Learning from Tradition: Health-Promoting Potential of Traditional Lactic Acid Fermentation to Drive Innovation in Fermented Plant-Based Dairy Alternatives
Abstract
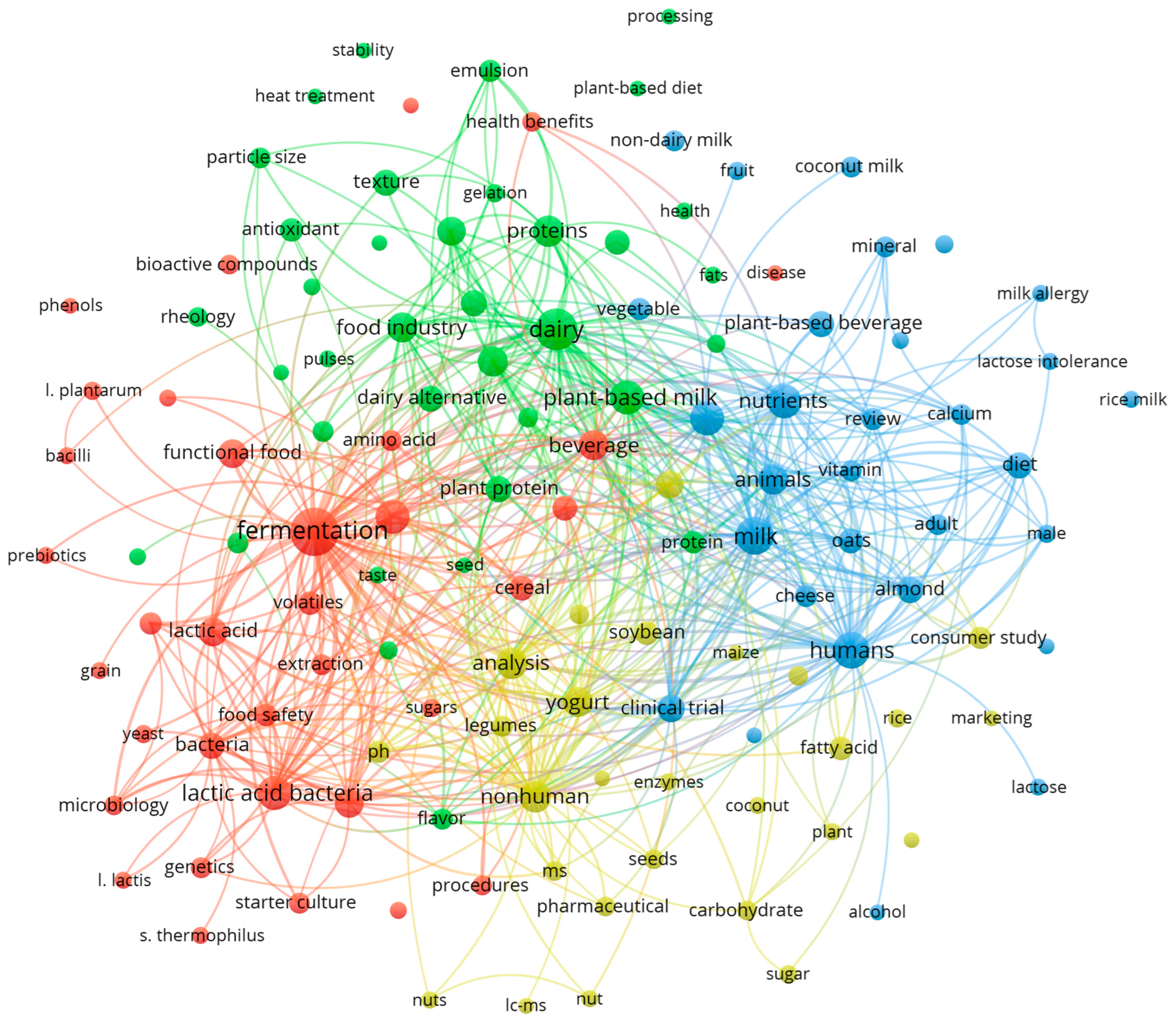
1. Introduction
2. A Renewed Interest in Plant-Based Dairy Alternative Foods
3. Fermentation in Traditional Foods
3.1. Undefined Mixed Cultures of Traditional Foods
3.2. Metabolic Processes during Fermentation
4. Impact of Lactic Acid Fermentation on Macronutrients and Relevance to Human Health
4.1. Carbohydrates
4.2. Proteins
4.3. Secondary Metabolites
4.4. Lipids
4.5. Vitamins
5. Current Trends in LAB-Fermented Plant-Based Dairy Alternatives, as Compared to Traditional Fermented Products
5.1. Fermentation Conditions
5.2. Fermenting Organisms
5.3. Safety Implications
6. Future Outlook
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tangyu, M.; Muller, J.; Bolten, C.J.; Wittmann, C. Fermentation of plant-based milk alternatives for improved flavour and nutritional value. Appl. Microbiol. Biotechnol. 2019, 103, 9263–9275. [Google Scholar] [CrossRef]
- Montemurro, M.; Pontonio, E.; Coda, R.; Rizzello, C.G. Plant-Based Alternatives to Yogurt: State-of-the-Art and Perspectives of New Biotechnological Challenges. Foods 2021, 10, 316. [Google Scholar] [CrossRef]
- Şanlier, N.; Gökcen, B.B.; Sezgin, A.C. Health benefits of fermented foods. Crit. Rev. Food Sci. Nutr. 2019, 59, 506–527. [Google Scholar] [CrossRef]
- Rul, F.; Béra-Maillet, C.; Champomier-Vergès, M.; El-Mecherfi, K.-E.; Foligné, B.; Michalski, M.; Milenkovic, D.; Savary-Auzeloux, I. Underlying evidence for the health benefits of fermented foods in humans. Food Funct. 2022, 13, 4804–4824. [Google Scholar] [CrossRef]
- Adebo, O.A.; Medina-Meza, I.G. Impact of Fermentation on the Phenolic Compounds and Antioxidant Activity of Whole Cereal Grains: A Mini Review. Molecules 2020, 25, 927. [Google Scholar] [CrossRef]
- Adebo, J.A.; Njobeh, P.B.; Gbashi, S.; Oyedeji, A.B.; Ogundele, O.M.; Oyeyinka, S.A.; Adebo, O.A. Fermentation of Cereals and Legumes: Impact on Nutritional Constituents and Nutrient Bioavailability. Fermentation 2022, 8, 63. [Google Scholar] [CrossRef]
- Frazzoli, C.; Grasso, G.; Husaini, D.C.; Ajibo, D.N.; Orish, F.C.; Orisakwe, O.E. Immune System and Epidemics: The Role of African Indigenous Bioactive Substances. Nutrients 2023, 15, 73. [Google Scholar] [CrossRef]
- Joye, I. Protein Digestibility of Cereal Products. Foods 2019, 8, 199. [Google Scholar] [CrossRef]
- Ramesh, S.V.; Shameena Beegum, P.P.; Pandiselvam, R.; Manikantan, M.R.; Hebbar, K.B. Plant-Based Milk Alternatives: Nutritional Potential and Challenges; Springer Nature: Singapore, 2022; pp. 91–106. [Google Scholar]
- Szabo, Z.; Koczka, V.; Marosvolgyi, T.; Szabo, E.; Frank, E.; Polyak, E.; Fekete, K.; Erdelyi, A.; Verzar, Z.; Figler, M. Possible biochemical processes underlying the positive health effects of plant-based diets—A narrative review. Nutrients 2021, 13, 2593. [Google Scholar] [CrossRef]
- Pawlak, R.; Kolasa, K.M. Embracing a plant-based diet: What Is It and What’s the Evidence? Nutr. Today 2017, 52, 155–161. [Google Scholar] [CrossRef]
- Szabó, Z.; Erdélyi, A.; Kisbenedek, A.G.; Ungár, T.; Polyák, E.L.; Szabó, S.S.; Kovács, R.E.; Raposa, L.B.; Figler, M. Plant-based diets: A review. Orv. Hetil. 2016, 157, 1859–1865. [Google Scholar] [CrossRef]
- Appleby, P.N.; Key, T.J. The long-term health of vegetarians and vegans. Proc. Nutr. Soc. 2016, 75, 287–293. [Google Scholar] [CrossRef]
- Ferrari, L.; Panaite, S.A.; Bertazzo, A.; Visioli, F. Animal- and Plant-Based Protein Sources: A Scoping Review of Human Health Outcomes and Environmental Impact. Nutrients 2022, 14, 5115. [Google Scholar] [CrossRef]
- Segovia-Siapco, G.; Sabaté, J. Health and sustainability outcomes of vegetarian dietary patterns: A revisit of the EPIC-Oxford and the Adventist Health Study-2 cohorts. Eur. J. Clin. Nutr. 2019, 72, 60–70. [Google Scholar] [CrossRef]
- McMichael, A.J.; Powles, J.W.; Butler, C.D.; Uauy, R. Food, livestock production, energy, climate change, and health. Lancet 2007, 370, 1253–1263. [Google Scholar] [CrossRef]
- Mascaraque, M. Trends to Watch in Plant-Based Milk—Euromonitor Insights. 2021. Available online: https://www.euromonitor.com/article/trends-to-watch-in-plant-based-milk (accessed on 10 February 2022).
- Obafemi, Y.D.; Oranusi, S.U.; Ajanaku, K.O.; Akinduti, P.A.; Leech, J.; Cotter, P.D. African fermented foods: Overview, emerging benefits, and novel approaches to microbiome profiling. npj Sci. Food 2022, 6, 15. [Google Scholar] [CrossRef]
- Songré-Ouattara, L.T.; Mouquet-Rivier, C.; Icard-Vernière, C.; Humblot, C.; Diawara, B.; Guyot, J.P. Enzyme activities of lactic acid bacteria from a pearl millet fermented gruel (ben-saalga) of functional interest in nutrition. Int. J. Food Microbiol. 2008, 128, 395–400. [Google Scholar] [CrossRef]
- Tou, E.H.; Mouquet-Rivier, C.; Picq, C.; Traorè, A.S.; Trèche, S.; Guyot, J.P. Improving the nutritional quality of ben-saalga, a traditional fermented millet-based gruel, by co-fermenting millet with groundnut and modifying the processing method. LWT—Food Sci. Technol. 2007, 40, 1561–1569. [Google Scholar] [CrossRef]
- Gotcheva, V.; Pandiella, S.S.; Angelov, A.; Roshkova, Z.G.; Webb, C. Microflora identification of the Bulgarian cereal-based fermented beverage boza. Process Biochem. 2000, 36, 127–130. [Google Scholar] [CrossRef]
- Muyanja, C.M.; Narvhus, J.A.; Treimo, J.; Langsrud, T. Isolation, characterisation, and identification of lactic acid bacteria from bushera: A Ugandan traditional fermented beverage. Int. J. Food Microbiol. 2003, 80, 201–210. [Google Scholar] [CrossRef]
- Kalui, C.M.; Mathara, J.M.; Kutima, P.M. Probiotic potential of spontaneously fermented cereal-based foods—A review. Afr. J. Biotechnol. 2010, 9, 2490–2498. [Google Scholar]
- Ilango, S.; Pandey, R.; Antony, U. Functional characterization and microencapsulation of probiotic bacteria from koozh. J. Food Sci. Technol. 2016, 53, 977–989. [Google Scholar] [CrossRef]
- Oguntoyinbo, F.A.; Tourlomousis, P.; Gasson, M.J.; Narbad, A. Analysis of bacterial communities of traditional fermented West African cereal foods using culture independent methods. Int. J. Food Microbiol. 2011, 145, 205–210. [Google Scholar] [CrossRef]
- Efiuvwevwere, B.J.O.; Akona, O. The microbiology o kunun-zaki, a cereal beverage from northern Nigeria, during the fermentation (production) process. World J. Microbiol. Biotechnol. 1995, 11, 491–493. [Google Scholar] [CrossRef]
- Pswarayi, F.; Gänzle, M.G. Composition and Origin of the Fermentation Microbiota of Mahewu, a Zimbabwean Fermented Cereal Beverage. Appl. Environ. Microbiol. 2019, 85, e03130-18. [Google Scholar] [CrossRef]
- Okeke, C.A.; Ezekiel, C.N.; Nwangburuka, C.C.; Sulyok, M.; Ezeamagu, C.O.; Adeleke, R.A.; Dike, S.K.; Krska, R. Bacterial diversity and mycotoxin reduction during maize fermentation (steeping) for ogi production. Front. Microbiol. 2015, 6, 1402. [Google Scholar] [CrossRef]
- Ben Omar, N.; Abriouel, H.; Keleke, S.; Sánchez Valenzuela, A.; Martínez-Cañamero, M.; Lucas López, R.; Ortega, E.; Gálvez, A. Bacteriocin-producing Lactobacillus strains isolated from poto poto, a Congolese fermented maize product, and genetic fingerprinting of their plantaricin operons. Int. J. Food Microbiol. 2008, 127, 18–25. [Google Scholar] [CrossRef]
- Ben Omar, N.; Ampe, F. Microbial community dynamics during production of the Mexican fermented maize dough pozol. Appl. Environ. Microbiol. 2000, 66, 3664–3673. [Google Scholar] [CrossRef]
- Khumalo, B.; Kidane, S.W.; Gadaga, T.H.; Shelembe, J.S. Isolation, identification, and characterization of predominant microorganisms in Swazi traditional fermented porridge (incwancwa) and optimization of fermentation conditions. CyTAJ. Food 2022, 20, 394–403. [Google Scholar] [CrossRef]
- Sengun, I.Y.; Nielsen, D.S.; Karapinar, M.; Jakobsen, M. Identification of lactic acid bacteria isolated from Tarhana, a traditional Turkish fermented food. Int. J. Food Microbiol. 2009, 135, 105–111. [Google Scholar] [CrossRef]
- Sekwati-Monang, B.; Gänzle, M.G. Microbiological and chemical characterisation of ting, a sorghum-based sourdough product from Botswana. Int. J. Food Microbiol. 2011, 150, 115–121. [Google Scholar] [CrossRef]
- Mugula, J.K.; Nnko, S.A.M.; Narvhus, J.A.; Sørhaug, T. Microbiological and fermentation characteristics of togwa, a Tanzanian fermented food. Int. J. Food Microbiol. 2003, 80, 187–199. [Google Scholar] [CrossRef]
- Mbugua, S. Isolation, and characterization of lactic acid bacteria during the traditional fermentation of uji. East Afr. Agric. For. J. 1984, 50, 36–43. [Google Scholar] [CrossRef]
- Oyewole, O.B.; Odunfa, S.A. Characterization, and distribution of lactic acid bacteria in cassava fermentation during fufu production. J. Appl. Bacteriol. 1990, 68, 145–152. [Google Scholar] [CrossRef]
- Oguntoyinbo, F.A.; Dodd, C.E.R. Bacterial dynamics during the spontaneous fermentation of cassava dough in gari production. Food Control. 2010, 21, 306–312. [Google Scholar] [CrossRef]
- Mehari, T.; Ashenafi, M. Microbiology of siljo, a traditional Ethiopian fermented legume product. World J. Microbiol. Biotechnol. 1995, 11, 338–342. [Google Scholar] [CrossRef]
- Zheng, J.; Wittouck, S.; Salvetti, E.; Franz, C.M.; Harris, H.M.; Mattarelli, P.; O’toole, P.W.; Pot, B.; Vandamme, P.; Walter, J. A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol. 2020, 70, 2782–2858. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Zaunmüller, T.; Eichert, M.; Richter, H.; Unden, G. Variations in the energy metabolism of biotechnologically relevant heterofermentative lactic acid bacteria during growth on sugars and organic acids. Appl. Microbiol. Biotechnol. 2006, 72, 421–429. [Google Scholar] [CrossRef]
- Narvhus, J.A.; Gadaga, T.H. The role of interaction between yeasts and lactic acid bacteria in African fermented milks: A review. Int. J. Food Microbiol. 2003, 86, 51–60. [Google Scholar] [CrossRef]
- Samtiya, M.; Aluko, R.E.; Puniya, A.K.; Dhewa, T. Enhancing Micronutrients Bioavailability through Fermentation of Plant-Based Foods: A Concise Review. Fermentation 2021, 7, 63. [Google Scholar] [CrossRef]
- Franz, C.M.A.P.; Huch, M.; Mathara, J.M.; Abriouel, H.; Benomar, N.; Reid, G.; Galvez, A.; Holzapfel, W.H. African fermented foods and probiotics. Int. J. Food Microbiol. 2014, 190, 84–96. [Google Scholar] [CrossRef]
- Achi, O.K.; Asamudo, N.U. Cereal-Based Fermented Foods of Africa as Functional Foods; Springer International Publishing: Berlin/Heidelberg, Germany, 2018; pp. 1–32. [Google Scholar]
- Todorov, S.; Holzapfel, W. Traditional cereal fermented foods as sources of functional microorganisms. In Advances in Fermented Foods and Beverages; Elsevier: Amsterdam, The Netherlands, 2015; pp. 123–153. [Google Scholar]
- Todorov, S.D. Diversity of bacteriocinogenic lactic acid bacteria isolated from boza, a cereal-based fermented beverage from Bulgaria. Food Control 2010, 21, 1011–1021. [Google Scholar] [CrossRef]
- Li, Q.; Gänzle, M.G. Host-adapted lactobacilli in food fermentations: Impact of metabolic traits of host adapted lactobacilli on food quality and human health. Curr. Opin. Food Sci. 2020, 31, 71–80. [Google Scholar] [CrossRef]
- Erkus, O.; De Jager, V.C.; Spus, M.; van Alen-Boerrigter, I.J.; Van Rijswijck, I.M.; Hazelwood, L.; Janssen, P.W.; Van Hijum, S.A.; Kleerebezem, M.; Smid, E.J. Multifactorial diversity sustains microbial community stability. ISME J. 2013, 7, 2126–2136. [Google Scholar] [CrossRef]
- de Vos, W.M. Systems solutions by lactic acid bacteria: From paradigms to practice. Microb. Cell Factories 2011, 10, S2. [Google Scholar] [CrossRef]
- Duar, R.M.; Lin, X.B.; Zheng, J.; Martino, M.E.; Grenier, T.; Pérez-Muñoz, M.E.; Leulier, F.; Gänzle, M.; Walter, J. Lifestyles in transition: Evolution and natural history of the genus Lactobacillus. FEMS Microbiol. Rev. 2017, 41 (Suppl. 1), S27–S48. [Google Scholar] [CrossRef] [PubMed]
- Peyer, L.C.; Zannini, E.; Arendt, E.K. Lactic acid bacteria as sensory biomodulators for fermented cereal-based beverages. Trends Food Sci. Technol. 2016, 54, 17–25. [Google Scholar] [CrossRef]
- Petrova, P.; Petrov, K. Lactic Acid Fermentation of Cereals and Pseudocereals: Ancient Nutritional Biotechnologies with Modern Applications. Nutrients 2020, 12, 1118. [Google Scholar] [CrossRef]
- Johansson, M.; Sanni, A.; Lönner, C.; Molin, G. Phenotypically based taxonomy using API 50CH of lactobacilli from Nigerian ogi, and the occurrence of starch fermenting strains. Int. J. Food Microbiol. 1995, 25, 159–168. [Google Scholar] [CrossRef]
- Velikova, P.; Stoyanov, A.; Blagoeva, G.; Popova, L.; Petrov, K.; Gotcheva, V.; Angelov, A.; Petrova, P. Starch utilization routes in lactic acid bacteria: New insight by gene expression assay. Starch Stärke 2016, 68, 953–960. [Google Scholar] [CrossRef]
- Singh, T.A.; Devi, K.R.; Ahmed, G.; Jeyaram, K. Microbial and endogenous origin of fibrinolytic activity in traditional fermented foods of Northeast India. Food Res. Int. 2014, 55, 356–362. [Google Scholar] [CrossRef]
- Lolkema, J.; Poolman, B.; Konings, W. Role of scalar protons in metabolic energy generation in lactic acid bacteria. J. Bioenerg. Biomembr. 1995, 27, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Savijoki, K.; Ingmer, H.; Varmanen, P. Proteolytic systems of lactic acid bacteria. Appl. Microbiol. Biotechnol. 2006, 71, 394–406. [Google Scholar] [CrossRef]
- Venegas-Ortega, M.G.; Flores-Gallegos, A.C.; Martínez-Hernández, J.L.; Aguilar, C.N.; Nevárez-Moorillón, G.V. Production of Bioactive Peptides from Lactic Acid Bacteria: A Sustainable Approach for Healthier Foods. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1039–1051. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Bayjanov, J.R.; Renckens, B.; Nauta, A.; Siezen, R.J. The proteolytic system of lactic acid bacteria revisited: A genomic comparison. BMC Genom. 2010, 11, 36. [Google Scholar] [CrossRef]
- Ziadi, M.; Bergot, G.; Courtin, P.; Chambellon, E.; Hamdi, M.; Yvon, M. Amino acid catabolism by Lactococcus lactis during milk fermentation. Int. Dairy J. 2010, 20, 25–31. [Google Scholar] [CrossRef]
- Marco, M.L.; Sanders, M.E.; Gänzle, M.; Arrieta, M.C.; Cotter, P.D.; De Vuyst, L.; Hill, C.; Holzapfel, W.; Lebeer, S.; Merenstein, D.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on fermented foods. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 196–208. [Google Scholar] [CrossRef]
- Setta, M.C.; Matemu, A.; Mbega, E.R. Potential of probiotics from fermented cereal-based beverages in improving health of poor people in Africa. J. Food Sci. Technol. 2020, 57, 3935–3946. [Google Scholar] [CrossRef]
- Siciliano, R.A.; Reale, A.; Mazzeo, M.F.; Morandi, S.; Silvetti, T.; Brasca, M. Paraprobiotics: A New Perspective for Functional Foods and Nutraceuticals. Nutrients 2021, 13, 1225. [Google Scholar] [CrossRef]
- Adeyemo, S.; Onilude, A. Reduction of oligosaccharide content of soybeans by the action of Lactobacillus plantarum isolated from fermented cereals. Afr. J. Biotechnol. 2014, 13, 3790–3796. [Google Scholar] [CrossRef]
- Pontonio, E.; Raho, S.; Dingeo, C.; Centrone, D.; Carofiglio, V.E.; Rizzello, C.G. Nutritional, Functional, and Technological Characterization of a Novel Gluten- and Lactose-Free Yogurt-Style Snack Produced With Selected Lactic Acid Bacteria and Leguminosae Flours. Front. Microbiol. 2020, 11, 1664. [Google Scholar] [CrossRef]
- Nyyssölä, A.; Ellilä, S.; Nordlund, E.; Poutanen, K. Reduction of FODMAP content by bioprocessing. Trends Food Sci. Technol. 2020, 99, 257–272. [Google Scholar] [CrossRef]
- Zannini, E.; Jeske, S.; Lynch, K.M.; Arendt, E.K. Development of novel quinoa-based yoghurt fermented with dextran producer Weissella cibaria MG1. Int. J. Food Microbiol. 2018, 268, 19–26. [Google Scholar] [CrossRef]
- Minervini, F.; De Angelis, M.; Surico, R.; Di Cagno, R.; Gänzle, M.; Gobbetti, M. Highly efficient synthesis of exopolysaccharides by Lactobacillus curvatus DPPMA10 during growth in hydrolyzed wheat flour agar. Int. J. Food Microbiol. 2010, 141, 130–135. [Google Scholar] [CrossRef]
- Vila-Real, C.; Pimenta-Martins, A.; Mbugua, S.; Hagrétou, S.-L.; Katina, K.; Maina, N.H.; Pinto, E.; Gomes, A.M.P. Novel synbiotic fermented finger millet-based yoghurt-like beverage: Nutritional, physicochemical, and sensory characterization. J. Funct. Foods 2022, 99, 105324. [Google Scholar] [CrossRef]
- Adeyemo, S.; Onilude, A. Weaning food fortification and improvement of fermented cereal and legume by metabolic activities of probiotics Lactobacillus plantarum. Afr. J. Food Sci. 2018, 12, 254–262. [Google Scholar] [CrossRef][Green Version]
- Montemurro, M.; Pontonio, E.; Gobbetti, M.; Rizzello, C.G. Investigation of the nutritional, functional and technological effects of the sourdough fermentation of sprouted flours. Int. J. Food Microbiol. 2019, 302, 4–58. [Google Scholar] [CrossRef] [PubMed]
- Lorusso, A.; Coda, R.; Montemurro, M.; Rizzello, C. Use of Selected Lactic Acid Bacteria and Quinoa Flour for Manufacturing Novel Yogurt-Like Beverages. Foods 2018, 7, 51. [Google Scholar] [CrossRef]
- Dusabe, A.; Chacha, M.; Vianney, J.M.; Raymond, J. Development of Plant-Based Yoghurt Rich in Bioavailable Essential Nutrients and Bioactive Compounds from Ingredients Available in East Africa. Curr. Res. Nutr. Food Sci. 2022, 10, 250–266. [Google Scholar] [CrossRef]
- Luana, N.; Rossana, C.; Curiel, J.A.; Kaisa, P.; Marco, G.; Rizzello, C.G. Manufacture and characterization of a yogurt-like beverage made with oat flakes fermented by selected lactic acid bacteria. Int. J. Food Microbiol. 2014, 185, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chelikani, V.; Serventi, L. Evaluation of chickpea as alternative to soy in plant-based beverages, fresh and fermented. LWT 2018, 97, 570–572. [Google Scholar] [CrossRef]
- Chiş, M.S.; Păucean, A.; Man, S.M.; Vodnar, D.C.; Teleky, B.-E.; Pop, C.R.; Stan, L.; Borsai, O.; Kadar, C.B.; Urcan, A.C.; et al. Quinoa Sourdough Fermented with Lactobacillus plantarum ATCC 8014 Designed for Gluten-Free Muffins—A Powerful Tool to Enhance Bioactive Compounds. Appl. Sci. 2020, 10, 7140. [Google Scholar] [CrossRef]
- Nissen, L.; Casciano, F.; Gianotti, A. Volatilome changes during probiotic fermentation of combined soy and rice drinks. Food Funct. 2021, 12, 3159–3169. [Google Scholar] [CrossRef]
- Fernando, W.M.A.D.B.; Flint, S.H.; Ranaweera, K.K.D.S.; Bamunuarachchi, A.; Johnson, S.K.; Brennan, C.S. The potential synergistic behaviour of inter- and intra-genus probiotic combinations in the pattern and rate of short chain fatty acids formation during fibre fermentation. Int. J. Food Sci. Nutr. 2018, 69, 144–154. [Google Scholar] [CrossRef]
- Ogodo, A.C.; Ugbogu, O.C.; Onyeagba, R.A.; Okereke, H.C. Microbiological quality, proximate composition and in vitro starch/protein digestibility of Sorghum bicolor flour fermented with lactic acid bacteria consortia. Chem. Biol. Technol. Agric. 2019, 6, 7. [Google Scholar] [CrossRef]
- Simwaka, J.; Chamba, M.; Huiming, Z.; Masamba, K.; Luo, Y. Effect of fermentation on physicochemical and antinutritional factors of complementary foods from millet, sorghum, pumpkin and amaranth seed flours. Int. Food Res. J. 2017, 24, 1869–1879. [Google Scholar]
- Rizzello, C.G.; Lorusso, A.; Russo, V.; Pinto, D.; Marzani, B.; Gobbetti, M. Improving the antioxidant properties of quinoa flour through fermentation with selected autochthonous lactic acid bacteria. Int. J. Food Microbiol. 2017, 241, 252–261. [Google Scholar] [CrossRef]
- Martinez-Villaluenga, C.; Peñas, E.; Frias, J. Bioactive peptides in fermented foods: Production and evidence for health effects. In Fermented Foods in Health and Disease Prevention; Frias, J., Martinez-Villaluenga, C., Peñas, E., Eds.; Academic Press: Boston, MA, USA, 2017; Chapter 2; pp. 23–47. [Google Scholar]
- Daliri, E.B.-M.; Lee, B.H.; Park, M.H.; Kim, J.-H.; Oh, D.-H. Novel angiotensin I-converting enzyme inhibitory peptides from soybean protein isolates fermented by Pediococcus pentosaceus SDL1409. LWT 2018, 93, 88–93. [Google Scholar] [CrossRef]
- Jang, E.K.; Kim, N.Y.; Ahn, H.J.; Ji, G. γ-Aminobutyric acid (GABA) production and angiotensin-I converting enzyme (ACE) inhibitory activity of fermented soybean containing sea tangle by the co-culture of Lactobacillus brevis with Aspergillus oryzae. J. Microbiol. Biotechnol. 2015, 25, 1315–1320. [Google Scholar] [CrossRef]
- Vallabha, S.V.; Tiku, P.K. Antihypertensive Peptides Derived from Soy Protein by Fermentation. Int. J. Pept. Res. Ther. 2014, 20, 161–168. [Google Scholar] [CrossRef]
- Cavazos, A.; de Mejia, E.G. Identification of bioactive peptides from cereal storage proteins and their potential role in prevention of chronic diseases. Compr. Rev. Food Sci. Food Saf. 2013, 12, 364–380. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, B.F.; Zougman, A.; Masse, R.; Mulligan, C. Production and characterization of bioactive peptides from soy hydrolysate and soy-fermented food. Food Res. Int. 2004, 37, 123–131. [Google Scholar] [CrossRef]
- Endo, A.; Sasaki, F.; Maeno, S.; Kanesaki, Y.; Hamaguchi, Y.; Torres, G.A.; Tomita, S.; Nakagawa, J. In vitro and in silico characterisation of Lactobacillus paraplantarum D2-1, a starter culture for soymilk fermentation. Int. J. Food Sci. Nutr. 2018, 69, 857–869. [Google Scholar] [CrossRef]
- Komatsuzaki, N.; Shima, J.; Kawamoto, S.; Momose, H.; Kimura, T. Production of γ-aminobutyric acid (GABA) by Lactobacillus paracasei isolated from traditional fermented foods. Food Microbiol. 2005, 22, 497–504. [Google Scholar] [CrossRef]
- Gupta, S.; Lee, J.J.L.; Chen, W.N. Analysis of Improved Nutritional Composition of Potential Functional Food (Okara) after Probiotic Solid-State Fermentation. J. Agric. Food Chem. 2018, 66, 5373–5381. [Google Scholar] [CrossRef]
- Hwang, C.E.; Kim, S.C.; Kim, D.H.; Lee, H.Y.; Suh, H.K.; Cho, K.M.; Lee, J.H. Enhancement of isoflavone aglycone, amino acid, and CLA contents in fermented soybean yogurts using different strains: Screening of antioxidant and digestive enzyme inhibition properties. Food Chem. 2021, 340, 128199. [Google Scholar] [CrossRef]
- Li, H.; Liu, Y.; Bao, Y.; Liu, X.; Zhang, H. Conjugated Linoleic Acid Conversion by Six Lactobacillus plantarum Strains Cultured in MRS Broth Supplemented with Sunflower Oil and Soymilk. J. Food Sci. 2012, 77, M330–M336. [Google Scholar] [CrossRef]
- Glenn-Davi, K.; Hurley, A.; Brennan, E.; Coughlan, J.; Shiels, K.; Moran, D.; Saha, S.K.; Zabetakis, I.; Tsoupras, A. Fermentation Enhances the Anti-Inflammatory and Anti-Platelet Properties of Both Bovine Dairy and Plant-Derived Dairy Alternatives. Fermentation 2022, 8, 292. [Google Scholar] [CrossRef]
- Khetarpaul, N.; Chauhan, B.M. Effect of pure sequential culture fermentation by yeasts and lactobacilli on HCl-extractability of minerals from pearl millet. Food Chem. 1991, 39, 347–355. [Google Scholar] [CrossRef]
- Sanni, A.; Onilude, A.; Adeleke, E. Preparation and characteristics of lactic acid fermented cowpea milk. Z. Für Lebensm. Forsch. A 1999, 208, 225–229. [Google Scholar] [CrossRef]
- Imade, E.E.; Omonigho, S.E.; Babalola, O.O.; Enagbonma, B.J. Lactic acid bacterial bacteriocins and their bioactive properties against food-associated antibiotic-resistant bacteria. Ann. Microbiol. 2021, 71, 1431–1440. [Google Scholar] [CrossRef]
- Kumar, V.; Sheoran, P.; Gupta, A.; Yadav, J.; Tiwari, S.K. Antibacterial property of bacteriocin produced by Lactobacillus plantarum LD4 isolated from a fermented food. Ann. Microbiol. 2016, 66, 1431–1440. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Wu, S.-J.; Shyu, Y.-T. Antioxidant properties of certain cereals as affected by food-grade bacteria fermentation. J. Biosci. Bioeng. 2014, 117, 449–456. [Google Scholar] [CrossRef]
- Khan, S.A.; Zhang, M.; Liu, L.; Dong, L.; Ma, Y.; Wei, Z.; Chi, J.; Zhang, R. Co-culture submerged fermentation by lactobacillus and yeast more effectively improved the profiles and bioaccessibility of phenolics in extruded brown rice than single-culture fermentation. Food Chem. 2020, 326, 126985. [Google Scholar] [CrossRef]
- Russo, P.; de Chiara, M.L.V.; Capozzi, V.; Arena, M.P.; Amodio, M.L.; Rascón, A.; Dueñas, M.T.; López, P.; Spano, G. Lactobacillus plantarum strains for multifunctional oat-based foods. LWT—Food Sci. Technol. 2016, 68, 288–294. [Google Scholar] [CrossRef]
- Binou, P.; Yanni, A.E.; Karathanos, V.T. Physical properties, sensory acceptance, postprandial glycemic response, and satiety of cereal based foods enriched with legume flours: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 2722–2740. [Google Scholar] [CrossRef]
- Kårlund, A.; Gómez-Gallego, C.; Korhonen, J.; Palo-Oja, O.-M.; El-Nezami, H.; Kolehmainen, M. Harnessing Microbes for Sustainable Development: Food Fermentation as a Tool for Improving the Nutritional Quality of Alternative Protein Sources. Nutrients 2020, 12, 1020. [Google Scholar] [CrossRef]
- Holzapfel, W. Use of starter cultures in fermentation on a household scale. Food Control 1997, 8, 241–258. [Google Scholar] [CrossRef]
- Kasubuchi, M.; Hasegawa, S.; Hiramatsu, T.; Ichimura, A.; Kimura, I. Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation. Nutrients 2015, 7, 2839–2849. [Google Scholar] [CrossRef]
- Ripari, V. Techno-functional role of exopolysaccharides in cereal-based, yogurt-like beverages. Beverages 2019, 5, 16. [Google Scholar] [CrossRef]
- Olano-Martin, E.; Mountzouris, K.C.; Gibson, G.R.; Rastall, R.A. In vitro fermentability of dextran, oligodextran and maltodextrin by human gut bacteria. Br. J. Nutr. 2000, 83, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Prete, R.; Alam, M.K.; Perpetuini, G.; Perla, C.; Pittia, P.; Corsetti, A. Lactic Acid Bacteria Exopolysaccharides Producers: A Sustainable Tool for Functional Foods. Foods 2021, 10, 1653. [Google Scholar] [CrossRef]
- Laiño, J.; Villena, J.; Kanmani, P.; Kitazawa, H. Immunoregulatory effects triggered by lactic acid bacteria exopolysaccharides: New insights into molecular interactions with host cells. Microorganisms 2016, 4, 27. [Google Scholar] [CrossRef] [PubMed]
- Bleau, C.; Monges, A.; Rashidan, K.; Laverdure, J.P.; Lacroix, M.; Van Calsteren, M.R.; Millette, M.; Savard, R.; Lamontagne, L. Intermediate chains of exopolysaccharides from Lactobacillus rhamnosus RW-9595M increase IL-10 production by macrophages. J. Appl. Microbiol. 2010, 108, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, M.; López, P.; Strahinic, I.; Suárez, A.; Kojic, M.; Fernández-García, M.; Topisirovic, L.; Golic, N.; Ruas-Madiedo, P. Characterisation of the exopolysaccharide (EPS)-producing Lactobacillus paraplantarum BGCG11 and its non-EPS producing derivative strains as potential probiotics. Int. J. Food Microbiol. 2012, 158, 155–162. [Google Scholar] [CrossRef]
- Chabot, S.; Yu, H.-L.; De Léséleuc, L.; Cloutier, D.; Van Calsteren, M.-R.; Lessard, M.; Roy, D.; Lacroix, M.; Oth, D. Exopolysaccharides from Lactobacillus rhamnosus RW-9595M stimulate TNF, IL-6 and IL-12 in human and mouse cultured immunocompetent cells, and IFNγ in mouse splenocytes. Lait 2001, 81, 683–697. [Google Scholar] [CrossRef]
- Martínez-Padilla, E.; Li, K.; Blok Frandsen, H.; Skejovic Joehnke, M.; Vargas-Bello-Pérez, E.; Lykke Petersen, I. In Vitro Protein Digestibility and Fatty Acid Profile of Commercial Plant-Based Milk Alternatives. Foods 2020, 9, 1784. [Google Scholar] [CrossRef]
- Lacroix, I.M.E.; Li-Chan, E.C.Y. Evaluation of the potential of dietary proteins as precursors of dipeptidyl peptidase (DPP)-IV inhibitors by an in silico approach. J. Funct. Foods 2012, 4, 403–422. [Google Scholar] [CrossRef]
- Nogata, Y.; Nagamine, T.; Yanaka, M.; Ohta, H. Angiotensin I converting enzyme inhibitory peptides produced by autolysis reactions from wheat bran. J. Agric. Food Chem. 2009, 57, 6618–6622. [Google Scholar] [CrossRef]
- Vecchi, B.; Añon, M.C. ACE inhibitory tetrapeptides from Amaranthus hypochondriacus 11S globulin. Phytochemistry 2009, 70, 864–870.nogata. [Google Scholar] [CrossRef] [PubMed]
- Stanisavljevic, N.; Vukotic, G.; Pastor, F.; Suznjevic, D.; Jovanovic, Z.; Strahinic, I.; Fira, D.; Radovic, S. Antioxidant activity of pea protein hydrolysates produced by batch fermentation with lactic acid bacteria. Arch. Biol. Sci. 2015, 67, 1033–1042. [Google Scholar] [CrossRef]
- Ziarno, M.; Cichońska, P. Lactic acid bacteria-fermentable cereal-and pseudocereal-based beverages. Microorganisms 2021, 9, 2532. [Google Scholar] [CrossRef]
- Waters, D.M.; Mauch, A.; Coffey, A.; Arendt, E.K.; Zannini, E. Lactic acid bacteria as a cell factory for the delivery of functional biomolecules and ingredients in cereal-based beverages: A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 503–520. [Google Scholar] [CrossRef] [PubMed]
- Yousif, N.E.; El Tinay, A.H. Effect of fermentation on protein fractions and in vitro protein digestibility of maize. Food Chem. 2000, 70, 181–184. [Google Scholar] [CrossRef]
- Duodu, K.; Taylor, J.; Belton, P.; Hamaker, B. Factors affecting sorghum protein digestibility. J. Cereal Sci. 2003, 38, 117–131. [Google Scholar] [CrossRef]
- Sarwar Gilani, G.; Wu, C.; Xiao, K.A. Cockell, Impact of Antinutritional Factors in Food Proteins on the Digestibility of Protein and the Bioavailability of Amino Acids and on Protein Quality. Br. J. Nutr. 2012, 108 (Suppl. 2), S315–S332. [Google Scholar] [CrossRef]
- Çabuk, B.; Nosworthy, M.G.; Stone, A.K.; Korber, D.R.; Tanaka, T.; House, J.D.; Nickerson, M.T. Effect of Fermentation on the Protein Digestibility and Levels of Non-Nutritive Compounds of Pea Protein Concentrate. Food Technol. Biotechnol. 2018, 56, 257–264. [Google Scholar] [CrossRef]
- Coda, R.; Melama, L.; Rizzello, C.G.; Curiel, J.A.; Sibakov, J.; Holopainen, U.; Pulkkinen, M.; Sozer, N. Effect of air classification and fermentation by Lactobacillus plantarum VTT E-133328 on faba bean (Vicia faba L.) flour nutritional properties. Int. J. Food Microbiol. 2015, 193, 34–42. [Google Scholar] [CrossRef]
- Gupta, R.K.; Gangoliya, S.S.; Singh, N.K. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J. Food Sci. Technol. 2015, 52, 676–684. [Google Scholar] [CrossRef]
- Đorđević, T.M.; Šiler-Marinković, S.S.; Dimitrijević-Branković, S.I. Effect of fermentation on antioxidant properties of some cereals and pseudo cereals. Food Chem. 2010, 119, 957–963. [Google Scholar] [CrossRef]
- Huynh, N.; Van Camp, J.; Smagghe, G.; Raes, K. Improved Release and Metabolism of Flavonoids by Steered Fermentation Processes: A Review. Int. J. Mol. Sci. 2014, 15, 19369–19388. [Google Scholar] [CrossRef] [PubMed]
- Yeo, S.K.; Ewe, J.A. 5—Effect of fermentation on the phytochemical contents and antioxidant properties of plant foods. In Advances in Fermented Foods and Beverages; Holzapfel, W., Ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 107–122. [Google Scholar]
- Senger, D.R.; Li, D.; Jaminet, S.-C.; Cao, S. Activation of the Nrf2 Cell Defense Pathway by Ancient Foods: Disease Prevention by Important Molecules and Microbes Lost from the Modern Western Diet. PLoS ONE 2016, 11, e0148042. [Google Scholar] [CrossRef] [PubMed]
- Sahab, N.R.; Subroto, E.; Balia, R.L.; Utama, G. γ-Aminobutyric acid found in fermented foods and beverages: Current trends. Heliyon 2020, 6, e05526. [Google Scholar] [CrossRef]
- Melini, F.; Melini, V.; Luziatelli, F.; Ficca, A.G.; Ruzzi, M. Health-promoting components in fermented foods: An up-to-date systematic review. Nutrients 2019, 11, 1189. [Google Scholar] [CrossRef]
- Diana, M.; Quílez, J.; Rafecas, M. Gamma-aminobutyric acid as a bioactive compound in foods: A review. J. Funct. Foods 2014, 10, 407–420. [Google Scholar] [CrossRef]
- Coda, R.; Rizzello, C.G.; Gobbetti, M. Use of sourdough fermentation and pseudo-cereals and leguminous flours for the making of a functional bread enriched of γ-aminobutyric acid (GABA). Int. J. Food Microbiol. 2010, 137, 236–245. [Google Scholar] [CrossRef]
- Bintsis, T. Lactic acid bacteria as starter cultures: An update in their metabolism and genetics. AIMS Microbiol. 2018, 4, 665–684. [Google Scholar] [CrossRef]
- Gorissen, L.; Leroy, F.; De Vuyst, L.; De Smet, S.; Raes, K. Bacterial production of conjugated linoleic and linolenic acid in foods: A technological challenge. Crit. Rev. Food Sci. Nutr. 2015, 55, 1561–1574. [Google Scholar] [CrossRef]
- Stanton, C.; Ross, R.P.; Fitzgerald, G.F.; Sinderen, D.V. Fermented functional foods based on probiotics and their biogenic metabolites. Curr. Opin. Biotechnol. 2005, 16, 198–203. [Google Scholar] [CrossRef]
- Saubade, F.; Hemery, Y.M.; Guyot, J.-P.; Humblot, C. Lactic acid fermentation as a tool for increasing the folate content of foods. Crit. Rev. Food Sci. Nutr. 2017, 57, 3894–3910. [Google Scholar] [CrossRef] [PubMed]
- Nie, Y.; Liu, Y.; Jiang, J.; Xiong, Y.L.; Zhao, X. Rheological, structural, and water-immobilizing properties of mung bean protein-based fermentation-induced gels: Effect of pH-shifting and oil imbedment. Food Hydrocoll. 2022, 129, 107607. [Google Scholar] [CrossRef]
- Saavedra Isusi, G.I.; Paz Puga, D.; van der Schaaf, U.S. Texturing Fermented Emulsion Gels from Soy Protein: Influence of the Emulsifying Agent—Soy Protein vs. Pectin Microgels—On Gel Microstructure, Rheology and Tribology. Foods 2022, 11, 294. [Google Scholar] [CrossRef]
- Vasilean, I.; Aprodu, I.; Garnai, M.; Munteanu, V.; Patrașcu, L. Preliminary Investigations into the Use of Amylases and Lactic Acid Bacteria to Obtain Fermented Vegetable Products. Foods 2021, 10, 1530. [Google Scholar] [CrossRef] [PubMed]
- Boeck, T.; Zannini, E.; Sahin, A.W.; Bez, J.; Arendt, E.K. Nutritional and Rheological Features of Lentil Protein Isolate for Yoghurt-Like Application. Foods 2021, 10, 1692. [Google Scholar] [CrossRef]
- Yang, M.; Li, N.; Tong, L.; Fan, B.; Wang, L.; Wang, F.; Liu, L. Comparison of physicochemical properties and volatile flavor compounds of pea protein and mung bean protein-based yogurt. LWT 2021, 152, 112390. [Google Scholar] [CrossRef]
- Jiang, Z.-Q.; Wang, J.; Stoddard, F.; Salovaara, H.; Sontag-Strohm, T. Preparation and Characterization of Emulsion Gels from Whole Faba Bean Flour. Foods 2020, 9, 755. [Google Scholar] [CrossRef]
- Klost, M.; Drusch, S. Structure formation and rheological properties of pea protein-based gels. Food Hydrocoll. 2019, 94, 622–630. [Google Scholar] [CrossRef]
- Hickisch, A.; Beer, R.; Vogel, R.F.; Toelstede, S. Influence of lupin-based milk alternative heat treatment and exopolysaccharide-producing lactic acid bacteria on the physical characteristics of lupin-based yogurt alternatives. Food Res. Int. 2016, 84, 180–188. [Google Scholar] [CrossRef]
- Wang, X.; Kong, X.; Zhang, C.; Hua, Y.; Chen, Y.; Li, X. Comparison of physicochemical properties and volatile flavor compounds of plant-based yoghurt and dairy yoghurt. Food Res. Int. 2023, 164, 112375. [Google Scholar] [CrossRef]
- Dąbrowski, G.; Paulauskienė, A.; Baltušnikienė, A.; Kłębukowska, L.; Czaplicki, S.; Konopka, I. Changes in Selected Quality Indices in Microbially Fermented Commercial Almond and Oat Drinks. Appl. Sci. 2022, 12, 9983. [Google Scholar] [CrossRef]
- Huang, K.; Liu, Y.; Zhang, Y.; Cao, H.; Luo, D.-k.; Yi, C.; Guan, X. Formulation of plant-based yoghurt from soybean and quinoa and evaluation of physicochemical, rheological, sensory and functional properties. Food Biosci. 2022, 49, 101831. [Google Scholar] [CrossRef]
- Raikos, V.; Juskaite, L.; Vas, F.; Hayes, H.E. Physicochemical properties, texture, and probiotic survivability of oat-based yogurt using aquafaba as a gelling agent. Food Sci. Nutr. 2020, 8, 6426–6432. [Google Scholar] [CrossRef]
- Brückner-Gühmann, M.; Vasil’eva, E.; Culetu, A.; Duta, D.; Sozer, N.; Drusch, S. Oat protein concentrate as alternative ingredient for non-dairy yoghurt-type product. J. Sci. Food Agric. 2019, 99, 5852–5857. [Google Scholar] [CrossRef]
- Salmerón, I.; Thomas, K.; Pandiella, S.S. Effect of potentially probiotic lactic acid bacteria on the physicochemical composition and acceptance of fermented cereal beverages. J. Funct. Foods 2015, 15, 106–115. [Google Scholar] [CrossRef]
- Sethi, S.; Tyagi, S.K.; Anurag, R.K. Plant-based milk alternatives an emerging segment of functional beverages: A review. J. Food Sci. Technol. 2016, 53, 3408–3423. [Google Scholar] [CrossRef] [PubMed]
- Craig, W.J.; Brothers, C.J. Nutritional Content and Health Profile of Non-Dairy Plant-Based Yogurt Alternatives. Nutrients 2021, 13, 4069. [Google Scholar] [CrossRef]
- Jeske, S.; Bez, J.; Arendt, E.K.; Zannini, E. Formation, stability, and sensory characteristics of a lentil-based milk substitute as affected by homogenisation and pasteurisation. Eur. Food Res. Technol. 2019, 245, 1519–1531. [Google Scholar] [CrossRef]
- Aydar, E.F.; Tutuncu, S.; Ozcelik, B. Plant-based milk substitutes: Bioactive compounds, conventional and novel processes, bioavailability studies, and health effects. J. Funct. Foods 2020, 70, 103975. [Google Scholar] [CrossRef]
- Tsafrakidou, P.; Michaelidou, A.-M.; Biliaderis, C.G. Fermented Cereal-based Products: Nutritional Aspects, Possible Impact on Gut Microbiota and Health Implications. Foods 2020, 9, 734. [Google Scholar] [CrossRef]
- De Vuyst, L.; De Vin, F.; Vaningelgem, F.; Degeest, B. Recent developments in the biosynthesis and applications of heteropolysaccharides from lactic acid bacteria. Int. Dairy J. 2001, 11, 687–707. [Google Scholar] [CrossRef]
- Degeest, B.; Mozzi, F.; De Vuyst, L. Effect of medium composition and temperature and pH changes on exopolysaccharide yields and stability during Streptococcus thermophilus LY03 fermentations. Int. J. Food Microbiol. 2002, 79, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Chien, M.-F.C. Exopolysaccharides production as affected by lactic acid bacteria and fermentation time. Food Chem. 2007, 100, 1419–1423. [Google Scholar] [CrossRef]
- Degeest, B.; Janssens, B.; De Vuyst, L. Exopolysaccharide (EPS) biosynthesis by Lactobacillus sakei 0–1: Production kinetics, enzyme activities and EPS yields. J. Appl. Microbiol. 2001, 91, 470–477. [Google Scholar] [CrossRef]
- Wang, K.; Niu, M.; Song, D.; Liu, Y.; Wu, Y.; Zhao, J.; Li, S.; Lu, B. Evaluation of biochemical and antioxidant dynamics during the co-fermentation of dehusked barley with Rhizopus oryzae and Lactobacillus plantarum. J. Food Biochem. 2020, 44, e13106. [Google Scholar] [CrossRef]
- Vaningelgem, F.; Zamfir, M.; Mozzi, F.; Adriany, T.; Vancanneyt, M.; Swings, J.; De Vuyst, L. Biodiversity of exopolysaccharides produced by Streptococcus thermophilus strains is reflected in their production and their molecular and functional characteristics. Appl. Environ. Microbiol. 2004, 70, 900–912. [Google Scholar] [CrossRef]
- Rabha, B.; Nadra, R.-S.; Ahmed, B. Effect of some fermentation substrates and growth temperature on exopolysaccharide production by Streptococcus thermophilus BN1. Int. J. Biosci. Biochem. Bioinform. 2012, 2, 44. [Google Scholar] [CrossRef]
- Gänzle, M.G. Lactic metabolism revisited: Metabolism of lactic acid bacteria in food fermentations and food spoilage. Curr. Opin. Food Sci. 2015, 2, 106–117. [Google Scholar] [CrossRef]
- Aderinola, T.A.; Duodu, K.G. Production, health-promoting properties and characterization of bioactive peptides from cereal and legume grains. BioFactors 2022, 48, 972–992. [Google Scholar] [CrossRef]
- Chai, K.F.; Voo, A.Y.H.; Chen, W.N. Bioactive peptides from food fermentation: A comprehensive review of their sources, bioactivities, applications, and future development. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3825–3885. [Google Scholar] [CrossRef]
- Cheigh, C.-I.; Choi, H.-J.; Park, H.; Kim, S.-B.; Kook, M.-C.; Kim, T.-S.; Hwang, J.-K.; Pyun, Y.-R. Influence of growth conditions on the production of a nisin-like bacteriocin by Lactococcus lactis subsp. lactis A164 isolated from kimchi. J. Biotechnol. 2002, 95, 225–235. [Google Scholar]
- Mataragas, M.; Metaxopoulos, J.; Galiotou, M.; Drosinos, E.H. Influence of pH and temperature on growth and bacteriocin production by Leuconostoc mesenteroides L124 and Lactobacillus curvatus L442. Meat Sci. 2003, 64, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Jakubczyk, A.; Karaś, M.; Baraniak, B.; Pietrzak, M. The impact of fermentation and in vitro digestion on formation angiotensin converting enzyme (ACE) inhibitory peptides from pea proteins. Food Chem. 2013, 141, 3774–3780. [Google Scholar] [CrossRef] [PubMed]
- Rekha, C.R.; Vijayalakshmi, G. Bioconversion of isoflavone glycosides to aglycones, mineral bioavailability and vitamin B complex in fermented soymilk by probiotic bacteria and yeast. J. Appl. Microbiol. 2010, 109, 1198–1208. [Google Scholar] [CrossRef]
- Hugenschmidt, S.; Schwenninger, S.M.; Lacroix, C. Concurrent high production of natural folate and vitamin B12 using a co-culture process with Lactobacillus plantarum SM39 and Propionibacterium freudenreichii DF13. Process Biochem. 2011, 46, 1063–1070. [Google Scholar] [CrossRef]
- Bolaños-Núñez, S.; Santiago-Urbina, J.A.; Guyot, J.-P.; Díaz-Ruiz, G.; Wacher, C. Microbial Interactions between Amylolytic and Non-Amylolytic Lactic Acid Bacteria Strains Isolated during the Fermentation of Pozol. Foods 2021, 10, 2607. [Google Scholar] [CrossRef]
- Bartula, K.; Begley, M.; Latour, N.; Callanan, M. Growth of food-borne pathogens Listeria and Salmonella and spore-forming Paenibacillus and Bacillus in commercial plant-based milk alternatives. Food Microbiol. 2023, 109, 104143. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, V.; Fragasso, M.; Romaniello, R.; Berbegal, C.; Russo, P.; Spano, G. Spontaneous Food Fermentations and Potential Risks for Human Health. Fermentation 2017, 3, 49. [Google Scholar] [CrossRef]
- Keisam, S.; Tuikhar, N.; Ahmed, G.; Jeyaram, K. Toxigenic and pathogenic potential of enteric bacterial pathogens prevalent in the traditional fermented foods marketed in the Northeast region of India. Int. J. Food Microbiol. 2019, 296, 21–30. [Google Scholar] [CrossRef]
- Holzapfel, W.H. Appropriate starter culture technologies for small-scale fermentation in developing countries. Int. J. Food Microbiol. 2002, 75, 197–212. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, N.; Battista, N.; Prete, R.; Corsetti, A. Health-Promoting Role of Lactiplantibacillus plantarum Isolated from Fermented Foods. Microorganisms 2021, 9, 349. [Google Scholar] [CrossRef] [PubMed]
- Cotter, P.D.; Hill, C.; Ross, R.P. Bacteriocins: Developing innate immunity for food. Nat. Rev. Microbiol. 2005, 3, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Yeğin, S.; Üren, A. Biogenic amine content of boza: A traditional cereal-based, fermented Turkish beverage. Food Chem. 2008, 111, 983–987. [Google Scholar] [CrossRef]
- Li, B.; Lu, S. The Importance of Amine-degrading Enzymes on the Biogenic Amine Degradation in Fermented Foods: A review. Process Biochem. 2020, 99, 331–339. [Google Scholar] [CrossRef]
- Gao, X.; Li, C.; He, R.; Zhang, Y.; Wang, B.; Zhang, Z.-H.; Ho, C.-T. Research advances on biogenic amines in traditional fermented foods: Emphasis on formation mechanism, detection and control methods. Food Chem. 2023, 405, 134911. [Google Scholar] [CrossRef]
- Alvarez, M.A.; Moreno-Arribas, M.V. The problem of biogenic amines in fermented foods and the use of potential biogenic amine-degrading microorganisms as a solution. Trends Food Sci. Technol. 2014, 39, 146–155. [Google Scholar] [CrossRef]

| Food * | Substrate | Dominating LAB Species ** | Preparation | Type | Origin | Ref. |
|---|---|---|---|---|---|---|
| Cereals | ||||||
| Ben-saalga | Millet | L. delbrueckii subsp. bulgaricus, Lp. plantarum, Lm. fermentum, Weisella confusa, L. amylolyticus, L. helveticus | Soaked, wet-milled, sieved, fermented, then cooked | Fermented gruel used to complement diets of infants and young children | Burkina Faso | [19,20] |
| Boza | Maize, Millet, and Rye | Lp. plantarum, L. acidophilus, Lm. fermentum, L. coprophilus, Lv. brevis, Lcc. raffinolactis, Leuc. mesenteroides | Boiled flour mixed with old batch, fermented for 24 h | Thick sweet–sour beverage with pale white-to-yellow colour | BulgariaTurkey | [21] |
| Bushera, Obushera | Millet, Sorghum | Lv. brevis, Lm. fermentum, Lp. plantarum, Lc. paracasei subsp. paracasei, Streptococcus thermophilus, L. delbrueckii | Boiled flour mixed with germinated unboiled flour as inoculant, fermented for 5 days at ambient temperature | Moderately thick, sweet–sour social drink and weaning food with pale brown colour | Uganda | [22] |
| Ikii | Maize | Lm. fermentum, Lp. plantarum, Weissella confusa, Lacticaseibacillus rhamnosus, Pediococcus sp. | Cooked, mixed with old batch, fermented for 72 h at ambient temperature | Thick porridge, popular among children, breastfeeding mothers, and aged populations and given to a sick and recovering person | Kenya | [23] |
| Koozh | Millet, Rice | Lp. plantarum, Bacillus amyloliquefaciens, Leuconostoc sp., Weissella sp. | Millet slurry, fermented overnight, mixed with rice porridge and cooked, fermented again, mixed with water before consumption | Fermented porridge as breakfast or lunch | India | [24] |
| Kunu-zaki | Millet | S. lutetiensis, Lm. fermentum, L. delbrueckii, Clostridium perfringens, Weissella confusa | Soaked grains, wet-milled, sieved, one part cooked mixed with uncooked part, fermented for 8 h | Sweet breakfast food drink | Nigeria | [25,26] |
| Mahewu | Maize, Millet, Sorghum | Lp. plantarum, Lv. brevis, Lm. fermentum, Pediococcus pentosaceus, Weissella confusa | Germinated ground millet mixed with cooked maise porridge, fermented 16–48 h | Creamy and sour beverage used as infant food | South africa | [27] |
| Ogi | Maize, Sorghum | Lp. plantarum, Lp. paraplantarum, P. acidilacti, P. pentosaceus, L. helveticus, Lm. fermentum, W. confusa, L. amylolyticus, Lcc. lactis | Soaked and pre-fermented grains, wet-milled, sieved, fermented for three days, then cooked until creamy | Fermented cereal pudding used as infant food | Nigeria | [25,28] |
| Poto poto | Maize | Lp. plantarum, L. gasseri, Enterococcus sp., Escherichia coli, L. acidophilus, L. delbrueckii, Bacillus sp., Lm. reuteri and Lc. casei | Soaked, milled, fermented for 10 h, dried for storage, boiled before consumption | Gruel used as weaning food | Congo | [29] |
| Pozol | Maize | Lp. plantarum, Lc. casei, L. delbrueckii, Lm. fermentum, Bifidobacterium sp. | Maize dough fermented for 3 days, soaked before consumption | Refreshing beverage, consumed for its curative properties and at religious ceremonies | Mexico | [30] |
| Swazi | Maize, Sorghum | Lp. plantarum, Leuconostoc sp. | Boiled, fermented for 72 h to 6 d | Liquid brown coloured beverage | Eswatini | [31] |
| Tarhana | Wheat | S. thermophilus, Lm. fermentum, Pediococcus pentosaceus, Leuconostoc pseudomesenteroides, Weissella cibaria, Lp. plantarum, L. delbrueckii spp. bulgaricus, Leuconostoc citreum, Lp. paraplantarum and L. casei | Soaked, fermented for 2 days, dried | Dried ingredient used for soups and stocks | Turkey | [32] |
| Ting | Sorghum | Lm. reuteri, Lm. fermentum, L. harbinensis, Lp. plantarum, Ll. parabuchneri, Lc. casei and L. coryniformis. | Sorghum slurry fermented 1–3 days, cooked to soft porridge | Fermented sour porridge | Botswana | [33] |
| Togwa | Maize, Millet | Lp. plantarum, Lv. brevis, Lm. fermentum, W. confusa, P. pentosaceus | Germinated ground millet mixed with cooked maise porridge, fermented 15 h | Industrially produced opaque, sweet beverage | Tanzania | [34] |
| Uji | Maize, Sorghum, Millet | Leuc. mesenteroides, S. faecalis, Lp. plantarum, Lv. brevis, P. cerevisiae | Mixed flour slurry, fermented for 3 days then cooked | Fermented porridge | Kenya | [35] |
| Tubers | ||||||
| Fufu | Cassava | Lp. plantarum, Leu. mesenteroides, Lm. fermentum, Lv. brevis, L. coprophilus, Lcc. lactis, L. bulgaricus | Steeped, fermented 3–4 days, mashed, sieved, expelled water is consumed | Liquid drink | Ghana | [36] |
| Garri | Cassava | Lp. plantarum, Lm. fermentum, L. pentosus, L. acidophilus, Lc. casei, Lc. mesenteroides | Mashed roots, fermented then dehydrated, reconstituted in water before consumption | Gruel | Nigeria | [37] |
| Legumes | ||||||
| Siljo | Faba beans | L. acidophilus, Lp. plantarum, L. delbruekii, Micrococcus spp., Bacillus spp. | Slurry | Ethiopia | [38] | |
| Macronutrient | Effect * | Function and Reported Improvement in Bioactive Properties | Reference |
|---|---|---|---|
| Carbohydrate | Reduction in fermentable oligo-, di-, monosaccharides and polyols (FODMAPS) a,i,j,k | Reduced risk for irritable bowel disease (IBD) | [65,66,67] |
| Production of exopolysaccharides (EPS) b,e,c,g,h | Prebiotic effect, immunogenic, anti-inflammatory | [68,69,70] | |
| Reduction in raffinose family oligosaccharides (raffinose, verbascose, stachyose) a,b,c,d,e,k,j,i | Reduced risk of indigestion and flatulence | [66,71,72] | |
| Production of organic acids and acidification a,b,c,d,e,f,h,k,j | Flavour, food safety | [66,68,70,72,73,74,75,76] | |
| Improvement of glycaemic index by the reduction of soluble carbohydrates a,e,k,j | Reduced blood glucose spikes during digestion | [66,77] | |
| Generation of short-chain fatty acids from fibre fermentation a,i | Regulative effect on gut microbiota, reducing the risk for dysbiosis and associated disease | [78,79] | |
| Protein | Improvement of in vitro protein digestibility a,b,c,d,e,k,h,j | Improved availability of essential amino acids | [66,68,70,72,80,81,82,83] |
| Release of bioactive peptides a,c,d,f,i | Antihypertensive, antithrombotic, antimicrobial, angiotensin-converting enzyme (ACE)-inhibitory activity, dipeptidyl peptidase-IV (DPP-IV)-inhibitory, hypolipidemic | [83,84,85,86,87,88] | |
| Conversion of glutamate to γ-amino butyric acid (GABA) b,c,d,h,i,j,e | Antidiabetic via the induction of hypotension; relaxation effect can alleviate anxiety | [70,72,83,89,90] | |
| Lipid | Conversion of linoleic acid to conjugated linoleic acid i | Antioxidant, anti-cancer, prevention of atherosclerosis, reduction of body fat | [89,91,92,93] |
| Modification of polar lipid fraction a | Anti-inflammatory, anti-platelet properties | [94] | |
| Secondary metabolites | Degradation of anti-nutritional factors (tannins, trypsin inhibitors, phytic acid) a,b,g,h,k,i,j | Antioxidant | [66,69,72,72,81,95,96] |
| Generation of bacteriocins b,h,m,n | Food safety | [47,97,98] | |
| Increase in polyphenol availability and bioactive properties (through hydrolysis, production, or modification) a,e,f,i,h,l | Antioxidant | [75,81,91,92,99,100] | |
| Generation of vitamins (riboflavin and folate) f,i | [89,101] |
| Year | Substrate | Fermentation Conditions | Culture Type | Organism(s) | Topic of Study | Reference |
|---|---|---|---|---|---|---|
| Legumes | ||||||
| 2022 | Mung bean protein isolate, quinoa, coconut oil (3% w/v) | 43 °C, 8 h | Co- | L. bulgaricus, S. thermophilus, Kefir microflora, B. lactis, B. longum, and B. infantis | Effects of pre-treatments on protein gelation | [138] |
| 2022 | Soy protein isolate (5% w/w), coconut oil (4% w/w), pectin microgel particle (1% w/w) | 43 °C, 8 h | Co- | L. delbrueckii subsp. bulgaricus and S. thermophilus | Effects of added fats and fibre on protein gelation | [139] |
| 2021 | Faba bean flour, (10.37% w/v) or chickpea flour (9.86% w/v) | 43 °C, 10 h | Co- | S. thermophilus, L. delbrueckii subsp. bulgaricus, Lc. casei, Lc. lactis subsp. cremoris, Lc. lactis subsp. lactis, Le. species, Lc. lactis subsp. lactis biovar. Diacetylactis | Sensory and nutritional assessment | [140] |
| 2021 | Lentil protein isolate (4.35% w/w), sucrose (0.75% w/v), sunflower oil (1.5% w/v) | 42 °C, 6 h | Co- | Commercial culture (Yoflex® Acidifix™, Chr. Hansen, Denmark) | Texture and functionality compared to soy and dairy yoghurt | [141] |
| 2021 | Pea, mung bean protein isolate (3% w/v), sucrose (5% w/v), sunflower oil (3% v/v) | 37 °C, 120–200 min | Co- | S. thermophilus, L. delbrueckii subsp. bulgaricus, Lp. plantarum, L. acidophilus (NCFM®), and Bifidobacterium lactis (HN019™) | Comparative texture analysis | [142] |
| 2020 | Rice (10% w/w), lentil (5% w/w), and chickpea (5% w/w) | 30 °C, 16 h | Mono- | Lp. plantarum DSM33326, Lv. brevis DSM33325 | Nutritional analysis and probiotic stability | [66] |
| 2020 | Faba bean flour or faba bean isolate, rapeseed oil (3% w/w) | 37 °C, 6 h | Co- | Commercial culture (ABT-1, Chr. Hansen, Denmark) | Effects of amylase pre-treatment and fractionation on texture | [143] |
| 2019 | Pea protein (10% w/w), rapeseed oil (4%), oat fibre (3%) | 43 °C, 18 h | Co- | L. delbrueckii subsp. bulgaricus, S. thermophilus | Mechanism of pea protein gelation | [144] |
| 2018 | Chickpea flour (4.80% total solids), | 42 °C, 16 h | Co- | S. thermophilus, L. delbrueckii subsp. bulgaricus, L. acidophilus | Sensory comparison to dairy yoghurt | [76] |
| 2016 | Lupin protein isolate (5% w/v), glucose (2% w/v), coconut oil (4% v/v) | 30 °C, 14–35 h | Mono- | Lp. plantarum TMW 1.460 a. 1.468, P. pentosaceus BGT B34, Lv. brevis BGT L150 (at 8 log10) | Texture promotion by EPS, effects of heat treatment | [145] |
| cereals | ||||||
| 2023 | Oats (33% w/w) | 42 °C, 6 h | Co- | S. thermophilus, L. delbrueckii ssp. bulgaricus, | Volatile analysis and comparison to dairy yoghurt | [146] |
| 2023 | Oats, almonds | 30/37 °C, 48 h | Co- | L. delbrueckii ssp. bulgaricus, Lp. plantarum ATCC 8014, Lp. plantarum PK 1.1. | Volatile analysis | [147] |
| 2022 | Rice flour, coconut, guar gum (0.05% w/v), date palm syrup (10% w/v) | 43 °C, 8–12 h | Co- | S. thermophilus, L. delbrueckii subsp. bulgaricus | Nutritional analysis and comparison to yoghurt | [74] |
| 2022 | Sorghum, finger millet | 30 °C, 8 h | Co- | Lp. plantarum 299 v, W. confusa/cibaria C2 | Effects of novel co-culture on nutritional benefits | [70] |
| 2022 | Quinoa flakes, soybean at differing ratios (14.5% w/w) | 42 °C, 12 h | Co- | L. delbrueckii subsp. bulgaricus and S. thermophilus | Optimised formulation based on sensory acceptance and nutritional value | [148] |
| 2022 | One of cooked rice (20% w/w), shredded coconut (20% w/w), almond (10% w/w) | 37 °C, 24 h | Co- | L. delbrueckii subsp. bulgaricus and Streptococcus thermophilus | Bioactive analysis, anti-inflammatory and anti-platelet properties of lipid fractions after fermentation | [94] |
| 2020 | Oat flour (12% w/w), aquafaba (3% w/v), coconut oil (5% w/v), lactose (5% w/v) | 43 °C, 6 h | Co- | B. lactis, L. acidophilus, L. delbrueckii subsp. bulgaricus, L. delbrueckii subsp. lactis, and S. thermophilus | Use of aquafaba as gelling agent | [149] |
| 2019 | Oat protein isolate (87% purity, 15% w/v), lactose (10% w/v) | 45 °C, 24 h | Co- | L. delbrueckii subsp. bulgaricus, S. thermophilus | Use of oat isolates as ingredient | [150] |
| 2018 | Quinoa flour (15% w/w), sucrose (10% w/v) | 30 °C, 24 h | Mono- | Weissella cibaria MG1 | Texture promotion by EPS, effects of proteolysis on protein digestibility | [68] |
| 2015 | Oat, barley, malt flour (15% w/w) | 37 °C, 10 h | Co- | L. acidophilus NCIMB 8821, Lp. plantarum NCIMB 8826, and Lm. reuteri NCIMB 11951 | Volatile analysis and acceptance based on used starter culture | [151] |
| 2014 | Oat flakes (25% w/w) | 30 °C, 12 h | Mono- | Lp. plantarum LP09 | Sensory analysis and optimised formulation | [75] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Horlacher, N.; Oey, I.; Agyei, D. Learning from Tradition: Health-Promoting Potential of Traditional Lactic Acid Fermentation to Drive Innovation in Fermented Plant-Based Dairy Alternatives. Fermentation 2023, 9, 452. https://doi.org/10.3390/fermentation9050452
Horlacher N, Oey I, Agyei D. Learning from Tradition: Health-Promoting Potential of Traditional Lactic Acid Fermentation to Drive Innovation in Fermented Plant-Based Dairy Alternatives. Fermentation. 2023; 9(5):452. https://doi.org/10.3390/fermentation9050452
Chicago/Turabian StyleHorlacher, Nicholas, Indrawati Oey, and Dominic Agyei. 2023. "Learning from Tradition: Health-Promoting Potential of Traditional Lactic Acid Fermentation to Drive Innovation in Fermented Plant-Based Dairy Alternatives" Fermentation 9, no. 5: 452. https://doi.org/10.3390/fermentation9050452
APA StyleHorlacher, N., Oey, I., & Agyei, D. (2023). Learning from Tradition: Health-Promoting Potential of Traditional Lactic Acid Fermentation to Drive Innovation in Fermented Plant-Based Dairy Alternatives. Fermentation, 9(5), 452. https://doi.org/10.3390/fermentation9050452





