Propionic Acid Fermentation—Study of Substrates, Strains, and Antimicrobial Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Microbial Cultures
2.2. Maintenance and Propagation of Microbial Cultures
2.3. Fermentation Experiment—Media and Culture Conditions
2.4. Fermentation Metabolites, pH and Biomass Analyses
2.5. Acid and Biomass Outcomes Calculations
2.6. Preparation of Fermentates for the in Vitro Antimicrobial Effects Studies
2.7. Determination of the Antimicrobial Effects of Fermentates
2.8. Determination of Minimum Inhibitory Concentrations (MICs) of Organic Acids
- Lactic and formic acids: 4, 6, 8, 10, 12, 20 mM;
- Propionic and acetic acids: 3, 4, 6, 8, 10, 12 mM;
- Blank samples without added acids (with added sterile water) were studied as a negative control to determine the normal growth kinetics of microorganisms without acid inhibition.
2.9. Statistical Analyzes
3. Results
3.1. Results of Propionic Acid Fermentation Study—Substrates and Bacterial Strains
3.2. Results of Propionic Acid Bacteria Antimicrobial Effects Study
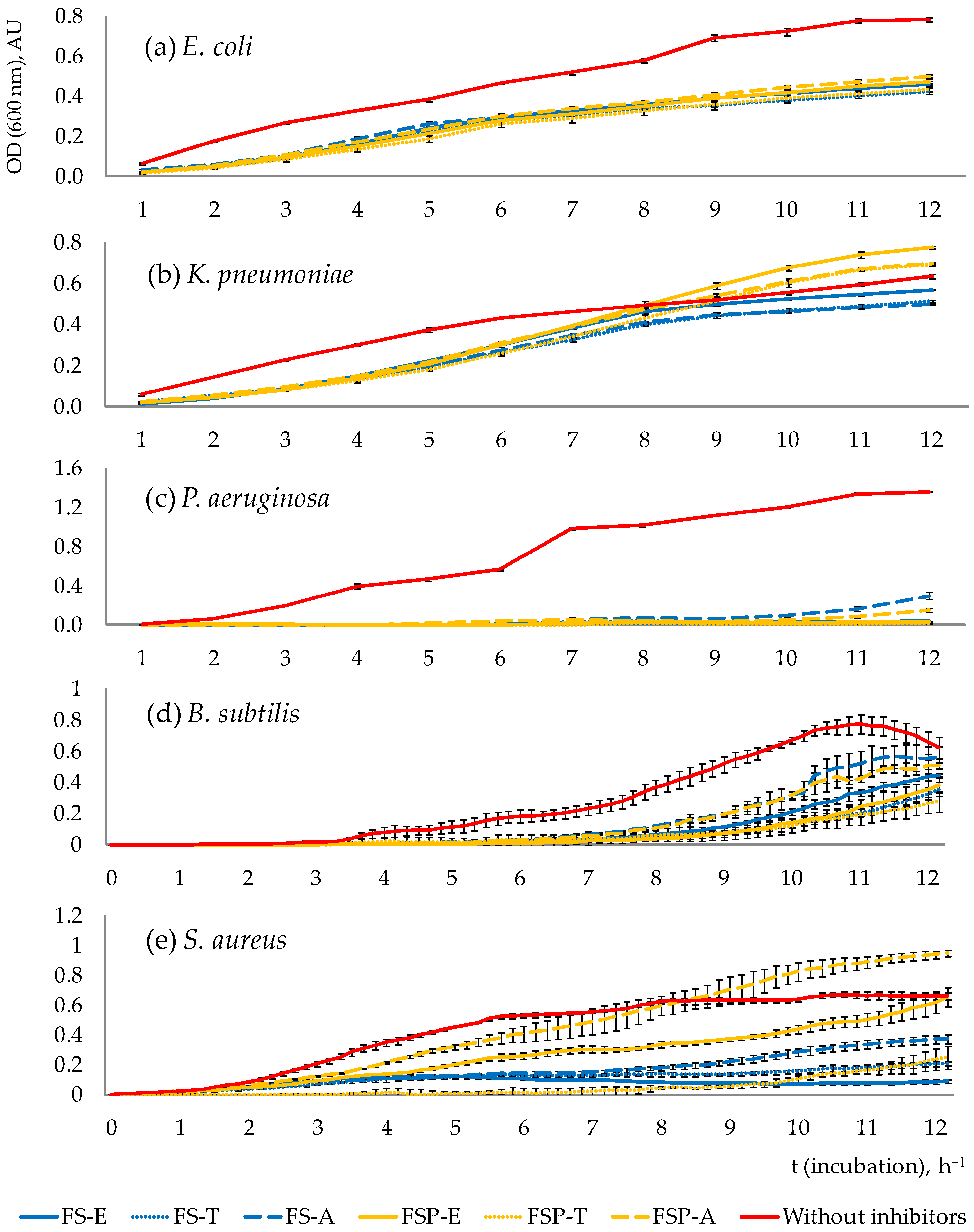
3.2.1. Pathogen Growth Dynamics throughout the Total Incubation Period (12 h)
3.2.2. The Survival Rate of Pathogens after 10 h of Incubation
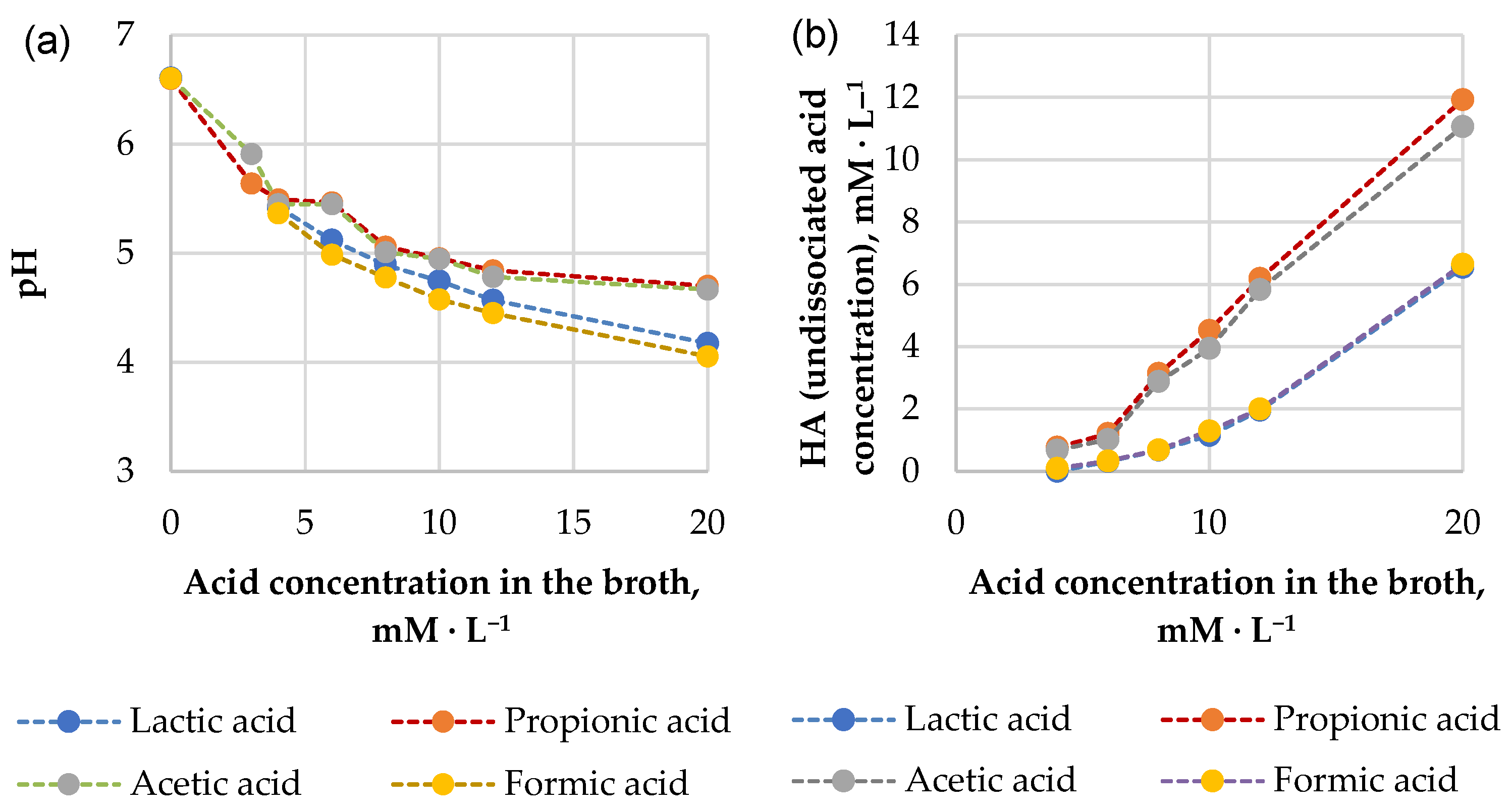
3.2.3. Antimicrobial Compounds Involved in the Inhibition of Pathogenic Microorganisms
3.2.4. Antimicrobial Effects of Pure Organic Acid Exposure
- E. coli: formic > propionic = acetic > lactic;
- K. pneumonia, P. aeruginosa, S. aureus: propionic = acetic = formic > lactic;
- B. subtilis: propionic > acetic > lactic = formic.
4. Discussion
4.1. Assessment of the Results of the Fermentation Study
4.2. Assessment of the Results of Antimicrobial Effects’Study
5. Conclusions
- The knowledge gained in the present study can help to model the acquirement of acid metabolites and biomass from several PAB strains. The results show that the fermentative production of propionic and acetic acid is considerably influenced by the added carbon source. Lactose broth fermentation resulted in a significant pH reduction, and propionic and acetic acids’ outcomes were significantly lower (p < 0.05) than when using lactate as a carbon source.
- Biomass production in lactose was significantly higher (p < 0.05) than in lactate-based broths.
- When comparing the propionic acid bacteria strains, significant differences were observed for the production of acids and resulting pH in both types of fermentates (p < 0.05). Acid production, lactose consumption, substrate conversion efficiency to acetic acid, as well as biomass production was strain-dependent. At the same time, lactose conversion efficiency to propionic acid and propionic acid yield were similar for all strains (p ≥ 0.05).
- Results show that antimicrobial efficacy is strain-dependent. The strongest inhibitory effect was demonstrated by fermentates of all tested PAB strains against Pseudomonas aeruginosa, by strain Acidipropionibacterium thoenii against Bacillus subtilis and by strains Propionibacterium freudenreichii and Acidipropionibacterium thoenii against Staphylococcus aureus.
- Bacteriocins are likely to play a significant role in the inhibition of Staphylococcus aureus and Klebsiella pneumonia, while Escherichia coli, Pseudomonas aeruginosa and Bacillus subtilis are inhibited by acids. To increase the antimicrobial activity of fermentates, we recommend acidifying the fermentate or fermentate-supplemented medium to at least pH 5.5.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zandona, E.; Blažić, M.; Režek Jambrak, A. Whey Utilization: Sustainable Uses and Environmental Approach. Food Technol. Biotechnol. 2021, 59, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Chourasia, R.; Phukon, L.C.; Abedin, M.M.; Padhi, S.; Singh, S.P.; Rai, A.K. Whey valorization by microbial and enzymatic bioprocesses for the production of nutraceuticals and value-added products. Bioresour. Technol. Rep. 2022, 19, 101144. [Google Scholar] [CrossRef]
- Lakstina, J.; Aboltina, I.; Vanaga, L.; Ciprovica, I.; Jonkus, D.; Zagorska, J.; Cinkmanis, I. The novel solution for acid whey permeate application in animal feeding. Rural Sustain. Res. 2020, 44, 1–7. [Google Scholar] [CrossRef]
- Russo, G.L.; Langellotti, A.L.; Verardo, V.; Martín-García, B.; Di Pierro, P.; Sorrentino, A.; Baselice, M.; Oliviero, M.; Sacchi, R.; Masi, P. Formulation of New Media from Dairy and Brewery Wastes for a Sustainable Production of DHA-Rich Oil by Aurantiochytrium mangrovei. Mar. Drugs 2022, 20, 39. [Google Scholar] [CrossRef] [PubMed]
- Sarenkova, I.; Saez-Orviz, S.; Ciprovica, I.; Rendueles, M.; Diaz, M. Lactobionic acid production by Pseudomonas taetrolens in a fed-batch bioreactor using acid whey as substrate. Int. J. Dairy Technol. 2022, 75, 361–371. [Google Scholar] [CrossRef]
- Caplice, E.; Fitzgerald, G.F. Food fermentations: Role of microorganisms in food production and preservation. Int. J. Food Microbiol. 1999, 50, 131–149. [Google Scholar] [CrossRef]
- Patel, A.; Shah, N. Current scenario of antimicrobial compounds produced by food grade bacteria in relation to enhance food safety and quality. J. Innov. Biol. 2014, 1, 189–194. [Google Scholar]
- Fernandez, B.; Vimont, A.; Desfossés-Foucault, É.; Daga, M.; Arora, G.; Fliss, I. Antifungal activity of lactic and propionic acid bacteria and their potential as protective culture in cottage cheese. Food Control 2017, 78, 350–356. [Google Scholar] [CrossRef]
- Quinto, E.J.; Caro, I.; Villalobos-Delgado, L.H.; Mateo, J.; De-Mateo-Silleras, B.; Redondo-Del-Río, M.P. Food Safety through Natural Antimicrobials. Antibiotics 2019, 8, 208. [Google Scholar] [CrossRef]
- Ajingi, Y.S.; Rodpan, S.; Usman, J.N.; Koga, Y.; Jongruja, N. Synergistic effect of nisin with acetic and propionic acids inactivates Bacillus subtilis on meat and potato. Biocatal. Agric. Biotechnol. 2022, 41, 102317. [Google Scholar] [CrossRef]
- Parada, J.L.; Caron, C.; Medeiros, A.B.; Soccol, C.R. Bacteriocins from lactic acid bacteria: Purification, properties and use as biopreservatives. Braz. Arch. Biol. Technol. 2007, 50, 512–542. [Google Scholar] [CrossRef]
- Poonam, P.S.D.; Tomar, S.K.; De, S.; Singh, R. Multifaceted attributes of dairy propionibacteria: A review. World J. Microbiol. Biotechnol. 2012, 28, 3081–3095. [Google Scholar] [CrossRef]
- Šimat, V.; Čagalj, M.; Skroza, D.; Gardini, F.; Tabanelli, G.; Montanari, C.; Hassoun, A.; Ozogul, F. Chapter Two—Sustainable sources for antioxidant and antimicrobial compounds used in meat and seafood products. In Advances in Food and Nutrition Research; Toldra, F., Ed.; Academic Press: Cambridge, MA, USA, 2021; Volume 97, pp. 55–118. [Google Scholar] [CrossRef]
- Alonso, S.; Rendueles, M.; Díaz, M. Microbial production of specialty organic acids from renewable and waste materials. Crit. Rev. Biotechnol. 2015, 35, 497–513. [Google Scholar] [CrossRef]
- Gonzalez-Garcia, R.A.; McCubbin, T.; Navone, L.; Stowers, C.; Nielsen, L.K.; Marcellin, E. Microbial Propionic Acid Production. Fermentation 2017, 3, 21. [Google Scholar] [CrossRef]
- Rutenberg, R.; Bernstein, S.; Fallik, E.; Paster, N.; Poverenov, E. The improvement of propionic acid safety and use during the preservation of stored grains. Crop Prot. 2018, 110, 191–197. [Google Scholar] [CrossRef]
- Ali, R.; Saravia, F.; Hille-Reichel, A.; Gescher, J.; Horn, H. Propionic acid production from food waste in batch reactors: Effect of pH, types of inoculum, and thermal pre-treatment. Bioresour. Technol. 2020, 319, 124166. [Google Scholar] [CrossRef]
- Antone, U.; Liepins, J.; Zagorska, J.; Cinkmanis, I. Fermentation of milk whey permeate with different dairy propionibacteria strains. In Rural Development: Challenges for Sustainable Bioeconomy and Climate Change, Proceedings of the 10th International Scientific Conference, Kaunas, Lithuania, 21–23 September 2021; Černiauskienė, J., Ed.; Vytautas Magnus University Agriculture Academy: Kaunas, Lithuania, 2021; pp. 1–7. [Google Scholar] [CrossRef]
- Piwowarek, K.; Lipińska, E.; Hać-Szymańczuk, E.; Kieliszek, M.; Ścibisz, I. Propionibacterium spp.—Source of propionic acid, vitamin B12, and other metabolites important for the industry. Appl. Microbiol. Biotechnol. 2018, 102, 515–538. [Google Scholar] [CrossRef]
- Teles, J.C.; Stolle, E.M.; Koloda, S.A.; Barana, A.C. Production of Propionic Acid by Propionibacterium acidipropionici from Agroindustrial Effluents. Braz. Arch. Biol. Technol. 2019, 62, e19180550. [Google Scholar] [CrossRef]
- Ranaei, V.; Pilevar, Z.; Khaneghah, A.M.; Hosseini, H. Propionic Acid: Method of Production, Current State and Perspectives. Food Technol. Biotechnol. 2020, 58, 115–127. [Google Scholar] [CrossRef]
- Coral, J. Propionic acid production by Propionibacterium sp. using low-cost carbon sources in submerged fermentation. Master’s Thesis, The University of Provence, the University of the Mediterranean Sea and Federal University of Parana, Curitiba, Brazil, 2008. [Google Scholar]
- Lyon, W. Characterization and Isolation of a Bacteriocin Produced by a Strain of Propionibacterium thoenii (A Dissertation Submitted to the Graduate Faculty in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy); Iowa State University: Ames, IA, USA, 1992. [Google Scholar] [CrossRef]
- Zarate, G.; Babot, J.D.; Argañaraz-Martínez, E.; Lorenzo-Pisarello, M.J.; Perez Chaia, A. Dairy propionibacteria: Technological importance and probiotic potential for application on human and animal nutrition. In Multidisciplinary Approaches on Food Science and Nutrition for the XXI Century; Filip, R., Ed.; Transworld Research Network: Kerala, India, 2011; pp. 175–213. [Google Scholar]
- Wyk, J.V.; Morkel, R.A.; Dolley, L. Metabolites of Propionibacterium: Techno- and biofunctional ingredients. In Alternative and Replacement Foods; Handbook of Food Bioengineering, Series; Grumezescu, A.M., Holban, A.M., Eds.; Academic Press: London, UK, 2018; Volume 17, pp. 205–260. [Google Scholar]
- Turgay, M.; Bachmann, H.; Irmler, S.; von Ah, U.; Hlich-Wyder, M.F.; Falentin, H.; Deutsch, S.; Jan, G.; Thierry, A. Propionibacterium spp. and Acidipropionibacterium spp.; Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2020; Available online: https://hal.inrae.fr/hal-02640422v1 (accessed on 17 September 2021). [CrossRef]
- EFSA BIOHAZ Panel (EFSA Panel on Biological Hazards); Koutsoumanis, K.; Allende, A.; Alvarez-Ordóñez, A.; Bolton, D.; Bover-Cid, S.; Chemaly, M.; Davies, R.; De Cesare, A.; Hilbert, F.; et al. Statement on the update of the list of QPS-recommended microbiological agents intentionally added to food or feed as notified to EFSA 16: Suitability of taxonomic units notified to EFSA until March 2022. EFSA J. 2022, 20, 7408. [Google Scholar] [CrossRef]
- Rabah, H.; Rosa do Carmo, F.L.; Jan, G. Dairy propionibacteria: Versatile probiotics. Microorganisms 2017, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Thierry, A.; Deutsch, S.M.; Falentin, H.; Dalmasso, M.; Cousin, F.J.; Jan, G. New insights into physiology and metabolism of Propionibacterium freudenreichii. Int. J. Food Microbiol. 2011, 149, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Hugenholtz, J.; Hunik, J.; Santos, H.; Smid, E. Nutraceutical production by propionibacteria. Le Lait 2002, 82, 103–112. [Google Scholar] [CrossRef]
- Holo, H.; Faye, T.; Brede, D.A.; Nilsen, T.; Ødegård, I.; Langsrud, T.; Brendehaug, J.; Nes, I.F. Bacteriocins of propionic acid bacteria. Le Lait 2002, 82, 59–68. [Google Scholar] [CrossRef]
- Faye, T.; Brede, D.A.; Langsrud, T.; Nes, I.F.; Holo, H. Prevalence of the genes encoding propionicin T1 and protease-activated antimicrobial peptide and their expression in classical propionibacteria. Appl. Environ. Microbiol. 2004, 70, 2240–2244. [Google Scholar] [CrossRef] [PubMed]
- Thierry, A.; Valence, F.; Deutsch, S.M.; Even, S.; Falentin, H.; Le Loir, Y.; Jan, G.; Gagnaire, V. Strain-to-strain differences within lactic and propionic acid bacteria species strongly impact the properties of cheese—A review. Dairy Sci. Technol. 2015, 95, 895–918. [Google Scholar] [CrossRef]
- MicroGARD® Fermentates. Danisco DuPont. 2022. Available online: https://www.dupontnutritionandbiosciences.com/products/microgard.html (accessed on 10 September 2022).
- Cultured Whey. Inhibit 3600 Dairy. Mezzoni Foods. 2022. Available online: https://www.mezzonifoods.com/shop/cultured-whey/ (accessed on 10 September 2022).
- Danilova, I.V.; Lee, H.; Tourova, T.P.; Ryzhkova, E.P.; Netrusov, A.I. Propionibacterium freudenreichii strains as antibacterial agents at neutral ph and their production on food-grade media fermented by some lactobacilli. J. Food Saf. 2012, 32, 48–58. [Google Scholar] [CrossRef]
- Vorobjeva, I.L. Propionibacteria; Kluver Academic: Dordrecht, The Netherlands, 1999. [Google Scholar]
- Acidipropionibacterium Acidipropionici. Leibniz Institute DSMZ-German Collection of Microorganisms and Cell Cultures GmbH. 2022. Available online: https://www.dsmz.de/microorganisms/medium/pdf/DSMZ_Medium91.pdf (accessed on 15 December 2022).
- Le Lay, C.; Coton, E.; Le Blay, G.; Chobert, J.M.; Haertlé, T.; Choiset, Y.; Van Long, N.N.; Meslet-Cladière, L.; Mounier, J. Identification and quantification of antifungal compounds produced by lactic acid bacteria and propionibacteria. Int. J. Food Microbiol. 2016, 239, 79–85. [Google Scholar] [CrossRef]
- Martynova, J.; Kokina, A.; Kibilds, J.; Liepins, J.; Scerbaka, R.; Vigants, A. Effects of acetate on Kluyveromyces marxianus DSM 5422 growth and metabolism. Appl. Microbiol. Biotechnol. 2016, 100, 4585–4594. [Google Scholar] [CrossRef]
- Andrews, J.M. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother. 2001, 48, 5–16. [Google Scholar] [CrossRef]
- Latham, K.G.; Ferguson, A.; Donne, S.W. Influence of ammonium salts and temperature on the yield, morphology and chemical structure of hydrothermally carbonized saccharides. SN Appl. Sci. 2019, 1, 1–13. [Google Scholar] [CrossRef]
- Lambert, R.J.; Stratford, M. Weak-acid preservatives: Modelling microbial inhibition and response. J. Appl. Microbiol. 1999, 86, 157–164. [Google Scholar] [CrossRef]
- Moresi, M.; Parente, E. Fermentation (Industrial)|Production of some organic acids (citric, gluconic, lactic, and propionic). In Encyclopedia of Food Microbiology, 2nd ed.; Batt, C.A., Tortorello, M.L., Eds.; Academic Press: Cambridge, MA, USA, 2014; pp. 804–815. [Google Scholar] [CrossRef]
- Frallicciardi, J.; Melcr, J.; Siginou, P.; Marrink, S.J.; Poolman, B. Membrane thickness, lipid phase and sterol type are determining factors in the permeability of membranes to small solutes. Nat. Commun. 2022, 13, 1605. [Google Scholar] [CrossRef]
- Hassan, A.N.; Frank, J.F. Ch.6 Starter cultures and their use. In Applied Dairy Microbiology, 2nd ed.; Marth, E.H., Steele, J., Eds.; Marcel Dekker Inc.: New York, NY, USA; CRC Press: Basel, Switzerland, 2001; p. 185. [Google Scholar] [CrossRef]
- Ciani, M.; Comitini, F.; Mannazzu, I. Ecological Processes. Fermentation. In Encyclopedia of Ecology; Jorgensen, S.E., Fath, B., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2018; pp. 310–321. [Google Scholar] [CrossRef]
- Lewis, V.P.; Yang, S.T. Propionic acid fermentation by Propionibacterium acidipropionici: Effect of growth substrate. Appl. Microbiol. Biotechnol. 1992, 37, 437–442. [Google Scholar] [CrossRef]
- Fitz, A. Über Spaltpilzgärung, IV. Ber. Deut. Chem. Ges. 1878, 11, 1896–1899. [Google Scholar]
- Guan, N.; Liu, L. Microbial response to acid stress: Mechanisms and applications. Appl. Microbiol. Biotechnol. 2020, 104, 51–65. [Google Scholar] [CrossRef]
- Theron, M.M.; Lues, J.F.R. Organic Acids and Food Preservation, 1st ed.; CRC Press: Basel, Switzerland, 2010. [Google Scholar] [CrossRef]
- Blanc, P.; Goma, G. Propionic acid fermentation: Improvement of performances by coupling continuous fermentation and ultrafiltration. Bioprocess. Biosyst. Eng. 1987, 2, 137–139. [Google Scholar] [CrossRef]
- Ibragimova, S.I.; Sakharova, Z.V. Sviaz’ mezhdu udel’noĭ skorost’iu rosta i kolichestvom potreblennogo substrata u Probionibacterium shermanii [Relation between specific growth rate and amount of consumed substrate in Propionibacterium shermanii]. Dokl. Akad. Nauk. SSSR 1974, 219, 988–989. [Google Scholar]
- Kusano, K.; Yamada, H.; Niwa, M.; Yamasato, K. Propionibacterium cyclohexanicum sp. nov., a new acid-tolerant omega-cyclohexyl fatty acid-containing propionibacterium isolated from spoiled orange juice. Int. J. Syst. Bact. 1997, 47, 825–831. [Google Scholar] [CrossRef]
- Thierry, A.; Berthier, F.; Gagnaire, V.; Kerjean, J.R.; Lopez, C.; Noël, Y. 10. Eye Formation and Swiss-Type Cheeses. In Technology of Cheesemaking, 2nd ed.; Law, B.A., Tamime, A.Y., Eds.; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2010; pp. 360–383. [Google Scholar]
- Zarate, G.; Perez Chaia, A.; Guillermo, O. Some characteristics of practical relevance of the β-galactosidase from potential probiotic strains of Propionibacterium acidipropionici. Anaerobe 2002, 8, 259–267. [Google Scholar] [CrossRef]
- Piveteau, P. Metabolism of lactate and sugars by dairy propionibacteria: A review. Le Lait 1999, 79, 23–41. [Google Scholar] [CrossRef]
- Johns, A.T. The mechanism of propionic acid formation by propionibacteria. J. Gen. Microbiol. 1951, 5, 337–345. [Google Scholar] [CrossRef][Green Version]
- Ahmadi, N.; Khosravi-Darani, K.; Mortazavian, A.M. An overview of biotechnological production of propionic acid: From upstream to downstream processes. Electron. J. Biotechnol. 2017, 28, 67–75. [Google Scholar] [CrossRef]
- Bücher, C.; Burtscher, J.; Domig, K.J. Propionic acid bacteria in the food industry: An update on essential traits and detection methods. Compr. Rev. Food Sci. Food Saf. 2021, 20, 4299–4323. [Google Scholar] [CrossRef] [PubMed]
- Boyaval, P.; Corre, C. Production of propionic acid. Le Lait 1995, 75, 453–461. [Google Scholar] [CrossRef]
- Pulgar, M.J.A. The ESKAPE Bacteria Group and Its Clinical Importance. CLOVER BioSoft. 2019. Available online: https://cloverbiosoft.com/the-eskape-bacteria-group-and-its-clinical-importance/ (accessed on 5 May 2022).
- Bushell, F.M.L.; Tonner, P.D.; Jabbari, S.; Schmid, A.K.; Lund, P.A. Synergistic impacts of organic acids and ph on growth of Pseudomonas aeruginosa: A comparison of parametric and bayesian non-parametric methods to model growth. Front. Microbiol. 2019, 9, 3196. [Google Scholar] [CrossRef] [PubMed]
- Gwiazdowska, D.; Trojanowska, K. Antimicrobial activity and stability of partially purified bacteriocins produced by Propionibacterium freudenreichii ssp. freudenreichii and ssp. shermanii. Le Lait 2006, 86, 141–154. [Google Scholar] [CrossRef]
- Warmińska-Radyko, I.; Łaniewska-Moroz, Ł. Antibacterial activity of different fractions from the culture of Propionibacterium acidipropionici. Pol. J. Food Nutr. Sci. 1999, 49, 23–29. [Google Scholar]
- Cobirka, M.; Tancin, V.; Slama, P. Epidemiology and classification of mastitis. Animals 2020, 10, 2212. [Google Scholar] [CrossRef]
- Rios, V.M. Predictive Food Microbiology—New Models for Safety and Quality Assessment of a Broad Range of Dairy Products. Ph.D. Thesis, Technical University of Denmark, Kongens Lyngby, Denmark, 2019. [Google Scholar]
- Arslan, S.; Eyi, A.; Özdemir, F. Spoilage potentials and antimicrobial resistance of Pseudomonas spp. isolated from cheeses. J. Dairy Sci. 2011, 94, 5851–5856. [Google Scholar] [CrossRef]
- Raposo, A.; Pérez, E.; de Faria, C.T.; Ferrús, M.A.; Carrascosa, C. Food spoilage by Pseudomonas spp.—An overview. In Foodborne Pathogens and Antibiotic Resistance; Singh, O.V., Ed.; John Wiley & Sons: Haboken, NJ, USA, 2016. [Google Scholar] [CrossRef]
- Lopez-Brea, S.G.; Gómez-Torres, N.; Arribas, M.Á. Spore-forming bacteria in dairy products. In Microbiology in Dairy Processing: Challenges and Opportunities; Poltronieri, P., Ed.; John Wiley & Sons: Haboken, NJ, USA, 2017; pp. 11–36. [Google Scholar] [CrossRef]
- Eghbal, N.; Liao, W.; Dumas, E.; Azabou, S.; Dantigny, P.; Gharsallaoui, A. Microencapsulation of natural food antimicrobials: Methods and applications. Appl. Sci. 2022, 12, 3837. [Google Scholar] [CrossRef]


| Microorganisms | Strain Abbreviation Used in the Article | Species | Strain No. and the Source |
|---|---|---|---|
| Propionibacteria | A | Acidipropionibacterium acidipropionici | DSM 20273 (DSMZ, Germany) |
| C | Acidipropionibacterium cyclohexanicum | DSM 16859 (DSMZ, Germany) | |
| E | Propionibacterium freudenreichii | DSM 4902 (DSMZ, Germany) | |
| J | Acidipropionibacterium jensenii | DSM 20535 (DSMZ, Germany) | |
| T | Acidipropionibacterium thoenii | DSM 20276 (DSMZ, Germany) | |
| Foodborne pathogens | E. coli | Escherichia coli | ATCC® 8739 (Manassas, VA, USA) |
| K. pneumoniae | Klebsiella pneumoniae | MSCL535 (Microbial Strain Collection of Latvia, Riga, Latvia) | |
| P. aeruginosa | Pseudomonas aeruginosa | ATCC® 9027™ (USA) | |
| B. subtilis | Bacillus subtilis subsp. spizizenii | ATCC® 6633™ (USA) | |
| S. aureus | Staphylococcus aureus | ATCC® 6538P (USA) |
| Broth Type | PAB Strain | Produced Acids, g·L−1 | P/A * Ratio | pH | ||||
|---|---|---|---|---|---|---|---|---|
| Propionic | Acetic | Succinic | Lactic | Pyruvic | ||||
| Lactate-based | A | (8.35 ± 0.21) a | (3.84 ± 0.09) a | (0.23 ± 0.03) a | n.d. | (0.15 ± 0.01) a | (2.17 ± 0.01) a | (6.47 ± 0.01) a |
| C | (8.08 ± 0.47) ab | (3.70 ± 0.25) ab | (0.30 ± 0.02) bd | n.d. | (0.27 ± 0.04) ac | (2.18 ± 0.02) a | (6.44 ± 0.06) ab | |
| E | (7.18 ± 0.36) b | (3.32 ± 0.14) b | (0.49 ± 0.01) ce | n.d. | (0.11 ± 0.00) bd | (2.16 ± 0.02) a | (6.50 ± 0.05) ab | |
| J | (7.90 ± 0.23) ab | (3.51 ± 0.08) b | (0.31 ± 0.01) db | n.d. | (0.24 ± 0.04) ca | (2.25 ± 0.02) b | (6.55 ± 0.05) b | |
| T | (8.61 ± 0.51) a | (3.87 ± 0.17) a | (1.34 ± 0.37) ec | n.d. | (0.11 ± 0.00) bd | (2.23 ± 0.04) ab | (6.46 ± 0.01) a | |
| Lactose-based | A | (4.20 ± 0.26) ab | (1.68 ± 0.01) abc | n.d. | n.d. | (0.09 ± 0.02) a | (2.50 ± 0.15) a | (4.26 ± 0.01) a |
| C | (4.26 ± 0.28) a | (1.84 ± 0.04) a | (0.51 ± 0.02) a | (2.40 ± 0.12) a | (0.13 ± 0.00) b | (2.31 ± 0.12) a | (3.84 ± 0.02) b | |
| E | (4.20 ± 0.10) a | (2.15 ± 0.06) b | (0.07 ± 0.01) b | n.d. | (0.14 ± 0.01) b | (1.96 ± 0.04) b | (4.27 ± 0.01) c | |
| J | (3.54 ± 0.03) b | (1.83 ± 0.05) ac | (0.07 ± 0.01) b | 0.00 | (0.11 ± 0.00) a | (1.94 ± 0.03) b | (4.36 ± 0.01) d | |
| T | (3.77 ± 0.38) ab | (1.43 ± 0.07) c | (0.16 ± 0.03) c | (0.17 ± 0.02) b | (0.82 ± 0.07) c | (2.64 ± 0.14) a | (4.06 ± 0.01) e | |
| Broth Type | PAB Strains | Consumed Substrate, g·L−1 | Acid Titers and Yields | Substrate Conversion Efficiency, % | Biomass Yield, g·g−1 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Propionic Acid | Acetic Acid | Propionic Acid | Acetic Acid | ||||||||
| Titer, g·L−1 | Yield, g·g−1 | Titer, g·L−1 | Yield, g·g−1 | ||||||||
| Theoretical * | Observed | Theoretical * | Observed | ||||||||
| Lactate-based | A | 15.94 | 8.74 | (8.35 ± 0.21) a | (0.52 ± 0.01) a | 3.54 | (3.84 ± 0.09) a | (0.24 ± 0.01) a | (95.56 ± 2.42) a | (108.52 ± 2.42) a | (0.06 ± 0.00) a |
| C | 15.94 | 8.74 | (8.08 ± 0.47) ab | (0.51 ± 0.03) ab | 3.54 | (3.70 ± 0.25) ad | (0.23 ± 0.02) ab | (92.53 ± 5.36) ab | (104.52 ± 7.04) ab | (0.03 ± 0.00) bc | |
| E | 15.94 | 8.74 | (7.18 ± 0.36) b | (0.45 ± 0.02) b | 3.54 | (3.32 ± 0.14) b | (0.21 ± 0.01) b | (82.16 ± 4.18) b | (93.87 ± 3.99) b | (0.03 ± 0.01) bc | |
| J | 15.94 | 8.74 | (7.90 ± 0.23) ab | (0.50 ± 0.01) ab | 3.54 | (3.51± 0.08) b | (0.22 ± 0.01) b | (91.45 ± 2.61) ab | (99.25 ± 2.29) b | (0.02 ± 0.00) d | |
| T | 15.94 | 8.74 | (8.61 ± 0.51) a | (0.54 ± 0.03) a | 3.54 | (3.87 ± 0.17) a | (0.24 ± 0.01) a | (98.52 ± 5.83) a | (109.19 ± 4.87) a | (0.09 ± 0.00) e | |
| Lactose-based | A | (10.13 ± 2.29) ab | (5.55 ± 1.25) ab | (4.20 ± 0.26) ab | (0.43 ± 0.12) a | (2.25 ± 0.51) ab | (1.68 ± 0.01) a | (0.17 ± 0.04) ab | (78.16 ± 22.32) a | (76.63 ± 17.54) ab | (0.14 ± 0.04) ab |
| C | (12.40 ± 1.67) a | (6.80 ± 0.91) a | (4.26 ± 0.28) a | (0.35 ± 0.08) a | (2.76 ± 0.37) a | (1.84 ± 0.04) bd | (0.15 ± 0.02) a | (64.11 ± 14.36) a | (67.07 ± 10.65) a | (0.12 ± 0.02) a | |
| E | (9.12 ± 0.53) b | (5.00 ± 0.29) b | (4.20 ± 0.10) a | (0.46 ± 0.02) a | (2.03 ± 0.12) b | (2.15 ± 0.06) c | (0.24 ± 0.01) b | (84.16 ± 3.27) a | (105.94 ± 3.45) b | (0.22 ± 0.02) b | |
| J | (8.58 ± 0.58) ab | (4.70 ± 0.32) ab | (3.54 ± 0.03) b | (0.41 ± 0.03) a | (1.91 ± 0.13) ab | (1.83± 0.05) bd | (0.21 ± 0.02) ab | (75.40 ± 5.66) a | (96.18 ± 8.90) ab | (0.43 ± 0.02) c | |
| T | (11.72 ± 4.92) ab | (6.43 ± 2.70) ab | (3.77 ± 0.38) ab | (0.36 ± 0.18) a | (2.61 ± 1.09) ab | (1.43 ± 0.07) e | (0.13 ± 0.06) ab | (65.68 ± 33.51) a | (60.65 ± 28.17) ab | (0.11 ± 0.05) a | |
| Tested Pathogens | PAB Strains Producing Fermentates | Survival Rate, % | Does Treatment with Proteinase Decrease the Inhibitory Effect? * | The Inhibition Potential of FS-Type Fermentates ** | ||
|---|---|---|---|---|---|---|
| Tested Fermentate Type | The Difference (FSP − FS), % | |||||
| FS | FSP | |||||
| E. coli | E | (58.08 ± 2.04) a | (58.26 ± 1.80) a | 0.18 | no | moderate |
| T | (52.45 ± 0.29) a | (53.84 ± 3.48) a | 1.38 | no | moderate | |
| A | (57.11 ± 0.97) a | (62.00 ± 1.24) b | 4.89 | yes | moderate | |
| K. pneumoniae | E | (94.33 ± 0.91) a | (121.56 ± 2.35) b | 27.23 | yes | weak |
| T | (84.68 ± 1.97) a | (108.85 ± 3.45) b | 24.17 | yes | weak | |
| A | (83.72 ± 0.99) a | (109.33 ± 1.44) b | 25.61 | yes | weak | |
| P. aeruginosa | E | (2.73 ± 0.60) a | (1.96 ± 0.25) a | −0.77 | no | strong |
| T | (2.06 ± 0.24) a | (1.05 ± 0.05) b | −1.01 | no | strong | |
| A | (7.70 ± 0.84) a | (4.09 ± 0.63) b | −3.62 | no | strong | |
| B. subtilis | E | (33.13 ± 2.80) a | (22.35 ± 4.17) b | −10.78 | no | moderate |
| T | (20.50 ± 2.73) a | (17.50 ± 3.59) a | −3.00 | no | strong | |
| A | (48.07 ± 2.63) a | (50.52 ± 8.23) a | 2.45 | no | moderate | |
| S. aureus | E | (11.42 ± 1.18) a | (70.06 ± 4.76) b | 58.65 | yes | strong |
| T | (25.21 ± 2.47) a | (108.78 ± 3.60) b | 83.57 | yes | strong | |
| A | (45.02 ± 4.14) a | (128.45 ± 8.99) b | 83.42 | yes | moderate | |
| Tested Microorganisms | MIC, mM·L−1 | |||
|---|---|---|---|---|
| Lactic Acid | Propionic Acid | Acetic Acid | Formic Acid | |
| E. coli | (9.9 ± 0.1) a | (7.0 ± 0.2) b | (6.8 ± 0.4) b | (6.0 ± 0.0) c |
| K. pneumoniae | (12.1 ± 0.5) a | (6.1 ± 0.1) b | (6.2 ± 0.1) b | (6.7 ± 0.7) b |
| P. aeruginosa | (8.1 ± 0.0) a | (5.7 ± 0.4) b | (5.5 ± 0.4) b | (5.3 ± 0.7) b |
| B. subtilis | (6.1 ± 0.1) a | (3.6 ± 0.3) b | (4.3 ± 0.1) c | (5.6 ± 0.6) a |
| S. aureus | (14.0 ± 0.2) a | (10.2 ± 0.9) b | (10.9 ± 0.8) b | (11.0 ± 0.3) b |
| Comparable Variants of Fermentate pH | The Acid Present in Fermentate | Acid Concentration, mM·L−1 | pKa | HA, mM·L−1 | A−, mM·L−1 |
|---|---|---|---|---|---|
| 6.5 (actual) | propionic | 100 | 4.87 | 2 | 98 |
| acetic | 60 | 4.76 | 1 | 59 | |
| 5.5 (advisable) | propionic | 100 | 4.87 | 19 | 81 |
| acetic | 60 | 4.76 | 9 | 51 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antone, U.; Ciprovica, I.; Zolovs, M.; Scerbaka, R.; Liepins, J. Propionic Acid Fermentation—Study of Substrates, Strains, and Antimicrobial Properties. Fermentation 2023, 9, 26. https://doi.org/10.3390/fermentation9010026
Antone U, Ciprovica I, Zolovs M, Scerbaka R, Liepins J. Propionic Acid Fermentation—Study of Substrates, Strains, and Antimicrobial Properties. Fermentation. 2023; 9(1):26. https://doi.org/10.3390/fermentation9010026
Chicago/Turabian StyleAntone, Unigunde, Inga Ciprovica, Maksims Zolovs, Rita Scerbaka, and Janis Liepins. 2023. "Propionic Acid Fermentation—Study of Substrates, Strains, and Antimicrobial Properties" Fermentation 9, no. 1: 26. https://doi.org/10.3390/fermentation9010026
APA StyleAntone, U., Ciprovica, I., Zolovs, M., Scerbaka, R., & Liepins, J. (2023). Propionic Acid Fermentation—Study of Substrates, Strains, and Antimicrobial Properties. Fermentation, 9(1), 26. https://doi.org/10.3390/fermentation9010026








