Dose-Response of Fruit Oligosaccharides on Rumen Fermentation Parameters, CH4 Emission and Skatole Content In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Experimental Diet
2.2. In Vitro Rumen Fermentation
2.3. Sampling
2.4. Chemical Analysis
2.5. Data Processing and Analysis
3. Results
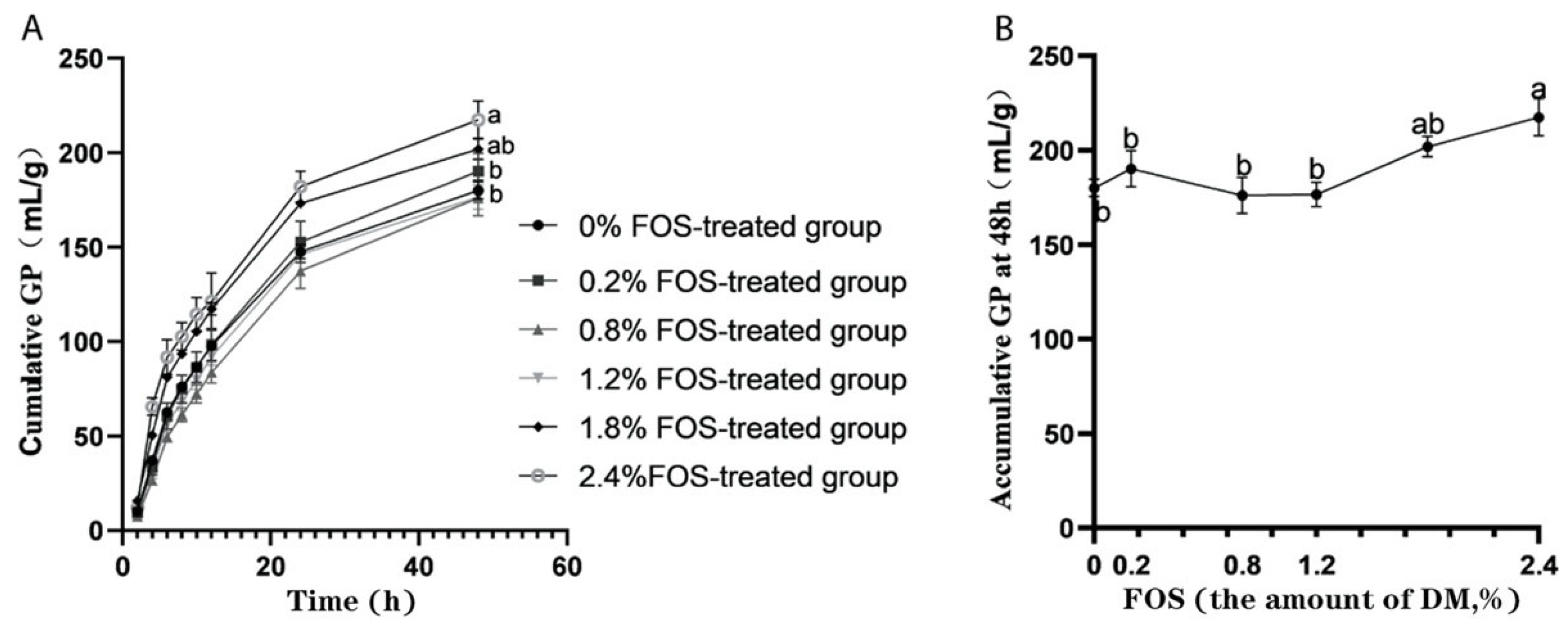
3.1. Effects of FOS Addition on GP In Vitro Rumen Fermentation
3.2. Effects of FOS Addition on CH4 Production In Vitro Rumen Fermentation
3.3. Effects of FOS Addition on Nutrient Degradation Rates of Rumen Fermentation
3.4. Effects of FOS Addition on the pH, and Contents of NH3-N and MCP In Vitro Rumen Fermentation
3.5. Effects of FOS Addition on the Contents of Skatole In Vitro Rumen Fermentation
3.6. Effects of FOS Addition on the Contents of VFA In Vitro Rumen Fermentation
4. Discussion
4.1. Effects of FOS Addition on GP and CH4 Production In Vitro Rumen Fermentation
4.2. Effects of FOS Addition on Nutrient Degradation Rates of Rumen Fermentation
4.3. Effects of FOS Addition on the pH, Contents of NH3-N and MCP In Vitro Rumen Fermentation
4.4. Effects of FOS Addition on the Contents of Skatole In Vitro Rumen Fermentation
4.5. Effects of FOS Addition on the Contents of VFA In Vitro Rumen Fermentation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhong, R.; Xiang, H.; Cheng, L.; Zhao, C.; Wang, F.; Zhao, X.; Fang, Y. Effects of Feeding Garlic Powder on Growth Performance, Rumen Fermentation, and the Health Status of Lambs Infected by Gastrointestinal Nematodes. Animals 2019, 9, 102. [Google Scholar] [CrossRef] [PubMed]
- Sakita, G.Z.; Lima, P.; Filho, A.; Bompadre, T.; Ovani, V.; Chaves, C.; Bizzuti, B.; Costa, W.; Paim, P.; Campioni, T. Treating tropical grass with fibrolytic enzymes from the fungus Trichoderma reesei: Effects on animal performance, digestibility and enteric methane emissions of growing lambs. Anim. Feed. Sci. Technol. 2022, 286, 115–253. [Google Scholar] [CrossRef]
- Matthews, C.; Crispie, F.; Lewis, E.; Reid, M.; O’Toole, P.W.; Cotter, P.D. The rumen microbiome: A crucial consideration when optimising milk and meat production and nitrogen utilisation efficiency. Gut Microbes 2019, 10, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Mi, J.; Peng, H.; Wu, Y.; Wang, Y.; Liao, X. Diversity and community of methanogens in the large intestine of finishing pigs. BMC Microbiol. 2019, 19, 83. [Google Scholar] [CrossRef]
- Hanum, F.; Atsuta, Y.; Daimon, H. Methane Production Characteristics of an Anaerobic Co-Digestion of Pig Manure and Fermented Liquid Feed. Molecules 2022, 27, 6509. [Google Scholar] [CrossRef]
- Zhang, X.M.; Wang, R.; Ma, Z.Y.; Wang, M.; Tan, Z.L. Methane emissions from ruminant gastrointestinal tract and mitigation strategies. J Integr Agric. 2020, 39, 732–742. [Google Scholar]
- Woodward, S.L.; Waghorn, G.C.; Lassey, K.R.; Laboyrie, P.G. Does feeding sulla (Hedysarum coronarium) reduce methane emissions from dairy cows? Proc. N. Z. Soc. Anim. Prod. 2002, 62, 227–230. [Google Scholar]
- Dias-Moreira, G.; Lima, P.M.T.; Borges, B.O.; Primavesi, O.; Longo, C.; McManus, C.; Abdalla, A.; Louvandini, H. Tropical tanniniferous legumes used as an option to mitigate sheep enteric methane emission. Trop. Anim. Health Prod. 2013, 45, 879–882. [Google Scholar] [CrossRef]
- Oshita, K.; Sun, X.; Taniguchi, M.; Takaoka, M.; Matsukawa, K.; Fujiwara, T. Emission of greenhouse gases from controlled incineration of cattle manure. Environ. Technol. 2012, 33, 1539–1544. [Google Scholar] [CrossRef]
- Popp, J.D.; McAllister, T.A.; Kastelic, J.P.; Majak, W.; Ayroud, M.; VanderKop, M.A.; Karren, D.; Yost, G.S.; Cheng, K.J. Effect of melengestrol acetate on development of 3-methylindole-induced pulmonary edema and emphysema in sheep. Can. J. Vet. Res. 1998, 62, 268–274. [Google Scholar]
- Watkins, P.J.; Kearney, G.; Rose, G.; Allen, D.; Ball, A.J.; Pethick, D.W.; Warner, R.D. Effect of branched-chain fatty acids, 3-methylindole and 4-methylphenol on consumer sensory scores of grilled lamb meat. Meat Sci. 2014, 96, 1088–1094. [Google Scholar] [CrossRef]
- Sharma, N.; Doerner, K.C.; Alok, P.C.; Choudhary, M. Skatole remediation potential of Rhodopseudomonas palustris WKU-KDNS3 isolated from an animal waste lagoon. Lett. Appl. Microbiol. 2015, 60, 298–306. [Google Scholar] [CrossRef]
- Jovanovic-Malinovska, R.; Kuzmanova, S.; Winkelhausen, E. Application of ultrasound for enhanced extraction of prebiotic oligosaccharides from selected fruits and vegetables. Ultrason. Sonochem. 2015, 22, 446–453. [Google Scholar] [CrossRef]
- Lordan, C.; Thapa, D.; Ross, R.P.; Cotter, P.D. Potential for enriching next-generation health-promoting gut bacteria through prebiotics and other dietary components. Gut Microbes 2020, 11, 1–20. [Google Scholar] [CrossRef]
- Bruzzese, E.; Volpicelli, M.; Squeglia, V.; Bruzzese, D.; Salvini, F.; Bisceglia, M.; Lionetti, P.; Cinquetti, M.; Iacono, G.; Amarri, S.; et al. A formula containing galacto- and fructo-oligosaccharides prevents intestinal and extra-intestinal infections: An observational study. Clin. Nutr. 2009, 28, 156–161. [Google Scholar] [CrossRef]
- Takahashi, J.; Iwasa, M. Entomological approach to the impact of ionophore-feed additives on greenhouse gas emissions from pasture land in cattle. J. Anim. Sci. Technol. 2021, 63, 16–24. [Google Scholar] [CrossRef]
- Fan, R.; Burghardt, J.P.; Huang, J.; Xiong, T.; Czermak, P. Purification of Crude Fructo-Oligosaccharide Preparations Using Probiotic Bacteria for the Selective Fermentation of Monosaccharide Byproducts. Front. Microbiol. 2021, 11, 620626. [Google Scholar] [CrossRef]
- Béghin, L.; Tims, S.; Roelofs, M.; Rougé, C.; Oozeer, R.; Rakza, T.; Chirico, G.; Roeselers, G.; Knol, J.; Rozé, J.C.; et al. Fermented infant formula (with Bifidobacterium breve C50 and Streptococcus thermophilus O65) with prebiotic oligosaccharides is safe and modulates the gut microbiota towards a microbiota closer to that of breastfed infants. Clin. Nutr. 2021, 40, 778–787. [Google Scholar] [CrossRef]
- Li, Z.; Bai, H.; Zheng, L.; Jiang, H.; Cui, H.; Cao, Y.; Yao, J. Bioactive polysaccharides and oligosaccharides as possible feed additives to manipulate rumen fermentation in Rusitec fermenters. Int. J. Biol. Macromol. 2018, 109, 1088–1094. [Google Scholar] [CrossRef]
- Fabiano, V.; Indrio, F.; Verduci, E.; Calcaterra, V.; Pop, T.L.; Mari, A.; Zuccotti, G.V.; Cullu Cokugras, F.; Pettoello-Mantovani, M.; Goulet, O. Term Infant Formulas Influencing Gut Microbiota: An Overview. Nutrients 2021, 13, 4200. [Google Scholar] [CrossRef]
- Rogier, R.; Ederveen, T.H.A.; Wopereis, H.; Hartog, A.; Boekhorst, J.; van Hijum, S.A.F.T.; Knol, J.; Garssen, J.; Walgreen, B.; Helsen, M.M.; et al. Supplementation of diet with non-digestible oligosaccharides alters the intestinal microbiota, but not arthritis development, in IL-1 receptor antagonist deficient mice. PLoS ONE 2019, 14, e0219366. [Google Scholar]
- Zhang, F.; Wang, Y.; Wang, H.; Nan, X.; Guo, Y.; Xiong, B. Calcium Propionate Supplementation Has Minor Effects on Major Ruminal Bacterial Community Composition of Early Lactation Dairy Cows. Front. Microbiol. 2022, 13, 847488. [Google Scholar] [CrossRef] [PubMed]
- Spiro, M.D.; Bowers, J.F.; Cosgrove, D.J. A comparison of oligogalacturonide- and auxin-induced extracellular alkalinization and growth responses in roots of intact cucumber seedlings. Plant Physiol. 2002, 130, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Menke, K.; Raab, L.; Salewski, A. The estimation of the digestibility and metabolizable energy content of ruminant feedingstuffs from the gas production when they are incubated with rumen liquor in vitro. J. Agric. Sci. 1979, 93, 217–222. [Google Scholar] [CrossRef]
- Li, J.; Yan, H.; Chen, J.; Duan, C.; Guo, Y.; Liu, Y.; Zhang, Y.; Ji, S. Correlation of Ruminal Fermentation Parameters and Rumen Bacterial Community by Comparing Those of the Goat, Sheep, and Cow In Vitro. Fermentation 2022, 8, 427. [Google Scholar] [CrossRef]
- Benetel, G.; Silva, T.D.S.; Fagundes, G.M.; Welter, K.C.; Melo, F.A.; Lobo, A.A.G.; Muir, J.P.; Bueno, I.C.S. Essential Oils as In Vitro Ruminal Fermentation Manipulators to Mitigate CH4 Emission by Beef Cattle Grazing Tropical Grasses. Molecules 2022, 27, 2227. [Google Scholar] [CrossRef]
- Lueangwattanapong, K.; Ammam, F.; Mason, P.M.; Whitehead, C.; McQueen-Mason, S.J.; Gomez, L.D.; Smith, J.A.C.; Thompson, I.P. Anaerobic digestion of Crassulacean Acid Metabolism plants: Exploring alternative feedstocks for semi-arid lands. Bioresour. Technol. 2020, 297, 122262. [Google Scholar] [CrossRef]
- Hansen-Møller, J. Rapid high-performance liquid chromatographic method for simultaneous determination of androstenone, Skatole and indole in back fat from pigs. J. Chromatogr. B Biomed. Appl. 1994, 661, 219–230. [Google Scholar] [CrossRef]
- Wang, M.; Tang, S.; Tan, Z. Modeling in vitro gas production kinetics: Derivation of Logistic–Exponential (LE) equations and comparison of models. Anim. Feed Sci. Technol. 2011, 165, 137–150. [Google Scholar] [CrossRef]
- Winichayakul, S.; Beechey-Gradwell, Z.; Muetzel, S.; Molano, G.; Crowther, T.; Lewis, S.; Xue, H.; Burke, J.; Bryan, G.; Roberts, N.J. In vitro gas production and rumen fermentation profile of fresh and ensiled genetically modified high-metabolizable energy ryegrass. J. Dairy Sci. 2020, 103, 2405–2418. [Google Scholar] [CrossRef]
- Peng, J.; Yang, L.; Zhang, L. Application of Fructooligosaccharides in Ruminant production. China Feed 2020, 651, 5–8. [Google Scholar]
- Zheng, C.; Ma, J.; Liu, T.; Wei, B.; Yang, H. Effects of Mannan Oligosaccharides on Gas Emission, Protein and Energy Utilization, and Fasting Metabolism in Sheep. Animals 2019, 28, 741. [Google Scholar] [CrossRef]
- Johnson, J.R.; Carstens, G.E.; Krueger, W.K.; Lancaster, P.A.; Brown, E.G.; Tedeschi, L.O.; Anderson, R.C.; Johnson, K.A.; Brosh, A. Associations between residual feed intake and apparent nutrient digestibility, in vitro CH4-producing activity, and volatile fatty acid concentrations in growing beef cattle1. J. Anim. Sci. 2019, 97, 3550–3561. [Google Scholar] [CrossRef] [PubMed]
- Van Niekerk, J.K.; Fischer-Tlustos, A.J.; Deikun, L.L.; Quigley, J.D.; Dennis, T.S.; Suarez-Mena, F.X.; Hill, T.M.; Schlotterbeck, R.L.; Guan, L.L.; Steele, M.A. Effect of amount of milk replacer fed and the processing of corn in starter on growth performance, nutrient digestibility, and rumen and fecal fibrolytic bacteria of dairy calves. J. Dairy Sci. 2020, 103, 2186–2199. [Google Scholar] [CrossRef]
- Zheng, C.; Zhou, J.; Zeng, Y.; Liu, T. Effects of mannan oligosaccharides on growth performance, nutrient digestibility, ruminal fermentation and hematological parameters in sheep. PeerJ 2021, 9, e11631. [Google Scholar] [CrossRef]
- Zheng, C.; Li, F.; Hao, Z.; Liu, T. Effects of adding mannan oligosaccharides on digestibility and metabolism of nutrients, ruminal fermentation parameters, immunity, and antioxidant capacity of sheep. J. Anim. Sci. 2018, 96, 284–292. [Google Scholar] [CrossRef]
- Kim, H.; Jung, E.; Lee, H.G.; Kim, B.; Cho, S.; Lee, S.; Kwon, I.; Seo, J. Essential oil mixture on rumen fermentation and microbial community—An in vitro study. Asian Australas. J. Anim. Sci. 2019, 32, 808–814. [Google Scholar] [CrossRef]
- Garcia Diaz, T.; Ferriani Branco, A.; Jacovaci, F.A.; Cabreira Jobim, C.; Bolson, D.C.; Pratti Daniel, J.L. Inclusion of live yeast and mannan-oligosaccharides in high grain-based diets for sheep: Ruminal parameters, inflammatory response and rumen morphology. PLoS ONE 2018, 13, e0193313. [Google Scholar] [CrossRef]
- Sun, X.; Cheng, L.; Jonker, A.; Munidasa, S.; Pacheco, D. A Review: Plant Carbohydrate Types-The Potential Impact on Ruminant CH4 Emissions. Front. Vet. Sci. 2022, 9, 880115. [Google Scholar] [CrossRef]
- Wang, B.; Jia, M.; Fang, L.; Jiang, L.; Li, Y. Effects of eucalyptus oil and anise oil supplementation on rumen fermentation characteristics, CH4 emission, and digestibility in sheep. J. Anim. Sci. 2018, 96, 3460–3470. [Google Scholar]
- Brunette, T.; Baurhoo, B.; Mustafa, A.F. Effects of replacing grass silage with forage pearl millet silage on milk yield, nutrient digestion, and ruminal fermentation of lactating dairy cows. J. Dairy Sci. 2016, 99, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Rabee, A.E.; Kewan, K.Z.; Sabra, E.A.; El Shaer, H.M.; Lamara, M. Rumen bacterial community profile and fermentation in Barki sheep fed olive cake and date palm byproducts. PeerJ 2021, 9, e12447. [Google Scholar] [CrossRef] [PubMed]
- Yulistiani, D.; Jelan, Z.A.; Liang, J.B.; Yaakub, H.; Abdullah, N. Effects of Supplementation of Mulberry (Morus alba) Foliage and Urea-rice Bran as Fermentable Energy and Protein Sources in Sheep Fed Urea-treated Rice Straw Based Diet. Asian-Australas. J. Anim. Sci. 2015, 28, 494–501. [Google Scholar] [CrossRef]
- Mapato, C.; Wanapat, M. New roughage source of Pennisetum purpureum cv. Mahasarakham utilization for ruminants feeding under global climate change. Asian-Australas. J. Anim. Sci. 2018, 31, 1890–1896. [Google Scholar] [CrossRef]
- Zhou, J.; Ding, Z.; Pu, Q.; Xue, B.; Yue, S.; Guan, S.; Wang, Z.; Wang, L.; Peng, Q.; Xue, B. Rumen Fermentation and Microbiome Responses to Enzymatic Hydrolysate of Cottonseed Protein Supplementation in Continuous In Vitro Culture. Animals 2022, 12, 2113. [Google Scholar] [CrossRef]
- Li, G.H.; Ling, B.M.; Qu, M.R. Effects of several oligosaccha-rides on ruminal fermentation in sheep: An in vitro experiment. Rev. Médecine Vétérinaire 2011, 162, 192–197. [Google Scholar]
- Gasiorek, M.; Stefanska, B.; Pruszynska-Oszmalek, E.; Taciak, M.; Komisarek, J.; Nowak, W. Effect of oat hay provision method on growth performance, rumen fermentation and blood metabolites of dairy calves during preweaning and postweaning periods. Animal 2020, 14, 2054–2062. [Google Scholar] [CrossRef]
- Chang, M.; Wang, F.; Ma, F.; Jin, Y.; Sun, P. Supplementation with galacto-oligosaccharides in early life persistently facilitates the microbial colonization of the rumen and promotes growth of preweaning Holstein dairy calves. Anim. Nutr. 2022, 10, 223–233. [Google Scholar] [CrossRef]
- Gao, J.; Xu, K.; Liu, H.; Liu, G.; Bai, M.; Peng, C.; Li, T.; Yin, Y. Impact of the Gut Microbiota on Intestinal Immunity Mediated by Tryptophan Metabolism. Front. Cell. Infect. Microbiol. 2018, 8, 13. [Google Scholar] [CrossRef]
- Shi, J.; Zhao, D.; Zhao, F.; Wang, C.; Zamaratskaia, G.; Li, C. Chicken-eaters and pork-eaters have different gut microbiota and tryptophan metabolites. Sci. Rep. 2021, 11, 11934. [Google Scholar] [CrossRef]
- Liu, H.Y.; Li, X.; Zhu, X.; Dong, W.G.; Yang, G.Q. Soybean oligosaccharides attenuate odour compounds in excreta by modulating the caecal microbiota in broilers. Animal 2021, 15, 100159. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.Q.; Yin, Y.; Liu, H.Y.; Liu, G.H. Effects of dietary oligosaccharide supplementation on growth performance, concentrations of the major odor-causing compounds in excreta, and the cecal microflora of broilers. Poult. Sci. 2016, 95, 2342–2351. [Google Scholar] [CrossRef]
- Xu, Z.R.; Hu, C.H.; Wang, M.Q. Effects of fructooligosaccharide on conversion of L-tryptophan to Skatole and indole by mixed populations of pig fecal bacteria. J. Gen. Appl. Microbiol. 2002, 48, 83–90. [Google Scholar] [CrossRef]
- Parnell, J.A.; Reimer, R.A. Prebiotic fibres dose-dependently increase satiety hormones and alter Bacteroidetes and Firmicutes in lean and obese JCR:LA-cp rats. Br. J. Nutr. 2012, 107, 601–613. [Google Scholar] [CrossRef]
- Grossi, S.; Dell’Anno, M.; Rossi, L.; Compiani, R.; Sgoifo Rossi, C.A. Supplementation of Live Yeast, Mannan Oligosaccharide, and Organic Selenium during the Adaptation Phase of Newly Arrived Beef Cattle: Effects on Health Status, Immune Functionality, and Growth Performance. Antibiotics 2021, 10, 1114. [Google Scholar] [CrossRef]


| Component | Contents |
|---|---|
| Ingredients, g/kg of DM | |
| Peanut seedling | 344 |
| Corn | 508 |
| Soybean meal (CP:44%) | 138 |
| Premix compound | 10 |
| Total | 1000 |
| Nutritive composition | |
| Metabolic energy, MJ/kg | 11.00 |
| CP, % | 12.29 |
| NDF, % | 34.63 |
| ADF, % | 20.56 |
| Ca, % | 0.68 |
| P, % | 0.28 |
| Items | FOS (mg/g DM) | SEM | p Value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.2 | 0.8 | 1.2 | 1.8 | 2.4 | Trt | L | Q | ||
| Lag time, h | 0.05 d | 0.18 c | 0.30 a | 0.25 b | −0.06 e | −0.38 f | 0.10 | <0.01 | 0.08 | 0.02 |
| Potential total GP, mL | 189.70 b | 204.50 ab | 197.50 b | 190.90 b | 208.31 ab | 221.64 a | 6.48 | <0.01 | 0.25 | 0.42 |
| GP rate, (%/h) | 0.06 ab | 0.06 ab | 0.05 b | 0.06 ab | 0.07 a | 0.07 a | 0.01 | <0.01 | 0.22 | 0.23 |
| Items | FOS (mg/g DM) | SEM | p Value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.2 | 0.8 | 1.2 | 1.8 | 2.4 | Trt | L | Q | ||
| Lag time, h | 0.57 a | 0.29 b | 0.27 b | 0.60 a | 0.17 c | 0.18 c | 0.08 | <0.01 | 0.64 | 0.44 |
| Potential CH4 production, mL | 66.32 a | 57.17 b | 38.20 d | 31.34 f | 34.82 e | 42.80 c | 5.61 | <0.01 | 0.39 | 0.02 |
| CH4 production rate, (%/h) | 0.07 ab | 0.06 bc | 0.06 bc | 0.05 c | 0.08 a | 0.08 a | 0.01 | <0.01 | 0.18 | 0.34 |
| Items | FOS (mg/g DM) | SEM | p Value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.2 | 0.8 | 1.2 | 1.8 | 2.4 | Trt | L | Q | ||
| DM | 60.3 | 62.11 | 60.67 | 61.41 | 62.99 | 63.10 | 1.26 | 0.32 | <0.01 | 0.70 |
| GE | 54.19 | 57.98 | 51.92 | 59.88 | 61.92 | 58.67 | 0.79 | 0.82 | <0.01 | 0.95 |
| CP | 61.15 b | 59.79 b | 60.70 b | 67.64 a | 71.14 a | 69.58 a | 0.65 | <0.01 | <0.01 | 0.39 |
| NDF | 41.73 b | 38.42 b | 36.33 b | 47.63 a | 49.08 a | 47.83 a | 1.46 | <0.01 | <0.01 | 0.58 |
| ADF | 28.88 b | 26.76 c | 22.10 d | 32.87 a | 35.43 a | 35.03 a | 1.01 | <0.01 | <0.01 | 0.90 |
| Items | FOS (mg/g DM) | SEM | p Value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.2 | 0.8 | 1.2 | 1.8 | 2.4 | Trt | L | Q | ||
| pH | 5.78 | 5.82 | 5.93 | 5.95 | 6.03 | 6.13 | 1.26 | 0.47 | <0.01 | 0.64 |
| NH3-N, mg/dL | 18.83 | 18.76 | 18.84 | 18.67 | 18.66 | 18.60 | 0.87 | 0.82 | <0.01 | 0.80 |
| MCP, μg/mL | 56.35 d | 62.93 c | 64.64 bc | 63.57 c | 70.92 ab | 74.73 a | 1.01 | <0.01 | <0.01 | 0.33 |
| Items | FOS (mg/g DM) | SEM | p Value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.2 | 0.8 | 1.2 | 1.8 | 2.4 | Trt | L | Q | ||
| Acetate, mmol/L | 21.42 b | 20.38 b | 22.09 ab | 23.60 a | 23.61 a | 23.95 a | 0.34 | 0.01 | 0.02 | 0.10 |
| Propionate, mmol/L | 12.98 bc | 12.13 c | 14.02 b | 15.67 a | 15.68 a | 16.05 a | 0.45 | 0.03 | <0.01 | 0.07 |
| Butyrate, mmol/L | 9.95 c | 8.61 d | 11.59 b | 14.20 a | 14.21 a | 14.79 a | 0.50 | <0.01 | 0.01 | 0.07 |
| Valeric acid, mmol/L | 2.07 cd | 1.97 d | 2.19 b | 2.38 ab | 2.39 ab | 2.43 a | 0.04 | 0.01 | 0.01 | 0.07 |
| Total VFA, mmol/L | 46.43 cd | 43.09 d | 49.89 bc | 55.86 ab | 55.89 ab | 57.22 a | 1.25 | <0.01 | 0.01 | 0.08 |
| Acetate/Propionate | 1.65 ab | 1.68 a | 1.58 abc | 1.51 bc | 1.51 bc | 1.49 c | 0.03 | 0.03 | 0.02 | 0.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Ji, S.; Yan, H.; Li, J.; Zhang, L.; Yan, D.; Duan, C.; Liu, Y.; Zhang, Y. Dose-Response of Fruit Oligosaccharides on Rumen Fermentation Parameters, CH4 Emission and Skatole Content In Vitro. Fermentation 2023, 9, 428. https://doi.org/10.3390/fermentation9050428
Wang L, Ji S, Yan H, Li J, Zhang L, Yan D, Duan C, Liu Y, Zhang Y. Dose-Response of Fruit Oligosaccharides on Rumen Fermentation Parameters, CH4 Emission and Skatole Content In Vitro. Fermentation. 2023; 9(5):428. https://doi.org/10.3390/fermentation9050428
Chicago/Turabian StyleWang, Liyan, Shoukun Ji, Hui Yan, Jinhui Li, Lishen Zhang, Dezhi Yan, Chunhui Duan, Yueqin Liu, and Yingjie Zhang. 2023. "Dose-Response of Fruit Oligosaccharides on Rumen Fermentation Parameters, CH4 Emission and Skatole Content In Vitro" Fermentation 9, no. 5: 428. https://doi.org/10.3390/fermentation9050428
APA StyleWang, L., Ji, S., Yan, H., Li, J., Zhang, L., Yan, D., Duan, C., Liu, Y., & Zhang, Y. (2023). Dose-Response of Fruit Oligosaccharides on Rumen Fermentation Parameters, CH4 Emission and Skatole Content In Vitro. Fermentation, 9(5), 428. https://doi.org/10.3390/fermentation9050428





