Seed Dormancy and Germination Characteristics of Scutellaria indica L. var. coccinea S.T.Kim & S.T.Lee., an Endemic Species Found on Jeju Island, South Korea
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Basic Characteristics of Seed
2.3. Phenology Experiments
2.4. Water Absorption Test
2.5. Laboratory Experiments
2.5.1. General Procedure
2.5.2. Effect of Light on Germination
2.5.3. Effect of Temperature on Germination
2.5.4. Effect of Cold Stratification on Germination
2.5.5. Effect of GA3 Treatment on Germination
2.6. Statistical Analysis
3. Results
3.1. Basic Characteristics of Seed
3.2. Phenology Experiment
3.3. Water Absorption Test
3.4. Laboratory Experiments
3.4.1. Effect of Light on Germination
3.4.2. Effect of Temperature on Germination
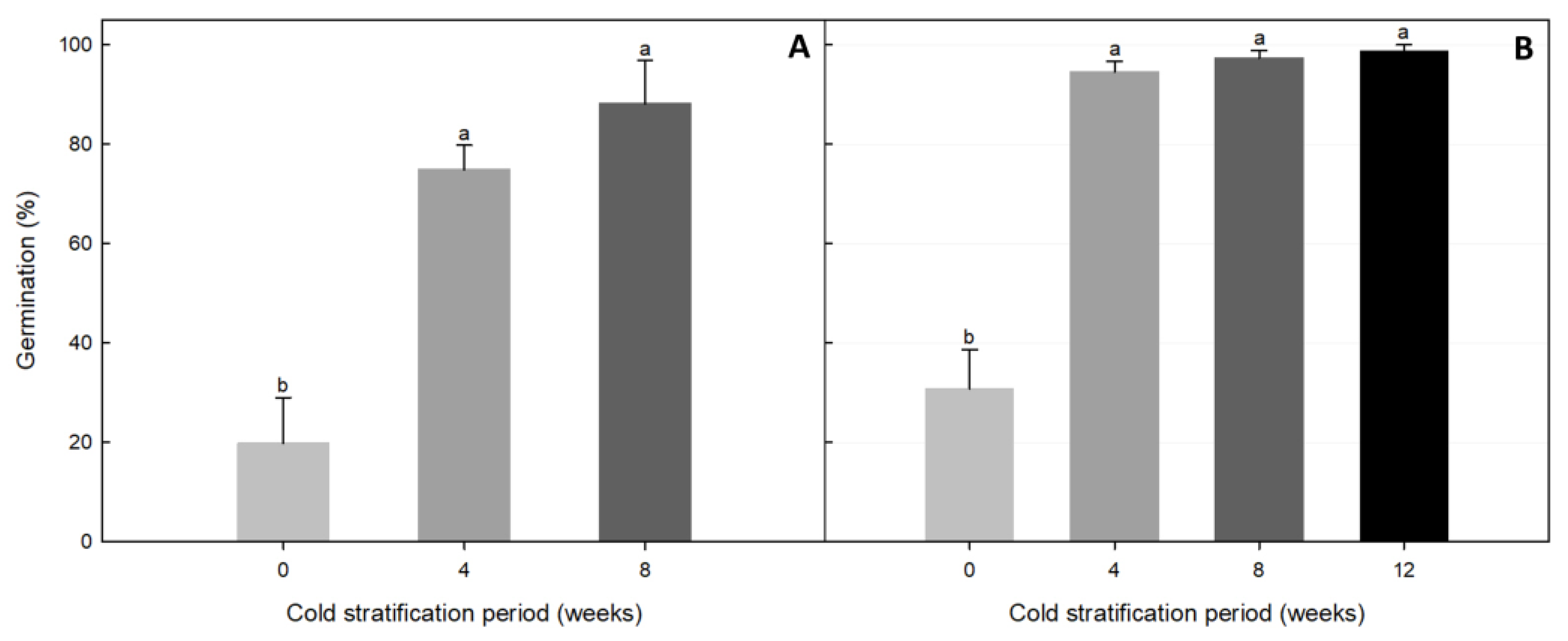
3.4.3. Effect of Cold Stratification on Germination
3.4.4. Effect of GA3 Treatment on Germination
4. Discussion
Practical Conservation Applications
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Noss, R.F. Indicators for monitoring biodiversity: A hierarchical approach. Conserv. Biol. 1990, 4, 355–364. [Google Scholar] [CrossRef]
- Redford, K.H.; Richter, B.D. Conservation of biodiversity in a world of use. Conserv. Biol. 1999, 13, 1246–1256. [Google Scholar] [CrossRef]
- Ramanatha Rao, V.; Hodgkin, T. Genetic diversity and conservation and utilization of plant genetic resources. Plant Cell Tissue Organ Cult. 2002, 68, 1–19. [Google Scholar] [CrossRef]
- Gitay, H.; Suárez, A.; Watson, R.T.; Dokken, D.J. Climate Change and Biodiversity (IPCC Technical Paper V); Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2002. [Google Scholar]
- Bélanger, J.; Pilling, D. The State of the World’s Biodiversity for Food and Agriculture; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2019. [Google Scholar]
- McNeely, J.A. Lessons from the past: Forests and biodiversity. Biodivers. Conserv. 1994, 3, 3–20. [Google Scholar] [CrossRef]
- McNeely, J.A.; Gadgil, M.; Leveque, C.; Padoch, C.; Redford, K. Human influence in biodiversity. In Global Biodiversity Assessment; Heywood, V.H., Ed.; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Sodhi, N.S.; Ehrlich, P.R. (Eds.) Conservation Biology for All; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Pyšek, P.; Jarošík, V.; Hulme, P.E.; Pergl, J.; Hejda, M.; Schaffner, U.; Vilà, M. A global assessment of invasive plant impacts on resident species, communities and ecosystems. Glob. Change Biol. 2012, 18, 1725–1737. [Google Scholar] [CrossRef]
- Corlett, R.T. Plant diversity in a changing world: Status, trends, and conservation needs. Plant Divers. 2016, 38, 10–16. [Google Scholar] [CrossRef]
- Kim, Y.S. Current Status and Development Strategies of Korean Botanical Gardens. Proc. Korean Soc. Environ. Ecol. Conf. 2005, 15, 6–11. [Google Scholar]
- Kim, Y.S. Conservation of plant diversity in Korea. Landsc. Ecol. Eng. 2006, 2, 163–170. [Google Scholar] [CrossRef]
- Lee, B.H. Destruction of nature and the crisis of biodiversity (Jayeon pahoe wa saengmul dayangseong-ui wigi). Sci. Technol. 1991, 24, 42–47. [Google Scholar]
- Hong, H.J.; Kim, C.K.; Lee, H.W.; Lee, W.K. Conservation, restoration, and sustainable use of biodiversity based on habitat quality monitoring: A case study on Jeju Island, South Korea (1989–2019). Land 2021, 10, 774. [Google Scholar] [CrossRef]
- Anderson, S. Area and endemism. Q. Rev. Biol. 1994, 69, 451–471. [Google Scholar] [CrossRef]
- Primack, R.B. A Primer of Conservation Biology, 5th ed.; Sinauer Associates: Sunderland, MA, USA, 2008. [Google Scholar]
- Burlakova, L.E.; Karatayev, A.Y.; Karatayev, V.A.; May, M.E.; Bennett, D.L.; Cook, M.J. Endemic species: Contribution to community uniqueness, effect of habitat alteration, and conservation priorities. Biol. Conserv. 2011, 144, 155–165. [Google Scholar] [CrossRef]
- Chung, G.Y.; Chang, K.S.; Chung, J.M.; Choi, H.J.; Paik, W.K.; Hyun, J.O. A checklist of endemic plants on the Korean Peninsula. Korean J. Plant Taxon. 2017, 47, 264–288. [Google Scholar] [CrossRef]
- Cho, Y.C.; Seol, J.; Lim, C.H. Climate-induced distribution dynamics and niche adaptation of South Korean endemic plants across the Korean Peninsula. Sci. Rep. 2024, 14, 22253. [Google Scholar] [CrossRef]
- Heywood, V.H. The future of plant conservation and the role of botanic gardens. Plant Divers. 2017, 39, 309–313. [Google Scholar] [CrossRef]
- Shin, H.H.; Kang, S.K. The trends in the application of the Global Strategy for Plant Conservation to arboretums and countermeasures. J. Korean Inst. Forest Recreat. 2011, 15, 91–102. [Google Scholar]
- Choi, S.R. Content Analysis and Recognition Survey on Biodiversity Education Programs in National Institute of Biological Resources. Master’s Thesis, Seoul National University, Seoul, Republic of Korea, 2018. [Google Scholar]
- Havens, K.; Vitt, P.; Maunder, M.; Guerrant, E.O.; Dixon, K. Ex situ plant conservation and beyond. BioScience 2006, 56, 525–531. [Google Scholar] [CrossRef]
- Paton, A. A global taxonomic investigation of Scutellaria (Labiatae). Kew Bull. 1990, 45, 399–450. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, S. The complete chloroplast genome of Scutellaria indica var. coccinea (Lamiaceae), an endemic taxon in Korea. Mitochondrial DNA B Resour. 2019, 4, 2539–2540. [Google Scholar] [CrossRef]
- Kim, S.T.; Lee, S.T. Two new varieties of Scutellaria indica L. from Korea. Korean J. Plant Taxon. 1994, 24, 73–78. [Google Scholar] [CrossRef]
- Nishikawa, K.; Furukawa, H.; Fujioka, T.; Fujii, H.; Mihashi, K.; Shimomura, K.; Ishimaru, K. Phenolics in tissue cultures of Scutellaria. Recent Res. Dev. Phytochem. 2000, 4, 55–60. [Google Scholar]
- Guo, X.R.; Wang, X.G.; Su, W.H.; Zhang, G.F.; Zhou, R. DNA barcodes for discriminating the medicinal plant Scutellaria baicalensis (Lamiaceae) and its adulterants. Biol. Pharm. Bull. 2011, 34, 1198–1203. [Google Scholar] [CrossRef]
- Joshee, N.; Tascan, A.; Medina-Bolivar, F.; Parajuli, P.; Rimando, A.M.; Shannon, D.A.; Adelberg, J.W. Scutellaria: Biotechnology, phytochemistry and its potential as a commercial medicinal crop. In Biotechnology for Medicinal Plants: Micropropagation and Improvement; Chandra, S., Lata, H., Varma, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 69–99. [Google Scholar]
- Cole, I.B.; Saxena, P.K.; Murch, S.J. Medicinal biotechnology in the genus Scutellaria. Vitro Cell. Dev. Biol. Plant 2007, 43, 318–327. [Google Scholar] [CrossRef]
- Shang, X.; He, X.; He, X.; Li, M.; Zhang, R.; Fan, P.; Zhang, Q.; Jia, Z. The genus Scutellaria: An ethnopharmacological and phytochemical review. J. Ethnopharmacol. 2010, 128, 279–313. [Google Scholar] [CrossRef]
- Wang, Z.L.; Wang, S.; Kuang, Y.; Hu, Z.M.; Qiao, X.; Ye, M. A comprehensive review on phytochemistry, pharmacology, and flavonoid biosynthesis of Scutellaria baicalensis. Pharm. Biol. 2018, 56, 465–484. [Google Scholar] [CrossRef]
- Korea Heritage Service. Sangumburi Crater. Jeju. Available online: https://www.heritage.go.kr/main/?v=1756202055699 (accessed on 6 April 2025).
- Korea Institute of Oriental Medicine (KIOM). Scutellaria indica var. coccinea. Available online: https://www.kmif.org/board/board.php?b_id=pds (accessed on 6 April 2025).
- Korea National Arboretum (KNA). Scutellaria indica var. coccinea. Available online: https://terms.naver.com/entry.naver?docId=3541232&cid=46694&categoryId=46694 (accessed on 6 April 2025).
- National Institute of Biological Resources (NIBR). Available online: https://species.nibr.go.kr/home/mainHome.do (accessed on 5 June 2025).
- Korea National Arboretum (KNA). Establishment of Conservation and Restoration Infrastructure for Rare Endemic Plants; Reported No. TRKO201500014027; Korea Forest Service: Daejeon, Republic of Korea, 2014. [Google Scholar]
- Abeli, T.; Dalrymple, S.; Godefroid, S.; Mondoni, A.; Müller, J.V.; Rossi, G.; Orsenigo, S. Ex situ collections and their potential for the restoration of extinct plants. Conserv. Biol. 2020, 34, 303–313. [Google Scholar] [CrossRef]
- Doijode, S.D. Seed conservation-An effective method for preservation of genetic variability in French bean (Phaseolus vulgaris L.). Indian J. Plant Genet. Resour. 2001, 14, 290–292. [Google Scholar]
- Baskin, C.C.; Baskin, J.M. Seeds: Ecology, Biogeography and Evolution of Dormancy and Germination, 2nd ed.; Academic Press: Cambridge, MA, USA; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Bewley, J.D. Seed germination and dormancy. Plant Cell 1997, 9, 1055–1066. [Google Scholar] [CrossRef]
- Baskin, J.M.; Baskin, C.C. A classification system for seed dormancy. Seed Sci. Res. 2004, 14, 1–16. [Google Scholar] [CrossRef]
- Adams, C.A.; Baskin, J.M.; Baskin, C.C. Trait stasis versus adaptation in disjunct relict species: Evolutionary changes in seed dormancy-breaking and germination requirements in a subclade of Aristolochia subgenus Siphisia (Piperales). Seed Sci. Res. 2005, 15, 161–173. [Google Scholar] [CrossRef]
- Baskin, J.M.; Baskin, C.C. Ecological life cycle and temperature relations of seed germination and bud growth of Scutellaria parvula. Bull. Torrey Bot. Club. 1982, 109, 1–6. [Google Scholar] [CrossRef]
- Yoshimura, H.; Arakaki, S.; Hamagawa, M.; Kitamura, Y.; Yokota, M.; Denda, T. Differentiation of germination characteristics in Scutellaria rubropunctata (Lamiaceae) associated with adaptation to rheophytic habitats in the subtropical Ryukyu Islands of Japan. J. Plant Res. 2019, 132, 359–368. [Google Scholar] [CrossRef]
- Gorai, M.; Gasmi, H.; Neffati, M. Factors influencing seed germination of medicinal plant Salvia aegyptiaca L. (Lamiaceae). Saudi J. Biol. Sci. 2011, 18, 255–260. [Google Scholar] [CrossRef]
- Bentekhici, M.; Mehdadi, Z.; Latreche, A. Seed germination behavior of Teucrium santae Quézel & Simonneau: A vulnerable and endemic Lamiaceae (Northwest Algeria). Folia Oecol. 2023, 50, 174–184. [Google Scholar]
- Oliveira, M.F.; Cruz, C.R.P.; de Oliveira, L.M. Morphometric characterization of the seeds and germination of six endemic Lamiaceae species in northeastern Brazil. Braz. J. Agric. 2018, 93, 69–79. [Google Scholar]
- Matthews, S.; Khajeh-Hosseini, M. Mean germination time as an indicator of emergence performance in soil of seed lots of maize (Zea mays). Seed Sci. Technol. 2006, 34, 339–347. [Google Scholar] [CrossRef]
- Matthews, S.; Khajeh-Hosseini, M. Length of the lag period of germination and metabolic repair explain vigour differences in seed lots of maize (Zea mays). Seed Sci. Technol. 2007, 35, 200–212. [Google Scholar] [CrossRef]
- Mavi, K.; Demir, I.; Matthews, S. Mean germination time estimates the relative emergence of seed lots of three cucurbit crops under stress conditions. Seed Sci. Technol. 2010, 38, 14–25. [Google Scholar] [CrossRef]
- Scott, S.J.; Jones, R.A.; Williams, W.A. Review of data analysis methods for seed germination. Crop Sci. 1984, 24, 1192–1199. [Google Scholar] [CrossRef]
- Salehzade, H.; Shishvan, M.I.; Ghiyasi, M.; Forouzin, F.; Siyahjani, A.A. Effect of seed priming on germination and seedling growth of wheat (Triticum aestivum L.). Res. J. Biol. Sci. 2009, 4, 629–631. [Google Scholar]
- Miller, S.A.; Bartow, A.; Gisler, M.; Ward, K.; Young, A.S.; Kaye, T.N. Can an ecoregion serve as a seed transfer zone? Evidence from a common garden study with five native species. Restor. Ecol. 2011, 19, 268–276. [Google Scholar] [CrossRef]
- Martin, A.C. The comparative internal morphology of seeds. Am. Midl. Nat. 1946, 36, 513–660. [Google Scholar] [CrossRef]
- An, K.; Yang, M.; Baskin, C.C.; Li, M.; Zhu, M.; Jiao, C.; Wu, H.; Zhang, P. Type 2 nondeep physiological dormancy in seeds of Fraxinus chinensis subsp. rhynchophylla (Hance) A.E. Murray. Forests 2022, 13, 1951. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. When breaking seed dormancy is a problem: Try a move-along experiment. Nat. Plants J. 2003, 4, 17–21. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. The natural history of soil seed banks of arable land. Weed Sci. 2006, 54, 549–557. [Google Scholar] [CrossRef]
- Baskin, J.M.; Baskin, C.C. Classification, biogeography, and phylogenetic relationships of seed dormancy. In Seed Conservation: Turning Science into Practice; Smith, R.D., Dickie, J.B., Linington, S.H., Pritchard, H.W., Probert, R.J., Eds.; Royal Botanic Gardens: Kew, UK, 2003; pp. 518–544. [Google Scholar]
- Nikolaeva, M.G. Factors controlling the seed dormancy pattern. In The Physiology and Biochemistry of Seed Dormancy and Germination; Khan, A.A., Ed.; North-Holland Publishing Company: Amsterdam, The Netherland, 1977; pp. 52–74. [Google Scholar]
- Baskin, J.M.; Nan, X.; Baskin, C.C. A comparative study of seed dormancy and germination in an annual and a perennial species of Senna (Fabaceae). Seed Sci. Res. 1998, 8, 501–512. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. Seeds: Ecology, Biogeography, and Evolution of Dormancy and Germination; Academic Press: Cambridge, MA, USA, 1998. [Google Scholar]
- Heather, A.E.; Pérez, H.E.; Wilson, S.B. Non-deep physiological dormancy in seeds of two Polygonella species with horticultural potential. HortScience 2010, 45, 1854–1858. [Google Scholar] [CrossRef]
- Szekely-Varga, Z.; Kentelky, E.; Cantor, M. Effect of gibberellic acid on the seed germination of Lavandula angustifolia Mill. Rom. J. Hortic. 2021, 2, 169–176. [Google Scholar] [CrossRef]
- Todorović, S.; Živković, S.; Giba, Z.; Grubišić, D.; Mišić, D. Basic seed germination characteristics of the endemic species Nepeta rtanjensis (Lamiaceae). Plant Species Biol. 2007, 22, 205–210. [Google Scholar] [CrossRef]
- Wang, J.Y.; Bu, Z.J.; Poschlod, P.; Yusup, S.; Zhang, J.Q.; Zhang, Z.X. Seed dormancy types and germination response of 15 plant species in temperate montane peatlands. Ecol. Evol. 2024, 14, e11671. [Google Scholar] [CrossRef]
- Liu, K.; Baskin, J.M.; Baskin, C.C.; Bu, H.; Liu, M.; Liu, W.; Du, G. Effect of storage conditions on germination of seeds of 489 species from high elevation grasslands of the eastern Tibet Plateau. Am. J. Bot. 2011, 98, 12–19. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. Germination ecophysiology of herbaceous plant species in a temperate region. Am. J. Bot. 1988, 75, 286–305. [Google Scholar] [CrossRef]
- Grime, J.P. Trait convergence and trait divergence in herbaceous plant communities: Mechanisms and consequences. J. Veg. Sci. 2006, 17, 255–260. [Google Scholar] [CrossRef]
- Soltani, E.; Baskin, C.C.; Baskin, J.M. A graphical method for identifying the six types of non-deep physiological dormancy in seeds. Plant Biol. 2017, 19, 673–682. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. Seed dormancy in wild flowers. In Flower Seeds: Biology and Technology; McDonald, M.B., Kwong, F.Y., Eds.; CABI Publishing: Wallingford, UK, 2005; pp. 163–185. [Google Scholar]
- Nur, M.; Baskin, C.C.; Lu, J.J.; Tan, D.Y.; Baskin, J.M. A new type of non-deep physiological dormancy: Evidence from three annual Asteraceae species. Seed Sci. Res. 2014, 24, 301–314. [Google Scholar] [CrossRef]
- Baskin, J.M.; Baskin, C.C. The annual dormancy cycle in buried weed seeds: A continuum. BioScience 1985, 35, 492–498. [Google Scholar] [CrossRef]
- Brändle, M.; Stadler, J.; Klotz, S.; Brandl, R. Distributional range size of weedy plant species is correlated to germination patterns. Ecology 2003, 84, 136–144. [Google Scholar] [CrossRef]
- Donohue, K.; Rubio de Casas, R.R.; Burghardt, L.; Kovach, K.; Willis, C.G. Germination, postgermination adaptation, and species ecological ranges. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 293–319. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. Cold stratification in winter is more than enough for seed dormancy-break of summer annuals. Seed Sci. Res. 2022, 32, 63–69. [Google Scholar] [CrossRef]
- Archibold, O.W. Ecology of World Vegetation; Springer Science and Business Media: New York, NY, USA, 2012. [Google Scholar]
- Gilliam, F.S. Forest ecosystems of temperate climatic regions: From ancient use to climate change. New Phytol. 2016, 212, 871–887. [Google Scholar] [CrossRef]
- Kim, J.H. Analysis of Plant Phenological Changes in Temperate Forests of Korea. Master’s Thesis, Seoul National University, Seoul, Republic of Korea, 2019. [Google Scholar]
- Ricklefs, R.E.; Latham, R.E. Intercontinental correlation of geographical ranges suggests stasis in ecological traits. Am. Nat. 1992, 139, 1305–1321. [Google Scholar] [CrossRef]
- Qian, H.; Ricklefs, R.E. Geographical distribution and ecological conservatism of disjunct genera. J. Ecol. 2004, 92, 253–265. [Google Scholar] [CrossRef]
- Angevine, M.W.; Chabot, B.F. Seed germination syndromes in higher plants. In Topics in Plant Population Biology; Solbrig, O.T., Jain, S., Johnson, G.B., Raven, P.H., Eds.; Columbia University Press: New York, NY, USA, 1979; pp. 188–206. [Google Scholar]
- Bewley, J.D.; Black, M. 1. Development, Germination, and Growth. In Physiology and Biochemistry of Seeds in Relation to Germination; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Thompson, K.; Grime, J.P. Seasonal variation in the seed banks of herbaceous species in ten contrasting habitats. J. Ecol. 1979, 67, 893–921. [Google Scholar] [CrossRef]
- Roberts, H.A. Seed banks in soils. Adv. Appl. Biol. 1981, 6, 1–55. [Google Scholar]
- Thompson, K.; Bakker, J.P.; Bekker, R.M. The Soil Seed Banks of North West Europe: Methodology, Density and Longevity; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Bakker, J.P.; Poschlod, P.; Strykstra, R.J.; Bekker, R.M.; Thompson, K. Seed banks and seed dispersal: Important topics in restoration ecology. Acta Bot. Neerl. 1996, 45, 461–490. [Google Scholar] [CrossRef]
- Williams, P.A. Ecology of the endangered herb Scutellaria novae-zelandiae. N. Z. J. Ecol. 1992, 16, 127–135. [Google Scholar]
- Walck, J.L.; Baskin, J.M.; Baskin, C.C.; Hidayati, S.N. Defining transient and persistent seed banks. Seed Sci. Res. 2005, 15, 189–196. [Google Scholar] [CrossRef]
- Finch-Savage, W.E.; Leubner-Metzger, G. Seed dormancy and the control of germination. New Phytol. 2006, 171, 501–523. [Google Scholar] [CrossRef]
- Lamichhane, J.R.; Dürr, C.; Schwanck, A.A.; Robin, M.H.; Sarthou, J.P.; Cellier, V.; Messean, A.; Aubertot, J.N.; Debaeke, P. Integrated management of damping-off diseases. A Review. Agron. Sustain. Dev. 2017, 37, 10. [Google Scholar] [CrossRef]
- Slot, M.; Poorter, L. Diversity of tropical tree seedling responses to drought. Biotropica 2007, 39, 683–690. [Google Scholar] [CrossRef]

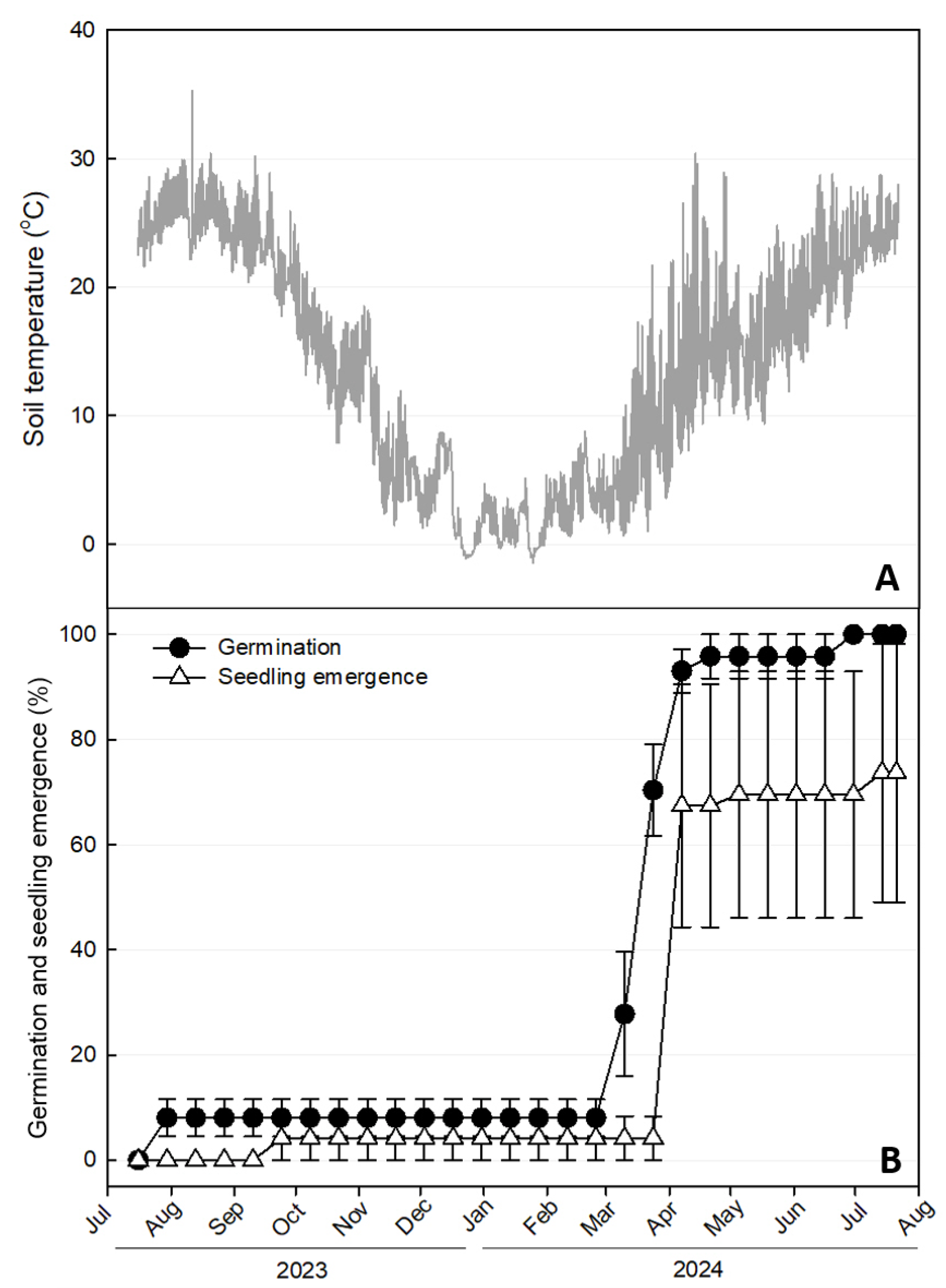
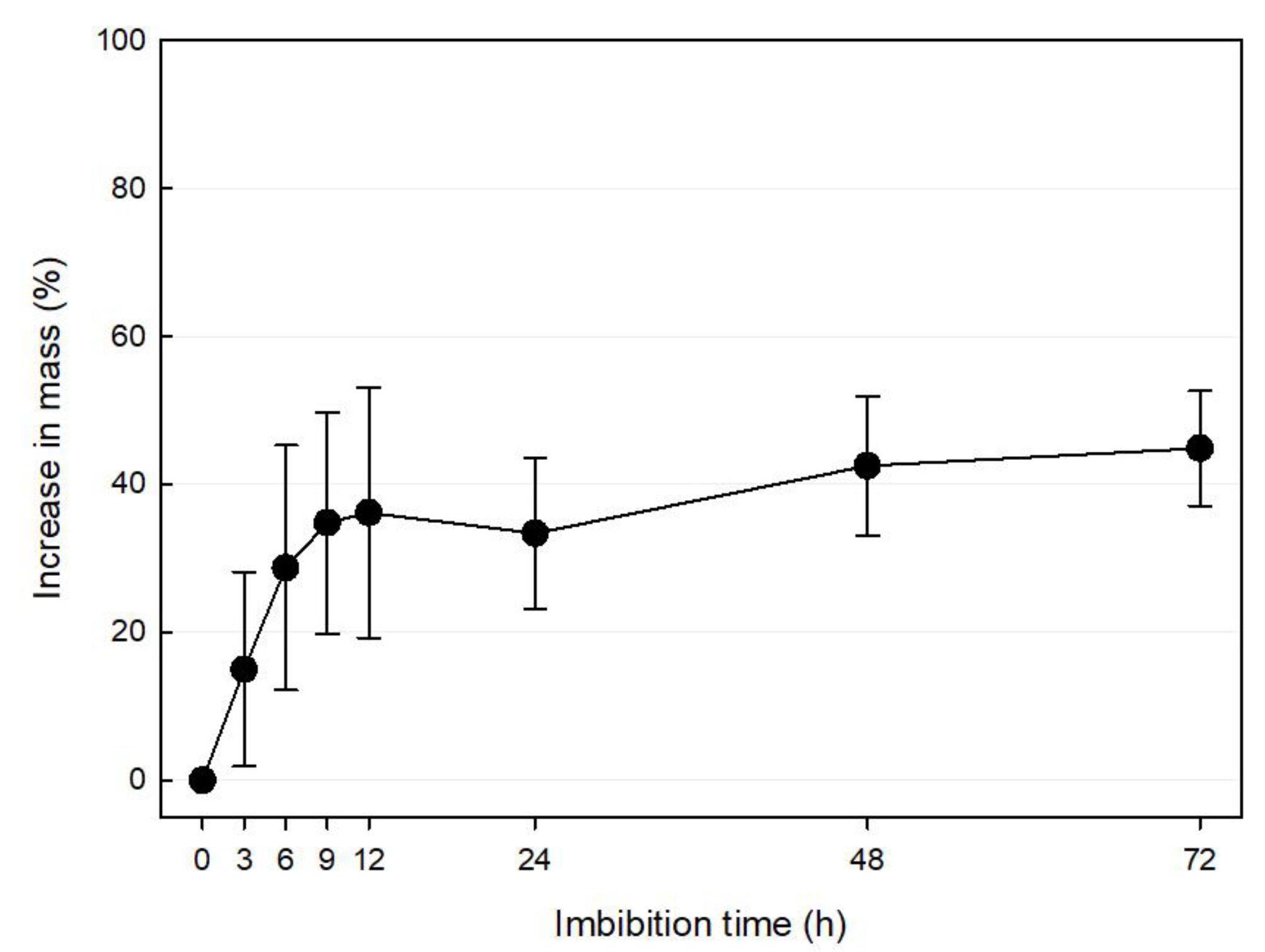
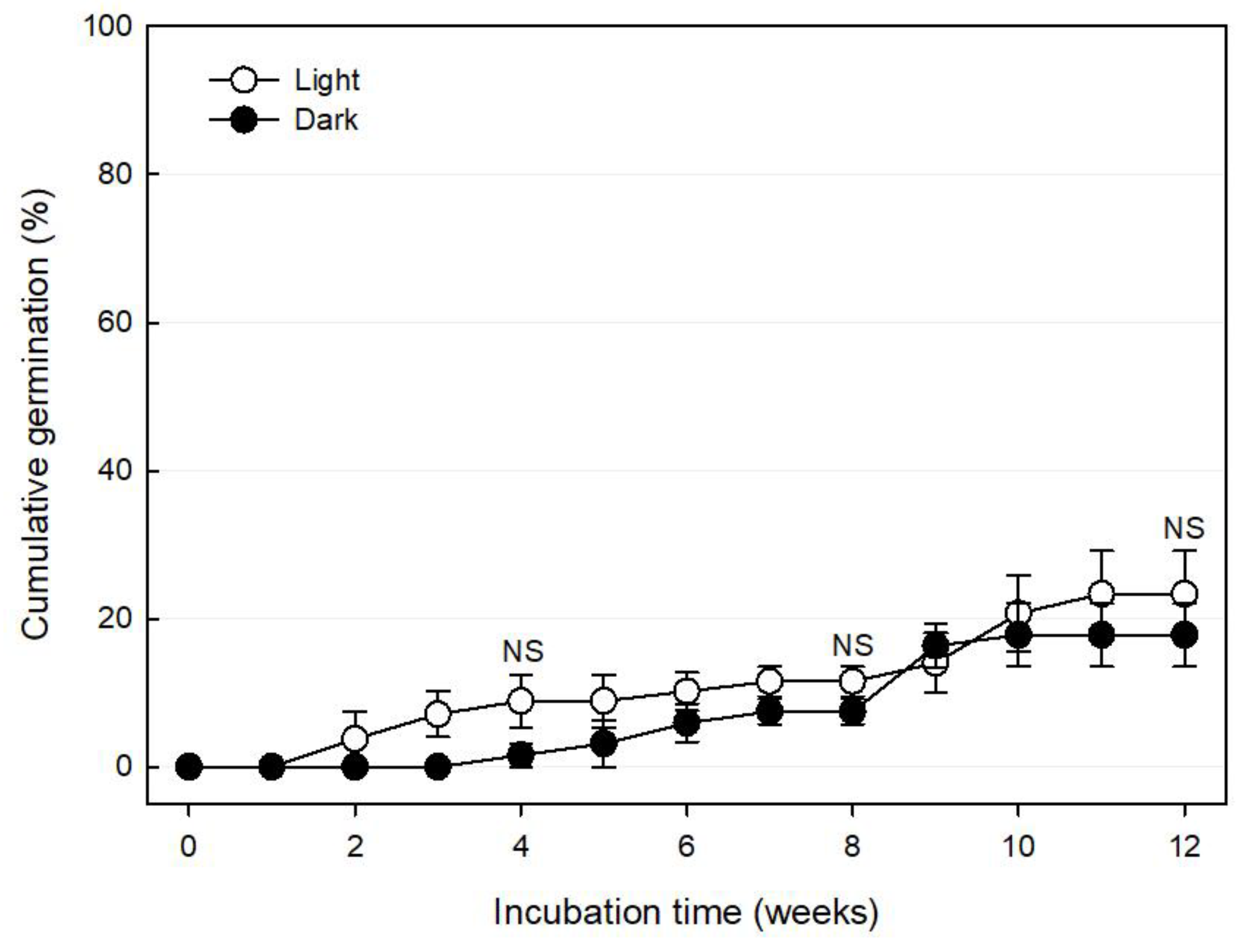
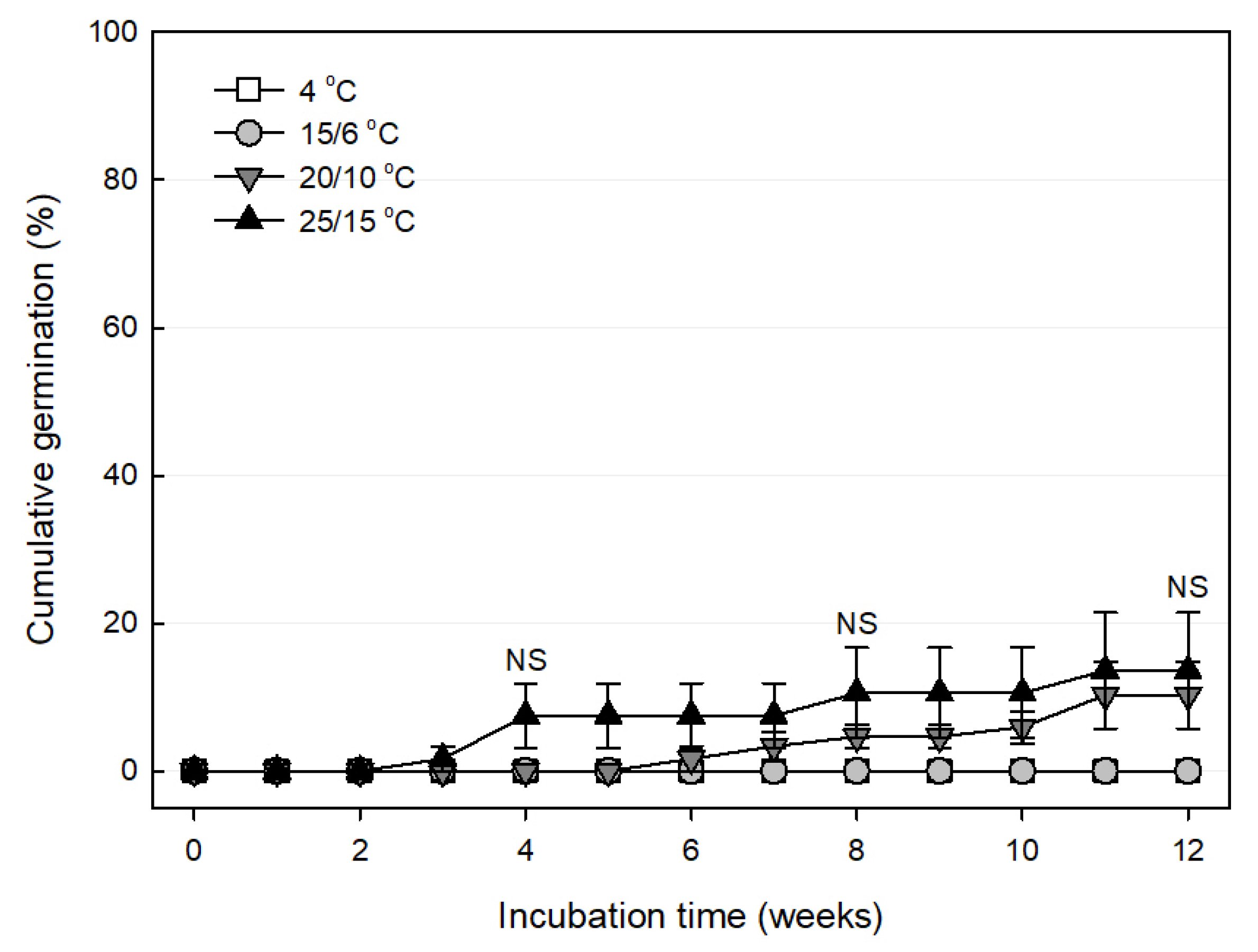

| Scientific Name | Collection Location | Collection Date | Length (mm) | Width (mm) | 100 Seed Weight (mg) |
|---|---|---|---|---|---|
| Scutellaria indica L. var. coccinea S.T.Kim & S.T.Lee | Hantaek Botanical Garden (37°09′ N, 127°41′ E) | 26 June 2021 | 1.27 ± 0.03 1 | 0.82 ± 0.02 | 51.17 ± 0.54 2 |
| Experiment | Cold Stratification Period (Weeks) | MGT (Days) | T50 (Days) | GI | |||
|---|---|---|---|---|---|---|---|
| 1 | 0 | 61.46 ± 6.33 1 | a 2 | NG 3 | 0.06 ± 0.01 | b | |
| 4 | 11.80 ± 0.82 | b | 7.25 ± 1.88 | a | 1.38 ± 0.05 | a | |
| 8 | 12.79 ± 0.58 | b | 10.50 ± 0.00 | a | 1.48 ± 0.19 | a | |
| 2 | 0 | 45.68 ± 4.82 | a | NG | 0.14 ± 0.02 | b | |
| 4 | 15.32 ± 0.27 | b | 10.50 ± 0.00 | a | 1.34 ± 0.03 | b | |
| 8 | 11.88 ± 1.42 | c | 6.13 ± 1.68 | b | 2.08 ± 0.07 | b | |
| 12 | 5.79 ± 1.14 | c | 2.63 ± 0.88 | c | 7.11 ± 1.12 | a | |
| DF | SS | MS | F-Value | p-Value | ||
|---|---|---|---|---|---|---|
| Cold stratification (Experiment 1) | Germination | 2 | 10,556.198 | 5278.099 | 21.38 | 0.0004 |
| MGT | 2 | 6438.010 | 3219.005 | 58.79 | <0.0001 | |
| T50 | 1 | 21.125 | 21.125 | 3.00 | 0.1340 | |
| GI | 2 | 5.078 | 2.539 | 36.48 | <0.0001 | |
| Cold stratification (Experiment 2) | Germination | 3 | 13,181.046 | 4393.682 | 62.37 | <0.0001 |
| MGT | 3 | 3793.997 | 1264.666 | 47.56 | <0.0001 | |
| T50 | 2 | 124.542 | 62.271 | 13.07 | 0.0022 | |
| GI | 3 | 112.995 | 37.665 | 9.16 | 0.0020 | |
| GA3 | Germination | 3 | 33,398.152 | 11,132.717 | 358.33 | <0.0001 |
| Seed Traits | Observation in S. indica var. coccinea |
|---|---|
| Embryo development | Embryo was foliate, bent, and fully developed (no morphological dormancy) |
| Water absorption | Permeable seed coat (no physical and combinational dormancy) |
| Effect of temperature regimes on germination | ≤20% germination (seeds were dormant) |
| Effect of cold stratification on germination | 4 °C, ≥8weeks → ≥80% germination at 25/15 °C |
| Effect of GA3 treatment on germination | GA3 100, 1000 mg·L−1 → ≥80% germination at 25/15 °C |
| Germination phenology | After exposure to cold winter temperatures, spring germination at low soil temperatures (≤10 °C) (temperature range at which seeds can germinate gradually increases from high to low) |
| Dormancy type | Type 2 non-deep physiological dormancy 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.H.; Kwon, H.C.; Lee, S.Y. Seed Dormancy and Germination Characteristics of Scutellaria indica L. var. coccinea S.T.Kim & S.T.Lee., an Endemic Species Found on Jeju Island, South Korea. Horticulturae 2025, 11, 1019. https://doi.org/10.3390/horticulturae11091019
Kim JH, Kwon HC, Lee SY. Seed Dormancy and Germination Characteristics of Scutellaria indica L. var. coccinea S.T.Kim & S.T.Lee., an Endemic Species Found on Jeju Island, South Korea. Horticulturae. 2025; 11(9):1019. https://doi.org/10.3390/horticulturae11091019
Chicago/Turabian StyleKim, Jae Hui, Hak Cheol Kwon, and Seung Youn Lee. 2025. "Seed Dormancy and Germination Characteristics of Scutellaria indica L. var. coccinea S.T.Kim & S.T.Lee., an Endemic Species Found on Jeju Island, South Korea" Horticulturae 11, no. 9: 1019. https://doi.org/10.3390/horticulturae11091019
APA StyleKim, J. H., Kwon, H. C., & Lee, S. Y. (2025). Seed Dormancy and Germination Characteristics of Scutellaria indica L. var. coccinea S.T.Kim & S.T.Lee., an Endemic Species Found on Jeju Island, South Korea. Horticulturae, 11(9), 1019. https://doi.org/10.3390/horticulturae11091019







