1. Introduction
Kveik yeasts have received more attention in the last few years. They genetically differ from domesticated
Saccharomyces cerevisiae yeast, which has been commonly used for generations in Norway for traditional Norwegian farmhouse brewing. Kveik yeasts are usually related to a certain family and are passed from generation to generation. The main characteristic is that they are non-purified. As they are thermo- and ethanol tolerant, and showing good flocculation properties, these strains of yeast are becoming more popular [
1]. According to the research Preiss et al. [
1] conducted using PCR fingerprint data, Kveik yeast strains form a genetically distinct group of ale yeasts.
The selection of beer yeasts over the span of centuries relied on the important properties of yeast to: efficiently ferment wort sugars (maltose and maltotriose); to reduce the production of phenolic off flavor (important for lager beers); and to show efficient flocculation [
2,
3,
4,
5,
6]. Domestication of brewing yeasts assumably occurred due to frequent reuse (repitching) and cross-sharing between brewers and families [
5,
7,
8,
9,
10]. Historically, families preserved these strains—some for more than 100 years—by continuously using them in brewing and breadmaking [
1,
10].
Typically, Norwegian farmhouse ale was produced from malted barley and hopped wort. However, it was also infused with juniper needles [
10].
Kveik yeasts can be stored in dried form for long periods (up to 1 year or more) [
10], and wort (containing higher extract values ∼1.080 SG/19.25 °Plato) is commonly inoculated at temperatures 28–40 °C [
11]. It is not unusual that fermentation time often lasts 1–2 days [
10,
12]. Dry storage of yeast means that yeast is collected (using wooden ring) with top and bottom cropping after primary fermentation, and dried until its next usage [
10]. Due to the above-mentioned properties, it is not unusual that they are getting more attention, especially since they are really easy to use during fermentation, and the produced beer is much like a lager produced with
Saccharomyces pastorianus (a traditional lager yeast).
S. pastorianus, however, is much more temperature sensitive, requires lower temperatures for fermentation (10–15 °C), diacetyl rest, longer fermentation (up to 10 days), and maturation time of 2–3 weeks. Kveik yeasts are much more robust and temperature and alcohol tolerant (some can withstand 16% ethanol) [
1], require less time to ferment, no diacetyl rest and no maturation/lagering time—characteristics that would surely be of interest to the brewing industry.
There are many Kveik yeasts, all named after the family that was using them, and they belong to different geographical areas in Norway (Ørjasæter, Gamlegrua, Espe, Gausemel, Otterdal, Sigmund, Midtbust, Ebbegarden, Tormodgarden) [
13]. The yeast used in this research was Lutra
® Kveik, isolated from Hornindal Kveik (OYL-091) culture [
14]. The aim of this research was to assess the fermentation characteristics of Lutra
® Kveik yeast fermented at two different temperatures (21 and 35 °C). After fermentation, the physical–chemical, sensory and aromatic profile was determined in order to assess which fermentation temperature offers a better beer.
2. Materials and Methods
2.1. Brewing with Lutra®
For this purpose, pilsner beer was brewed. One hundred and twenty L of wort containing pilsner malt (22.5 kg), pale malt (3 kg) and dextrin malt (1.5 kg) was prepared. The pilsner and pale malt were Badass (Boortmalt, Nova gradiška, Croatia) and the dextrin malt was Simpsons Malt (Northumberland, UK). Malts were purchased from the local brewing and malting equipment store. Reverse osmosis water was adjusted for lager production by adding 3 g calcium chloride, 8.1 g Epsom salt, 4.8 g gypsum and 30 mL of 80% (v/v) lactic acid. Mashing was performed at 65 °C for 60 min, and mash out was performed at 75 °C for 10 min. Boiling was carried out at 100 °C for 15 min. Three hundred g of hops was added during boiling time (60 min). Hops were added as follows: 45 min before the end of boil, 60 g of Chinook was added; 10 min before the end, 90 g of Hallertauer Mittelfrueh was added; and 5 min before the end, Saphir hops (150 g) was added to the boil. Cooling was carried out so that the temperature was first reduced at 35 °C and 20 L of wort was separated into three fermentation vessels marked as 2. By continuing cooling, the temperature was set to 21 °C, and 20 L of wort was discharged into three fermentation vessels marked as 1. All fermenting vessels contained the same wort, just chilled to different temperatures. Inoculation followed.
Lutra® Kveik yeast (OYL-071) was obtained from Omega Yeast (Omega Yeast Labs, LLC, Chicago, IL, USA) and added to the wort after it was divided into marked vessels for fermentation. Upon inoculation, vessels were stored at different temperatures, one at 21 °C (sample 1; in a cooling chamber) and the other at 35 °C (sample 2; ClimaCell 111, MMM Medcenter GmbH, Planegg, Germany). These temperatures were chosen as the minimal and the maximal temperature designated by the producer. At the end of fermentation, beer was transferred to kegs, carbonated and stored in a cool place (4 °C).
2.2. Physical–Chemical Analysis
Before the inoculation wort (after mashing and after boiling), samples were analyzed using Anton Paar Beer analyser (Anton Paar GmbH, Graz, Austria). Fermentation was followed via portable EasyDens Anton Paar (Anton Paar GmbH, Graz, Austria). Polyphenols, color and bitterness analysis were conducted according to [
15]. All analyses were performed in triplicate for all fermentation vessels.
2.3. Aroma Profile
Determination of beer’s volatile compounds (acetaldehyde, ethyl-acetate, n–propanol, isobutanol, 3-methyl-butanol, 2-phenyl-ethanol, isoamyl-acetate, 2-phenylethyl-acetate, dimethyl-sulfide, 2,3–butanedione, 2,3-pentanedione) was performed by using gas chromatography (Shimadzu GC-2030 quadrupol mass spectrophotometer GCMS-TQ 8050 NX (Shimadzu, Kyoto, Japan) coupled with a headspace/SPME autosampler (AOC-6000 Plus, Shimadzu, Kyoto, Japan) with MS detector. The analytical column used was capillary (SH-Rxi-5Sil-MS (30 m, 0.25 mm ID, 0.25 µm) Shimadzu, Kyoto, Japan). Beer samples (10 mL) were added to separate vials 20 mL, in which 1 g NaCl (≥99%, Sigma-Aldrich, St. Louis, MO, USA) was previously added, closed immediately and set for analyses. The analyses were performed in triplicate according to the EBC® methods 9.39 and 9.24.2. The integration of obtained diagrams was performed using GCMS Solution Version 4.53SP1.
2.4. Sensory Evaluation
2.4.1. Evaluation of Beer Characteristics
Sensory evaluation was conducted by 20 untrained consumers (15 males and 5 females). Panels consisted of beer consumers from different age groups (aged 23–57 years old). The materials used in ranking tests for intensity and sensory descriptors were adjusted from the general evaluation sheer for beer [
16]. The intensity scales producing the best discrimination between samples and the most reproducible results were chosen. The scoring of each sensory attribute was performed on a five-point intensity scale, where 1 point means ‘fault’ and 5 points means ‘excellent’. Descriptors were used to anchor/explain the points of the scale. Tasting tests were performed in an appropriate room and beer samples were kept at room temperature for 10 min before the test. As a control sample, lager beer was used. Samples were poured into a clean glass and covered with a watch glass to prevent volatile compounds from escaping the glass. All the beer samples were numbered, and every sample was tested in triplicate. Evaluators were offered flat mineral water between the samples, together with plane white bread. All beers, including the control sample (lager) were served as draught beer.
2.4.2. Drinkability Test
After the sensory evaluation, a drinkability test was performed. The drinkability test included the two pseudo-lager beers, with lager beer as a control sample. The same panelists who participated in sensory evaluation were called again (20 consumers, aged 23–57). Panelists were initially offered to try all beers (200 mL in a glass marked with numbers), in a relaxed atmosphere (a bar-like environment with a person waiting on the participants). After the initial tasting of all beers, panelists were left on their own to choose between the samples. The test lasted for 2 h and was conducted in the afternoon (6–8 PM). The consumers were engaged in a conversation and offered snacks (beer pretzels and peanuts) and they only had to note in the report the number of beer glasses consumed for each beer. Each panelist was always given 300 mL of beer in the glass. Leftover volumes were subtracted from the reports for each panelist. The initial 200 mL was not included in the result. The final result was obtained by summing up the number of fully drank glasses for each beer. All beers, including the control sample (lager), were served as draught beer.
2.5. Statistical Analysis
Analysis of variance (ANOVA) and Fisher’s least significant difference test (LSD) were conducted, with the least statistical significance set to p < 0.05. Statistica 13.1. (TIBCO Software Inc., Palo Alto, CA, USA) was the software of choice for this data set. PCA (principal component analysis) was also carried out using Statistica 13.1. (TIBCO Software Inc., Palo Alto, CA, USA). PCA was applied on an autoscaled data matrix made up of the 2 produced beers and one control sample (sample 1; sample 2; lager), and the mean values of the 11 identified volatiles, and 7 sensory attributes (grainy, honey-like, caramel-like, smokey, grassy, hay-like, yeasty, fruity, flowery, spicy, hoppy) as variables.
3. Results and Discussion
The results in
Table 1 show basic physical–chemical quality indicators for wort after mashing and after boiling. It is evident that data are in accordance with lager beer requirements, with enough extract (14.6 °P) for fermentation. Bitterness after boiling was 33 BU, which is acceptable for lager beers. The pH values of wort after mashing were also within the desirable values 5.8–5.4 [
16,
17].
Table 2 presents a follow-up of fermentation parameters. From this, it can be seen that fermentation in vessel stored at 35 °C was finished in two days, while fermentation in a vessel stored at 21 °C lasted for 9 days. Somewhat lower alcohol content was obtained in fermentation vessel stored at 21 °C (4.1%) than in a fermentation vessel stored at 35 °C (4.4%). Fermentation for sample 2 was finished the next day following inoculation, and it was evident through final gravity values which dropped to 2.5 °P. Fermentation in sample 1 had much slower pace and lasted for 9 days. The end was evident due to a final gravity drop to 3.0 °P. This is not in accordance with results obtained by [
18], who reported that Lutra
® ferments at the same speed at 22 °C and 35 °C. Additionally, the fermenting time for Lutra
® at 22 °C is 88 h, while in our research, fermentation lasted for 216 h (9 days). At 35 °C, fermentation lasted for 76 h, as reported by [
18], and fermentation in our study lasted for a short time: only 24 h. Fermentation at 35 °C was vigorous. Even though there are differences between data, the conclusion is that Lutra
® finishes faster at 35 °C than at 22 °C. The lower alcohol content is a result of slightly lower attenuation in case of fermentation conducted at 22 °C.
As can be seen from the physical–chemical analysis of beer shown in
Table 3, the samples do not differ much, but there are slight discrepancies. The most evident difference between samples can be designated to differences in specific gravity and apparent extract. However, this is due to the lower alcohol content (4.1%) and higher real extract content (4.59 °P). Differences between values for original gravity in
Table 2 and apparent extract (related to final gravity in
Table 3) are due to the fact that the beer was not filtered, and the remaining yeast continued to spend the remaining extract in the keg. However, this should be examined further by monitoring the dissolved oxygen during fermentation and kegging [
19]. Polyphenols were slightly higher in sample 1, which is probably related to the pronounced bitterness in sample 1 as well. Sample 2 showed better clarity values in comparison to sample 1. This could be due the longer lagering time, since the fermentation ended much quicker than in sample 1. pH values differ, probably due to the difference in alcohol (which is a weak acid and is higher in sample 2) and extract content (lower in sample 2) in finished beer.
Table 4 shows the results of GC-MS analysis of evaluated beers. To establish a better correlation of why certain beer was declared better, we have conducted an analysis of 11 aroma compounds related to beer. Since we produce a pseudo-lager, we included a regular commercial lager beer (also used for sensory evaluation and for the drinkability test) into the analysis just to affirm the similarities and differences in aroma compounds between the pseudo-lager produced using Kveik and lager beer. Acetaldehyde (ACE) in beer is described as a sour, tart green apple flavor, reminiscent of dry cider [
16]. The presence of ACE above the threshold (10–20 mgL
−1) results in pronounced above-mentioned off flavors [
20]. ACE concentrations showed significant differences between samples, with sample 1 (fermented at 21 °C) having the highest concentration of this compound (4.42 mgL
−1). This can be explained by the fact that the fermentation in this case lasted longer than in sample 2 and yeast cells probably started to die out, which could result in elevated levels of ACE [
21]. However, in all samples, the concentrations of ACE were below the threshold, and thus not detectable for a consumer.
Ethyl-acetate (EAC) has a fruity, ester-like, rum-like off flavor and is usually a fermentation by-product [
16]. EAC was quantified in higher levels in sample 2 (18.99 mgL
−1) which is due to its higher temperature and more abrupt fermentation pace, according to [
22]. The flavor threshold for ethyl-acetate in beer is 5–10 mgL
−1, according to [
23], which most lager beers are restricted to. However, another source reported a higher value (33 mgL
−1) [
24]. Lager in this case had over 10 mgL
−1 (11.66 mgL
−1), but this is still acceptable. In any case, values for EAC were within the limits if we consider the 33 mgL
−1 as a threshold. Research conducted by Engan [
25] reported concentrations of volatile compounds in Norwegian beers, and in this research, EAC concentration was above 16 mgL
−1. This result is close to the values we obtained using Norwegian Kveik yeast.
The compound n-propanol showed higher levels in sample 2 (25.79 mgL
−1), which is significantly higher than in other samples (lager 8.89 mgL
−1 and sample 1 13.36 mgL
−1). Propanol is a higher alcohol (containing more carbon atoms than ethanol), also known as fusel alcohol. Higher alcohols are important flavor contributors. Concentration of higher alcohol in beer is greatly influenced by the yeast strain used and the fermentation conditions, commonly temperature. Ales tend to have fourfold higher concentrations of propanol than lagers [
26]. This is in accordance with our research, since propanol levels were notably higher in samples that underwent fermentation at 35 °C. Similar, or even higher values for propanol concentrations were reported in research by [
27], who used Kveik yeast as well. A reported threshold for propanol is 800 mgL
−1 [
28,
29,
30].
Isobutanol also belongs to higher alcohols, and its values were significantly different for all samples. It was found in abundance in sample 2 (35.05 mgL
−1) in comparison to sample 1, where it reached 14.87 mgL
−1. Lager beer used as a control sample contained 4.92 mgL
−1 of isobutanol. Higher concentrations in sample 2 are, again, a consequence of faster fermentation at higher temperature (35 °C). Similar to sample 2, values were reported by [
27], where they ranged 38–57 mgL
−1.
3-methylbutanol (or isoamyl alcohol) showed the lowest value in sample 1 (29.67 mgL
−1), while sample 2 had the highest level of this compound (57.40 mgL
−1). These values are significantly lower than the ones obtained by [
27]. 3-methylbutanol is usually described as malty, bitter or alcoholic or solventy [
16]. Pronounced levels of this compound in beer are related to higher fermentation temperatures, as other higher alcohols are too [
16].
Belonging to higher alcohols, 2-phenylethanol levels in beer are also correlated with fermentation pace. In this research, levels of this compound were again highest in sample 2 (42.59 mgL
−1), while lager contained 21.47 mgL
−1. Lowest levels of this higher alcohol, which is described as having a floral, alcohol-like, honey-like and sweet aroma [
16], were determined in sample 1 and amounted to 19.70 mgL
−1.
Isoamyl acetate is another fermentation by-product that gives beer a banana-like, fruity, apple or pear flavor [
16]. This important ester’s threshold ranges from 0.6–1.2. mgL
−1. Values obtained in our research were below this threshold.
2-phenyletil acetate, also an ester, typically gives an aroma reminiscent of roses or honey and is often described as tasting like raspberry or even guava. In most cases, it is minimally present and according to its threshold is 3.8 mgL
−1 [
28,
29,
30]. All samples showed significantly lower levels than the designated threshold.
Dimethylsulfide (DMS) originates from malt and usually evaporates during the boiling stage. It is reminiscent of cooked cabbage, sweetcorn or cooked vegetables. The threshold for DMS in beer is 30 µgL
−1 [
31]. Samples produced using Kveik yeast showed significantly high levels of DMS, well above 30 µgL
−1 (sample 1 68.92 and sample 2 55.16), while lager samples showed values within the prescribed threshold (19.37). Higher values of DMS in sample 1 (fermented at 21 °C) are expected, since according to [
32,
33], lower fermentation temperatures result in higher DMS values.
2,3-butanedione, generally regarded as diacetyl (vicinal diketone), is commonly described as having a buttery, creamy, butterscotch flavor [
16]. Diacetyl is common in lager beers, and its threshold is 0.15 mgL
−1 [
34]. All samples showed significantly lower values than the threshold. This is not unusual for Kveik, and it does not require a diacetyl rest.
2,3-pentanedione is a vicinal diketone as well. It is described similarly to diacetyl, as buttery, cloying, honey or creamy. Its threshold is 10 times higher than for diacetyl [
34]. According to this, all samples had lower values than the designated threshold.
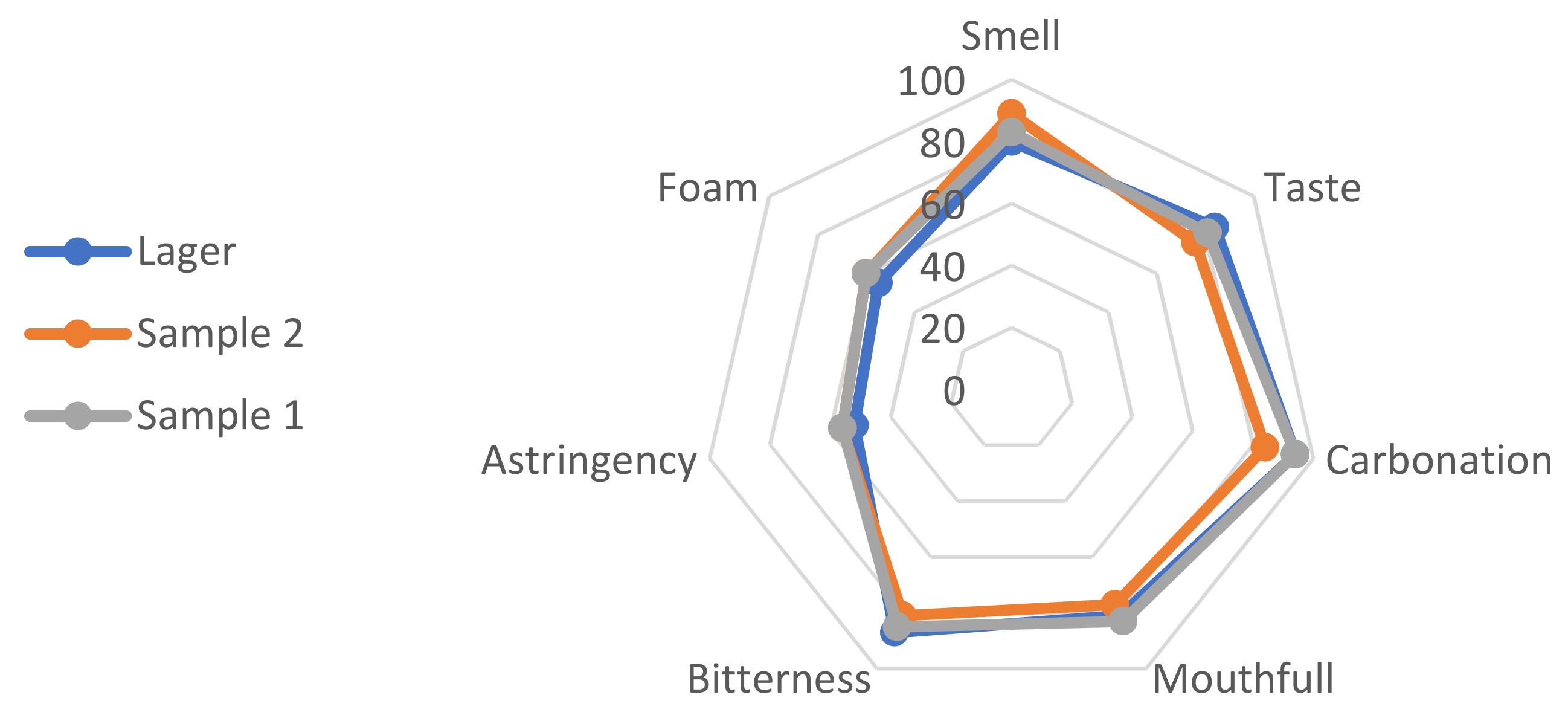
Sensory analysis was performed in order to evaluate the similarity of the produced pseudo-lager with conventional lager beer, which was used as a control sample. According to the results presented in
Figure 1, sample 2 had a slightly better smell than sample 1, but taste, carbonation, mouthful, bitterness and astringency all went in favor of sample 1. Foam was rated as good quality in all samples; slightly lower values were designated for lager beer. Both samples were recognized as lager beers.
As can be noticed in
Figure 2, there are variations between samples. All beers were set on different parts of the biplot. Lager was set at the negative F1 and F2 semi-axis, sample 1 was on the negative F1 and positive F2 semi-axis and sample 2 was set at the positive F1 and F2 semi-axis. This clearly indicates that there are distinguished differences between samples. Similarly, as reported by [
19], there is a strong connection between volatile compounds analysis and data obtained via sensory analysis. Lager beer, for example, has very few designated attributes (astringency, 2,3-pentadione, foam), while sample 2 has many (ethyl-acetate, isobutanol, n-propanol, 2-phenilyethanol, isoamyl acetate, bitterness, taste...). For sample 1, smell can be related to 2,3-butanedione, acetaldehyde and DMS.
A drinkability test (
Table 5) was performed in order to determine which temperature of fermentation would provide a better, more pleasant beer. According to the results, consumers were inclined to sample 1, as they drank 12.3 L of beer, and sample 2 was a bit less desirable, with 11 L of consumed beer. Lager, the control sample, was in between, with 11.7 L of beer.