Fermentative Production of Volatile Metabolites Using Brettanomyces bruxellensis from Fruit and Vegetable By-Products
Abstract
1. Introduction
2. Materials and Methods
2.1. By-Products from New Zealand Industries
2.2. Yeast Strain
2.3. Submerged Solid-Substrate and Liquid Fermentations
2.4. Headspace Solid-Phase Microextraction (HS-SPME) Coupled with Gas Chromatography–Mass Spectrometry (GC-MS)
2.5. SPME-GCMS Data Processing
2.6. Identification Confirmation and Quantification of Volatile Compounds
2.7. Gas Chromatography–Mass Spectrometry Conditions
3. Results
3.1. Substrate Composition
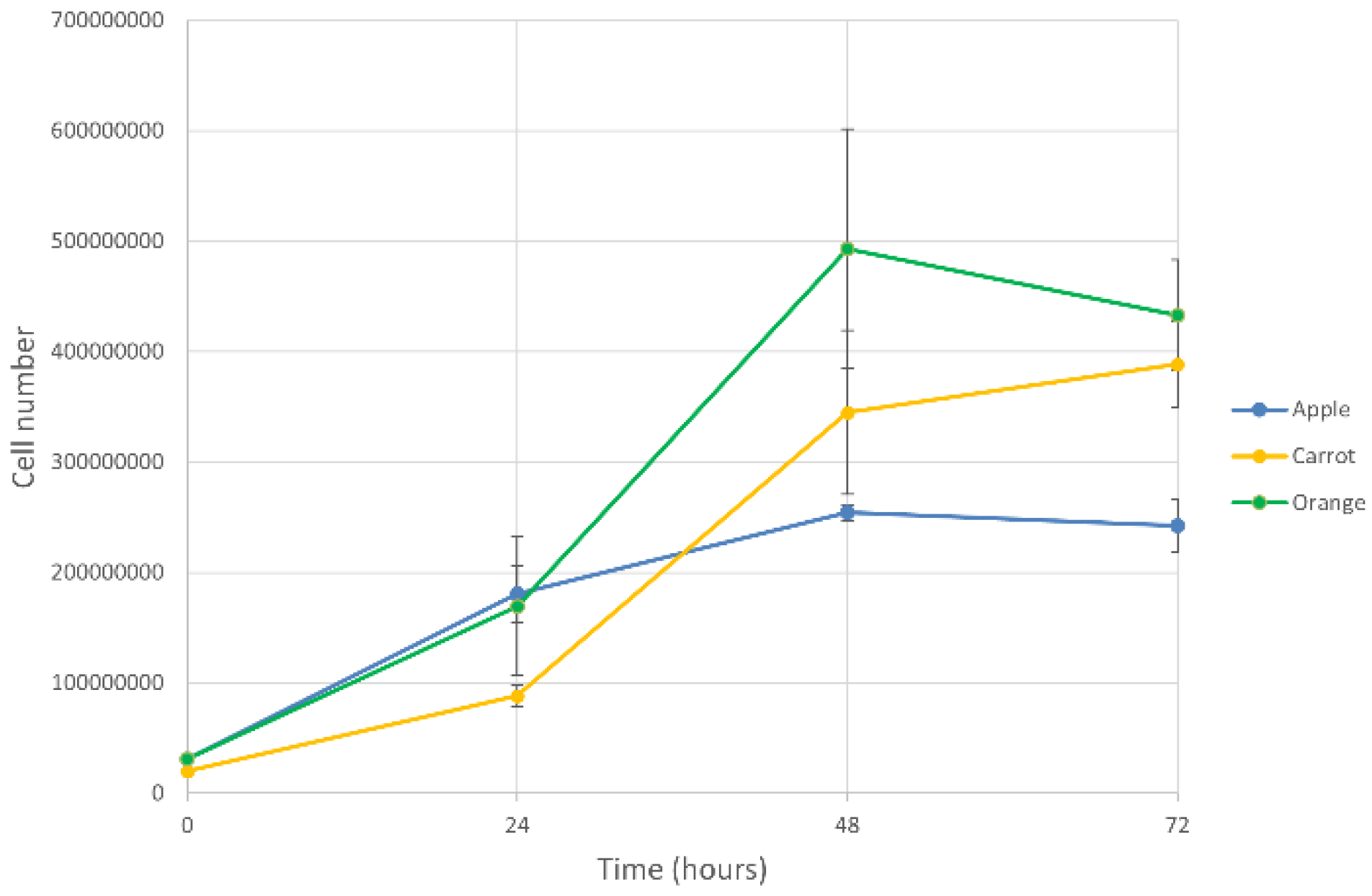
3.2. Fermentation Performance
3.3. Profiling of Volatile Compounds
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goddard, M.R. Microbiology: Mixing Wine, Chocolate, and Coffee. Curr. Biol. Dispatches 2016, 26, R275–R277. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Button, J.E.; Dutton, R.J. Cheese microbes. Curr. Biol. 2012, 22, R587–R589. [Google Scholar] [CrossRef] [PubMed]
- Hugenholtz, J. Traditional biotechnology for new foods and beverages. Curr. Opin. Biotechnol. 2013, 24, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Igrejas, G.; Falco, V.; Santos, T.P.; Torres, C.; Oliveira, A.M.P.; Pereira, J.E.; Amaral, J.S.; Poeta, P. Chemical composition, antioxidant and antimicrobial activity of phenolic compounds extracted from wine industry by-products. Food Control. 2018, 92, 516–522. [Google Scholar] [CrossRef]
- Kowalska, H.; Czajkowska, K.; Cichowska, J.; Lenart, A. What’s new in biopotential of fruit and vegetable by-products applied in the food processing industry. Trends Food Sci. Technol. 2017, 67, 150–159. [Google Scholar]
- Schieber, A.; Stintzing, F.C.; Carle, R. By-products of plant food processing as a source of functional compounds—Recent developments. Trends Food Sci. Technol. 2001, 12, 401–413. [Google Scholar]
- Brar, S.K.; Dhillon, G.S.; Soccol, C.R. Biotransformation of Waste Biomass into High Value Biochemicals, 1st ed.; Springer Science & Business Media: New York, NY, USA, 2013; 504p. [Google Scholar]
- Wedral, D.; Shewfelt, R.; Frank, J. The challenge of Brettanomyces in wine. LWT-Food Sci. Technol. 2010, 43, 1474–1479. [Google Scholar] [CrossRef]
- Steensels, J.; Daenen, L.; Malcorps, P.; Derdelinckx, G.; Verachtert, H.; Verstrepen, K.J. Brettanomyces yeasts—From spoilage organisms to valuable contributors to industrial fermentations. Int. J. Food Microbiol. 2015, 206, 24–38. [Google Scholar]
- Snowdon, E.M.; Bowyer, M.C.; Grbin, P.R.; Bowyer, P.K. Mousy off-Flavor: A review. J. Agric. Food Chem. 2006, 54, 6465–6474. [Google Scholar] [CrossRef]
- Renouf, V.; Lonvaud-Funel, A.; Coulon, J. The origin of Brettanomyces bruxellensis in wines: A review. J. Int. Sci. Vigne Vin 2007, 41, 161–173. [Google Scholar] [CrossRef]
- Schifferdecker, A.J.; Dashko, S.; Ishchuk, O.P.; Piškur, J. The wine and beer yeast Dekkera bruxellensis. Yeast 2014, 31, 323–332. [Google Scholar] [CrossRef] [PubMed]
- De Barros Pita, W.; Tiukova, I.; Leite, F.C.B.; Passoth, V.; Simões, D.A.; De Morais, M.A. The influence of nitrate on the physiology of the yeast Dekkera bruxellensis grown under oxygen limitation. Yeast 2013, 30, 111–117. [Google Scholar] [CrossRef]
- Granucci, N. Fruit Residues: Low-Cost Substrates for Development of New Food Products. Ph.D. Thesis, The University of Auckland, Auckland, New Zealand, 2018. [Google Scholar]
- Villas-Boas, S.G.; Esposito, E.; Mendonça, M.M. Novel lignocellulolytic ability of Candida utilis during solid-substrate cultivation on apple pomace. World J. Microbiol. Biotechnol. 2002, 18, 541–545. [Google Scholar] [CrossRef]
- Olivier, F.A.B.; Bang, K.W.; Zarate, E.; Kinzurik, M.; Chudakova, D.; Ganley, A.R.D.; Villas-Boas, S.G. Aerial warfare: An inducible production of volatile bioactive metabolites in a novel species of Scytinostroma sp. Fungal Genet. Biol. 2021, 158, 103646. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.L. Use of dinitrosalicilic reagent for determination of reducing sugars. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Lindsay, M.A.; Granucci, N.; Greenwood, D.R.; Villas-Boas, S.G. Identification of new natural sources of flavour and aroma metabolites from solid-state fermentation of agro-industrial by-products. Metabolites 2022, 12, 157. [Google Scholar] [CrossRef]
- Schwab, W.; Davidovich-Rikanati, R.; Lewinsohn, E. Biosynthesis of plant-derived flavor compounds. Plant J. 2008, 54, 712–732. [Google Scholar] [CrossRef]
- Beltran, G.; Esteve-Zarzoso, B.; Rozes, N.; Mas, A.; Guillamon, J.M. Influence of the timing of nitrogen additions during synthetic grape must fermentations on fermentation kinetics and nitrogen consumption. J. Agric. Food Chem. 2005, 53, 996–1002. [Google Scholar] [CrossRef]
- Deed, N.K.; van Vuuren, H.J.J.; Gardner, R.C. Effects of nitrogen catabolite repression and di-ammonium phosphate addition during wine fermentation by a commercial strain of S. cerevisiae. Appl. Microbiol. Biotechnol. 2011, 89, 1537–1549. [Google Scholar] [CrossRef]
- Baydar, H. Oil-bearing rose (Rosa Damascena Mill.), Cultivation and rose oil industry in Turkey. Ind. Crops Prod. 2006, 14, 13–17. [Google Scholar]
- Etschmann, M.; Bluemke, W.; Sell, D.; Schrader, J. Biotechnological production of 2-phenylethanol. Appl. Microbiol. Biotechnol. 2002, 59, 1–8. [Google Scholar] [PubMed]
- Mei, J.; Min, H.; Lü, Z. Enhanced biotransformation of L-phenylalanine to 2-phenylethanol using an in situ product adsorption technique. Process Biochem. 2009, 44, 886–890. [Google Scholar] [CrossRef]
- Mitri, S.; Koubaa, M.; Maroun, R.G.; Rossignol, T.; Nicaud, J.-M.; Louka, N. Bioproduction of 2-phenylethanol through yeast fermentation on synthetic media and on agro-industrial waste and by-products: A review. Foods 2022, 11, 109. [Google Scholar] [CrossRef] [PubMed]
- Conde-Báez, L.; Castro-Rosas, J.; Villagómez-Ibarra, J.R.; Páez-Lerma, J.B.; Gómez-Aldapa, C. Evaluation of waste of the cheese industry for the production of aroma of roses (phenylethyl alcohol). Waste Biomass Valorization 2017, 8, 1343–1350. [Google Scholar] [CrossRef]
- Yusef, C. Fermentation (Industrial): Production of oils and fatty acids. In Encyclopedia of Food Microbiology; Robinson, A., Batt, C., Patel, P., Eds.; Academic Press: London, UK, 1999; pp. 663–673. [Google Scholar]
- Ghosh, S.; Kebaara, B.W.; Atkin, A.L.; Nickerson, K.W. Regulation of aromatic alcohol production in Candida albicans. Appl. Environ. Microbiol. 2008, 74, 7211–7218. [Google Scholar] [CrossRef]
- Celińska, E.; Kubiak, P.; Białas, W.; Dziadas, M.; Grajek, W. Yarrowia lipolytica: The novel and promising 2-phenylethanol producer. J. Ind. Microbiol. Biotechnol. 2013, 40, 389–392. [Google Scholar] [CrossRef]
- Akita, O.; Ida, T.; Obata, T.; Hara, S. Mutants of Saccharomyces cerevisiae producing a large quantity of phenethyl alcohol and phenethyl acetate. J. Ferment. Bioeng. 1990, 69, 125–128. [Google Scholar] [CrossRef]
- Ayrapaa, T. The formation of phenlethyl alcohol from 14C-labelled phenylalanine. J. Inst. Brew. 1965, 71, 341–347. [Google Scholar] [CrossRef]
- Liu, S.Q. Impact of yeast and bacteria on beer appearance and flavour. In Brewing Microbiology; Woodhead Publishing: Sawston, UK, 2015; pp. 357–374. [Google Scholar]
- Kim, B.; Cho, B.-R.; Hahn, J.-S. Metabolic engineering of Saccharomyces cerevisiae for the production of 2-phnylethanol via Ehrlich pathway. Biotechnol. Bioeng. 2014, 111, 115–124. [Google Scholar] [CrossRef]
- Garavaglia, J.; Flôres, S.H.; Pizzolato, T.M.; Peralba, M.C.; Ayub, M.A.Z. Bioconversion of L-phenylalanine into 2-phenylethanol by Kluyveromyces marxianus in grape must cultures. World J. Microbiol. Biotechnol. 2007, 23, 1273–1279. [Google Scholar] [CrossRef]

| Components (%DM) | Apple Pomace | Orange Pomace | Carrot Pomace |
|---|---|---|---|
| Moisture | 79.0 | 78.0 | 87.5 |
| Nitrogen | 0.6 | 0.7 | 1.1 |
| Reducing sugars | 37.3 | 33.0 | 10.9 |
| Fibre | 39.5 | 34.3 | 53.4 |
| Ash | 1.5 | 3.2 | 6.2 |
| pH (units) | 3.4 | 4.0 | 5.1 |
| Compound | Substrate | Fold Change * | Descriptor |
|---|---|---|---|
| 2-Methylbutan-1-ol | Carrot | 887.8 | Black truffle |
| 3-Methylbutan-1-ol- | Carrot | 797.9 | Fusel, banana |
| Orange | 230.8 | ||
| Acetic acid | Apple | 6.1 | Vinegar |
| Phenylmethanol | Apple | 5.0 | Sweet, floral, balsamic |
| 2-Methylbutanoic acid | Carrot | 392.2 | Acidic, cheesy |
| Orange | 543.5 | ||
| Ethyl 2-methylbutanoate | Orange | 256.6 | Fruity, green apple |
| Ethyl decanoate | Apple | 6.4 | Waxy, sweet, fruity |
| Methyl decanoate | Carrot | 2623.4 | Oily, wine-like |
| Ethyl dodecanoate | Orange | 68.8 | Sweet, waxy |
| Ethyl acetate | Apple | 5.9 | Ethereal, fruity |
| Ethyl heptanoate | Orange | 103.8 | Pineapple |
| Methyl heptanoate | Carrot | 370.4 | Orris, currant |
| Ethyl hexanoate | Orange | 87.3 | Sweet, fruity, pineapple |
| Methyl hexanoate | Carrot | 206.7 | Pineapple |
| 3-Methylbutyl acetate | Apple | 5.2 | Banana, fruity |
| 3-Methylbutyl hexanoate | Carrot | 107.6 | Apple, pineapple |
| Methyl pentanoate | Carrot | 899.5 | Fruity |
| Ethyl nonanoate | Apple | 55.7 | Waxy, fruity |
| Methyl nonanoate | Carrot | 351.8 | Wine, coconut |
| (3S)-7-Hydroxy-3,7-dimethyloctanal | Apple | 15.4 | Floral, lily, green |
| 3-Methylbutyl decanoate | Orange | 57.4 | Waxy, banana |
| 4-Ethyl-2-methoxyphenol | Apple | 91.1 | Smoky, spicy, clove, wine taint |
| Orange | 8358.9 | ||
| 2-Phenylethyl acetate | Apple | 22.4 | Floral, honey |
| Orange | 313.0 | ||
| 2-Phenylethan-1-ol (phenylethyl alcohol) | Carrot | 325.7 | Floral, rose |
| 2-Methylpropanoic acid | Orange | 295.9 | Acidic, sour, cheesy |
| Compound | Value (kg) | Annual Consumption (Tonnes) | Substrate | Yield (g/kg) 3 |
|---|---|---|---|---|
| Phenylethyl alcohol | USD 500.00 1 | 14,000 2 | Carrot pomace | 2.68 ± 0.280 |
| Synthetic medium | 0.01 ± 0.004 | |||
| Synthetic medium plus L-phenylalanine | 1.27 ± 0.080 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lindsay, M.A.; Granucci, N.; Greenwood, D.R.; Villas-Boas, S.G. Fermentative Production of Volatile Metabolites Using Brettanomyces bruxellensis from Fruit and Vegetable By-Products. Fermentation 2022, 8, 457. https://doi.org/10.3390/fermentation8090457
Lindsay MA, Granucci N, Greenwood DR, Villas-Boas SG. Fermentative Production of Volatile Metabolites Using Brettanomyces bruxellensis from Fruit and Vegetable By-Products. Fermentation. 2022; 8(9):457. https://doi.org/10.3390/fermentation8090457
Chicago/Turabian StyleLindsay, Melodie A., Ninna Granucci, David R. Greenwood, and Silas G. Villas-Boas. 2022. "Fermentative Production of Volatile Metabolites Using Brettanomyces bruxellensis from Fruit and Vegetable By-Products" Fermentation 8, no. 9: 457. https://doi.org/10.3390/fermentation8090457
APA StyleLindsay, M. A., Granucci, N., Greenwood, D. R., & Villas-Boas, S. G. (2022). Fermentative Production of Volatile Metabolites Using Brettanomyces bruxellensis from Fruit and Vegetable By-Products. Fermentation, 8(9), 457. https://doi.org/10.3390/fermentation8090457








