Performance of Two Citrus Species Grafted to Different Rootstocks in the Presence of Huanglongbing Disease in Puerto Rico
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Area and Description
2.2. Variables Measured
2.3. Statistical Analysis
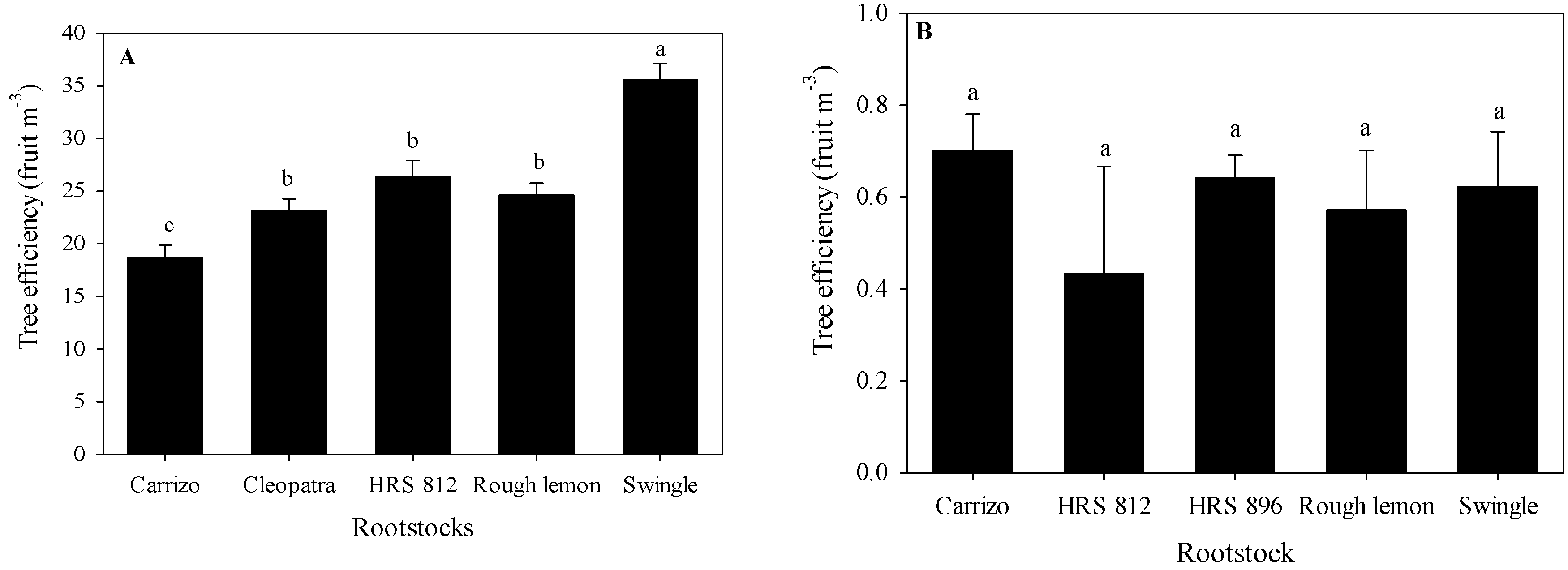
3. Results
3.1. Soil Chemical Properties
3.2. Tissue Nutrient and Trace Element Concentrations
3.3. Tree Response
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chung, K.R.; Brlansky, R.H. Citrus Diseases Exotic to Florida: Huanglongbing (Citrus Greening); Plant Pathology Department Fact Sheet PP-210; Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida: Gainesville, FL, USA, 2005. [Google Scholar]
- Batool, A.; Iftikhar, Y.; Mughal, S.M.; Khan, M.M.; Jaskani, M.J.; Abbas, M.; Khan, I.A. Citrus Greening Disease–A major cause of citrus decline in the world–A Review. Hort. Sci. (Prague) 2007, 34, 159–166. [Google Scholar] [CrossRef]
- Halbert, S.E.; Núñez, C.A. Distribution of the Asian citrus psyllid, Diaphorina citri Kuwayama (Rhynchota: Psyllidae) in the Caribbean basin. Fla. Entomol. 2004, 87, 401–402. [Google Scholar] [CrossRef]
- Alvarado Ortíz, A.N.; Estévez de Jensen, C.; Abreu, E.; Román, F.; Almodóvar, W. “Citrus Greening” en Puerto Rico: Muestreo Preliminar. Sociedad Puertorriqueña de Ciencias Agrícolas; Memorias de la Reunión Annual: Hato Rey, PR, USA, 2010; p. 32. [Google Scholar]
- Estévez de Jensen, C.; Vitoreli, A.; Román, F. Citrus greening in commercial orchards in Puerto Rico. Phytopathology 2010, 100, S34. [Google Scholar]
- Marroquiín-Guzmán, M.R.; Estévez de Jensen, C. Dissemination of Citrus greening in Puerto Rico. J. Agric. Univ. P. R. 2013, 97, 119–134. [Google Scholar]
- Román-Pérez, F.M.; González-Vélez, A.; Pagán, J.; Estévez de Jensen, C.; Rosa, E. New Approaches in the Production of Citrus in Puerto Rico; Abstracts Caribbean Food Crops Society: St. Thomas, VI, USA, 2014; Volume 50, p. 59. [Google Scholar]
- Méndez, J. Mensaje del Presidente; Décimo Cuarta Asamblea Annual; Productores de cítricas de la montaña, Inc.: Lares, PR, USA, 2016. [Google Scholar]
- Ingreso Bruto Agrícola; Departamento de Agricultura, División estadísticas agrícolas: Estado Libre Asociado de Puerto Rico, USA, 2015.
- Arce, S.C.; Rivera, D. New Media Components and Fertilization to Accelerate the Growth of Citrus Rootstocks Grown in a Greenhouse. Horticulturae 2018, 4, 10. [Google Scholar] [CrossRef]
- Zamora-Echevarría, J.L. Manejo Nutricional del “Citrus greening” y su Costo; El Frutal: Mayagüez, PR, USA, 2013; Volume 9, No. 1. [Google Scholar]
- Castle, W.S.; Tucker, D.P.H.; Krezdorn, A.H.; Youtsey, C.O. Rootstocks for Florida Citrus, 2nd ed.; University of Florida Institute of Food and Agricultural Sciences: Gainesville, FL, USA, 1993; 92p. [Google Scholar]
- Román-Pérez, F.M.; González-Vélez, A. Liberación de los patrones de cítricas “Swingle Citrumelo”, “Carrizo” y “HRS 812” para Puerto Rico. J. Agric. Univ. P. R. 2013, 97, 101–106. [Google Scholar]
- Román-Pérez, F.M.; González-Vélez, A.; Macchiavelli, R. Efecto de cuatro patrones en la producción y calidad de la china “Hamlin” (Citrus sinensis [L.] Osb.) en tres localidades de Puerto Rico. J. Agric. Univ. P. R. 2011, 95, 25–34. [Google Scholar]
- Bowman, K.D.; Rouse, R.E. US-812 Citrus Rootstock. HortScience 2006, 41, 832–836. [Google Scholar]
- Albrecht, U.; Bowman, K.D. Tolerance of trifoliate citrus hybrids to Candidatus Liberibacter asiaticus. Sci. Hortic. 2012, 147, 71–80. [Google Scholar] [CrossRef]
- Bové, J.M. Huanglongbing: A destructive, newly emerging, century-old disease of citrus. J. Plant Pathol. 2006, 88, 7–37. [Google Scholar]
- Spann, T.M.; Atwood, R.A.; Yates, J.D.; Rogers, M.E.; Brlansky, R.H. Dooryard Citrus Production: Citrus Greening Disease; Institute of Food and Agricultural Sciences, University of Florida: Gainesville, FL, USA, 2010; EDIS HS1131. [Google Scholar]
- Gottwald, T.R.; Graham, J.H.; Irey, M.S.; Mc Collum, T.G.; Wood, B.W. Inconsequential effect of nutritional treatments on Huanglongbing control, fruit quality, bacterial titer and disease progress. Crop Prot. 2012, 36, 73–82. [Google Scholar] [CrossRef]
- Morris, A.; Muraro, R. Economic Evaluation of Citrus Greening Management and Control Strategies; Florida Cooperative Extension Service, University of Florida: Gainesville, FL, USA, 2008; EDIS, Doc. FE712; p. 7. [Google Scholar]
- Beinroth, F.; Engel, R.; Lugo, J.; Santiago, C.; Ríos, S.; Brannon, G. Updated Taxonomic Classification of the Soils of Puerto Rico; Bulletin 303; University of Puerto Rico, Mayagüez Campus, College of Agricultural Sciences Agricultural Experiment Station: San Juan, PR, USA, 2002; 38p. [Google Scholar]
- Román-Pérez, F.M.; González-Vélez, A.; Macchiavelli, R.; Estevez de Jensen, C. Comportamiento de la lima Tahití (Citrus Latifolia Tan) en cinco patrones y dos zonas productoras de Puerto Rico. J. Agric. Univ. P. R. 2017, 101, 225–236. [Google Scholar]
- Estación Experimental Agrícola. Conjunto Tecnológico Para la Producción de Cítricas; UPR-Estación Experimental Agrícola: Lajas, PR, USA, 1987; Publicación 113. [Google Scholar]
- Rouse, B. Rehabilitation of HLB Infected Citrus trees using severe pruning and nutritional sprays. Proc. Fla. State Hortic. Soc. 2013, 126, 51–54. [Google Scholar]
- Thomas, G.W. Soil pH and soil acidity. In Methods of Soil Analysis, Part 3-Chemical Methods; Sparks, D.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1996; pp. 475–490. [Google Scholar]
- Warncke, D.; Brown, J.R. Potassium and other basic cations. In Recommended Chemical Soil Test Procedures for North Central Region; NCR Publication No. 221; Missouri Agricultural Station: Columbia, MO, USA, 1998; pp. 31–33. [Google Scholar]
- Jackson, M.L. Soil Chemical Analysis; Prentice-Hall: Englewood Cliffs, NJ, USA, 1958. [Google Scholar]
- Hossner, L.R. Dissolution for total elemental analysis. In Methods of Soil Analysis, Part 3—Chemical Methods; Sparks, D.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1996; pp. 49–64. [Google Scholar]
- Fallahi, E.; Mousavi, Z. Performance of Orlando tangelo trees on ten rootstocks in Arizona. J. Am. Soc. Hortic. Sci. 1991, 116, 2–5. [Google Scholar]
- Junior, D.; Quaggio, J.; Cantallera, H.; Marcelli, R.; Bachiega, F. Nutrient Management for High Citrus Yield in Tropical Soils. Better Crops 2012, 96, 4–7. [Google Scholar]
- Lautner, S.; Fromm, J. Calcium-dependent physiological processes in trees. Plant Biol. 2010, 12, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Mills, H.A.; Jones, J.B., Jr. Plant Analysis Hanbook II; Micro-Macro Pub.: Athens, GA, USA, 1996. [Google Scholar]
- Sechler, A.; Schuenzel, E.L.; Cooke, P.; Donnua, S.; Thaveechai, N.; Postnikova, E.; Schaad, N.W. Cultivation of ‘Candidatus Liberibacter asiaticus’, ‘Ca. L. africanus’, and ‘Ca. L. americanus’ associated with Huanglongbing. Phytopathology 2009, 99, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, U.; McCollum, G.; Bowman, K.D. Influence of rootstock variety on Huanglongbing disease development in field-grown sweet orange (Citrus sinensis [L.] Osbeck) trees. Sci. Hortic. 2012, 138, 210–220. [Google Scholar] [CrossRef]
- Piña-Dumoulin, G.; Laborem, E.G.; Monteverde, E.; Magaña-Lemus, S.; Espinoza, M.; Rangel, L. Growth, Production and Fruit Quality of Persian Lime on 11 Rootstocks. Agron. Trop. 2006, 56, 434–449. [Google Scholar]
| Scion | Rootstocks | OM z | pH | Ca | K | Mg | Na | P | S | NO3-N |
|---|---|---|---|---|---|---|---|---|---|---|
| -%- | -1:1- | ------------------------- mg kg−1 --------------------- | -ppm- | |||||||
| Tahiti lime | Carrizo | 5.93 | 5.18 | 485 | 184 | 90.8 ay | 16.8 a | 15.6 | 80.3 c | 11.0 b |
| Cleopatra | 5.40 | 5.00 | 521 | 258 | 74.0 b | 13.8 ab | 14.0 | 139 a | 28.5 a | |
| - | HRS 812 | 5.58 | 4.85 | 360 | 230 | 54.3 c | 11.3 b | 21.0 | 146 a | 20.0 a |
| - | Rough lemon | 5.40 | 5.18 | 534 | 189 | 69.5 b | 11.5 b | 15.0 | 105 b | 10.5 b |
| - | Swingle | 5.53 | 5.30 | 485 | 218 | 76.0 b | 12.3 ab | 15.5 | 82.3 c | 9.0 b |
| - | Pr > F | 0.60 | 0.76 | 0.65 | 0.50 | 0.09 | 0.04 | 0.39 | 0.08 | 0.03 |
| Nova mandarin | Carrizo | 5.05 a | 4.70 | 486 | 272 | 77.8 | 10.0 b | 22.0 | 86.0 | 14.5 |
| HRS 812 | 4.65 b | 4.95 | 501 | 206 | 81.0 | 10.5 ab | 13.5 | 83.5 | 16.5 | |
| - | HRS 896 | 4.55 b | 4.83 | 506 | 240 | 84.5 | 14.8 a | 19.3 | 62.8 | 15.3 |
| - | Rough lemon | 4.53 b | 4.80 | 432 | 289 | 81.8 | 10.5 ab | 12.3 | 104 | 20.5 |
| - | Swingle | 4.58 b | 4.93 | 502 | 232 | 81.5 | 11.3 ab | 13.8 | 69.8 | 14.5 |
| Pr > F | 0.09 | 0.85 | 0.90 | 0.13 | 0.99 | 0.09 | 0.57 | 0.66 | 0.64 | |
| Scion | Rootstocks | N z | Ca | K | Mg | Na | P | S | Mn |
|---|---|---|---|---|---|---|---|---|---|
| - | - | ---%--- | --------------------------------------- mg kg−1 ------------------------------------ | ||||||
| Tahiti | Carrizo | 2.65 aby | 3.16 | 1.56 b | 0.360 ab | 0.030 | 0.158 | 0.255 | 48.3 b |
| lime | Cleopatra | 2.48 b | 3.04 | 1.59 b | 0.383 a | 0.030 | 0.138 | 0.223 | 101 a |
| - | HRS 812 | 2.67 ab | 3.07 | 1.60 b | 0.325 abc | 0.038 | 0.155 | 0.268 | 66.0 ab |
| - | Rough lemon | 2.49 b | 3.06 | 1.83 a | 0.303 bc | 0.028 | 0.150 | 0.213 | 74.8 ab |
| - | Swingle | 2.88 a | 2.74 | 1.76 a | 0.275 c | 0.033 | 0.160 | 0.245 | 71.8 ab |
| - | Pr > F | 0.05 | 0.64 | 0.09 | 0.003 | 0.29 | 0.50 | 0.51 | 0.03 |
| Nova | Carrizo | 2.29 b | 2.46 a | 1.20 | 0.282 ab | 0.145 | 0.130 a | 0.213 | 69.8 b |
| mandarin | HRS 812 | 2.60 a | 2.74 a | 1.13 | 0.295 ab | 0.135 | 0.130 a | 0.245 | 113 ab |
| - | HRS 896 | 2.62 a | 2.15 b | 1.27 | 0.233 c | 0.123 | 0.128 a | 0.208 | 93.5 b |
| - | Rough lemon | 2.33 b | 2.29 b | 1.06 | 0.315 a | 0.160 | 0.105 b | 0.215 | 165 a |
| - | Swingle | 2.31 b | 2.22 b | 1.24 | 0.243 bc | 0.123 | 0.123 a | 0.230 | 121 ab |
| - | Pr > F | 0.05 | 0.03 | 0.77 | 0.007 | 0.22 | 0.08 | 0.32 | 0.007 |
| Scion | Rootstocks | Total Fruit Number | Total Fruit Weight | Tree Height | Tree Diameter | Tree Canopy Volume |
|---|---|---|---|---|---|---|
| -# tree- | ---kg--- | --m-- | --m2-- | --m3-- | ||
| Tahiti | Carrizo | 684 | 19.3 | 3.6 ay | 21.2 a | 41.8 a |
| lime | Cleopatra | 1032 | 18.6 | 3.7 a | 22.4 a | 44.6 a |
| - | HRS 812 | 532 | 6.12 | 2.7 c | 14.8 c | 23.0 c |
| - | Rough lemon | 797 | 20.4 | 3.5 a | 19.1 b | 37.0 b |
| - | Swingle | 930 | 8.07 | 3.0 b | 16.1 c | 26.1 c |
| - | Pr > F | 0.330 | 0.230 | 0.016 | 0.039 | 0.015 |
| Nova | Carrizo | 13.0 | 1.51 | 2.22 | 4.06 | 4.76 |
| mandarin | HRS 812 | 11.5 | 1.24 | 2.42 | 4.27 | 5.59 |
| - | HRS 896 | 9.63 | 1.46 | 2.38 | 4.97 | 6.42 |
| - | Rough lemon | 12.0 | 1.56 | 2.13 | 4.53 | 5.48 |
| - | Swingle | 14.5 | 1.79 | 2.39 | 4.57 | 5.75 |
| - | Pr > F | 0.964 | 0.970 | 0.17 | 0.56 | 0.388 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tirado-Corbalá, R.; Rivera-Ocasio, D.; Segarra-Carmona, A.; Román-Paoli, E.; González, A. Performance of Two Citrus Species Grafted to Different Rootstocks in the Presence of Huanglongbing Disease in Puerto Rico. Horticulturae 2018, 4, 38. https://doi.org/10.3390/horticulturae4040038
Tirado-Corbalá R, Rivera-Ocasio D, Segarra-Carmona A, Román-Paoli E, González A. Performance of Two Citrus Species Grafted to Different Rootstocks in the Presence of Huanglongbing Disease in Puerto Rico. Horticulturae. 2018; 4(4):38. https://doi.org/10.3390/horticulturae4040038
Chicago/Turabian StyleTirado-Corbalá, Rebecca, Dania Rivera-Ocasio, Alejandro Segarra-Carmona, Elvin Román-Paoli, and Agenol González. 2018. "Performance of Two Citrus Species Grafted to Different Rootstocks in the Presence of Huanglongbing Disease in Puerto Rico" Horticulturae 4, no. 4: 38. https://doi.org/10.3390/horticulturae4040038
APA StyleTirado-Corbalá, R., Rivera-Ocasio, D., Segarra-Carmona, A., Román-Paoli, E., & González, A. (2018). Performance of Two Citrus Species Grafted to Different Rootstocks in the Presence of Huanglongbing Disease in Puerto Rico. Horticulturae, 4(4), 38. https://doi.org/10.3390/horticulturae4040038





