3.2.1. Fermented and Dried Beans
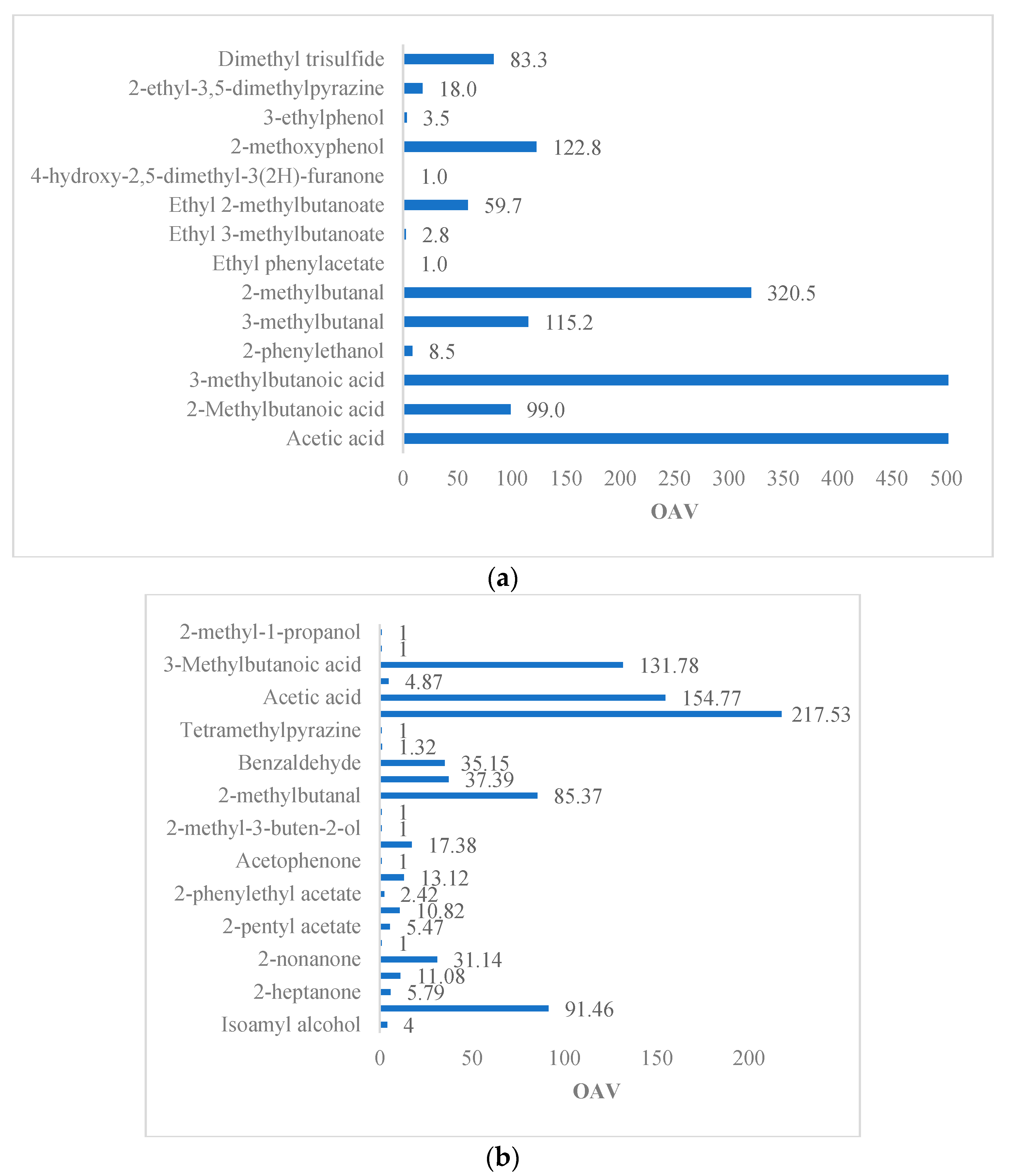
Table 3 indicates the contribution of the individual compounds to the overall aroma of the beans; their OAV > 1. 20 and 36% out of the compounds impart undesirable aromas. Acetic acid, 2-methylbutanoic acid, 3-methylbutanoic acid, and 2-methylpropanoic acid were common in both studies. They differ by approximately 55 times for acetic acid and 48 times for 3-methylbutanoic acid due to the fermentation method used (wood box vs. pile fermentation), days fermented (5 d vs. 3 d with prefermentation), and the origin of the beans (Costa Rica vs. Ecuador). These compounds are present in the over-fermented beans from leucine metabolism; such acids are undesirable in any cocoa matrix, imparting vinegar, pungent, sweaty, and rancid flavors [
5,
42]; it was reported in separate and complementary studies that box fermentation offers less aeration compared to the heap method. It was expected that the acetic acid concentration in the heap method would be higher, as aeration drives the emergence of acetic acid bacteria; however, the box method offers a higher concentration. The prefermentation of the beans helped to reduce their moisture and sugar content, thus decreasing the acid content at the end of fermentation. Prefermentation was absent in the box fermentation study. 2-methoxyphenol and 3-ethylphenol are also compounds that impart an unpleasant smoky flavor and odor and have been reported as a crucial marker of this aroma. In this study, these compounds are very significant, presenting concentrations of 221.00 and 7.66 µg/kg and an OAV of 122.8 and 3.5, respectively. Ref. [
13] proposed the maximum tolerable concentration based on the rounded threshold value of 2 µg/kg for 3-ethylphenol. Sometimes, the aroma imparted by 2-methoxyphenol goes unnoticed as it is masked by the more odorous compounds in cocoa. Ref. [
13] reported a concentration of 70 µg/kg for 2-methoxyphenol, and the reason for this depends on compound concentration and the authentic cocoa odors contributing to the pleasant aroma of cocoa masking the foul odor.
Table 3 shows the difference between the two studies on fruit aromas. The highest amount of these aromas is present in the study of [
5], who used beans of the Ecuadorian trinitario variety in their study. In contrast, Ref. [
42] proposed a single relevant fruit aroma, as is the case for ethyl phenylacetate. In both studies, the same variety (trinitario) was analyzed; however, there were large differences in the amount of its volatile compounds. The difference may be due to geographical origin, postharvest techniques, the chemical composition of the beans, and the method used for their quantification [
22]. Fruity volatile compounds, such as isoamyl acetate, 2-nonanone, 2-heptanol, 2-heptanone, 2-pentyl acetate, and ethyl, in descending order of their OAV, contribute to the overall aroma of the dried beans. These fruity aroma compounds are most relevant in the trinitario cocoa variety from Ecuador, known for aromatic cocoa. The esters synthesized during fermentation are dependent on environmental precursors, as well as on aeration and the presence of alcohol, such as ethanol. For example, the production of isoamyl acetate by yeasts (
Pichia fermentants) uses isoamyl alcohol as a precursor [
69]. According to
Table 2, this compound is also odor active (OAV = 4), and its concentration exceeds its OTV; therefore, it remains in the aromatic profile and serves as a precursor for other compounds. 2-pentyl acetate (OAV = 5.47) and ethyl acetate (OAV = 1) can act as the precursors of secondary alcohols, such as 2-pentanol and 2-heptanol [
70]. The latter contributes its relevant citrus aroma (OAV = 11.08) to Ecuadorian trinitario cocoa beans.
Floral aromas in both studies include, in descending order, linalool, 2-phenylethanol, 2-phenylacetaldehyde, 2-phenylethyl acetate, acetophenone, and 2-methyl-3-buten-2-ol. Ref. [
71] proposed linalool as a grade indicator (fine or basic grade) in some varieties. A linalool/benzaldehyde ratio of greater than 0.3 indicates fine-grade cocoa. Our review showed linalool and benzaldehyde OAVs of 17.38 and 35.15, respectively, with both compounds aromatically active at a 0.3 ratio. Ref. [
20] reported a 0.56-to-0.89 linalool/benzaldehyde ratio in roasted Criollo cocoa beans. As linalool is a biosynthesis product, its creation depends on plant varieties, growing, and fermentation conditions. During the roasting process, the linalool content decreases slightly due to volatility, but the relative difference between basic and fine-grade cocoa remains [
71].
Schluter et al. (2020) [
42] reported a high OAV with a prevalence of chocolate and malt flavors, likely related to the high concentrations of isoleucine, leucine, and phenylalanine during fermentation. They attributed such aromas to Strecker aldehydes, such as 2- and 3-methylbutanal (chocolate, malt flavor) and phenylacetaldehyde (honey flavor), respectively. On the contrary, 2-phenylethyl acetate, trimethylpyrazine, ethyl acetate, acetophenone, 2-methyl-3-buten-2-ol, 2-octanol, tetramethylpyrazine, ethanol, and 2-methyl-1-propanol show a low OAV, which, individually, would not contribute to their respective aromas, but, as a whole, would present a range of odors from wine to floral and honey.
Figure 2 illustrates the key aroma profile in both cocoa samples. The aroma profile of the trinitario Sacha Gold variety (
Figure 2b) is broader and has a distinct fine diversity of aromas representing processed chocolate.
Figure 2a, on the other hand, presents a limited aroma profile that is not appreciated by the chocolate industry. In both studies, a total of four pyrazines are observed, including 2-ethyl-3,5 and 3,6-dimethylpyrazine, trimethylpyrazine, and tetramethylpyrazine, for which the OAVs are less than 18. During fermentation, both trimethyl and tetramethylpyrazines appeared; however, they represent only 0.6% of the total volatiles [
72]. During grain roasting, the number and concentration of pyrazines notably rose: 2,5-dimethylpyrazine (DMP), 2,6-DMP, 2-ethylpyrazine, 2,3-DMP, 2,3,4-trimethylpyrazine (TrMP) and 2,3,5,6-tetramethylpyrazine (TMP) [
62], evidenced in the following section.
3.2.2. Roasted Beans
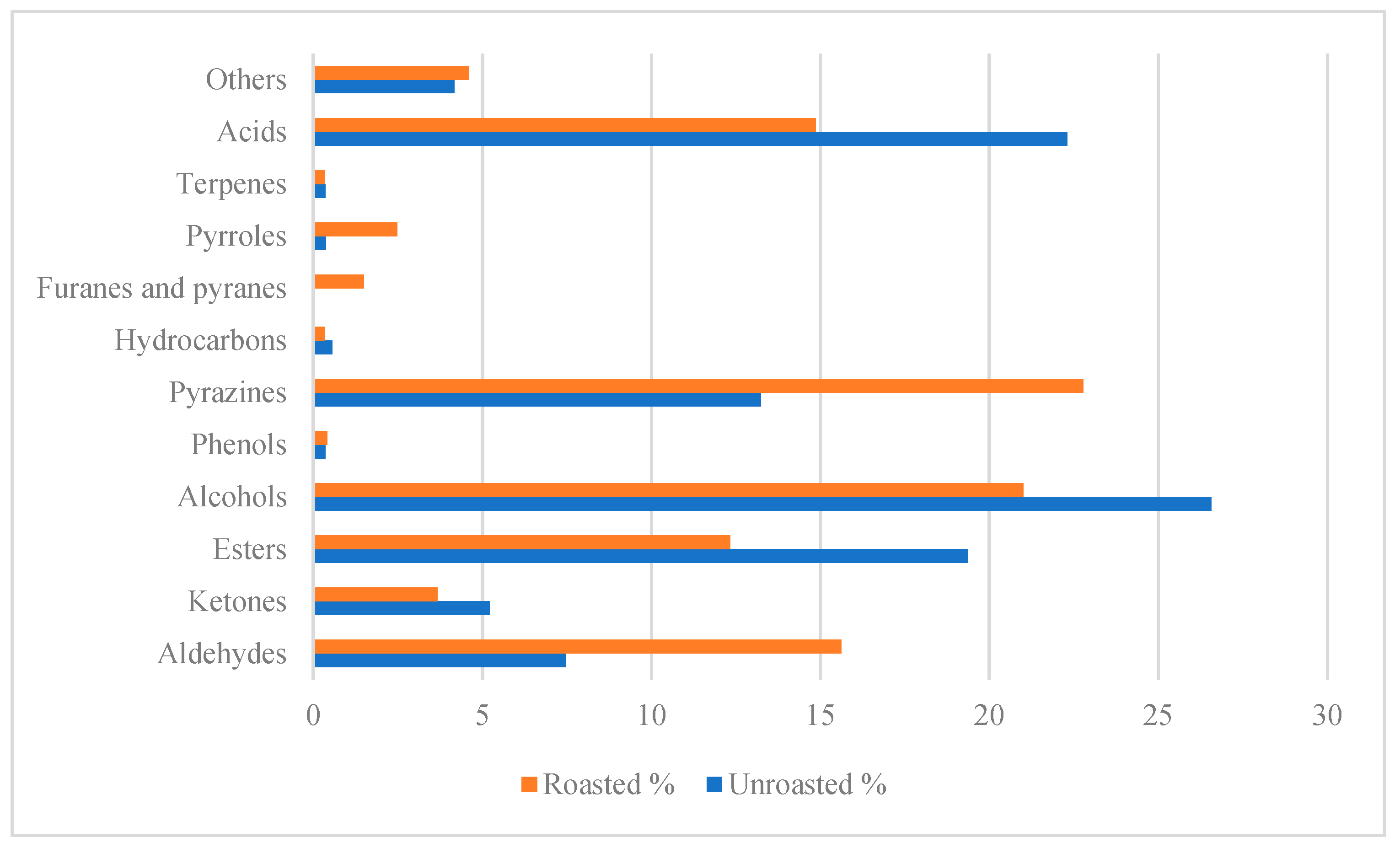
Roasting the beans boosts the intensity of some of the volatile compounds present in dried beans, generating the appearance of new compounds. Ref. [
6] reported 31 compounds in forastero cocoa beans roasted at 95 °C, 71 compounds in beans (of the same variety) roasted at 140 °C, while [
9] reported 78 compounds in forastero cocoa beans roasted at 160 °C. They found 34 compounds that had increased in their concentration during roasting, nine compounds that had increased in concentration up to 140 °C and had then decreased or were undetectable; A total of 26 new compounds were added, and the concentration of nine compounds decreased significantly. After roasting, the distribution of the different classes of compounds changed, increasing in pyrazines (22.79%) and aldehydes (15.62%) and forming new compounds from Maillard reactions, such as pyrroles, furans, and pyrans [
22]. Some pyrrole derivatives are formed at moderate roasting temperatures and relatively high humidity [
40]. These compounds come from sugar precursor degradation in cocoa and decrease during roasting. Ref. [
73] reported that these volatiles are a useful indicator for which their level can be used to monitor the early stages of roasting.
The presence of undesirable compounds,
Figure 3, in both studies represents 42% and 27% of the total aroma profile, indicating the presence of acidic, musty/earthy, burnt, and smoky flavors [
74]. Its high intensity and overall contribution are reflected in the OAVs ranging from 1.77 for 2-methoxyphenol to 4920 for acetic acid. The concentration of acetic acid is more than half in the unroasted beans when roasted at 130 °C for 30 mins. Roasting at higher temperatures or for a longer period does not significantly affect the acetic acid concentration. On the contrary, the concentration is higher when roasted at 160 °C for 30 min [
75]. Other compounds, such as sulfur derivatives, are also present in cocoa beans roasted at temperatures >160 °C. These compounds generate unpleasant smoky, onion, cabbage, and gasoline notes that persist in the final chocolates, albeit in lower concentrations [
39,
44].
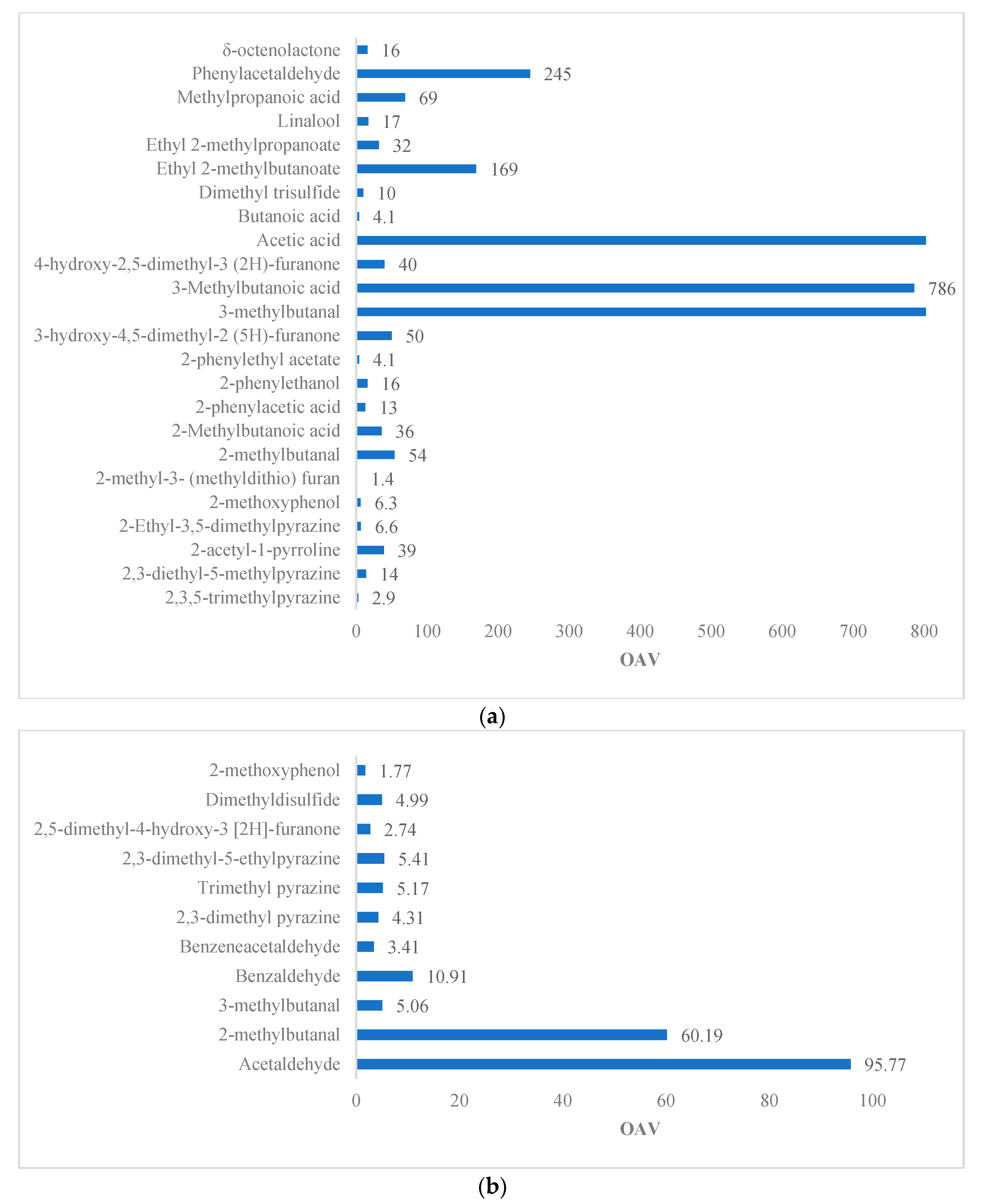
A percentage greater than 50% of the compounds in
Table 4 indicates a contribution of desirable aromas, such as fruity, floral, cocoa, and chocolate, including the Strecker aldehydes (2- and 3-methylbutanal, phenylacetaldehyde) present in dry beans in smaller quantities. 3-methylbutanal increases its contribution during roasting, especially at 95 and 110 °C, while at 125 °C, there is a return to the initial values. This is due to the prevalence of the volatilization phenomenon compared to that of generation [
76]. Furthermore, the concentration of benzaldehyde increases during fermentation [
5] and during roasting, making it susceptible to losses at temperatures above 125 °C [
76]. Throughout the processing of the beans, the concentration of this compound decreases. In dry beans, the concentration is 1.08 µg/g, and its OAV is 18.08, whereas, in roasted beans, it drops to 1.01 µg/g and presents OAV = 16.90 [
9,
21].
Other compounds that turn up after roasting are 4-hydroxy-2,5-dimethyl-3 (2H)-furanone, 2-acetyl-1-pyrroline, 2,3-dimethyl-5-ethylpyrazine, trimethylpyrazine, and 2,3-dimethylpyrazine. 2-acetyl-1-pyrroline, which is typically associated with roasting (Parker, 2015), exudes a popcorn and cracker-like aroma, having a low threshold in oil 0,1 ng/g and a concentration above OTV = 39 ng/g. Therefore, the contribution to the overall aroma of the roasted beans is significant (
Table 3). Likewise, the pyrazines generated during roasting at temperatures above 100 °C exude notes of cocoa, baked potato, and chocolate. Ref. [
45] reported that simple unsubstituted or monosubstituted pyrazines have a roasted, biscuit aroma and relatively high aroma thresholds, but as the substitution increases, the odor threshold decreases. It is evident from
Table 3 that some of the pyrazines have low OTVs: 2,3-diethyl-5-methylpyrazine and 2-ethyl-3,5-dimethylpyrazines, and high OTVs: 2,3,5-trimethylpyrazine and 2,3-dimethyl-5-ethylpyrazine. These compounds exude baked potato, chocolate, earthy, popcorn, and cocoa aromas, respectively. However, their OTVs are inversely related to their OAVs. There are a few fruity and floral aromas, as shown in
Table 3, including linalool, 2-phenylethanol, and 2-phenylethyl acetate, in descending order of their aromatic contribution. This deficiency in fruity aromas is common in the forastero variety [
28]. Finally, the 59 volatile compounds present in the roasted beans include alkyl pyrazines and aldehydes from reactions between oligopeptides derived from vicilin class globulin (7S) and reducing sugars of the cocoa beans [
36]. The author reported eight unidentified volatile compounds likely to expel specific cocoa notes.
3.2.3. Cocoa Liquor
The aroma of cocoa liquor is an important characteristic affecting its quality; therefore, the affecting factors are the origin of the cocoa bean and the postharvest processing (fermentation and drying), roasting, and storage [
19]. The origin of the cocoa bean is vital in determining the aroma of all its cocoa products and is related to genetic and environmental factors. In this regard, only a few aromas are characteristic of cocoa liquor from different origins, including 3-methylbutanal (malty), linalool (floral), β-phenylethyl alcohol (pink), benzaldehyde (almond-like), benzeneacetaldehyde (pink), β-phenylethyl acetate (fruity), 3-methylbutyl benzoate (fruity), 2,5-6-dimethylpyrazine (potato-like), ethylpyrazine (popcorn), trimethylpyrazine (roasted), 3-ethyl-2,5-dimethylpyrazine (roasted), tetramethylpyrazine (nutty), 3,5-diethyl-2-methylpyrazine (cocoa), furfural (potato), acetic acid (acid), 3-methylbutanoic acid (stench), and dimethyltrisulfide (onion), which are considered the key active aroma compounds contributing to the overall cocoa liquor odor of the forastero variety. However, the intensity of each aroma among the different cocoa liquors is distinct [
77]. Likewise, Ref. [
78] reported that the specific aroma of bulk cocoa liquor included sweet, nutty, caramel, and chocolate notes associated with trimethylpyrazine, tetramethylpyrazine, 2,3-butanediol, dodecanoic acid, β-phenylethyl alcohol, 2-acetylpyrrole, and benzeneacetaldehyde.
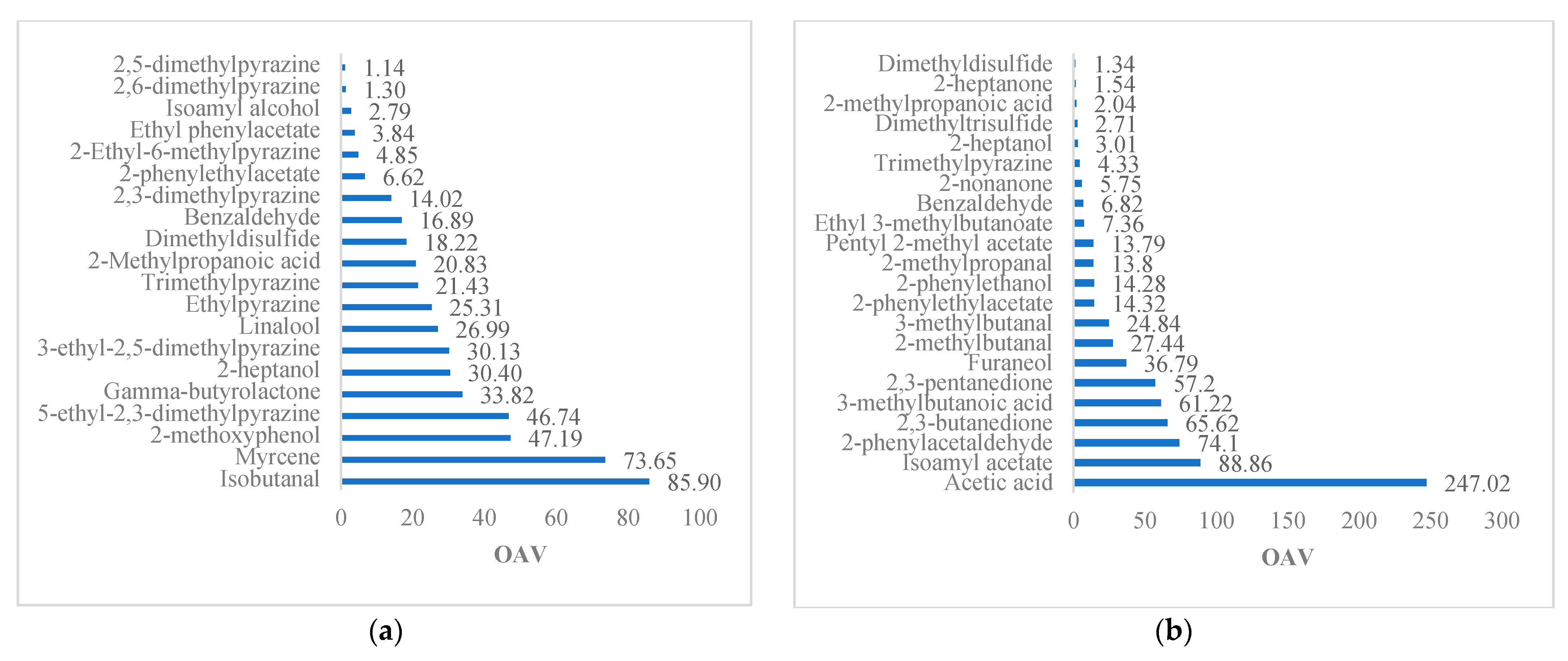
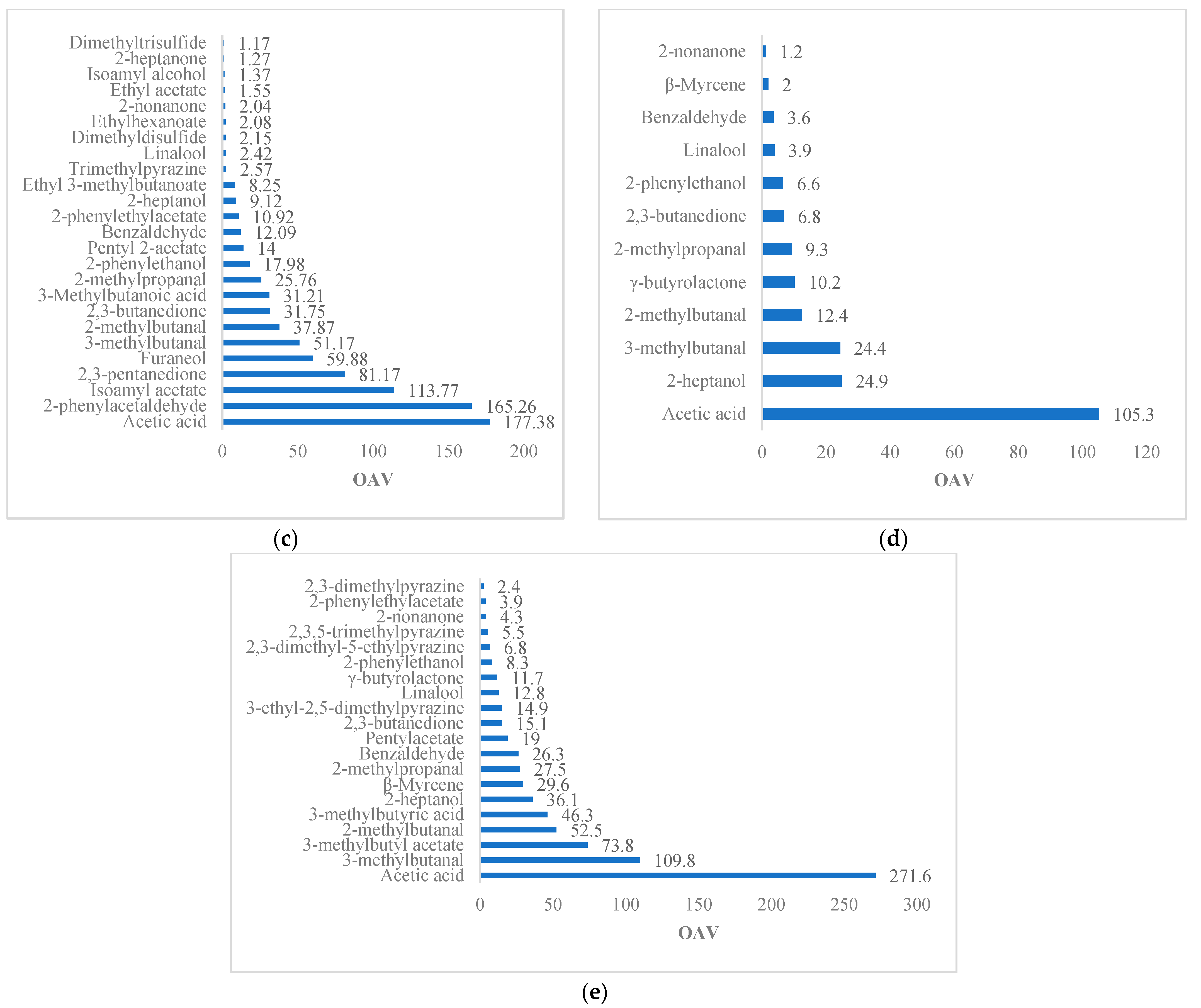
On the whole, the compounds of the cocoa liquors studied represent 57.89 to 65.79% of OAV >1, and 32.61 to 42.11% represent OAV < 1. Among these compounds, fruity, floral, chocolate, buttery, and undesirable aromas are highlighted (
Table 5). Several compounds account for 17 to 40% of fruit flavors with a VAO > 1, including isoamyl acetate (banana), 2-heptanol (citrus), ethyl phenylacetate, isoamyl alcohol (banana), furaneol (strawberry), pentyl 2-acetate (orange), ethyl 3-methylbutanoate, 2-nonanone, 2-heptanol, 2-heptanone (coconut), pentylacetate, and 3-methylbutyl acetate. From 14 to 25% of the floral compounds, 2-phenylethyl alcohol, linalool, 2-phenylethylacetate, acetophenone, 2-propanone, 2-phenylacetaldehyde, and β-myrcene are included. From 20 to 33% provide chocolate notes, including 2- and 3-methylbutanal, 5-ethyl-2,3-dimethylpyrazine, trimethylpyrazine, 2,3,5-trimethylpyrazine, benzaldehyde, 2-ethyl-5 and 6-methylpyrazine, 2,6-dimethylpyrazine, 2,5-dimethylpyrazine, tetramethylpyrazine, methylpyrazine, and 2-methylpropanal. From 3 to 14% provide creamy and buttery tones, such as ethylpyrazine, furfural, 3-hydroxy-2-butanone, 2,3-butanedione, 2,3-pentanedione, and 2-methylpropanoic acid. Finally, the compounds providing caramel and earthy notes in the 3% to 15% range include gamma-butyrolactone and 3-ethyl-2,5-dimethylpyrazine, respectively.
Figure 4 details the set of OAV > 1 compounds described above, which is common among the studies of Streker aldehydes, linalool, isoamyl acetate, and 2-heptanol.
The compounds with an OAV < 1 include isoamyl alcohol, ethyl acetate, ethylhexanoate, limonene, acetophenone, linalool, 2,5-dimethylpyrazine, 2,6-dimethylpyrazine, 2-ethyl-5-methylpyrazine, tetramethylpyrazine, propionic acid, 2-butanone, ethanol, 2-butanol, 2-methyl-1-propanol, 2-pentylfuran, 3-hydroxy-2-butanone and ethanol, with fruity, floral, chocolate, buttery, and mainly undesirable aromas. The compounds exclusive to the study include rose oxide and β-myrcene that reveal floral notes in cocoa of the forastero and national variety, respectively.
Smoky, acidic, hammy, or musty aromas are the common compounds in cocoa liquors, and are present throughout the postharvest process [
79]. From 8 to 24% of the total active odor aromas (OAV > 1) correspond to undesirable compounds in cocoa liquors and dried beans, such as acetic acid, 2,3-pentanedione, 2- and 3-methylbutanoic acid, and 2-methoxyphenol. In addition, 2,3-butanedione, dimethyl di- and trisulfide, methylpropanoic acid, and butanoic acid are in roasted beans and persist in cocoa liquor. During chocolate processing with these liquors, the OAV partially decreases or is undetected. For instance, in acetic acid and 2-nonanone, the OAV decreases by 5 to 14 times, and the concentration of 2,3-butanedione is undetected, so their OAV cannot be calculated [
12]. The same occurs with butyric acid and 3-methylbutyric acid, which are only detected in cocoa liquors and disappear in processed chocolate. It is worth mentioning that although acids present a lower number of compounds in cocoa liquors, they have the highest VAOs, affecting the liquor’s pH. Cocoa liquor with a low pH (4.75 to 5.19) is more likely to have off-flavors.
3.2.4. Chocolate
The flavor of chocolate depends on the way the series of processes described above are carried out. Conching is the last of these processes, whereby the manufacturer can obtain the flavor and aroma required for a particular product. However, this process cannot correct previous mistakes, like an unpleasant smoky or moldy flavor due to poor drying, nor can it turn an inferior cocoa taste into a perfect one [
80]. Specifically, the function of conching is to evaporate volatile acids, achieve adequate viscosity, remove excess moisture, and develop a desirable color [
81,
82], as well as remove off-flavors and aromas while retaining desirable ones [
80]. Chocolate producers often use different conching temperatures and times depending on cocoa bean varieties and the origin of the chocolate products with the desired aromatic properties [
83]. In particular, levels of the most important odors decrease significantly by rising conching duration (from 6 to 10 h at 80 °C). Prolonged times reduce most pyrazines (including 2,5-dimethylpyrazine, 2-ethyl-5-methylpyrazine, and 2,3,5-trimethylpyrazine) and the levels of alcohol, acid, aldehydes, and small esters [
25]. Similarly, [
11] reported that prolonged conching reduces volatile acid concentration, alcohol, 3-methylbutanal, benzaldehyde, and several lesser volatile pyrazines, like trimethylpyrazine, tetramethylpyrazine, and acetylpyrrole. They also noted that this treatment increases the furfural content and does not affect the isobutanal, 2-methylbutanal, and phenylacetaldehyde levels because of the additional reaction compensation to form Strecker aldehydes during conching. Other components with significant contributions to chocolate (
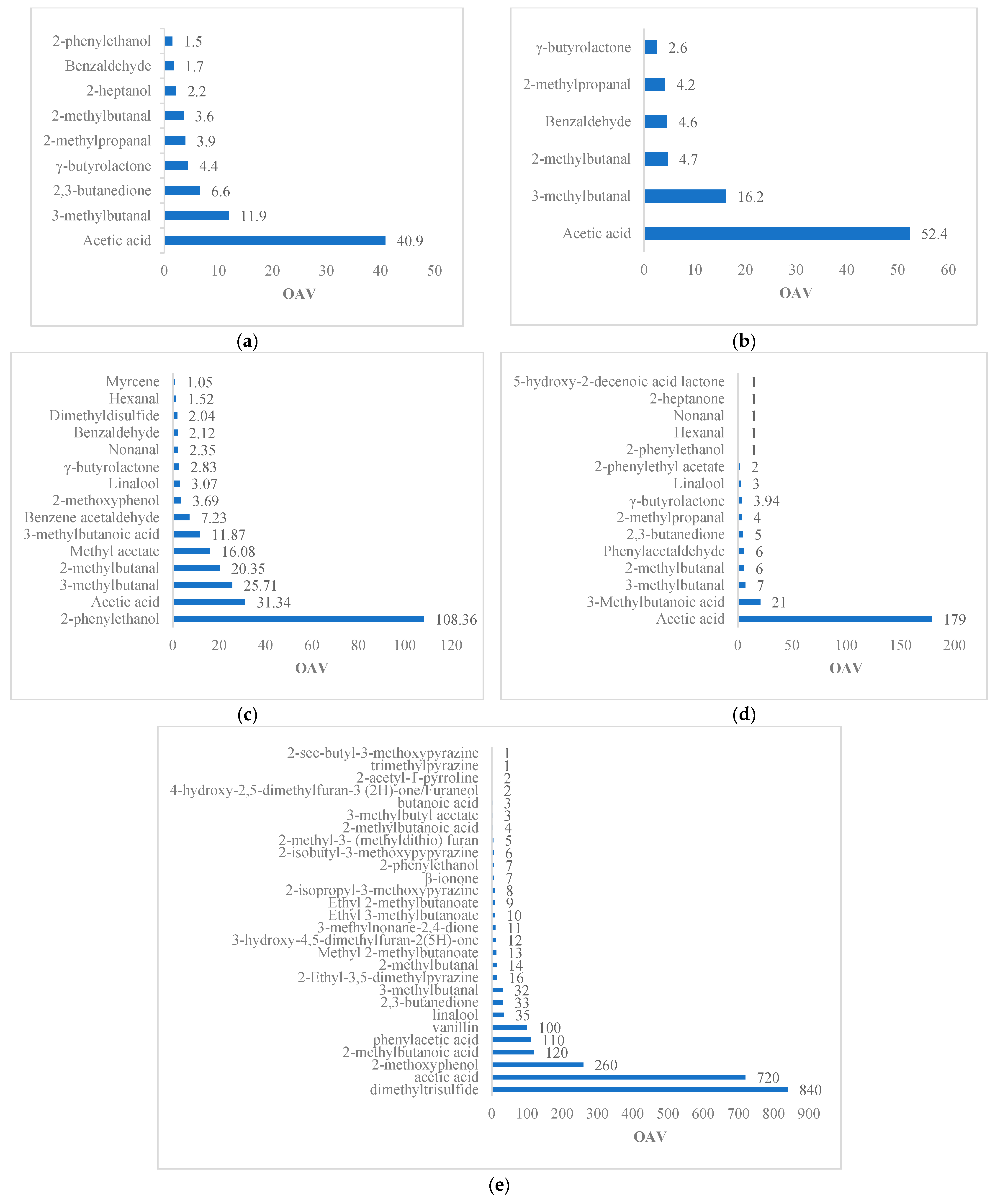
Figure 5) and that are highly odorous based on their low odor thresholds are 2-methylbutanal (2.2 ng/g), 3,5-diethyl-2-methylpyraniza, furaneol (27 ng/g), 2,3-diethyl-5-methylpyrazine (7.2 ng/g), ethyl 2- and 3-methylbutanoate (0.37 and 0.98 ng/g), 2-methylpropanal (3.4 ng/g), 3-isobutyl-methoxypyrazine (0.04 ng/g), 3-isopropyl-2-methoxypyrazine (0.01 ng/g), and linalool (37 ng/g) [
10,
11,
12,
44,
84].
Other major aroma compounds remaining in chocolates include 2- and 3-methylbutanal (chocolate, malt), benzaldehyde (roasted almonds, malt), gamma-butyrolactone (sweet, caramel), 2-methylpropanal (unroasted cocoa, malt), linalool (flowery, fruity, tea-like), acetic acid (bitter, vinegar), and 2-phenylethanol (honey, rose) (
Figure 5). Ref. [
84] reported similar compounds, such as 3-methylbutanal, 2-methylpropanal, phenylacetaldehyde, tetramethylpyrazine, 2-acetyl-1-pyrroline, trimethylpyrazine, 3-methylbutanoic acid, acetic acid, and vanillin. The uncommon compound is vanillin, which is a highly odorous compound with OAV = 100 and was found only in chocolate with 90% cocoa (
Figure 5e). In fact, the most uncommon aromatic compounds among the studies are found in this chocolate. Likewise, 2-acetyl-1-pyrroline (popcorn-like aroma) is found in roasted beans (OAV = 39) [
6], cocoa liquor (OAV = 207.55) and chocolate (OAV= 396.23) [
85]. Seyfried and Granvogl (2019) [
10] reported OAV = 2 in 90% cocoa chocolate, and this was unidentified in 99% cocoa chocolate. This compound is highly volatile and mainly generated during roasting.
The greater the availability of the compounds, the more complex the chocolate aroma is due to a wide volatile matrix. The studies in this paper show that acetic acid is the most abundant compound, and the rest of the compounds represent a mixture from different families (acids, alcohols, pyrans, aldehydes, esters, furans, and pyrazines).
Table 6 shows that the chocolates made with 40 and 90% cocoa have an almost complete distribution of aromas ranging from fruity to undesirable in a range of 13 to 25%.
Many of the compounds that remain in chocolate have an increased concentration along the processing chain or remained unchanged, as has been reported; the influence of the manufacturing process is greater than that of the difference in the cocoa production area, providing a diversity of aroma profiles [
86]. Particularly, criollo cocoa roasting increases acetophenone, tetramethylpyrazine, 2,3,5-trimethylpyrazine, and 2,5-dimethylpyrazine concentration, whereas 2-heptanol, phenylethyl alcohol, 2,3-butanedione, 2-phenylphenylpyrazine, 2,3-butanedione, 2,3-butanedione and 2,3-butanedione, 2,3-butanedione, 2-phenyl-2-butenal, 5-methyl-2-phenyl-2-hexanal, ethyl octanoate, ethyl phenylacetate, ethyl decanoate, and trans-linalool oxide remain stable during roasting [
2]. In addition, some of the samples’ volatile compounds are affected by each brand formulation, masking or enhancing a specific volatile. Ref. [
26] considered three of the predominant masses of chocolates, namely, mass 33, 43, and 61, which were identified as methanol, a fragment of diverse origin, and acetic acid, respectively, and because of their high concentration, he suggested these three masses can have a huge impact on the fingerprint analysis that differentiates them by regions and brands.