Effects of Different Additives on the Chemical Composition, Fermentation Profile, In Vitro and In Situ Digestibility of Paper mulberry Silage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Paper Mulberry Silage
2.2. Chemical Composition and Fermentation Profile Detection
2.3. In Vitro Fermentation and In Situ Digestibility Experiment
2.4. Calculations
2.4.1. Gas Production Parameters Calculation for the In Vitro Fermentation
2.4.2. The In Situ Digestibility Rate Calculation
2.5. Statistical Analysis
3. Results
3.1. Chemical Composition and Fermentation Profile of Silage
3.2. In Vitro Ruminal GP Characteristics, Ammonia-N, and VFA
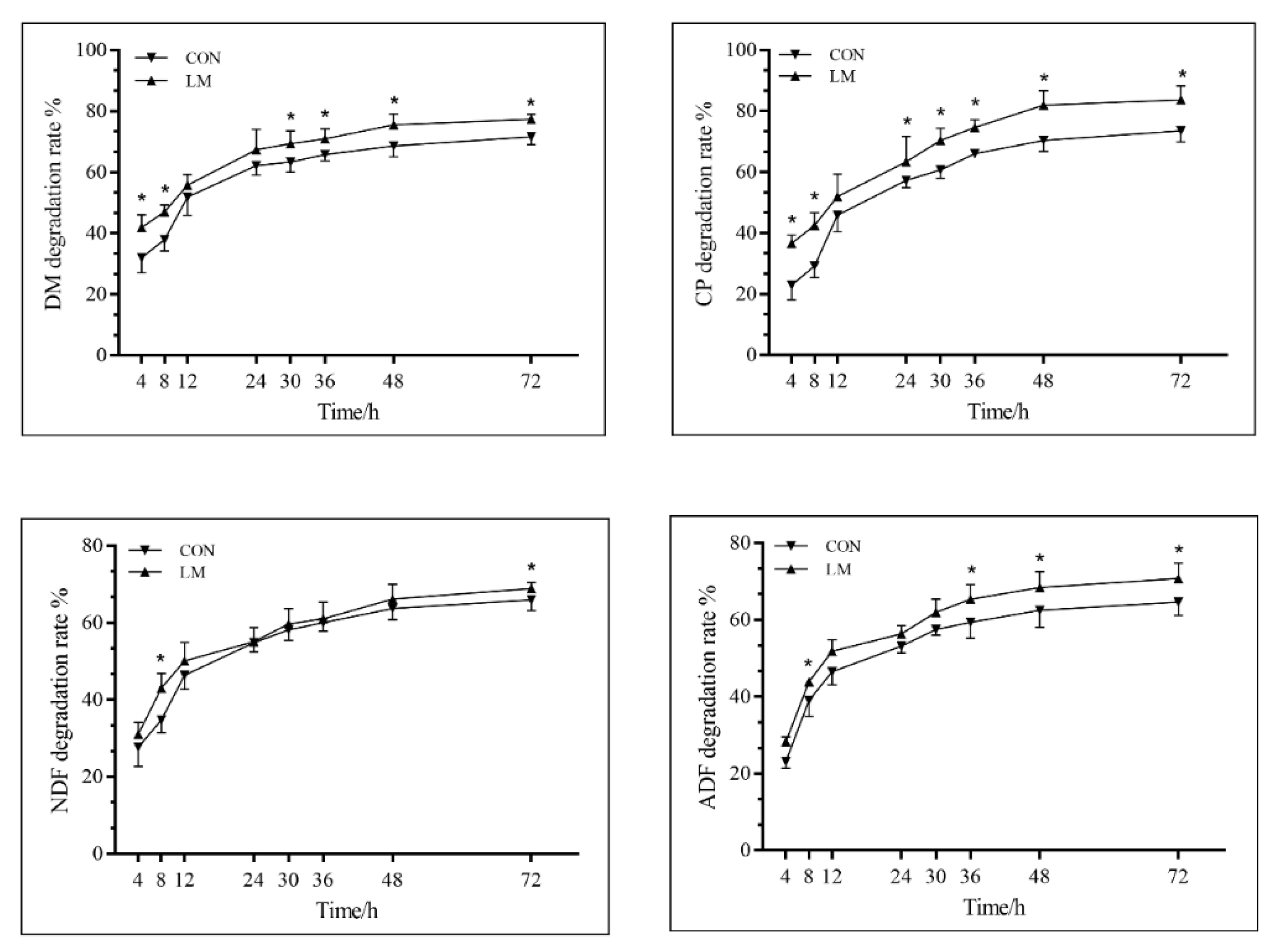
3.3. In Situ Ruminal Degradation Characteristics
4. Discussion
4.1. Chemical Composition and Fermentation Profile of Silage
4.2. In Vitro Ruminal GP Characteristics and Ammonia-N and VFA
4.3. In Situ Ruminal Degradation Characteristics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peng, X.; Wang, Y.; He, R.; Zhao, M.; Shen, S. Global transcriptomics identification and analysis of transcriptional factors in different tissues of the paper mulberry. BMC Plant Biol. 2014, 14, 194. [Google Scholar] [CrossRef] [PubMed]
- Li, G.Y.; Hu, N.; Ding, D.X.; Zheng, J.F.; Liu, Y.L.; Wang, Y.D.; Nie, X.Q. Screening of plant species for phytoremediation of uranium, thorium, barium, nickel, strontium and lead contaminated soils from a uranium mill tailings repository in South China. Bull. Environ. Contam. Toxicol. 2011, 86, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Penailillo, J.; Olivares, G.; Moncada, X.; Payacan, C.; Chang, C.S.; Chung, K.F.; Matthews, P.J.; Seelenfreund, A.; Seelenfreund, D. Sex Distribution of Paper Mulberry (Broussonetia papyrifera) in the Pacific. PLoS ONE 2016, 11, e0161148. [Google Scholar] [CrossRef]
- Si, B.; Tao, H.; Zhang, X.; Guo, J.; Cui, K.; Tu, Y.; Diao, Q. Effect of Broussonetia papyrifera L. (paper mulberry) silage on dry matter intake, milk composition, antioxidant capacity and milk fatty acid profile in dairy cows. Asian-Australas. J. Anim. Sci. 2018, 31, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Grant, R.J.; Ferraretto, L.F. Silage review: Silage feeding management: Silage characteristics and dairy cow feeding behavior. J. Dairy Sci. 2018, 101, 4111–4121. [Google Scholar] [CrossRef] [PubMed]
- Muck, R.E.; Nadeau, E.M.G.; McAllister, T.A.; Contreras-Govea, F.E.; Santos, M.C.; Kung, L., Jr. Silage review: Recent advances and future uses of silage additives. J. Dairy Sci. 2018, 101, 3980–4000. [Google Scholar] [CrossRef]
- Babaeinasab, Y.; Rouzbehan, Y.; Fazaeli, H.; Rezaei, J. Chemical composition, silage fermentation characteristics, and in vitro ruminal fermentation parameters of potato-wheat straw silage treated with molasses and lactic acid bacteria and corn silage. J. Anim. Sci. 2015, 93, 4377–4386. [Google Scholar] [CrossRef]
- Abbasi, M.; Rouzbehan, Y.; Rezaei, J.; Jacobsen, S.E. The effect of lactic acid bacteria inoculation, molasses or wilting on the fermentation quality and nutritive value of amaranth (Amaranthus hypochondriaus) silage. J. Anim. Sci. 2018, 96, 3983–3992. [Google Scholar] [CrossRef]
- Broderick, G.A.; Kang, J.H. Automated Simultaneous Determination of Ammonia and Total Amino Acids in Ruminal Fluid and In Vitro Media. J. Dairy Sci. 1980, 63, 64–75. [Google Scholar] [CrossRef]
- Yuan, X.J.; Wen, A.Y.; Wang, J.; Desta, S.T.; Dong, Z.H.; Shao, T. Effects of four short-chain fatty acids or salts on fermentation characteristics and aerobic stability of alfalfa (Medicago sativa L.) silage. J. Sci. Food Agric. 2018, 98, 328–335. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 17th ed.; AOAC International: Rockville, MD, USA, 2000. [Google Scholar]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Murphy, R.P. A method for the extraction of plant samples and the determination of total soluble carbohydrates. J. Agric. Food Chem. 1958, 9, 715–717. [Google Scholar] [CrossRef]
- Bai, S.; Cao, Z.J.; Cao, B.B.; Yang, H.J.; Li, S.L.; Liu, J.X. Effects of different forage combinations in total mixed rations on in vitro gas production kinetics, ruminal and milk fatty acid profiles of lactating cows. Anim. Sci. J. 2018, 89, 1261–1270. [Google Scholar] [CrossRef]
- Menke, K.H.; Stengass, H. Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid. Anim. Res. Dev. 1988, 28, 7–55. [Google Scholar]
- Zhang, D.F.; Yang, H.J. In vitro ruminal methanogenesis of a hay-rich substrate in response to different combination supplements of nitrocompounds; pyromellitic diimide and 2-bromoethanesulphonate. Anim. Feed Sci. Technol. 2011, 163, 20–32. [Google Scholar] [CrossRef]
- Mehrez, A.Z.; Orskov, E.R.; Mcdonald, I. Rates of Rumen Fermentation in Relation to Ammonia Concentration. Br. J. Nutr. 1977, 38, 437–443. [Google Scholar] [CrossRef]
- Groot, J.C.J.; Cone, J.W.; Williams, B.A.; Debersaques, F.M.A.; Lantinga, E.A. Multiphasic analysis of gas production kinetics for in vitro fermentation of ruminant feeds. Anim. Feed Sci. Technol. 1996, 64, 77–89. [Google Scholar] [CrossRef]
- Orskov, E.R.; McDonald, I. Estimation of Protein Degradability in the Rumen from Incubation Measurements Weighted According to Rate of Passage. J. Agric. Sci. 1979, 92, 499–503. [Google Scholar] [CrossRef]
- Batajoo, K.K.; Shaver, R.D. Impact of nonfiber carbohydrate on intake, digestion, and milk production by dairy cows. J. Dairy Sci. 1994, 77, 1580–1588. [Google Scholar] [CrossRef]
- Rezaei, J.; Rouzbehan, Y.; Fazaeli, H. Nutritive value of fresh and ensiled amaranth (Amaranthus hypochondriacus) treated with different levels of molasses. Anim. Feed Sci. Technol. 2009, 151, 153–160. [Google Scholar] [CrossRef]
- Yang, J.; Tan, H.; Cai, Y. Characteristics of lactic acid bacteria isolates and their effect on silage fermentation of fruit residues. J. Dairy Sci. 2016, 99, 5325–5334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, V.P.; Pereira, O.G.; Leandro, E.S.; Da Silva, T.C.; Ribeiro, K.G.; Mantovani, H.C.; Santos, S.A. Effects of lactic acid bacteria with bacteriocinogenic potential on the fermentation profile and chemical composition of alfalfa silage in tropical conditions. J. Dairy Sci. 2016, 99, 1895–1902. [Google Scholar] [CrossRef]
- Xia, C.; Liang, Y.; Bai, S.; He, Y.; Muhammad, A.U.R.; Su, H.; Cao, B. Effects of harvest time and added molasses on nutritional content, ensiling characteristics and in vitro degradation of whole crop wheat. Asian-Australas. J. Anim. Sci. 2018, 31, 354. [Google Scholar] [CrossRef] [PubMed]
- Hashemzadeh-Cigari, F.; Khorvash, M.; Ghorbani, G.; Ghasemi, E.; Taghizadeh, A.; Kargar, S.; Yang, W. Interactive effects of molasses by homofermentative and heterofermentative inoculants on fermentation quality, nitrogen fractionation, nutritive value and aerobic stability of wilted alfalfa (Medicago sativa L.) silage. J. Anim. Physiol. Anim. Nutr. 2014, 98, 290–299. [Google Scholar] [CrossRef]
- Raffrenato, E.; Fievisohn, R.; Cotanch, K.W.; Grant, R.J.; Chase, L.E.; Van Amburgh, M.E. Effect of lignin linkages with other plant cell wall components on in vitro and in vivo neutral detergent fiber digestibility and rate of digestion of grass forages. J. Dairy Sci. 2017, 100, 8119–8131. [Google Scholar] [CrossRef]
- Satter, L.D. Protein supply from undegraded dietary protein. J. Dairy Sci. 1986, 69, 2734–2749. [Google Scholar] [CrossRef]
- Li, Q.; Xue, B.; Zhao, Y.; Wu, T.; Liu, H.; Yi, X.; Sun, C.; Wang, Z.; Zou, H.; Yan, T. In situ degradation kinetics of 6 roughages and the intestinal digestibility of the rumen undegradable protein. J. Anim. Sci. 2018, 96, 4835–4844. [Google Scholar] [CrossRef] [PubMed]
| Groups 1 | DM | Chemical Composition 2 (% DM) | |||||
|---|---|---|---|---|---|---|---|
| CP | NDF | ADF | EE | ASH | WSC | ||
| Fresh | 26.76 ± 0.36 | 18.24 ± 1.03 | 35.68 ± 1.42 | 34.12 ± 0.87 | 3.45 ± 0.57 | 9.86 ± 0.45 b | 3.32 ± 0.03 a |
| CON | 27.35 ± 0.27 | 18.19 ± 1.97 | 39.15 ± 0.78 | 32.46 ± 1.38 | 3.85 ± 0.75 | 11.51 ± 0.33 a | 2.26 ± 0.02 b |
| LAB | 27.13 ± 0.33 | 19.03 ± 0.34 | 40.15 ± 0.92 | 33.05 ± 1.09 | 4.12 ± 0.03 | 12.23 ± 0.79 a | 1.68 ± 0.02 c |
| MOL | 26.86 ± 0.38 | 18.53 ± 0.16 | 44.37 ± 1.29 | 32.84 ± 2.06 | 4.18 ± 0.02 | 12.29 ± 0.15 a | 3.17 ± 0.03 a |
| LM | 26.94 ± 0.23 | 19.78 ± 1.38 | 41.39 ± 2.49 | 33.75 ± 1.58 | 3.80 ± 0.20 | 11.63 ± 0.39 a | 2.56 ± 0.04 b |
| Groups 1 | pH | NH3-N (% TN) | Organic Acid 2 (% DM) | |||
|---|---|---|---|---|---|---|
| LA | AA | PA | BA | |||
| CON | 5.26 ± 0.06 a | 7.78 ± 0.13 a | 6.72 ± 0.98 c | 3.75 ± 0.21 a | 1.32 ± 0.13 ab | 0.0007 ± 0.12 |
| LAB | 4.44 ± 0.02 c | 2.07 ± 0.20 b | 10.21 ± 0.14 b | 1.48 ± 0.54 b | 0.90 ± 0.78 ab | ND |
| MOL | 4.93 ± 0.05 b | 7.17 ± 0.57 a | 7.73 ± 0.54 c | 3.70 ± 0.23 a | 1.55 ± 0.26 a | ND |
| LM | 4.19 ± 0.05 d | 1.49 ± 0.22 b | 12.0 ± 0.99 a | 0.91 ± 0.04 b | 0.48 ± 0.22 b | ND |
| Groups 1 | CON | LAB | MOL | LM |
|---|---|---|---|---|
| IVDMD (%) | 75.95 ± 0.85 | 78.66 ± 2.26 | 75.91 ± 0.95 | 75.63 ± 6.32 |
| pH | 6.97 ± 0.02 a | 6.97 ± 0.07 a | 6.88 ± 0.04 ab | 6.80 ± 0.11 b |
| NH3-N (mg/dL) | 12.94 ± 0.20 b | 13.64 ± 2.08 ab | 13.52 ± 0.42 b | 14.34 ± 0.43 a |
| Acetic acid (mmol/L) | 47.61 ± 3.10 | 49.06 ± 1.44 | 48.77 ± 2.64 | 48.28 ± 0.66 |
| Propionic acid (mmol/L) | 19.64 ± 2.19 | 20.40 ± 0.88 | 19.22 ± 0.89 | 19.73 ± 0.24 |
| Butyric acid (mmol/L) | 7.61 ± 3.13 b | 9.45 ± 0.37 a | 8.95 ± 0.41 ab | 9.18 ± 0.28 a |
| Isobutyric acid (mmol/L) | 0.99 ± 0.13 | 1.11 ± 0.13 | 0.94 ± 0.14 | 0.85 ± 0.03 |
| Valeric acid (mmol/L) | 2.19 ± 0.18 | 2.20 ± 0.10 | 2.15 ± 0.13 | 2.21 ± 0.06 |
| Isovaleric acid (mmol/L) | 2.72 ± 0.66 | 2.12 ± 0.06 | 2.00 ± 0.11 | 2.06 ± 0.07 |
| TVFA (mmol/L) | 81.78 ± 6.18 | 84.35 ± 2.79 | 82.02 ± 3.97 | 82.31 ± 1.20 |
| Groups 1 | CON | LAB | MOL | LM |
|---|---|---|---|---|
| GP48 (mL/g) | 76.86 ± 3.31 b | 82.15 ± 0.59 ab | 84.16 ± 3.84 a | 87.05 ± 4.85 a |
| A | 80.28 ± 10.23 | 82.95 ± 0.67 | 85.18 ± 12.59 | 86.55 ± 4.82 |
| B | 1.35 ± 0.08 c | 1.66 ± 0.10 b | 1.54 ± 0.06 b | 1.83 ± 0.06 a |
| C | 5.45 ± 0.91 a | 3.51 ± 0.31 b | 3.30 ± 0.20 b | 3.16 ± 0.30 b |
| AGPR (mL/h) | 4.22 ± 0.81 b | 6.06 ± 0.54 a | 6.56 ± 0.67 a | 7.05 ± 0.88 a |
| Items 1 | a (%) | b (%) | c (%) | a + b (%) | ED (%) | |
|---|---|---|---|---|---|---|
| DM | LM | 32.56 ± 4.89 a | 47.52 ± 2.08 b | 6.02 ± 1.38 | 80.08 ± 5.57 a | 52.51 ± 2.32 a |
| CON | 16.64 ± 3.13 b | 54.09 ± 2.31 a | 7.87 ± 0.66 | 70.73 ± 1.86 b | 43.96 ± 3.16 b | |
| CP | LM | 25.26 ± 2.39 a | 62.55 ± 2.60 | 5.40 ± 3.22 | 87.10 ± 3.42 | 48.30 ± 2.66 |
| CON | 17.57 ± 0.60 b | 65.87 ± 5.72 | 6.23 ± 1.45 | 82.03 ± 4.40 | 45.51 ± 5.26 | |
| NDF | LM | 22.55 ± 3.46 | 46.34 ± 3.53 | 6.68 ± 3.98 | 68.88 ± 2.75 | 42.67 ± 0.71 |
| CON | 21.42 ± 2.90 | 45.81 ± 2.84 | 5.44 ± 0.95 | 67.22 ± 3.45 | 41.29 ± 2.29 | |
| ADF | LM | 14.09 ± 0.41 | 53.34 ± 0.44 | 8.97 ± 0.91 | 67.43 ± 0.61 a | 44.00 ± 0.92 a |
| CON | 10.24 ± 2.24 | 52.47 ± 1.60 | 7.94 ± 0.34 | 62.72 ± 1.33 b | 38.13 ± 1.75 b | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Hao, Y.; Luo, C.; Wang, Q.; Wang, Z.; Li, D.; Yuan, J.; Cao, Z.; Yang, H.; Li, S. Effects of Different Additives on the Chemical Composition, Fermentation Profile, In Vitro and In Situ Digestibility of Paper mulberry Silage. Fermentation 2022, 8, 435. https://doi.org/10.3390/fermentation8090435
Wang W, Hao Y, Luo C, Wang Q, Wang Z, Li D, Yuan J, Cao Z, Yang H, Li S. Effects of Different Additives on the Chemical Composition, Fermentation Profile, In Vitro and In Situ Digestibility of Paper mulberry Silage. Fermentation. 2022; 8(9):435. https://doi.org/10.3390/fermentation8090435
Chicago/Turabian StyleWang, Wei, Yangyi Hao, Chenglong Luo, Qianqian Wang, Zhonghan Wang, Dong Li, Jing Yuan, Zhijun Cao, Hongjian Yang, and Shengli Li. 2022. "Effects of Different Additives on the Chemical Composition, Fermentation Profile, In Vitro and In Situ Digestibility of Paper mulberry Silage" Fermentation 8, no. 9: 435. https://doi.org/10.3390/fermentation8090435
APA StyleWang, W., Hao, Y., Luo, C., Wang, Q., Wang, Z., Li, D., Yuan, J., Cao, Z., Yang, H., & Li, S. (2022). Effects of Different Additives on the Chemical Composition, Fermentation Profile, In Vitro and In Situ Digestibility of Paper mulberry Silage. Fermentation, 8(9), 435. https://doi.org/10.3390/fermentation8090435







