Limosilactobacillus fermentum MG5091 and Lactococcus lactis MG4668 and MG5474 Suppress Muscle Atrophy by Regulating Apoptosis in C2C12 Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Cell-Free Supernatants (CFSs) from Lactobacillus and Lactococcus Strains
2.2. Cell Culture and Myotube Differentiation
2.3. Cell Viability
2.4. Western Blotting
2.5. Morphological Characterization of LAB Strains
2.6. Characterization of Carbohydrate Fermentation
2.7. Hemolyltic Activity
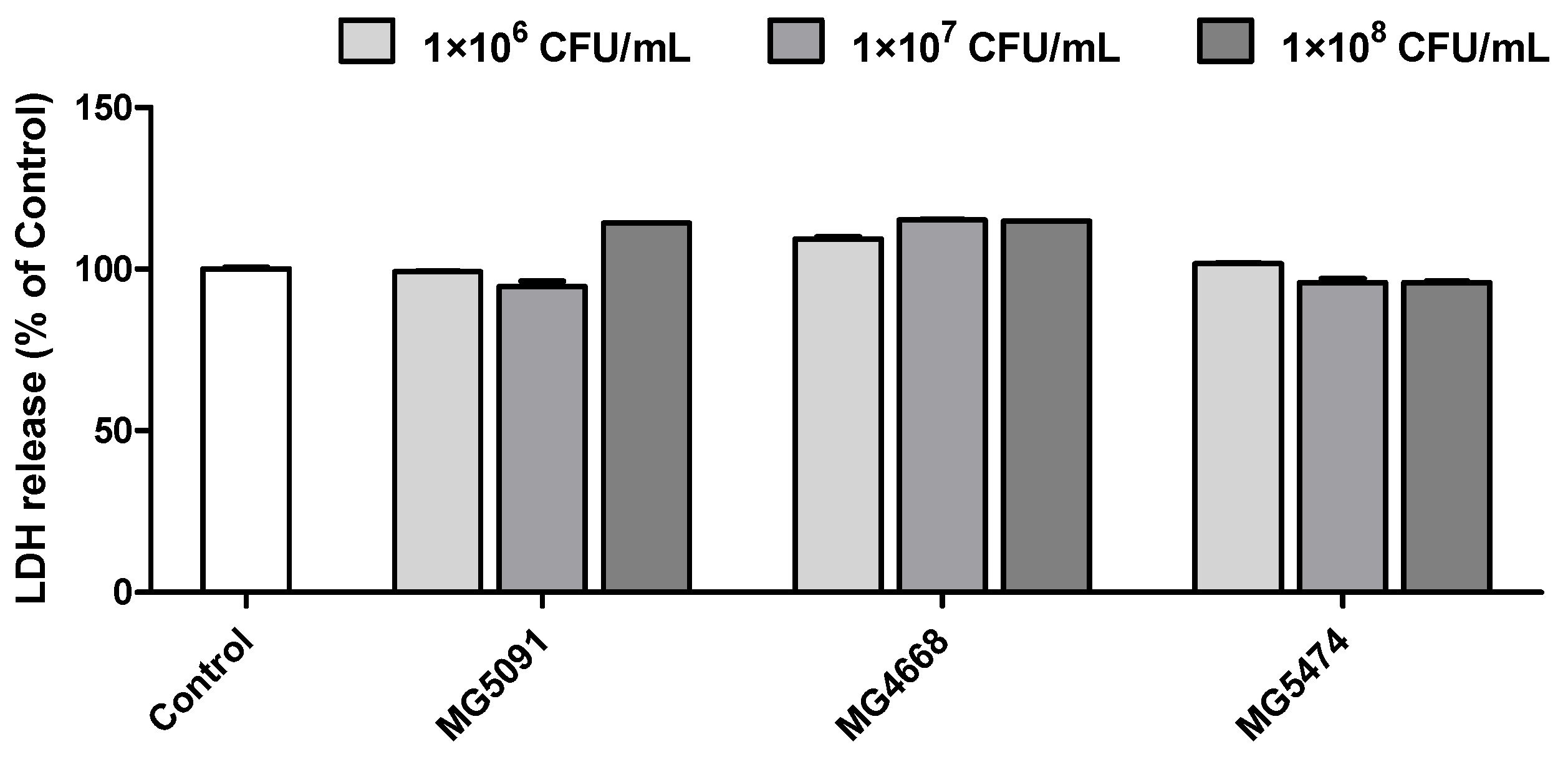
2.8. Lactate Dehydrogenase (LDH) Release
2.9. Survival in the Gastrointestinal Tract (GIT)
2.10. Adhesion Assay on Intestinal Epithelial Cells
2.11. Statistical Analysis
3. Results
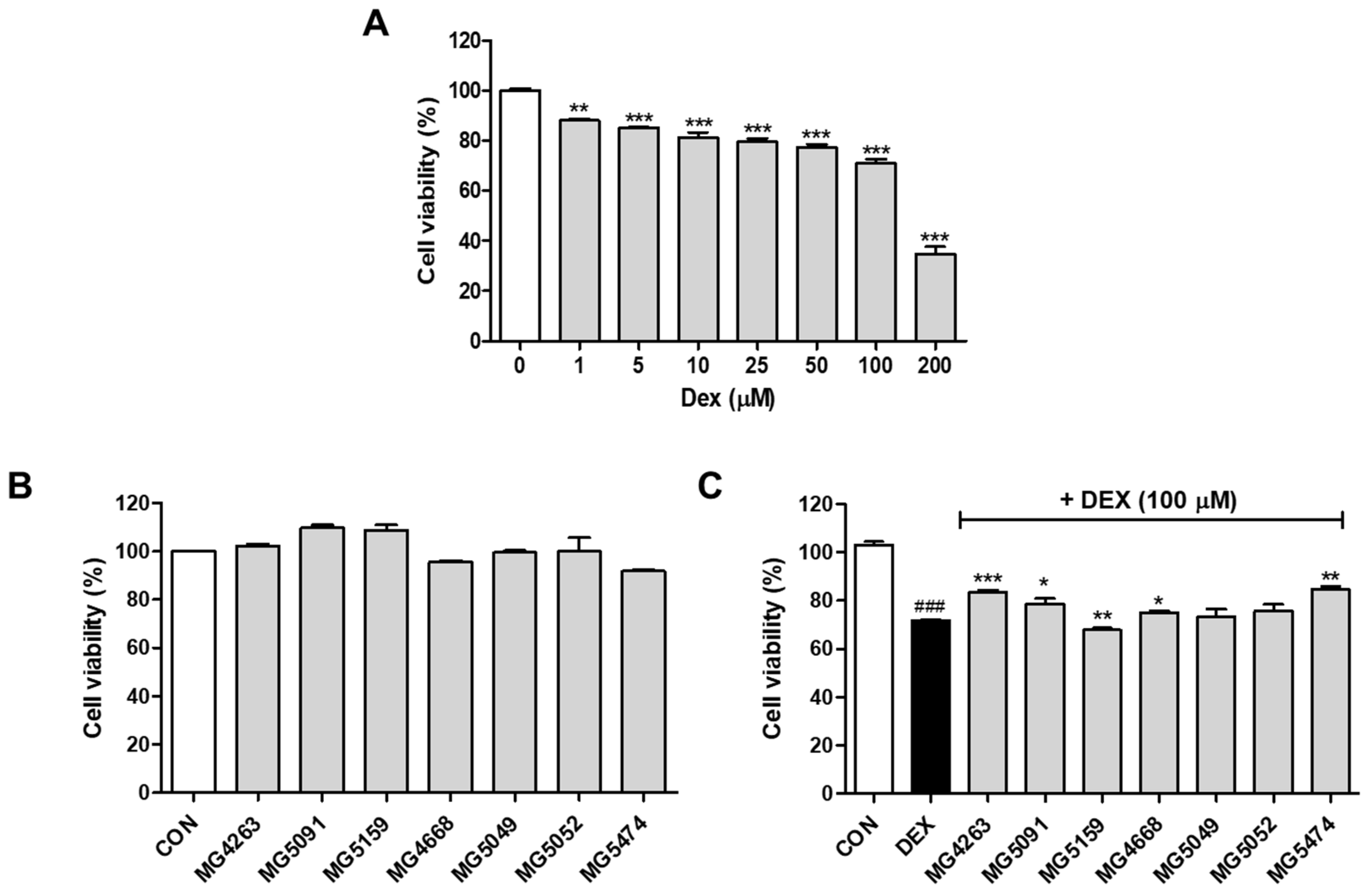
3.1. Cytotoxicity of the LAB Strains in DEX-Treated C2C12 Cells
3.2. Inhibitory Effects of the LAB Strains on Muscle Atrophy Protein Expression Levels in DEX-Treated C2C12 Cells
3.3. Inhibitory Effects of the LAB Strains on Apoptotic Protein Expression Levels in DEX-Treated C2C12 Cells
3.4. Identification of the LAB Strains
3.5. Cytotoxicity of the LAB Strains in HT-29 Cells
3.6. Viability in Simulated Gastrointestinal Fluid and Adhesion of LAB Strains to HT-29 Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abiri, B.; Vafa, M. The role of nutrition in attenuating age-related skeletal muscle atrophy. In Reviews on New Drug Targets in Age-Related Disorders, 1st ed.; Guest, P.C., Ed.; Springer: Berlin/Heidelberg, Germany, 2020; Volume 1260, pp. 297–318. [Google Scholar]
- Shiota, C.; Abe, T.; Kawai, N.; Ohno, A.; Teshima-Kondo, S.; Mori, H.; Terao, J.; Tanaka, E.; Nikawa, T. Flavones inhibit LPS-induced atrogin-1/MAFbx expression in mouse C2C12 skeletal myotubes. J. Nutr. Sci. Vitaminol. 2015, 61, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-W.; Chang, Y.B.; Park, C.W.; Han, S.H.; Suh, H.J.; Ahn, Y. Protein hydrolysate from Spirulina platensis prevents dexamethasone-induced muscle atrophy via Akt/Foxo3 signaling in C2C12 myotubes. Mar. Drugs 2022, 20, 365. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.P. Molecular regulation of skeletal muscle mass. Clin. Exp. Pharmacol. Physiol. 2010, 37, 378–384. [Google Scholar] [CrossRef]
- Lee, J.H.; Jang, J.Y.; Kwon, Y.H.; Lee, S.H.; Park, C.; Choi, Y.H.; Kim, N.D. Effects of Rosemary Extract on C2C12 Myoblast Differentiation and 5-Aminoimidazole-4-carboxamide Ribonucleoside (AICAR)-Induced Muscle Cell Atrophy. Appl. Sci. 2023, 13, 986. [Google Scholar] [CrossRef]
- Frontera, W.R. What is it about old muscles? J. Physiol. 2017, 595, 4581. [Google Scholar] [CrossRef] [PubMed]
- Miki, Y.; Morioka, T.; Shioi, A.; Fujimoto, K.; Sakura, T.; Uedono, H.; Kakutani, Y.; Ochi, A.; Mori, K.; Shoji, T. Oncostatin M induces C2C12 myotube atrophy by modulating muscle differentiation and degradation. Biochem. Biophys. Res. Commun. 2019, 516, 951–956. [Google Scholar] [CrossRef]
- Hah, Y.-S.; Lee, W.K.; Lee, S.; Kim, E.J.; Lee, J.H.; Lee, S.-J.; Ji, Y.H.; Kim, S.G.; Lee, H.-H.; Hong, S.Y. β-Sitosterol attenuates dexamethasone-induced muscle atrophy via regulating FoxO1-dependent signaling in C2C12 cell and mice model. Nutrients 2022, 14, 2894. [Google Scholar] [CrossRef]
- Yin, L.; Li, N.; Jia, W.; Wang, N.; Liang, M.; Yang, X.; Du, G. Skeletal muscle atrophy: From mechanisms to treatments. Pharmacol. Res. 2021, 172, 105807. [Google Scholar] [CrossRef]
- Bann, D.; Chen, H.; Bonell, C.; Glynn, N.W.; Fielding, R.A.; Manini, T.; King, A.C.; Pahor, M.; Mihalko, S.L.; Gill, T.M. Socioeconomic differences in the benefits of structured physical activity compared with health education on the prevention of major mobility disability in older adults: The LIFE study. J. Epidemiol. Community Health 2016, 70, 930–933. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Lee, J.Y.; Park, J.Y.; Jeong, Y.; Kang, C.H. Anti-Inflammatory Response in TNFα/IFNγ-Induced HaCaT Keratinocytes and Probiotic Properties of Lacticaseibacillus rhamnosus MG4644, Lacticaseibacillus paracasei MG4693, and Lactococcus lactis MG5474. J. Microbiol. Biotechnol. 2023, 33, 1–11. [Google Scholar]
- Wang, Y.; Liu, Q.; Quan, H.; Kang, S.-G.; Huang, K.; Tong, T. Nutraceuticals in the Prevention and Treatment of the Muscle Atrophy. Nutrients 2021, 13, 1914. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.A.K.; Sarker, M.; Li, T.; Yin, J. Probiotic species in the modulation of gut microbiota: An overview. BioMed Res. Int. 2018, 2018. [Google Scholar] [CrossRef]
- Zhao, J.; Huang, Y.; Yu, X. A narrative review of gut-muscle axis and sarcopenia: The potential role of gut microbiota. Int. J. Gen. Med. 2021, 14, 1263–1273. [Google Scholar] [CrossRef] [PubMed]
- Mancin, L.; Wu, G.D.; Paoli, A. Gut microbiota–bile acid–skeletal muscle axis. Trends Microbiol. 2022, 31, 254–269. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-K.; Guevarra, R.B.; Kim, Y.-T.; Kwon, J.; Kim, H.; Cho, J.H.; Kim, H.B.; Lee, J.-H. Role of probiotics in human gut microbiome-associated diseases. JMB 2019, 29, 1335–1340. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Yang, X.; Zheng, L.; Wang, Z.; Wu, L.; Jiang, J.; Yang, T.; Ma, L.; Fu, Z. Lactobacillus and Bifidobacterium improves physiological function and cognitive ability in aged mice by the regulation of gut microbiota. Mol. Nutr. Food Res. 2019, 63, 1900603. [Google Scholar] [CrossRef]
- Bindels, L.B.; Beck, R.; Schakman, O.; Martin, J.C.; De Backer, F.; Sohet, F.M.; Dewulf, E.M.; Pachikian, B.D.; Neyrinck, A.M.; Thissen, J.-P. Restoring specific lactobacilli levels decreases inflammation and muscle atrophy markers in an acute leukemia mouse model. PLoS ONE 2012, 7, e37971. [Google Scholar] [CrossRef]
- Chen, Y.-M.; Wei, L.; Chiu, Y.-S.; Hsu, Y.-J.; Tsai, T.-Y.; Wang, M.-F.; Huang, C.-C. Lactobacillus plantarum TWK10 supplementation improves exercise performance and increases muscle mass in mice. Nutrients 2016, 8, 205. [Google Scholar] [CrossRef]
- Lee, K.; Kim, H.J.; Kim, S.A.; Park, S.D.; Shim, J.J.; Lee, J.L. Exopolysaccharide from lactobacillus plantarum HY7714 protects against skin aging through skin–gut axis communication. Molecules 2021, 26, 1651. [Google Scholar] [CrossRef]
- Park, J.-Y.; Lee, J.Y.; Kim, Y.; Kang, C.-H. Latilactobacillus sakei Wikim0066 Protects Skin through MMP Regulation on UVB-Irradiated In Vitro and In Vivo Model. Nutrients 2023, 15, 726. [Google Scholar] [CrossRef]
- Kang, C.-H.; Han, S.H.; Kim, J.-S.; Kim, Y.; Jeong, Y.; Park, H.M.; Paek, N.-S. Inhibition of nitric oxide production, oxidative stress prevention, and probiotic activity of lactic acid bacteria isolated from the human vagina and fermented food. Microorganisms 2019, 7, 109. [Google Scholar] [CrossRef] [PubMed]
- Buxton, R. Blood agar plates and hemolysis protocols. Am. Soc. Microbiol. 2005, 15, 1–9. [Google Scholar]
- Lee, J.; Kim, S.; Kang, C.-H. Immunostimulatory Activity of Lactic Acid Bacteria Cell-Free Supernatants through the Activation of NF-κB and MAPK Signaling Pathways in RAW 264.7 Cells. Microorganisms 2022, 10, 2247. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.H.; Kim, J.-S.; Kwon, H.-J.; Kang, C.-H. The Effect of a Glutathione (GSH)-Containing Cryo-Protectant on the Viability of Probiotic Cells Using a Freeze-Drying Process. Fermentation 2023, 9, 187. [Google Scholar] [CrossRef]
- Bonaldo, P.; Sandri, M. Cellular and molecular mechanisms of muscle atrophy. Dis. Model. Mech. 2013, 6, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Sani, A.; Hasegawa, K.; Yamaguchi, Y.; Panichayupakaranant, P.; Pengjam, Y. Inhibitory effects of curcuminoids on dexamethasone-induced muscle atrophy in differentiation of C2C12 cells. Phytomed. Plus 2021, 1, 100012. [Google Scholar] [CrossRef]
- de Lacerda, J.R.M.; da Silva, T.F.; Vollú, R.E.; Marques, J.M.; Seldin, L. Generally recognized as safe (GRAS) Lactococcus lactis strains associated with Lippia sidoides Cham. are able to solubilize/mineralize phosphate. Springerplus 2016, 5, 1–7. [Google Scholar] [CrossRef]
- Lee, M.-C.; Tu, Y.-T.; Lee, C.-C.; Tsai, S.-C.; Hsu, H.-Y.; Tsai, T.-Y.; Liu, T.-H.; Young, S.-L.; Lin, J.-S.; Huang, C.-C. Lactobacillus plantarum TWK10 improves muscle mass and functional performance in frail older adults: A randomized, double-blind clinical trial. Microorganisms 2021, 9, 1466. [Google Scholar] [CrossRef]
- Chang, Y.B.; Ahn, Y.; Suh, H.J.; Jo, K. Yeast hydrolysate ameliorates dexamethasone-induced muscle atrophy by suppressing MuRF-1 expression in C2C12 cells and C57BL/6 mice. J. Funct. Foods 2022, 90, 104985. [Google Scholar] [CrossRef]
- Choi, R.-Y.; Kim, B.S.; Ban, E.-J.; Seo, M.; Lee, J.H.; Kim, I.-W. Mealworm Ethanol Extract Enhances Myogenic Differentiation and Alleviates Dexamethasone-Induced Muscle Atrophy in C2C12 Cells. Life 2022, 13, 58. [Google Scholar] [CrossRef]
- Li, F.; Luo, T.; Rong, H.; Lu, L.; Zhang, L.; Zheng, C.; Yi, D.; Peng, Y.; Lei, E.; Xiong, X. Maternal rodent exposure to di-(2-ethylhexyl) phthalate decreases muscle mass in the offspring by increasing myostatin. J. Cachexia Sarcopenia Muscle 2022, 13, 2740–2751. [Google Scholar] [CrossRef]
- Ryu, B.; Oh, S.; Yang, H.-W.; Sosorburam, B.; Chung, D.-M.; Seo, M.; Park, S.-J.; Byun, K.; Jeon, Y.-J. Diphlorethohydroxycarmalol derived from Ishige okamurae improves behavioral and physiological responses of muscle atrophy induced by dexamethasone in an in-vivo model. Pharmaceutics 2022, 14, 719. [Google Scholar] [CrossRef]
- Kim, J.W.; Ku, S.-K.; Han, M.H.; Kim, K.Y.; Kim, S.G.; Kim, G.-Y.; Hwang, H.J.; Kim, B.W.; Kim, C.M.; Choi, Y.H. The administration of Fructus Schisandrae attenuates dexamethasone-induced muscle atrophy in mice. Int. J. Mol. Med. 2015, 36, 29–42. [Google Scholar] [CrossRef]
- Chen, C.; Yang, J.-S.; Lu, C.-C.; Chiu, Y.-J.; Chen, H.-C.; Chung, M.-I.; Wu, Y.-T.; Chen, F.-A. Effect of quercetin on dexamethasone-induced C2C12 skeletal muscle cell injury. Molecules 2020, 25, 3267. [Google Scholar] [CrossRef] [PubMed]
- Carotenuto, F.; Coletti, D.; Di Nardo, P.; Teodori, L. α-linolenic acid reduces TNF-induced apoptosis in C2C12 myoblasts by regulating expression of apoptotic proteins. Eur. J. Transl. Myol. 2016, 26, 6033. [Google Scholar] [CrossRef]
- Jin, Y.; Duan, L.X.; Xu, X.L.; Ge, W.J.; Li, R.F.; Qiu, X.J.; Song, Y.; Cao, S.S.; Wang, J.G. Mechanism of apoptosis induction in human hepatocellular carcinoma cells following treatment with a gecko peptides mixture. Biomed. Rep. 2016, 5, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Castañeda, F.; Kinne, R.K. Apoptosis induced in HepG2 cells by short exposure to millimolar concentrations of ethanol involves the Fas-receptor pathway. J. Cancer Res. Clin. Oncol. 2001, 127, 418–424. [Google Scholar] [CrossRef]
- Kim, H.; Kim, J.-S.; Kim, Y.; Jeong, Y.; Kim, J.-E.; Paek, N.-S.; Kang, C.-H. Antioxidant and probiotic properties of Lactobacilli and Bifidobacteria of human origins. Biotechnol. Bioprocess Eng. 2020, 25, 421–430. [Google Scholar] [CrossRef]
- Silva, C.; Domingos-Lopes, M.; Magalhães, V.; Freitas, D.; Coelho, M.; Rosa, H.; Dapkevicius, M. Latin-style fresh cheese enhances lactic acid bacteria survival but not Listeria monocytogenes resistance under in vitro simulated gastrointestinal conditions. J. Dairy Sci. 2015, 98, 4377–4383. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.H.; Kim, Y.; Kim, J.-S.; Jeong, Y.; Park, H.M.; Kim, J.W.; Kim, J.-E.; Kim, H.; Paek, N.-S.; Kang, C.-H. Evaluating the cryoprotective encapsulation of the lactic acid bacteria in simulated gastrointestinal conditions. Biotechnol. Bioprocess Eng. 2020, 25, 287–292. [Google Scholar] [CrossRef]



| LAB | Strain | NCBI Accession Number | Origin |
|---|---|---|---|
| Limosilactobacillus fermentum | MG4263 | OP102570.1 | Human |
| MG5091 | OP102518.1 | Food | |
| MG5159 | MN435579.1 | ||
| Lactococcus lactis | MG4668 | OP035505.1 | Human |
| MG5049 | OP102495.1 | Food | |
| MG5052 | OP102497.1 | ||
| MG5474 | ON619520.1 |
| Carbohydrates | MG5091 | MG4668 | MG5474 |
|---|---|---|---|
| L-Arabinose | + | + | + |
| D-Ribose | + | + | + |
| D-Xylose | - | + | + |
| D-Galactose | + | + | + |
| D-Glucose | + | + | + |
| D-Fructose | + | + | + |
| D-Mannose | - | + | + |
| D-Mannitol | - | + | - |
| N-Acetylglucosamine | - | + | + |
| Amygdalin | - | + | - |
| Arbutin | - | + | + |
| Esculin | + | + | + |
| Salicin | - | + | + |
| D-Cellobiose | - | + | + |
| D-Maltose | + | + | + |
| D-Lactose | - | + | + |
| D-Melibiose | + | + | + |
| D-Sucrose | + | + | + |
| D-Trehalose | + | + | + |
| D-Raffinose | + | + | + |
| Starch | - | + | + |
| Gentiobiose | - | + | + |
| Gluconate | + | + | + |
| 5-keto-gluconate | + | - | - |
| Experiments | L. fermentum | Lc. lactis | ||
|---|---|---|---|---|
| MG5091 | MG4668 | MG5474 | ||
| GIT (Log CFU/mL) | Initial | 8.14 ± 0.02 | 7.59 ± 0.03 | 7.52 ± 0.01 |
| SGF | 7.99 ± 0.03 | 2.78 ± 0.01 | 7.18 ± 0.02 | |
| SIF | 7.88 ± 0.01 | 2.85 ± 0.00 | 7.05 ± 0.00 | |
| Reference | - 1 | [25] | [25] | |
| Adhesion (Log CFU/mL) | Initial | 9.19 ± 0.02 | 8.66 ± 0.07 | 8.41 ± 0.02 |
| Adherent | 6.99 ± 0.10 | 6.41 ± 0.03 | 7.55 ± 0.07 | |
| Adhesion rate (%) | 76.0 ± 1.12 | 74.0 ± 0.33 | 89.7 ± 0.77 | |
| Reference | - | - | [12] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.-Y.; Lee, J.Y.; Kim, Y.; Kang, C.-H. Limosilactobacillus fermentum MG5091 and Lactococcus lactis MG4668 and MG5474 Suppress Muscle Atrophy by Regulating Apoptosis in C2C12 Cells. Fermentation 2023, 9, 659. https://doi.org/10.3390/fermentation9070659
Park J-Y, Lee JY, Kim Y, Kang C-H. Limosilactobacillus fermentum MG5091 and Lactococcus lactis MG4668 and MG5474 Suppress Muscle Atrophy by Regulating Apoptosis in C2C12 Cells. Fermentation. 2023; 9(7):659. https://doi.org/10.3390/fermentation9070659
Chicago/Turabian StylePark, Jeong-Yong, Ji Yeon Lee, YongGyeong Kim, and Chang-Ho Kang. 2023. "Limosilactobacillus fermentum MG5091 and Lactococcus lactis MG4668 and MG5474 Suppress Muscle Atrophy by Regulating Apoptosis in C2C12 Cells" Fermentation 9, no. 7: 659. https://doi.org/10.3390/fermentation9070659
APA StylePark, J.-Y., Lee, J. Y., Kim, Y., & Kang, C.-H. (2023). Limosilactobacillus fermentum MG5091 and Lactococcus lactis MG4668 and MG5474 Suppress Muscle Atrophy by Regulating Apoptosis in C2C12 Cells. Fermentation, 9(7), 659. https://doi.org/10.3390/fermentation9070659






