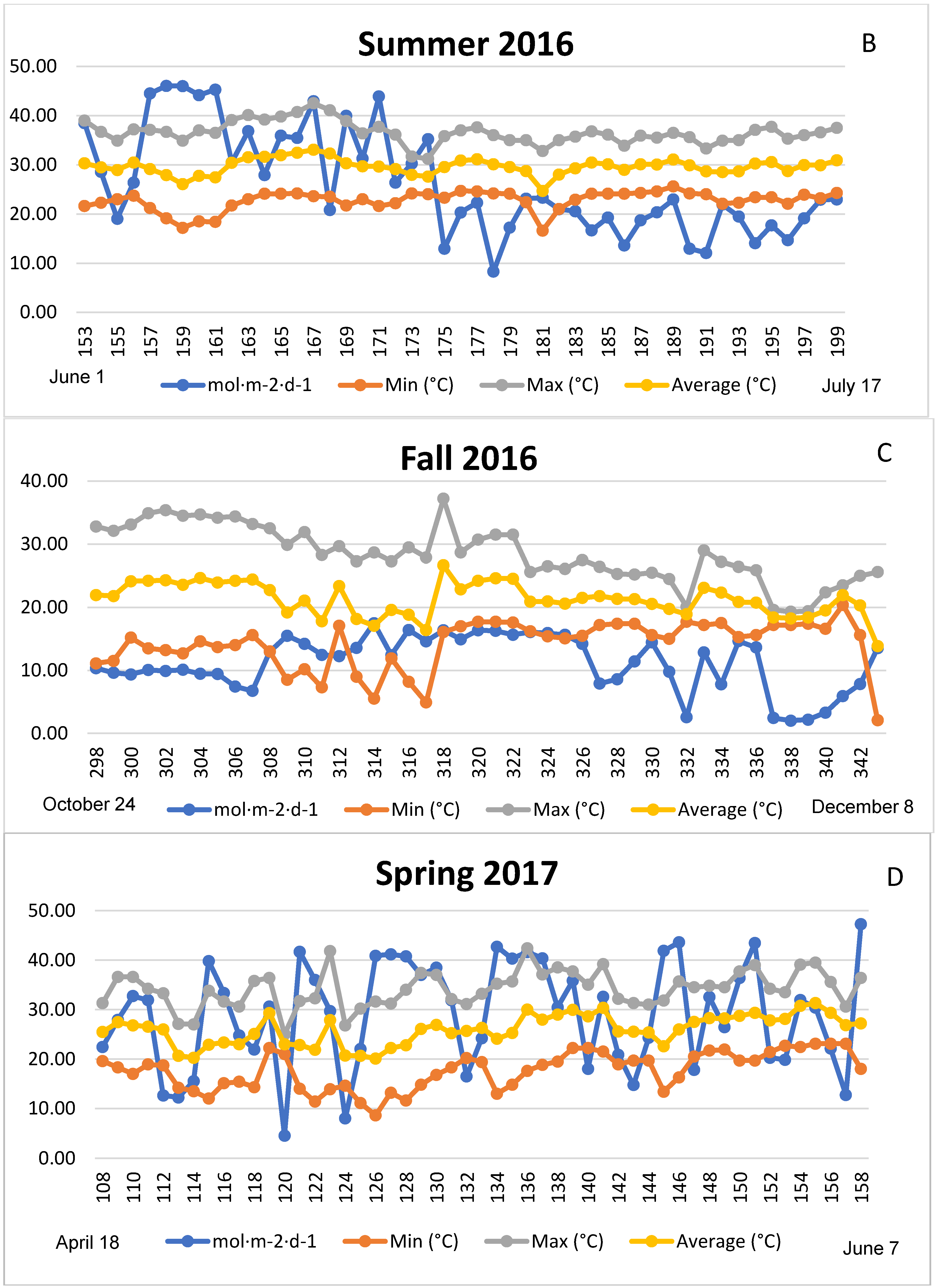
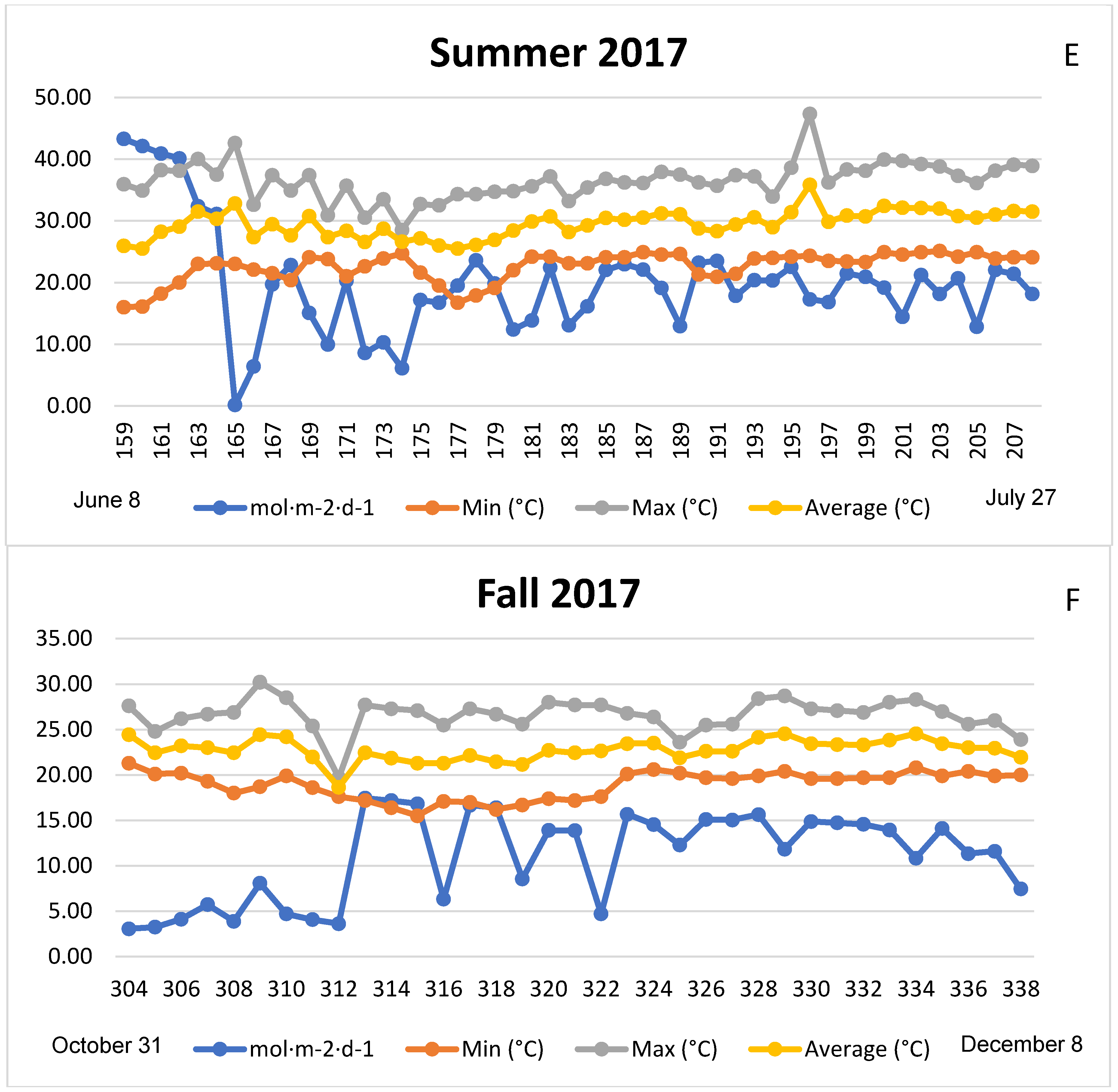
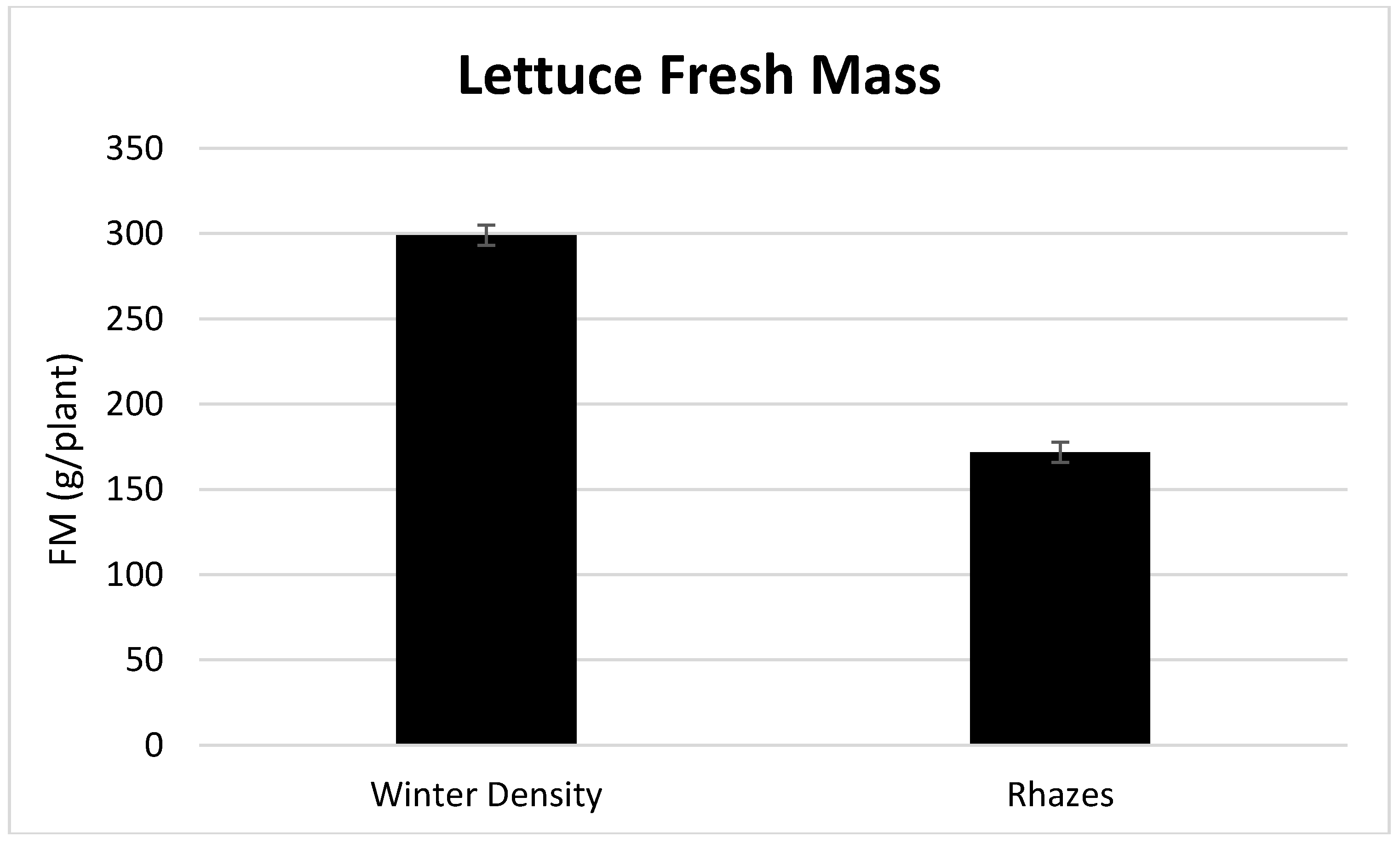
3.1. Season, Cultivar, and Treatment Effects on Plant Growth and Biomass Production
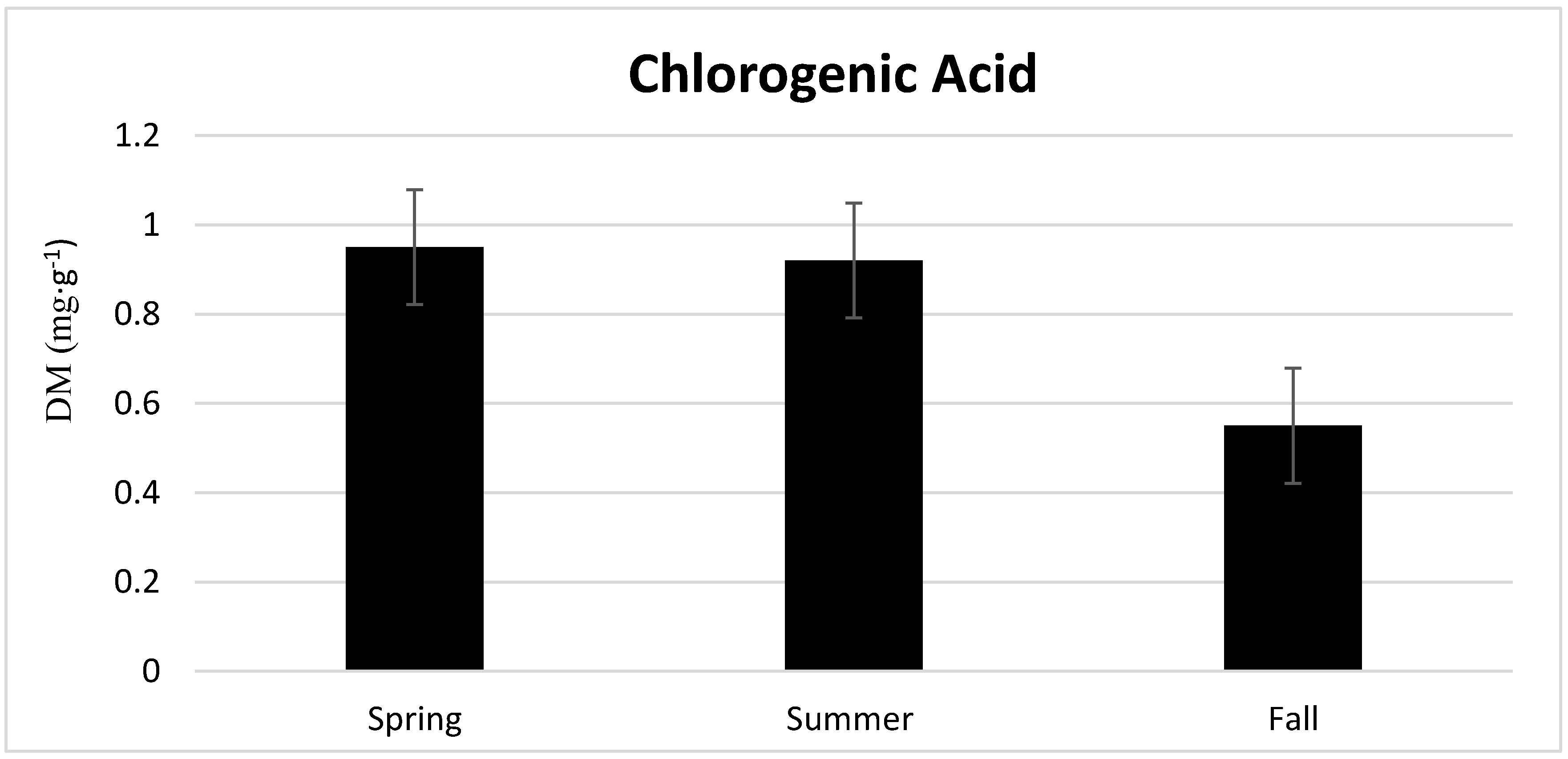
3.2. Season, Cultivar, and Treatment Effect on Lettuce Quality
3.3. Season, Cultivar, and Treatment Effects on Leaf Mineral Content
4. Discussion
4.1. Season, Cultivar, and Treatment Effect on Plant Growth and Biomass Production
4.2. Season, Cultivar, and Treatment Effect on Lettuce Quality
4.3. Season, Cultivar, and Treatment Effects on Leaf Mineral Content
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Romani, A.; Pinelli, P.; Galardi, C.; Sani, G.; Cimato, A.; Heimler, D. Polyphenols in Greenhouse and Open-Air-Grown Lettuce. Food Chem. 2002, 79, 337–342. [Google Scholar] [CrossRef]
- Husain, S.R.; Cilurd, J.; Cillard, P. Hydroxyl radical scavenging activity of Flavonoids. Phytochemistry 1987, 26, 2489–2491. [Google Scholar] [CrossRef]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Phenolic Compounds in Brassica Vegetables. Molecules 2011, 16, 251–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gruda, N. Impact of environmental factors on product quality of greenhouse vegetables for fresh consumption. Crit. Rev. Plant Sci. 2005, 24, 227–247. [Google Scholar] [CrossRef]
- Jensen, M. Hydroponics Worldwide. Acta Hortic. 1999, 481, 719–729. [Google Scholar] [CrossRef]
- Zbeetnoff, C. The North American Greenhouse Vegetable Industry. Farm Credit Canada and AgriSuccess; Agro-Environmental Consulting: White Rock, BC, Canada, 2006. [Google Scholar]
- Rappaport, L.; Wittwer, S.H. Night temperature and photoperiod effects on flowering of leaf lettuce. Proc. Am. Soc. Hortic. Sci. 1956, 68, 279–282. [Google Scholar]
- Jenni, S. Rib Discoloration: A Physiological Disorder Induced by Heat Stress in Crisphead Lettuce. HortScience 2005, 40, 2031–2035. [Google Scholar]
- Wissemeier, A.H.; Zühlke, G. Relation between climatic variables, growth and the incidence of tipburn in field-grown lettuce as evaluated by simple, partial and multiple regression analysis. Sci. Hortic. 2002, 93, 193–204. [Google Scholar] [CrossRef]
- Zanin, G.; Ponchia, G.; Sambo, P. Yield and quality of vegetables grown in a floating system for readyto-eat produce. Acta Hortic. 2009, 807, 433–438. [Google Scholar] [CrossRef]
- Scuderi, D.; Restuccia, C.; Chisari, M.; Barbagallo, R.N.; Caggia, C.; Giuffrida, F. Salinity of nutrient solution influences the shelf-life of fresh-cut lettuce grown in floating system. Postharvest Biol. Technol. 2011, 59, 132–137. [Google Scholar] [CrossRef]
- Abou-Hadid, A.F.; Abd-Elmoniem, E.M.; El-Shinawy, M.Z.; Abou-Elsoud, M. Electrical conductivity effect on growth and mineral composition of lettuce plants in hydroponic system. Acta Hortic. 1996, 434, 59–66. [Google Scholar] [CrossRef]
- Sgherri, C.; Perez-Lopez, U.; Micaelli, F.; Miranda-Apodaca, J.; Mena-Petite, A.; Munoz-Rueda, A.; Quartacci, M.F. Elevated CO2 and salinity are responsible for phenolics-enrichment in two differently pigmented lettuces. Plant Physiol. Biochem. 2017, 115, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Fonseca, J.M.; Choi, J.H.; Kubota, C.; Dae, Y.K. Salt in irrigation water affects the nutritional and visual properties of romaine lettuce (Lactuca sativa L.). J. Agric. Food Chem. 2008, 56, 3772–3776. [Google Scholar] [CrossRef] [PubMed]
- Neocleous, D.; Koukounaras, A.; Siomos, A.S.; Vasilakakis, M. Assessing the salinity effects on mineral composition and nutritional quality of green and red ‘baby’ lettuce. J. Food Qual. 2014, 37, 1–8. [Google Scholar] [CrossRef]
- Fallovo, C.; Rouphael, Y.; Cardarelli, M.; Rea, E.; Battistelli, A.; Colla, G. Yield and quality of leafy lettuce in response to nutrient solution composition and growing season. J. Food Agric. Environ. 2009, 7, 456–462. [Google Scholar]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Calif. Agric. Exp. Stn. Circ. 1950, 347, 1–32. [Google Scholar]
- Neugart, S.; Zietz, M.; Schreiner, M.; Rohn, S.; Kroh, L.W.; Krumbein, A. Structurally different flavonol glycosides and hydroxycinnamic acid derivatives respond differently to moderate UV-B radiation exposure. Physiol. Plant. 2012, 145, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Becker, C.; Kläring, H.P.; Kroh, L.W.; Krumbein, A. Temporary reduction of radiation does not permanently reduce flavonoid glycosides and phenolic acids in red lettuce. Plant Physiol. Biochem. 2013, 72, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Barickman, T.C.; Kopsell, D.A.; Sams, C.E. Selenium influences glucosinolate and isothiocyanates and increases sulfur uptake in Arabidopsis thaliana and rapid-cycling Brassica oleracea. J. Agric. Food. Chem. 2013, 61, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Li, H.; Yu, J.; Liu, H.; Cao, Z.; Manukovsky, N.S.; Liu, H. Interaction effects of light intensity and nitrogen concentration on growth, photosynthetic characteristics and quality of lettuce (Lactuca sativa L. Var. youmaicai). Sci. Hortic. 2017, 214, 51–57. [Google Scholar] [CrossRef]
- Gaudreau, L.; Charbonneau, J.; Canda, A.; Gv, Q. Photoperiod and Photosynthetic Photon Flux Influence Growth and Quality of Greenhouse-grown Lettuce. HortScience 1994, 29, 1285–1289. [Google Scholar]
- Glenn, E.P. Seasonal effects of radiation and temperature on growth of greenhouse lettuce in a high insolation desert environment. Sci. Hortic. 1984, 22, 9–21. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G. Growth, yield, fruit quality and nutrient uptake of hydroponically cultivated zucchini squash as affected by irrigation systems and growing seasons. Sci. Hortic. 2005, 105, 177–195. [Google Scholar] [CrossRef]
- Bres, W.; Weston, L. A Nutrient Accumulation and Tipburn in NFT-grown Lettuce at Several Potassium and pH Levels. HortScience 1992, 27, 790–792. [Google Scholar]
- Jenni, S.; Truco, M.J.; Michelmore, R.W. Quantitative trait loci associated with tipburn, heat stress-induced physiological disorders, and maturity traits in crisphead lettuce. Theor. Appl. Genet. 2013, 126, 3065–3079. [Google Scholar] [CrossRef] [PubMed]
- Bunning, M.L.; Kendall, P.A.; Stone, M.B.; Stonaker, F.H.; Stushnoff, C. Effects of Seasonal Variation on Sensory Properties and Total Phenolic Content of 5 Lettuce Cultivars. J. Food Sci. 2010, 75, 156–161. [Google Scholar] [CrossRef] [PubMed]
- García-Macías, P.; Ordidge, M.; Vysini, E.; Waroonphan, S.; Battery, N.H.; Gordon, M.H.; Hadley, P.; John, P.; Lovegrove, J.A.; Wagstaffe, A. Changes in the flavonoid and phenolic acid contents and antioxidant activity of red leaf lettuce (Lollo Rosso) due to cultivation under plastic films varying in ultraviolet transparency. J. Agric. Food Chem. 2007, 55, 10168–10172. [Google Scholar] [CrossRef] [PubMed]
- Oh, M.; Carey, E.E.; Rajashekar, C.B. Plant Physiology and Biochemistry Environmental stresses induce health-promoting phytochemicals in lettuce. Plant Physiol. Biochem. 2009, 47, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Barickman, T.C.; Horgan, T.E.; Wheeler, J.R.; Sams, C.E. Elevated Levels of Potassium in Greenhouse-grown Red Romaine Lettuce Impacts Mineral Nutrient and Soluble Sugar Concentrations. HortScience 2016, 51, 504–509. [Google Scholar]
- Hu, Y.; Schmidhalter, U. Drought and salinity: A comparison of their effects on mineral nutrition of plants. J. Plant Nutr. Soil Sci. 2005, 168, 541–549. [Google Scholar] [CrossRef]
- Anderson, T.S.; Martini, M.R.; de Villiers, D.; Timmons, M.B. Growth and Tissue Elemental Composition Response of Butterhead Lettuce (Lactuca sativa, cv. Flandria) to Hydroponic Conditions at Different pH and Alkalinity. Horticulturae 2017, 3, 41. [Google Scholar] [CrossRef]

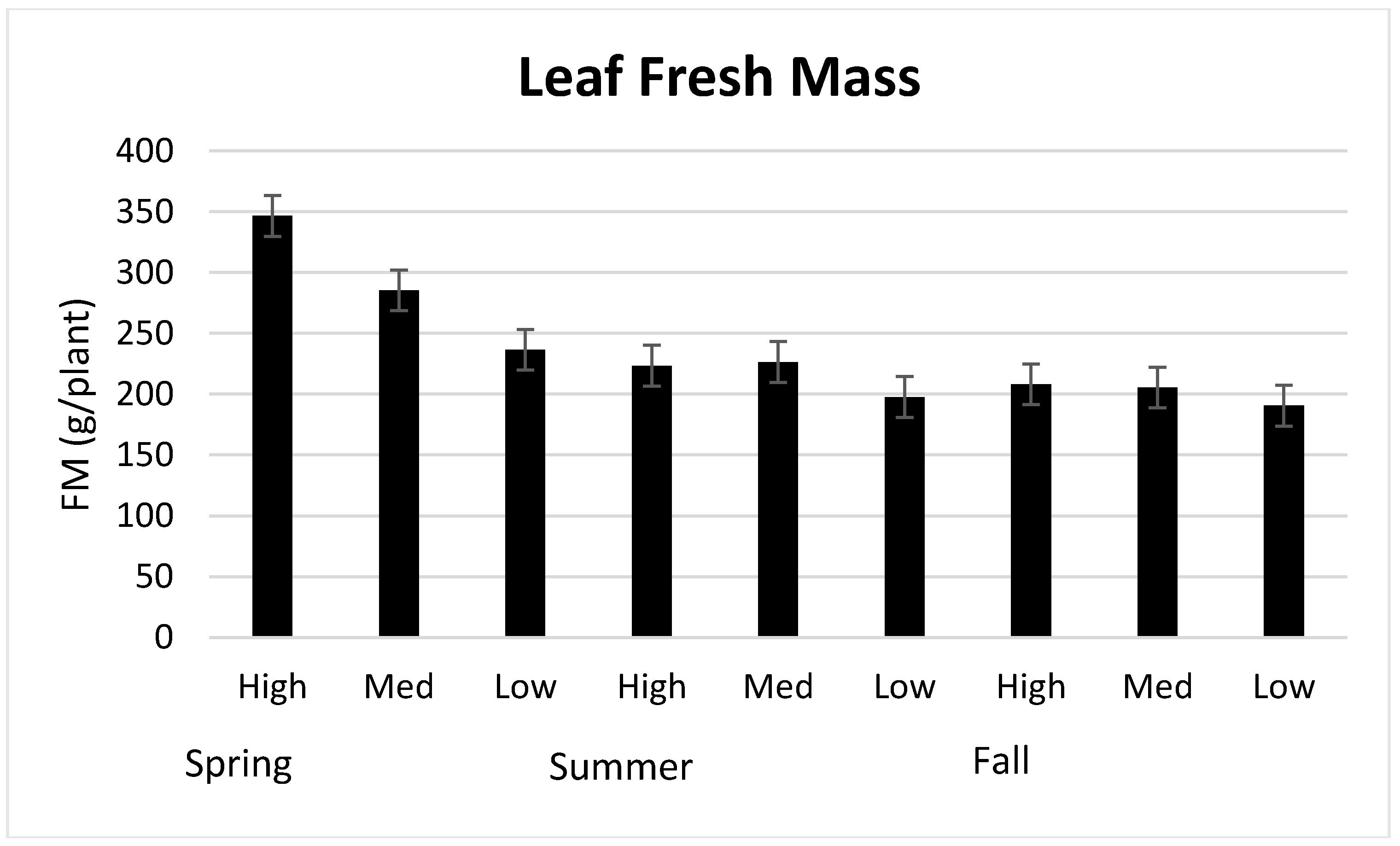
| Treatments | Leaf DM (g) | DM:FM (g) a | Leaf Water % |
|---|---|---|---|
| Spring | 12.37 a | 0.0413 b | 95.86 a |
| Summer | 9.97 b | 0.0460 a | 95.41 b |
| Fall | 8.26 c | 0.0410 b | 95.92 a |
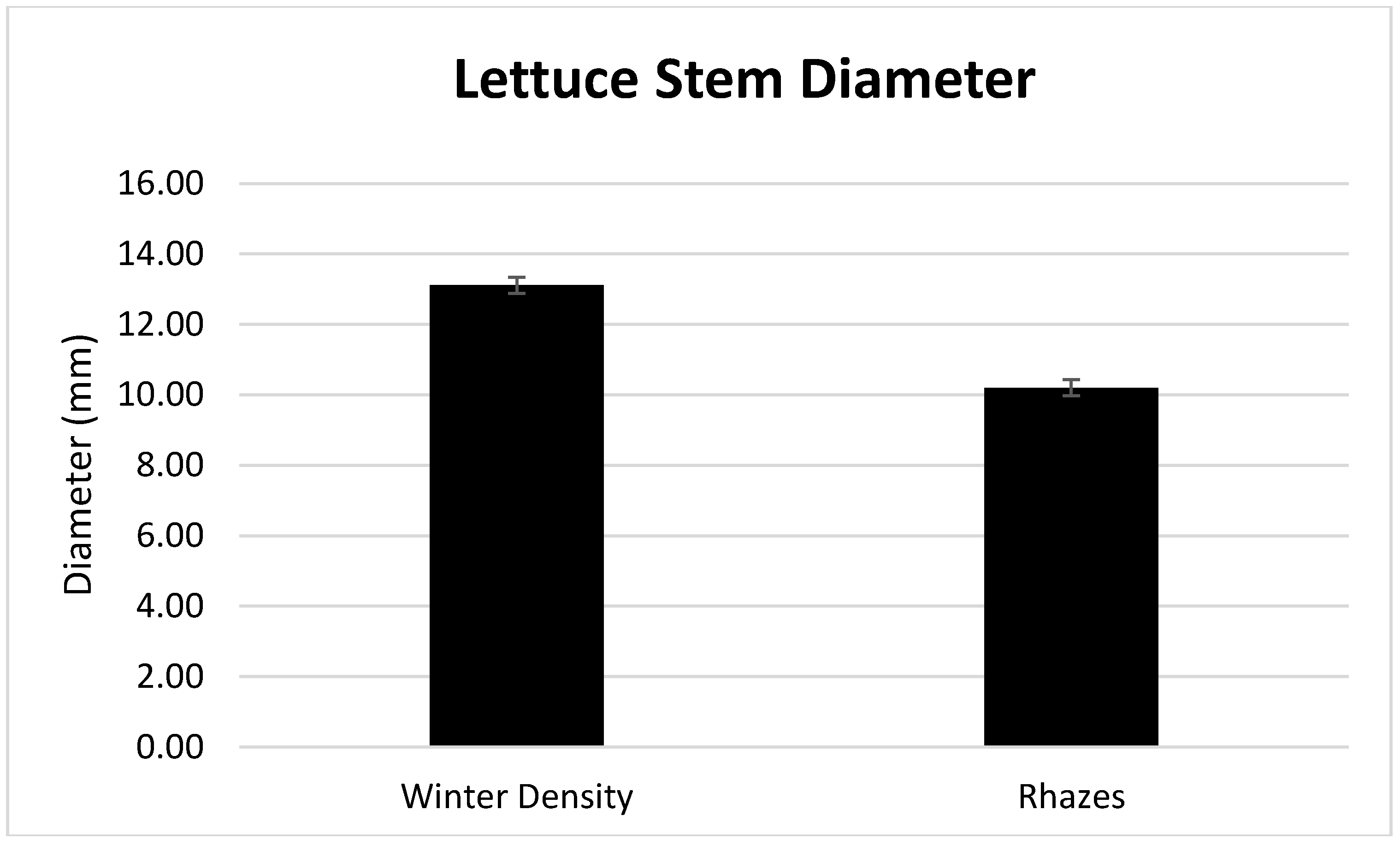
| Winter Density | 13.36 a | 0.0444 a | 95.56 b |
| Rhazes | 7.04 b | 0.0410 b | 95.90 a |
| High b | 11.44 a | 0.0433 ab | 95.61 b |
| Med | 9.77 b | 0.0408 b | 95.93 a |
| Low | 9.38 b | 0.0440 a | 95.64 b |
| P-Value c,d | |||
| Season | *** | ** | *** |
| Cultivar | *** | ** | ** |
| Electro-Conductivity | ** | ns | * |
| Season | Cultivar | Root FM (g) | Root DM (g) | Root DM:FM (g) a | Root Water % |
|---|---|---|---|---|---|
| Spring | Winter Density | 60.08 a | 2.42 a | 0.04 c | 0.96 a |
| Summer | Winter Density | 30.00 b | 1.47 b | 0.05 b | 0.95 b |
| Fall | Winter Density | 23.73 c | 1.22 c | 0.05 b | 0.95 b |
| Spring | Rhazes | 31.11 b | 1.32 bc | 0.05 b | 0.95 b |
| Summer | Rhazes | 12.28 d | 0.79 d | 0.06 a | 0.94 c |
| Fall | Rhazes | 13.88 d | 0.68 d | 0.05 b | 0.95 b |
| P-Value b,c | *** | ** | ** | ** |
| Season | Cultivar | Concentrations of Phenolics and Flavonoids (mg·g−1 DM) a,b | |||||
|---|---|---|---|---|---|---|---|
| chlo | chic | qgluc | qglucor | luteolin | qmal | ||
| Spring | Winter Density | 0.83 b | 11.34 d | 0.87 b | 1.01 c | 1.15 d | 3.56 d |
| Summer | Winter Density | 0.95 ab | 15.26 d | 1.51 b | 0.55 c | 0.91 d | 4.63 d |
| Fall | Winter Density | 0.52 c | 33.85 b | 1.05 b | 1.20 c | 1.56 d | 5.93 cd |
| Spring | Rhazes | 1.08 a | 23.79 c | 3.85 a | 9.25 a | 9.86 a | 15.33 bc |
| Summer | Rhazes | 0.90 ab | 28.31 bc | 3.98 a | 5.19 b | 5.40 c | 18.93 b |
| Fall | Rhazes | 0.58 c | 65.52 a | 1.34 b | 6.50 b | 7.73 b | 46.90 a |
| P-Value c | ns | ** | * | * | * | *** | |
| Elemental Nutrient Concentrations a | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Season | Cultivar | (mg·g−1) (µg·g−1) | ||||||||||
| Mg | P | S | K | Ca | Fe | B | Mn | Cu | Zn | Mo | ||
| Spring | Winter Density | 5.30 a | 6.22 bc | 5.22 a | 48.73 c | 18.02 a | 120.53 abc | 37.16 a | 58.31 ab | 4.56 a | 22.39 b | 0.97 a |
| Summer | Winter Density | 4.44 b | 6.46 b | 5.83 a | 46.26 c | 14.89 b | 138.57 a | 28.77 b | 40.59 b | 2.85 b | 23.58 b | 0.78 b |
| Fall | Winter Density | 4.11 bc | 6.49 b | 1.36 b | 48.87 c | 13.08 c | 108.08 bc | 30.50 b | 47.20 b | 2.83 b | 24.81 b | 0.51 c |
| Spring | Rhazes | 3.34 e | 5.66 c | 4.39 a | 48.13 c | 13.83 bc | 98.68 c | 34.43 a | 40.55 b | 5.37 a | 22.90 b | 0.64 bc |
| Summer | Rhazes | 3.73 cd | 6.68 b | 5.86 a | 53.56 b | 14.12 bc | 128.74 ab | 30.02 b | 55.39 ab | 3.04 b | 28.36 ab | 0.54 c |
| Fall | Rhazes | 3.62 de | 8.73 a | 1.32 b | 57.60 a | 13.73 bc | 127.34 ab | 37.22 a | 74.98 a | 5.25 a | 32.53 a | 0.51 c |
| P-Value b,c | *** | *** | ns | ** | *** | ns | ** | * | ns | ns | * | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sublett, W.L.; Barickman, T.C.; Sams, C.E. The Effect of Environment and Nutrients on Hydroponic Lettuce Yield, Quality, and Phytonutrients. Horticulturae 2018, 4, 48. https://doi.org/10.3390/horticulturae4040048
Sublett WL, Barickman TC, Sams CE. The Effect of Environment and Nutrients on Hydroponic Lettuce Yield, Quality, and Phytonutrients. Horticulturae. 2018; 4(4):48. https://doi.org/10.3390/horticulturae4040048
Chicago/Turabian StyleSublett, William L., T. Casey Barickman, and Carl E. Sams. 2018. "The Effect of Environment and Nutrients on Hydroponic Lettuce Yield, Quality, and Phytonutrients" Horticulturae 4, no. 4: 48. https://doi.org/10.3390/horticulturae4040048
APA StyleSublett, W. L., Barickman, T. C., & Sams, C. E. (2018). The Effect of Environment and Nutrients on Hydroponic Lettuce Yield, Quality, and Phytonutrients. Horticulturae, 4(4), 48. https://doi.org/10.3390/horticulturae4040048






