Antifungal Agent Chitooligosaccharides Derived from Solid-State Fermentation of Shrimp Shell Waste by Pseudonocardia antitumoralis 18D36-A1
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Shrimp Shell Waste and Colloidal Chitin
2.2. Isolated Actinomycetes
2.3. Solid-State Fermentation
2.4. Extraction and Antifungal Activity
2.4.1. Microscopic Analysis of Fungicidal Activity
2.4.2. Liquid Chromatography–Mass Spectrophotometry
3. Results and Discussion
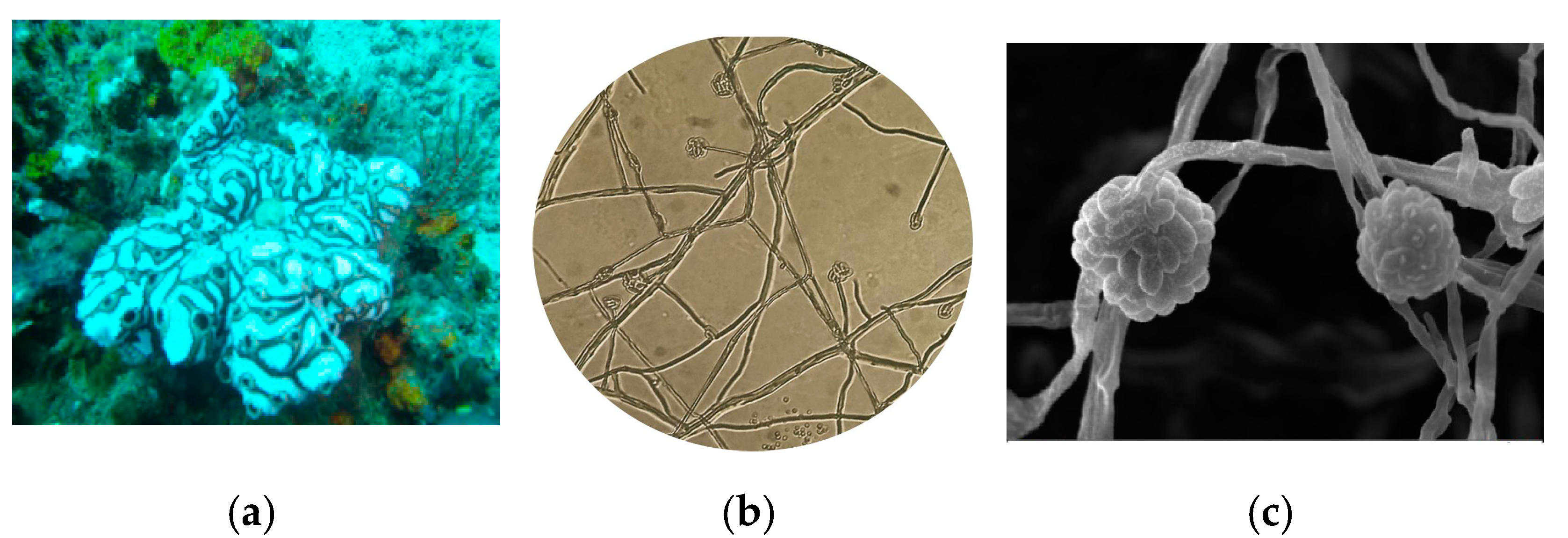
3.1. Isolation of Actinomycetes
3.2. Solid-State Fermentation
3.3. Extraction and Antifungal Activity
3.4. Characterization
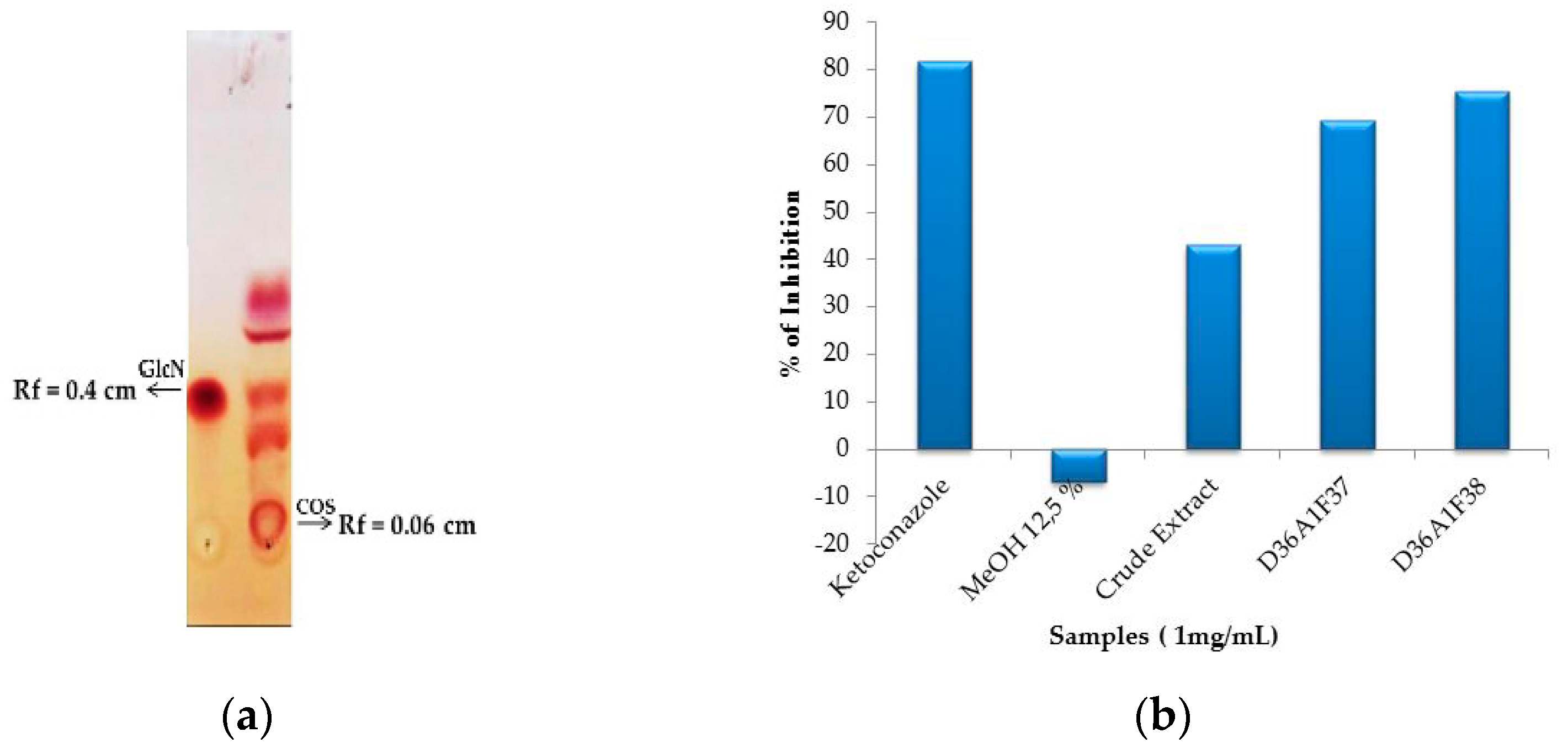
3.4.1. Analysis of Fungicidal Activity
3.4.2. Analysis of FTIR and Liquid Chromatography–Mass Spectrophotometry
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Azis, Y.; Mutamima, A.; Herman, S.; Alfarisi, C.D.; Ikrima, H. Isotherms Studies of Equilibrium Sorption of Cu2+ Unto Hydrochloric Acid Modified Shrimp Shell Waste. Mater. Today 2022, 3, 154–196. [Google Scholar]
- Wiradana, P.A.; Nur, A.; Sani, M.D.; Affandi, M.; Putranto, T.W.C. A Short Review on Status, Trends and Prospects of Jerbung Shrimp (Fenneropenaeus merguiensis de Man) in Indonesia. Ecol. Environ. Conserv. 2020, 26, 1657–1664. [Google Scholar]
- Abun, A.; Rusmana, D.; Widjastuti, T.; Haetami, K. Effect of Use of Prebiotics Bls (Bacillus Licheniformis, Lactobacillus sp. and Saccharomyces Cerevisiae) Based on Shrimp Waste on Protein Efficiency Ratio in Indonesia Local Chicken. Sci. Pap. Ser. D Anim. Sci. Int. Sess. Sci. Commun. Fac. Anim. Sci. 2019, 62, 37–43. [Google Scholar]
- Zargar, V.; Asghari, M.; Dashti, A. A Review on Chitin And Chitosan Polymers: Structure, Chemistry, Solubility, Derivatives, and Applications. ChemBioEng Rev. 2015, 2, 204–226. [Google Scholar] [CrossRef]
- Rahayu, A.P.; Islami, A.F.; Saputra, E.; Sulmartiwi, L.; Rahmah, A.U.; Kurnia, K.A. The Impact of the Different Types of Acid Solution on the Extraction and Adsorption Performance of Chitin from Shrimp Shell Waste. Int. J. Biol. Macromol. 2022, 194, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Fu, X.; Yan, Q.; Guo, Y.; Liu, Z.; Jiang, Z. Cloning, Expression, Purification and Application of a Novel Chitinase from a Thermophilic Marine Bacterium Paenibacillus barengoltzii. Food Chem. 2016, 192, 1041–1048. [Google Scholar] [CrossRef]
- El Sheikha, A.F.; Ray, R.C. Bioprocessing of Horticultural Wastes by Solid-State Fermentation into Value-Added/Innovative Bioproducts: A review. Food Rev. Int. 2022, 1–57. [Google Scholar] [CrossRef]
- Mohan, K.; Ganesan, A.R.; Ezhilarasi, P.N.; Kondamareddy, K.K.; Rajan, D.K.; Sathishkumar, P.; Rajarajeswaran, J.; Conterno, L. Green and eco-friendly approaches for the extraction of chitin and chitosan: A review. Carbohydr. Polym. 2022, 287, 119349. [Google Scholar] [CrossRef]
- Smith, P.J.; Ortiz-Soto, M.E.; Roth, C.; Barnes, W.J.; Seibel, J.; Urbanowicz, B.R.; Pfrengle, F. Enzymatic Synthesis of Artificial Polysaccharides. ACS Sustain. Chem. Eng. 2020, 8, 11853–11871. [Google Scholar] [CrossRef]
- Yahmed, N.B.; Carrere, H.; Marzouki, M.N.; Smaali, I. Enhancement of Biogas Production from Ulva sp. by Using Solid-State Fermentation as Biological Pretreatment. Algal Res. 2017, 27, 206–214. [Google Scholar] [CrossRef]
- Núñez Pérez, J.; Chávez Arias, B.S.; de la Vega Quintero, J.C.; Zárate Baca, S.; Pais-Chanfrau, J.M. Multi-Objective Statistical Optimization of Pectinolytic Enzymes Production by an Aspergillus sp. on Dehydrated Coffee Residues in Solid-State Fermentation. Fermentation 2022, 8, 170. [Google Scholar] [CrossRef]
- Bisly, A.A.; Hettiarachchy, N.S.; Kumar, T.K.S.; Lay, J.O., Jr. Antioxidant Activities of Solid-State Fermentation Derived Proteins and Peptides From Heat-Stabilized Defatted Rice Bran. J. Am. Oil Chem. Soc. 2022, 99, 215–228. [Google Scholar] [CrossRef]
- Setiawan, A.; Widyastuti, W.; Irawan, A.; Wijaya, O.S.; Laila, A.; Setiawan, W.A.; Juliasih, N.L.G.R.; Nonaka, K.; Arai, M.; Hendri, J. Solid State Fermentation of Shrimp Shell Waste Using Pseudonocardia carboxydivorans 18A13O1 to Produce Bioactive Metabolites. Fermentation 2021, 7, 247. [Google Scholar] [CrossRef]
- Javed, Z.; Tripathi, G.D.; Mishra, M.; Dashora, K. Actinomycetes–The Microbial Machinery for the Organic-Cycling, Plant Growth, and Sustainable Soil Health. Biocatal. Agric. Biotechnol. 2021, 31, 101893. [Google Scholar] [CrossRef]
- Gaber, Y.; Mekasha, S.; Vaaje-Kolstad, G.; Eijsink, V.G.; Fraaije, M.W. Characterization of a Chitinase from the Cellulolytic actinomycete Thermobifida fusca. Biochim. Biophys. Acta (BBA) Proteins Proteom. 2016, 1864, 1253–1259. [Google Scholar] [CrossRef] [PubMed]
- Malecki, P.H.; Bejger, M.; Rypniewski, W.; Vorgias, C.E. The Crystal Structure of a Streptomyces thermoviolaceus Thermophilic Chitinase Known for Its Refolding Efficiency. Int. J. Mol. Sci. 2020, 21, 2892. [Google Scholar] [CrossRef] [Green Version]
- Lv, C.; Gu, T.; Ma, R.; Yao, W.; Huang, Y.; Gu, J.; Zhao, G. Biochemical Characterization of a GH19 Chitinase from Streptomyces Alfalfae and Its Applications in Crystalline Chitin Conversion and Biocontrol. Int. J. Biol. Macromol. 2021, 167, 193–201. [Google Scholar] [CrossRef]
- Leong, C.; Buttafuoco, A.; Glatz, M.; Bosshard, P.P. Antifungal Susceptibility Testing of Malassezia spp. with an Optimized Colorimetric Broth Microdilution Method. J. Clin. Microbiol. 2017, 55, 1883–1893. [Google Scholar] [CrossRef] [Green Version]
- Setiawan, A.; Setiawan, F.; Juliasih, N.L.G.R.; Widyastuti, W.; Laila, A.; Setiawan, W.A.; Djailani, F.M.; Mulyono, M.; Hendri, J.; Arai, M. Fungicide Activity of Culture Extract from Kocuria palustris 19C38A1 against Fusarium oxysporum. J. Fungi 2022, 8, 280. [Google Scholar] [CrossRef]
- Xia, Z.F.; Guan, T.W.; Ruan, J.S.; Huang, Y.; Zhang, L.L. Longimycelium tulufanense gen. nov., sp. nov., a Filamentous Actinomycete of the Family Pseudonocardiaceae. Int. J. Syst. Evol. Microbiol. 2013, 63, 2813–2818. [Google Scholar] [CrossRef] [Green Version]
- Bharvad, P.B.; Algotar, H.J. Chitinase. In Practical Handbook on Agricultural Microbiology; Humana: New York, NY, USA, 2022; pp. 301–308. [Google Scholar]
- Suresh, P.V. Biodegradation of Shrimp Processing Bio-Waste and Concomitant Production of Chitinase Enzyme and N-acetyl-D-glucosamine by Marine Bacteria: Production and Process Optimization. World J. Microbiol. Biotechnol. 2012, 28, 2945–2962. [Google Scholar] [CrossRef]
- Jassim, Y.A. Screening for Antimicrobial Activities and Enzymatic Activities Production of Some Actinomycetes spp. Isolated from different Soil Samples from Hilla Province. Med. Microbiol. 2022, 1, 23–29. [Google Scholar]
- Gasmi, M.; Kitouni, M.; Carro, L.; Pujic, P.; Normand, P.; Boubakri, H. Chitinolytic Actinobacteria Isolated from an Algerian Semi-Arid Soil: Development of an Antifungal Chitinase-Dependent Assay and GH18 Chitinase Gene Identification. Ann. Microbiol. 2019, 69, 395–405. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, N.; He, J.; Li, Y.; Gao, X.; Huang, L.; Yan, X. Expression and Characterization of a Novel Chitinase with Antifungal Activity from a Rare Actinomycete, Saccharothrix yanglingensis Hhs. 015. Protein Expr. Purif. 2018, 143, 45–51. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, A.M.; Mesquita, M.d.S.; da Silva, G.C.; de Oliveira Lima, E.; de Medeiros, P.L.; Paiva, P.M.G.; Souza, I.A.D.; Napoleão, T.H. Evaluation of Toxicity and Antimicrobial Activity of an Ethanolic Extract From Leaves of Morus alba L. (Moraceae). Evid. Based Complement. Altern. Med. 2015, 2015, 513978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olicón-Hernández, D.R.; Hernández-Lauzardo, A.N.; Pardo, J.P.; Peña, A.; Velázquez-del Valle, M.G.; Guerra-Sánchez, G. Influence of Chitosan and Its Derivatives on Cell Development and Physiology of Ustilago maydis. Int. J. Biol. Macromol. 2015, 79, 654–660. [Google Scholar] [CrossRef]
- Silva, N.S.D.; Araújo, N.K.; Daniele-Silva, A.; Oliveira, J.W.d.F.; Medeiros, J.M.d.; Araújo, R.M.; Ferreira, L.D.S.; Rocha, H.A.O.; Silva-Junior, A.A.; Silva, M.S.; et al. Antimicrobial Activity of Chitosan Oligosaccharides with Special Attention to Antiparasitic Potential. Mar. Drugs 2021, 19, 110. [Google Scholar] [CrossRef]
- Ke, Y.; Ding, B.; Zhang, M.; Dong, T.; Fu, Y.; Lv, Q.; Wang, X. Study on Inhibitory Activity and Mechanism of Chitosan oligosaccharides on Aspergillus flavus and Aspergillus fumigatus. Carbohydr. Polym. 2022, 275, 118673. [Google Scholar] [CrossRef]
- Vidhate, R.P.; Bhide, A.J.; Gaikwad, S.M.; Giri, A.P. A Potent Chitin-Hydrolyzing Enzyme from Myrothecium verrucaria Affects Growth and Development of Helicoverpa armigera and Plant Fungal Pathogens. Int. J. Biol. Macromol. 2019, 141, 517–528. [Google Scholar] [CrossRef]
- Vasilieva, T.; Sigarev, A.; Kosyakov, D.; Ul’yanovskii, N.; Anikeenko, E.; Chuhchin, D.; Ladesov, A.; Hein, A.M.; Miasnikov, V. Formation of Low Molecular Weight Oligomers from Chitin and Chitosan Stimulated by Plasma-Assisted Processes. Carbohydr. Polym. 2017, 163, 54–61. [Google Scholar] [CrossRef]
- Li, K.; Xing, R.; Liu, S.; Qin, Y.; Li, P. Access to N-acetylated chitohexaose with Well-Defined Degrees of Acetylation. BioMed Res. Int. 2017, 2017, 2486515. [Google Scholar] [CrossRef] [PubMed]
- Kang, L.; Jiang, S.; Ma, L. Enzymatic Production of High Molecular Weight Chitooligosaccharides Using Recombinant Chitosanase from Bacillus thuringiensis BMB171. Microbiol. Biotechnol. Lett. 2018, 46, 45–50. [Google Scholar] [CrossRef]
- Tokuyasu, K.; Ono, H.; Mitsutomi, M.; Hayashi, K.; Mori, Y. Synthesis of a chitosan tetramer derivative, β-d-GlcNAc-(1→ 4)-β-d-GlcNAc-(1→ 4)-β-d-GlcNAc-(1→ 4)-d-GlcN through a partial N-acetylation Reaction by chitin deacetylase. Carbohydr. Res. 2000, 325, 211–215. [Google Scholar] [CrossRef]
- Uchiyama, T.; Katouno, F.; Nikaidou, N.; Nonaka, T.; Sugiyama, J.; Watanabe, T. Roles of the Exposed Aromatic Residues in Crystalline Chitin Hydrolysis by chitinase a from Serratia marcescens 2170. J. Biol. Chem. 2001, 276, 41343–41349. [Google Scholar] [CrossRef] [Green Version]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Widyastuti, W.; Setiawan, F.; Al Afandy, C.; Irawan, A.; Laila, A.; Juliasih, N.L.G.R.; Setiawan, W.A.; Arai, M.; Hendri, J.; Setiawan, A. Antifungal Agent Chitooligosaccharides Derived from Solid-State Fermentation of Shrimp Shell Waste by Pseudonocardia antitumoralis 18D36-A1. Fermentation 2022, 8, 353. https://doi.org/10.3390/fermentation8080353
Widyastuti W, Setiawan F, Al Afandy C, Irawan A, Laila A, Juliasih NLGR, Setiawan WA, Arai M, Hendri J, Setiawan A. Antifungal Agent Chitooligosaccharides Derived from Solid-State Fermentation of Shrimp Shell Waste by Pseudonocardia antitumoralis 18D36-A1. Fermentation. 2022; 8(8):353. https://doi.org/10.3390/fermentation8080353
Chicago/Turabian StyleWidyastuti, Widyastuti, Fendi Setiawan, Chasya Al Afandy, Arik Irawan, Aspita Laila, Ni Luh Gede Ratna Juliasih, Wawan Abdullah Setiawan, Masayoshi Arai, John Hendri, and Andi Setiawan. 2022. "Antifungal Agent Chitooligosaccharides Derived from Solid-State Fermentation of Shrimp Shell Waste by Pseudonocardia antitumoralis 18D36-A1" Fermentation 8, no. 8: 353. https://doi.org/10.3390/fermentation8080353
APA StyleWidyastuti, W., Setiawan, F., Al Afandy, C., Irawan, A., Laila, A., Juliasih, N. L. G. R., Setiawan, W. A., Arai, M., Hendri, J., & Setiawan, A. (2022). Antifungal Agent Chitooligosaccharides Derived from Solid-State Fermentation of Shrimp Shell Waste by Pseudonocardia antitumoralis 18D36-A1. Fermentation, 8(8), 353. https://doi.org/10.3390/fermentation8080353










