Probiotics as a Friendly Antibiotic Alternative: Assessment of Their Effects on the Health and Productive Performance of Poultry
Abstract
1. Introduction
2. Probiotics and Growth Performance
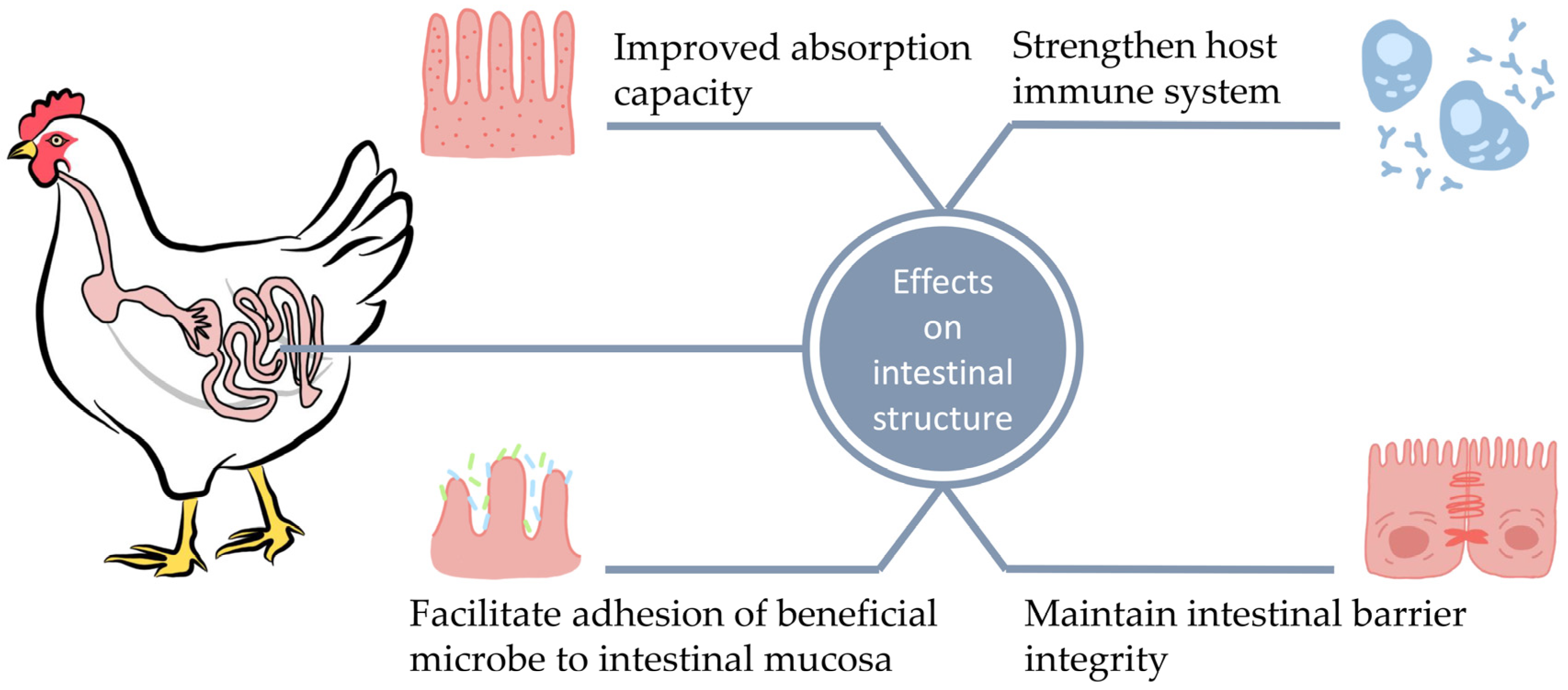
3. Probiotics and Intestinal Morphology
4. Probiotics and Immune Response
5. Mode of Action Probiotics
5.1. Probiotics and Competitive Exclusion
5.2. Probiotics and Organic Acid (Acidity and pH)
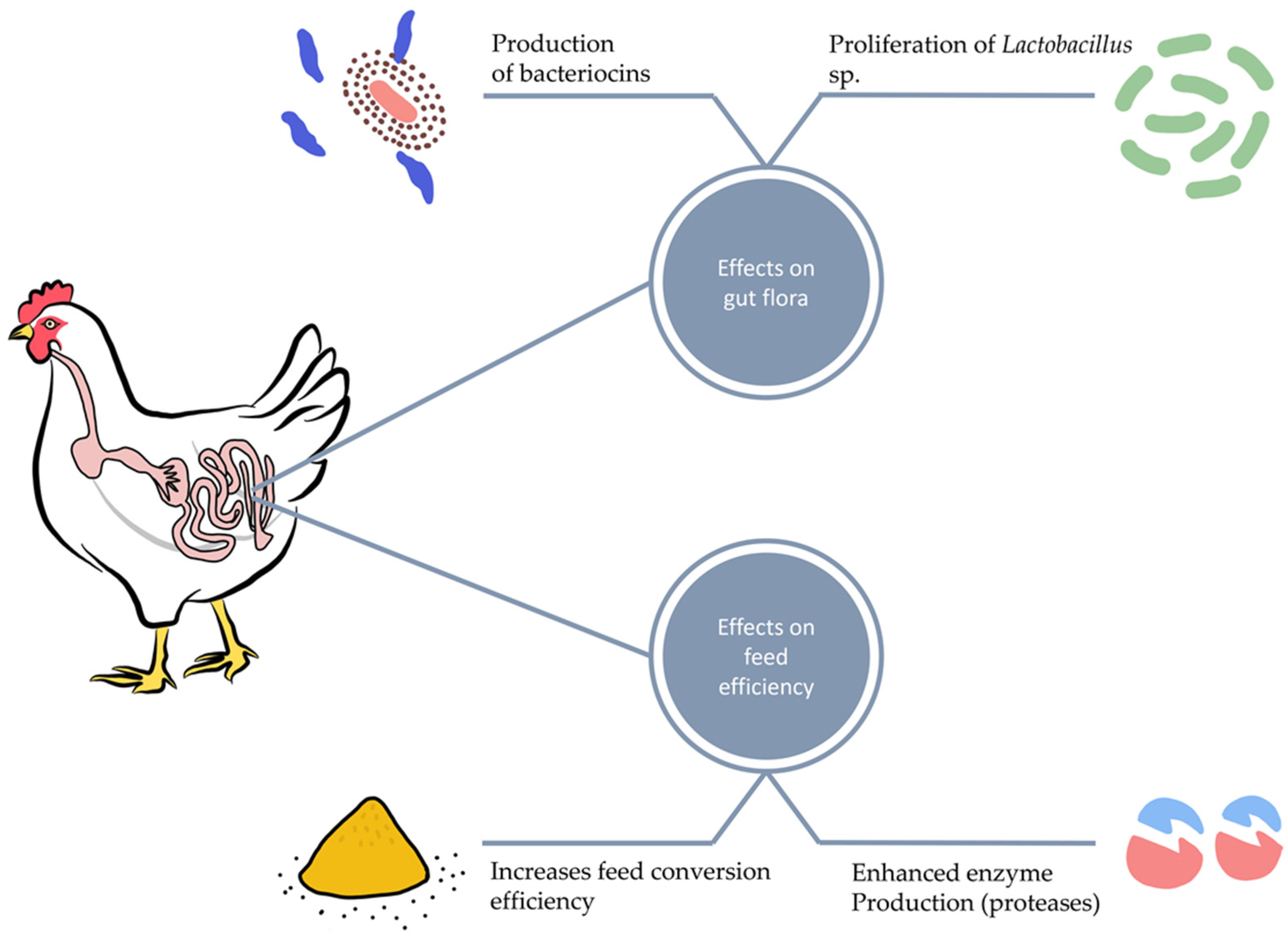
5.3. Probiotics and Gut Microbiota
5.4. Application and Validation of Probiotics Secondary Metabolites
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Neveling, D.P.; Dicks, L.M. Probiotics: An antibiotic replacement strategy for healthy broilers and productive rearing. Probiotics Antimicrob. Proteins 2021, 13, 1–11. [Google Scholar] [CrossRef]
- Agyare, C.; Boamah, V.E.; Zumbi, C.N.; Osei, F.B. Antibiotic use in poultry production and its effects on bacterial resistance. In Antimicrobial Resistance—A Global Threat; IntechOpen: London, UK, 2018; pp. 33–51. [Google Scholar]
- Neveling, D.P.; van Emmenes, L.; Ahire, J.; Pieterse, E.; Smith, C.; Dicks, L. Effect of a multi-species probiotic on the colonisation of Salmonella in broilers. Probiotics Antimicrob. Proteins 2020, 12, 896–905. [Google Scholar] [CrossRef]
- Shurson, G.C.; Urriola, P.E.; van de Ligt, J.L. Can we effectively manage parasites, prions, and pathogens in the global feed industry to achieve One Health? Transbound. Emerg. Dis. 2022, 69, 4–30. [Google Scholar] [CrossRef]
- Mehdi, Y.; Létourneau-Montminy, M.-P.; Gaucher, M.-L.; Chorfi, Y.; Suresh, G.; Rouissi, T.; Brar, S.K.; Côté, C.; Ramirez, A.A.; Godbout, S. Use of antibiotics in broiler production: Global impacts and alternatives. Anim. Nutr. 2018, 4, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Gadde, U.; Kim, W.; Oh, S.; Lillehoj, H.S. Alternatives to antibiotics for maximizing growth performance and feed efficiency in poultry: A review. Anim. Health Res. Rev. 2017, 18, 26–45. [Google Scholar] [CrossRef]
- Lourenco, J.M.; Rothrock Jr, M.J.; Fluharty, F.L.; Callaway, T.R. The successional changes in the gut microbiome of pasture-raised chickens fed soy-containing and soy-free diets. Front. Sustain. Food Syst. 2019, 3, 35. [Google Scholar] [CrossRef]
- Żbikowska, K.; Michalczuk, M.; Dolka, B. The use of bacteriophages in the poultry industry. Animals 2020, 10, 872. [Google Scholar] [CrossRef] [PubMed]
- Peterson, E.; Kaur, P. Antibiotic resistance mechanisms in bacteria: Relationships between resistance determinants of antibiotic producers, environmental bacteria, and clinical pathogens. Front. Microbiol. 2018, 9, 2928. [Google Scholar] [CrossRef]
- Zhao, X.; Yu, Z.; Ding, T. Quorum-sensing regulation of antimicrobial resistance in bacteria. Microorganisms 2020, 8, 425. [Google Scholar] [CrossRef]
- Grenni, P.; Ancona, V.; Caracciolo, A.B. Ecological effects of antibiotics on natural ecosystems: A review. Microchem. J. 2018, 136, 25–39. [Google Scholar] [CrossRef]
- Christaki, E.; Marcou, M.; Tofarides, A. Antimicrobial resistance in bacteria: Mechanisms, evolution, and persistence. J. Mol. Evol. 2020, 88, 26–40. [Google Scholar] [CrossRef] [PubMed]
- Shafiq, M.; Huang, J.; Shah, J.M.; Ali, I.; Rahman, S.U.; Wang, L. Characterization and resistant determinants linked to mobile elements of ESBL-producing and mcr-1-positive Escherichia coli recovered from the chicken origin. Microb. Pathog. 2021, 150, 104722. [Google Scholar] [CrossRef] [PubMed]
- Pal, M.; Kerorsa, G.B.; Marami, L.M.; Kandi, V. Epidemiology, pathogenicity, animal infections, antibiotic resistance, public health significance, and economic impact of staphylococcus aureus: A comprehensive review. Am. J. Public Health Res. 2020, 8, 14–21. [Google Scholar]
- Hayford, O. Isolation and Characterisation of Multi-Drug Resistant Pseudomonas aeruginosa from Clinical, Environmental and Poultry Litter Sources in Ashanti Region of Ghana. Master’s Thesis, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana, 2017. [Google Scholar]
- Aniokette, U.; Iroha, C.; Ajah, M.; Nwakaeze, A. Occurrence of multi-drug resistant Gram-negative bacteria from poultry and poultry products sold in Abakaliki. Int. J. Aric. Sci. Food Technol. 2016, 2, 119–124. [Google Scholar]
- Sharma, S.; Galav, V.; Agrawal, M.; Faridi, F.; Kumar, B. Multi-drug resistance pattern of bacterial flora obtained from necropsy samples of poultry. J. Anim. Health Prod. 2017, 5, 165–171. [Google Scholar]
- Varga, C.; Guerin, M.T.; Brash, M.L.; Slavic, D.; Boerlin, P.; Susta, L. Antimicrobial resistance in fecal Escherichia coli and Salmonella enterica isolates: A two-year prospective study of small poultry flocks in Ontario, Canada. BMC Vet. Res. 2019, 15, 464. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, H.; Zheng, S.; Wang, Z.; Sheng, H.; Shi, C.; Shi, X.; Niu, Q.; Yang, B. Prevalence, serotype, antibiotic susceptibility, and genotype of Salmonella in eggs from poultry farms and marketplaces in Yangling, Shaanxi province, China. Front. Microbiol. 2020, 11, 1482. [Google Scholar] [CrossRef] [PubMed]
- Shafiq, M.; Yao, F.; Bilal, H.; Rahman, S.U.; Zeng, M.; Ali, I.; Zeng, Y.; Li, X.; Yuan, Y.; Jiao, X. Synergistic Activity of Tetrandrine and Colistin against mcr-1-Harboring Escherichia coli. Antibiotics 2022, 11, 1346. [Google Scholar] [CrossRef]
- Mingmongkolchai, S.; Panbangred, W. Bacillus probiotics: An alternative to antibiotics for livestock production. J. Appl. Microbiol. 2018, 124, 1334–1346. [Google Scholar] [CrossRef]
- Muhammad, J.; Khan, S.; Su, J.Q.; Hesham, A.E.-L.; Ditta, A.; Nawab, J.; Ali, A. Antibiotics in poultry manure and their associated health issues: A systematic review. J. Soils Sediments 2020, 20, 486–497. [Google Scholar] [CrossRef]
- Yaqoob, M.U.; Wang, G. An updated review on probiotics as an alternative of antibiotics in poultry. Anim. Biosci. 2022, 35, 1109–1120. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Moneim, A.-M.E.; Shehata, A.M.; Khidr, R.E.; Paswan, V.K.; Ibrahim, N.S.; El-Ghoul, A.A.; Aldhumri, S.A.; Gabr, S.A.; Mesalam, N.M.; Elbaz, A.M. Nutritional manipulation to combat heat stress in poultry—A comprehensive review. J. Therm. Biol. 2021, 98, 102915. [Google Scholar] [CrossRef]
- Coman, M.M.; Mazzotti, L.; Silvi, S.; Scalise, A.; Orpianesi, C.; Cresci, A.; Verdenelli, M.C. Antimicrobial activity of SYNBIO® probiotic formulation in pathogens isolated from chronic ulcerative lesions: In Vitro studies. J. Appl. Microbiol. 2020, 128, 584–597. [Google Scholar] [CrossRef] [PubMed]
- Adedokun, S.A.; Olojede, O.C. Optimizing gastrointestinal integrity in poultry: The role of nutrients and feed additives. Front. Vet. Sci. 2019, 5, 348. [Google Scholar] [CrossRef]
- Al-Khalaifah, H. Benefits of probiotics and/or prebiotics for antibiotic-reduced poultry. Poult. Sci. 2018, 97, 3807–3815. [Google Scholar] [CrossRef]
- Wengerska, K.; Czech, A.; Knaga, S.; Drabik, K.; Próchniak, T.; Bagrowski, R.; Gryta, A.; Batkowska, J. The Quality of Eggs Derived from Japanese Quail Fed with the Fermented and Non-Fermented Rapeseed Meal. Foods 2022, 11, 2492. [Google Scholar] [CrossRef] [PubMed]
- Shehata, A.A.; Yalçın, S.; Latorre, J.D.; Basiouni, S.; Attia, Y.A.; Abd El-Wahab, A.; Visscher, C.; El-Seedi, H.R.; Huber, C.; Hafez, H.M. Probiotics, prebiotics, and phytogenic substances for optimizing gut health in poultry. Microorganisms 2022, 10, 395. [Google Scholar] [CrossRef]
- Flores, C.; Duong, T.; Askelson, T.; Dersjant-Li, Y.; Gibbs, K.; Awati, A.; Lee, J. Effects of direct fed-microorganisms and enzyme blend co-administration on growth performance in broilers fed diets with or without antibiotics. J. Appl. Poult. Res. 2019, 28, 1181–1188. [Google Scholar] [CrossRef]
- Cervera, C.; Carmona, J.F. Nutrition and climatic environment. In Nutrition of the Rabbit; CAB International: Wallingford, UK, 2020; pp. 289–307. [Google Scholar]
- Angelakis, E. Weight gain by gut microbiota manipulation in productive animals. Microb. Pathog. 2017, 106, 162–170. [Google Scholar] [CrossRef]
- Flores, C.; Askelson, T.; Dersjant-Li, Y.; Gibbs, K.; Awati, A.; Duong, T.; Lee, J. Effect of Direct-Fed Microorganisms and enzyme blend co-administration on broilers fed US commercial-type, diets with or without agp. J. Appl. Poult. Res. 2018, 28, 1181–1188. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, H.; Chen, Y.; Yang, M.; Zhang, L.; Lu, Z.; Zhou, Y.; Wang, T. Bacillus amyloliquefaciens supplementation alleviates immunological stress in lipopolysaccharide-challenged broilers at early age. Poult. Sci. 2015, 94, 1504–1511. [Google Scholar] [CrossRef]
- Mazanko, M.S.; Gorlov, I.F.; Prazdnova, E.V.; Makarenko, M.S.; Usatov, A.V.; Bren, A.B.; Chistyakov, V.A.; Tutelyan, A.V.; Komarova, Z.B.; Mosolova, N.I. Bacillus probiotic supplementations improve laying performance, egg quality, hatching of laying hens, and sperm quality of roosters. Probiotics Antimicrob. Proteins 2018, 10, 367–373. [Google Scholar] [CrossRef]
- Zhen, W.; Shao, Y.; Gong, X.; Wu, Y.; Geng, Y.; Wang, Z.; Guo, Y. Effect of dietary Bacillus coagulans supplementation on growth performance and immune responses of broiler chickens challenged by Salmonella enteritidis. Poult. Sci. 2018, 97, 2654–2666. [Google Scholar] [CrossRef] [PubMed]
- Pender, C.M.; Kim, S.; Potter, T.D.; Ritzi, M.M.; Young, M.; Dalloul, R.A. In ovo supplementation of probiotics and its effects on performance and immune-related gene expression in broiler chicks. Poult. Sci. 2017, 96, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Forte, C.; Acuti, G.; Manuali, E.; Proietti, P.C.; Pavone, S.; Trabalza-Marinucci, M.; Moscati, L.; Onofri, A.; Lorenzetti, C.; Franciosini, M.P. Effects of two different probiotics on microflora, morphology, and morphometry of gut in organic laying hens. Poult. Sci. 2016, 95, 2528–2535. [Google Scholar] [CrossRef]
- Cheng, Y.; Chen, Y.; Li, X.; Yang, W.; Wen, C.; Kang, Y.; Wang, A.; Zhou, Y. Effects of synbiotic supplementation on growth performance, carcass characteristics, meat quality and muscular antioxidant capacity and mineral contents in broilers. J. Sci. Food Agric. 2017, 97, 3699–3705. [Google Scholar] [CrossRef]
- Mikulski, D.; Jankowski, J.; Mikulska, M.; Demey, V. Effects of dietary probiotic (Pediococcus acidilactici) supplementation on productive performance, egg quality, and body composition in laying hens fed diets varying in energy density. Poult. Sci. 2020, 99, 2275–2285. [Google Scholar] [CrossRef] [PubMed]
- Mountzouris, K.; Tsitrsikos, P.; Palamidi, I.; Arvaniti, A.; Mohnl, M.; Schatzmayr, G.; Fegeros, K. Effects of probiotic inclusion levels in broiler nutrition on growth performance, nutrient digestibility, plasma immunoglobulins, and cecal microflora composition. Poult. Sci. 2010, 89, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Martínez, E.A.; Babot, J.D.; Lorenzo-Pisarello, M.J.; Apella, M.C.; Chaia, A.P. Feed supplementation with avian Propionibacterium acidipropionici contributes to mucosa development in early stages of rearing broiler chickens. Benef. Microbes. 2016, 7, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Hassanein, S.M.; Soliman, N.K. Effect of probiotic (Saccharomyces cerevisiae) adding to diets on intestinal microflora and performance of Hy-Line layers hens. J. Am. Sci. 2010, 6, 159–169. [Google Scholar]
- Alagawany, M.; El-Hack, A.; Mohamed, E.; Farag, M.R.; Sachan, S.; Karthik, K.; Dhama, K. The use of probiotics as eco-friendly alternatives for antibiotics in poultry nutrition. Environ. Sci. Pollut. Res. 2018, 25, 10611–10618. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, L.; Lv, Y.; Chen, Q.; Feng, J.; Zhao, X. Lactobacillus plantarum restores intestinal permeability disrupted by Salmonella infection in newly-hatched chicks. Sci. Rep. 2018, 8, 2229. [Google Scholar] [CrossRef] [PubMed]
- Olnood, C.G.; Beski, S.S.; Choct, M.; Iji, P.A. Use of Lactobacillus johnsonii in broilers challenged with Salmonella sofia. Anim. Nutr. 2015, 1, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Murugesan, G.R.; Persia, M.E. Influence of a direct-fed microbial and xylanase enzyme on the dietary energy uptake efficiency and performance of broiler chickens. J. Sci. Food Agric. 2015, 95, 2521–2527. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Raza, A.; Ahmad, M.A.; Li, L. Nutrient sensing mechanism of short-chain fatty acids in mastitis control. Microb. Pathog. 2022, 170, 105692. [Google Scholar] [CrossRef]
- Anwar, H.; Rahman, Z.U. Efficacy of protein, symbiotic and probiotic supplementation on production performance and egg quality characteristics in molted layers. Trop. Anim. Health Prod. 2016, 48, 1361–1367. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Li, C.; Kuang, M.; Shah, A.U.; Shafiq, M.; Ahmad, M.A.; Abdalmegeed, D.; Li, L.; Wang, G. Nrf2 Activation and NF-Kb & caspase/bax signaling inhibition by sodium butyrate alleviates LPS-induced cell injury in bovine mammary epithelial cells. Mol. Immunol. 2022, 148, 54–67. [Google Scholar]
- Rajput, D.S.; Zeng, D.; Khalique, A.; Rajput, S.S.; Wang, H.; Zhao, Y.; Sun, N.; Ni, X. Pretreatment with probiotics ameliorate gut health and necrotic enteritis in broiler chickens, a substitute to antibiotics. AMB Express 2020, 10, 220. [Google Scholar] [CrossRef]
- Arsène, M.M.; Davares, A.K.; Andreevna, S.L.; Vladimirovich, E.A.; Carime, B.Z.; Marouf, R.; Khelifi, I. The use of probiotics in animal feeding for safe production and as potential alternatives to antibiotics. Vet. World 2021, 14, 319. [Google Scholar] [CrossRef]
- Kogut, M.H. The effect of microbiome modulation on the intestinal health of poultry. Anim. Feed Sci. Technol. 2019, 250, 32–40. [Google Scholar] [CrossRef]
- Yadav, S.; Jha, R. Strategies to modulate the intestinal microbiota and their effects on nutrient utilization, performance, and health of poultry. J. Anim. Sci. Biotechnol. 2019, 10, 2. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.; Yu, Y.-H.; Hsiao, F.S.-H.; Su, C.-H.; Liu, H.-C.; Tobin, I.; Zhang, G.; Cheng, Y.-H. Influence of Heat Stress on Poultry Growth Performance, Intestinal Inflammation, and Immune Function and Potential Mitigation by Probiotics. Animals 2022, 12, 2297. [Google Scholar] [CrossRef] [PubMed]
- Jha, R.; Das, R.; Oak, S.; Mishra, P. Probiotics (direct-fed microbials) in poultry nutrition and their effects on nutrient utilization, growth and laying performance, and gut health: A systematic review. Animals 2020, 10, 1863. [Google Scholar] [CrossRef] [PubMed]
- Khalique, A.; Zeng, D.; Shoaib, M.; Wang, H.; Qing, X.; Rajput, D.S.; Pan, K.; Ni, X. Probiotics mitigating subclinical necrotic enteritis (SNE) as potential alternatives to antibiotics in poultry. AMB Express 2020, 10, 50. [Google Scholar] [CrossRef]
- Robert, H.; Payros, D.; Pinton, P.; Theodorou, V.; Mercier-Bonin, M.; Oswald, I.P. Impact of mycotoxins on the intestine: Are mucus and microbiota new targets? J. Toxicol. Environ. Health Part B 2017, 20, 249–275. [Google Scholar] [CrossRef]
- Bezirtzoglou, E.E.V. Intestinal cytochromes P450 regulating the intestinal microbiota and its probiotic profile. Microb. Ecol. Health Dis. 2012, 23, 1. [Google Scholar] [CrossRef] [PubMed]
- Barkhidarian, B.; Roldos, L.; Iskandar, M.M.; Saedisomeolia, A.; Kubow, S. Probiotic supplementation and micronutrient status in healthy subjects: A systematic review of clinical trials. Nutrients 2021, 13, 3001. [Google Scholar] [CrossRef]
- Meyer, M.M.; Fries-Craft, K.A.; Bobeck, E.A. Composition and inclusion of probiotics in broiler diets alter intestinal permeability and spleen immune cell profiles without negatively affecting performance. J. Anim. Sci. 2020, 98, skz383. [Google Scholar] [CrossRef]
- Yu, Y.-H.; Wu, C.-M.; Chen, W.-J.; Hua, K.-F.; Liu, J.-R.; Cheng, Y.-H. Effectiveness of Bacillus licheniformis-Fermented Products and Their Derived Antimicrobial Lipopeptides in Controlling Coccidiosis in Broilers. Animals 2021, 11, 3576. [Google Scholar] [CrossRef]
- Nochi, T.; Jansen, C.A.; Toyomizu, M.; Eden, W. The well-developed mucosal immune systems of birds and mammals allow for similar approaches of mucosal vaccination in both types of animals. Front. Nutr. 2018, 5, 60. [Google Scholar] [CrossRef]
- Rostagno, M.H. Effects of heat stress on the gut health of poultry. J. Anim. Sci. 2020, 98, skaa090. [Google Scholar] [CrossRef] [PubMed]
- Haykin, H.; Rolls, A. The neuroimmune response during stress: A physiological perspective. Immunity 2021, 54, 1933–1947. [Google Scholar] [CrossRef]
- Nguyen, J.; Pepin, D.M.; Tropini, C. Cause or effect? The spatial organization of pathogens and the gut microbiota in disease. Microbes Infect. 2021, 23, 104815. [Google Scholar] [CrossRef]
- Cristofori, F.; Dargenio, V.N.; Dargenio, C.; Miniello, V.L.; Barone, M.; Francavilla, R. Anti-inflammatory and immunomodulatory effects of probiotics in gut inflammation: A door to the body. Front. Immunol. 2021, 12, 178. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Cheng, Y.; Guan, L.; Zhou, Z.; Li, X.; Shi, D.; Xiao, Y. Bacillus amyloliquefaciens TL downregulates the ileal expression of genes involved in immune responses in broiler chickens to improve growth performance. Microorganisms 2021, 9, 382. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.-H.; Horng, Y.-B.; Dybus, A.; Yu, Y.-H. Bacillus licheniformis-fermented products improve growth performance and intestinal gut morphology in broilers under Clostridium perfringens challenge. Poult. Sci. J. 2021, 58, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Jacquier, V.; Nelson, A.; Jlali, M.; Rhayat, L.; Brinch, K.; Devillard, E. Bacillus subtilis 29784 induces a shift in broiler gut microbiome toward butyrate-producing bacteria and improves intestinal histomorphology and animal performance. Poult. Sci. 2019, 98, 2548–2554. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Shafi, M.E.; Qattan, S.Y.; Batiha, G.E.; Khafaga, A.F.; Abdel-Moneim, A.M.E.; Alagawany, M. Probiotics in poultry feed: A comprehensive review. J. Anim. Physiol. Anim. Nutr. 2020, 104, 1835–1850. [Google Scholar] [CrossRef] [PubMed]
- Yitbarek, A.; Echeverry, H.; Munyaka, P.; Rodriguez-Lecompte, J. Innate immune response of pullets fed diets supplemented with prebiotics and synbiotics. Poult. Sci. 2015, 94, 1802–1811. [Google Scholar] [CrossRef]
- Hatab, M.; Elsayed, M.; Ibrahim, N. Effect of some biological supplementation on productive performance, physiological and immunological response of layer chicks. J. Radiat. Res. Appl. Sci. 2016, 9, 185–192. [Google Scholar] [CrossRef]
- Manafi, M.; Khalaji, S.; Hedayati, M.; Pirany, N. Efficacy of Bacillus subtilis and bacitracin methylene disalicylate on growth performance, digestibility, blood metabolites, immunity, and intestinal microbiota after intramuscular inoculation with Escherichia coli in broilers. Poult. Sci. 2017, 96, 1174–1183. [Google Scholar] [CrossRef] [PubMed]
- Bajagai, Y.S.; Klieve, A.V.; Dart, P.J.; Bryden, W.L. Probiotics in Animal Nutrition: Production, Impact and Regulation; FAO: Rome, Italy, 2016. [Google Scholar]
- Zaefarian, F.; Abdollahi, M.; Ravindran, V. Particle size and feed form in broiler diets: Impact on gastrointestinal tract development and gut health. Worlds Poult. Sci. J. 2016, 72, 277–290. [Google Scholar]
- Hrnčár, C.; Gašparovič, M.; Weis, J.; Arpášová, H.; Pistová, V.; Fik, M.; Bujko, J. Effect of three-strain probiotic on productive performance and carcass characteristics of broiler chickens. Sci. Pap. J. Anim. Sci. Biotechnol. 2016, 49, 149–154. [Google Scholar]
- Ebeid, T.; Al-Homidan, I.; Fathi, M.; Al-Jamaan, R.; Mostafa, M.; Abou-Emera, O.; El-Razik, M.A.; Alkhalaf, A. Impact of probiotics and/or organic acids supplementation on growth performance, microbiota, antioxidative status, and immune response of broilers. Ital. J. Anim. Sci. 2021, 20, 2263–2273. [Google Scholar] [CrossRef]
- Dittoe, D.K.; Ricke, S.C.; Kiess, A.S. Organic acids and potential for modifying the avian gastrointestinal tract and reducing pathogens and disease. Front. Vet. Sci. 2018, 5, 216. [Google Scholar] [CrossRef]
- Ke, A.; Parreira, V.R.; Goodridge, L.; Farber, J.M. Current and future perspectives on the role of probiotics, prebiotics, and synbiotics in controlling pathogenic Cronobacter spp. in infants. Front. Microbiol. 2021, 12, 755083. [Google Scholar] [CrossRef]
- Nari, N.; Ghasemi, H.; Hajkhodadadi, I.; Farahani, A.K. Intestinal microbial ecology, immune response, stress indicators, and gut morphology of male broiler chickens fed low-phosphorus diets supplemented with phytase, butyric acid, or Saccharomyces boulardii. Livest. Sci. 2020, 234, 103975. [Google Scholar] [CrossRef]
- Ng, W.K.; Koh, C.B. The utilization and mode of action of organic acids in the feeds of cultured aquatic animals. Rev. Aquac. 2017, 9, 342–368. [Google Scholar] [CrossRef]
- Marsh, P.D.; Head, D.A.; Devine, D.A. Dental plaque as a biofilm and a microbial community—Implications for treatment. J. Oral Biosci. 2015, 57, 185–191. [Google Scholar] [CrossRef]
- Ayyash, M.M.; Abdalla, A.K.; AlKalbani, N.S.; Baig, M.A.; Turner, M.S.; Liu, S.-Q.; Shah, N.P. Invited review: Characterization of new probiotics from dairy and nondairy products—Insights into acid tolerance, bile metabolism and tolerance, and adhesion capability. J. Dairy Sci. 2021, 104, 8363–8379. [Google Scholar] [CrossRef]
- Rodrigues, I.; Choct, M. The foregut and its manipulation via feeding practices in the chicken. Poult. Sci. 2018, 97, 3188–3206. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.A.; Sylte, M.J.; Looft, T. In-feed bacitracin methylene disalicylate modulates the turkey microbiota and metabolome in a dose-dependent manner. Sci. Rep. 2019, 9, 8212. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.H.T.; Everaert, N.; Bindelle, J. Review on the effects of potential prebiotics on controlling intestinal enteropathogens Salmonella and Escherichia coli in pig production. J. Anim. Physiol. Anim. Nutr. 2018, 102, 17–32. [Google Scholar] [CrossRef]
- Arif, M.; Akteruzzaman, M.; Islam, S.S.; Das, B.C.; Siddique, M.P.; Kabir, S.L. Dietary supplementation of Bacillus-based probiotics on the growth performance, gut morphology, intestinal microbiota and immune response in low biosecurity broiler chickens. Vet. Anim. Sci. 2021, 14, 100216. [Google Scholar] [CrossRef] [PubMed]
- Foligne, B.; Nutten, S.; Grangette, C.; Dennin, V.; Goudercourt, D.; Poiret, S.; Dewulf, J.; Brassart, D.; Mercenier, A.; Pot, B. Correlation between in vitro and in vivo immunomodulatory properties of lactic acid bacteria. World J. Gastroenterol. WJG 2007, 13, 236. [Google Scholar] [CrossRef] [PubMed]
- Wanasawaeng, N.J.P.S.W.; Chansiripornchai, N. Efficacy of competitive exclusion to reduce Salmonella in broiler chickens. Thai. J. Vet. Med. 2019, 49, 385–391. [Google Scholar]
- El-Moneim, A.E.-M.E.A.; El-Wardany, I.; Abu-Taleb, A.M.; Wakwak, M.M.; Ebeid, T.A.; Saleh, A.A. Assessment of in ovo administration of Bifidobacterium bifidum and Bifidobacterium longum on performance, ileal histomorphometry, blood hematological, and biochemical parameters of broilers. Probiotics Antimicrob. Proteins 2020, 12, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Abou-Kassem, D.; Elsadek, M.; Abdel-Moneim, A.; Mahgoub, S.; Elaraby, G.; Taha, A.; Elshafie, M.; Alkhawtani, D.; Abd El-Hack, M.; Ashour, E. Growth, carcass characteristics, meat quality, and microbial aspects of growing quail fed diets enriched with two different types of probiotics (Bacillus toyonensis and Bifidobacterium bifidum). Poult. Sci. 2021, 100, 84–93. [Google Scholar] [CrossRef]
- Abdelqader, A.; Abuajamieh, M.; Hayajneh, F.; Al-Fataftah, A.-R. Probiotic bacteria maintain normal growth mechanisms of heat stressed broiler chickens. J. Therm. Biol. 2020, 92, 102654. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Hoseinifar, S.H.; Sun, Y.-Z.; Wang, A.; Zhou, Z. Probiotics as means of diseases control in aquaculture, a review of current knowledge and future perspectives. Front. Microbiol. 2018, 9, 2429. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Liu, C.; Fang, H.; Zhang, D. Bacillus subtilis: A universal cell factory for industry, agriculture, biomaterials and medicine. Microb. Cell Factories 2020, 19, 1–12. [Google Scholar] [CrossRef]
- Gao, Y.; Niu, M.; Yu, X.; Bao, T.; Wu, Z.; Zhao, X. Horizontally Acquired Polysaccharide-Synthetic Gene Cluster From Weissella cibaria Boosts the Probiotic Property of Lactiplantibacillus plantarum. Front. Microbiol. 2021, 12, 1668. [Google Scholar] [CrossRef] [PubMed]
- Elmi, V.A.; Moradi, S.; Harsini, S.G.; Rahimi, M. Effects of Lactobacillus acidophilus and natural antibacterials on growth performance and Salmonella colonization in broiler chickens challenged with Salmonella enteritidis. Livest. Sci. 2020, 233, 103948. [Google Scholar] [CrossRef]
- Nishiyama, T.; Ashida, N.; Nakagawa, K.; Iwatani, S.; Yamamoto, N. Dietary Bacillus subtilis C-3102 supplementation enhances the exclusion of Salmonella enterica from chickens. Poult. Sci. J. 2021, 58, 138–145. [Google Scholar] [CrossRef]
- Wang, J.; Wan, C.; Shuju, Z.; Yang, Z.; Celi, P.; Ding, X.; Bai, S.; Zeng, Q.; Mao, X.; Xu, S. Differential analysis of gut microbiota and the effect of dietary Enterococcus faecium supplementation in broiler breeders with high or low laying performance. Poult. Sci. 2021, 100, 1109–1119. [Google Scholar] [CrossRef] [PubMed]
- Han, G.G.; Song, A.A.; Kim, E.B.; Yoon, S.-H.; Bok, J.-D.; Cho, C.-S.; Kil, D.Y.; Kang, S.-K.; Choi, Y.-J. Improved antimicrobial activity of Pediococcus acidilactici against Salmonella Gallinarum by UV mutagenesis and genome shuffling. Appl. Microbiol. Biotechnol. 2017, 101, 5353–5363. [Google Scholar] [CrossRef]
- Kanwal, H.; Di Cerbo, A.; Zulfiqar, F.; Sabia, C.; Nawaz, A.; Siddiqui, F.M.; Aqeel, M.; Ghazanfar, S. Probiotic Characterization and Population Diversity Analysis of Gut-Associated Pediococcus acidilactici for Its Potential Use in the Dairy Industry. Appl. Sci. 2021, 11, 9586. [Google Scholar] [CrossRef]
- Penha Filho, R.A.C.; Díaz, S.J.A.; Fernando, F.S.; Chang, Y.-F.; Andreatti Filho, R.L.; Junior, A.B. Immunomodulatory activity and control of Salmonella Enteritidis colonization in the intestinal tract of chickens by Lactobacillus based probiotic. Vet. Immunol. Immunopathol. 2015, 167, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Yang, K.; Zhang, A.; Chang, W.; Zheng, A.; Chen, Z.; Cai, H.; Liu, G. Effects of Lactobacillus acidophilus on the growth performance, immune response, and intestinal barrier function of broiler chickens challenged with Escherichia coli O157. Poult. Sci. 2021, 100, 101323. [Google Scholar] [CrossRef] [PubMed]
- Richards-Rios, P. Understanding the Chicken Intestinal Microbiome: Towards a Rational Approach to Feed-Based Interventions. Doctoral Dissertation, The University of Liverpool, Liverpool, UK, 2020. [Google Scholar]
- Zhang, S.; Zhong, G.; Shao, D.; Wang, Q.; Hu, Y.; Wu, T.; Ji, C.; Shi, S. Dietary supplementation with Bacillus subtilis promotes growth performance of broilers by altering the dominant microbial community. Poult. Sci. 2021, 100, 100935. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Zaki, R.; Negm, E.; Mahmoud, M.; Cheng, H. Effects of dietary supplementation of a probiotic (Bacillus subtilis) on bone mass and meat quality of broiler chickens. Poult. Sci. 2021, 100, 100906. [Google Scholar] [CrossRef]
- Keerqin, C.; Rhayat, L.; Zhang, Z.-H.; Gharib-Naseri, K.; Kheravii, S.; Devillard, E.; Crowley, T.; Wu, S.-B. Probiotic Bacillus subtilis 29,784 improved weight gain and enhanced gut health status of broilers under necrotic enteritis condition. Poult. Sci. 2021, 100, 100981. [Google Scholar] [CrossRef] [PubMed]
- Neijat, M.; Shirley, R.; Barton, J.; Thiery, P.; Welsher, A.; Kiarie, E. Effect of dietary supplementation of Bacillus subtilis DSM29784 on hen performance, egg quality indices, and apparent retention of dietary components in laying hens from 19 to 48 weeks of age. Poult. Sci. 2019, 98, 5622–5635. [Google Scholar] [CrossRef] [PubMed]
- Zaghari, M.; Sarani, P.; Hajati, H. Comparison of two probiotic preparations on growth performance, intestinal microbiota, nutrient digestibility and cytokine gene expression in broiler chickens. J. Appl. Anim. Res. 2020, 48, 166–175. [Google Scholar] [CrossRef]
- Khan, S.; Yang, R.; Khalid, A.; Wang, Z. Probiotic Feeding Effect of Bacillus Subtilis on Broilers Chicks’ Microflora, TLRs and Interleukin Gene Expression; Research Square, Anhui Agricultural University: Hefei, China, 2021. [Google Scholar]
- Hasan, M.R. Effect of Probiotics Instead of Antibiotics as Growth Promoters in Broiler Production; Department of Animal Production and Management, Sher-e-bangla Agricultural: Dhaka, Bangladesh, 2019. [Google Scholar]
- Cheng, Y.H.; Zhang, N.; Han, J.C.; Chang, C.W.; Hsiao, F.S.H.; Yu, Y.H. Optimization of surfactin production from Bacillus subtilis in fermentation and its effects on Clostridium perfringens-induced necrotic enteritis and growth performance in broilers. J. Anim. Physiol. Anim. Nutr. 2018, 102, 1232–1244. [Google Scholar] [CrossRef]
- Cheng, Y.-H.; Horng, Y.-B.; Chen, W.-J.; Hua, K.-F.; Dybus, A.; Hsiao, F.-H.; Yu, Y.-H. Development and validation the efficacy of Bacillus-based fermented products as an antibiotics alternative in domestic animals. Acta Sci. Pol. Zootech. 2021, 20, 23–34. [Google Scholar] [CrossRef]
- Duangnumsawang, Y.; Zentek, J.; Goodarzi Boroojeni, F. Development and functional properties of intestinal mucus layer in poultry. Front. Immunol. 2021, 12, 745849. [Google Scholar] [CrossRef]
- He, T.; Long, S.; Mahfuz, S.; Wu, D.; Wang, X.; Wei, X.; Piao, X. Effects of probiotics as antibiotics substitutes on growth performance, serum biochemical parameters, intestinal morphology, and barrier function of broilers. Animals 2019, 9, 985. [Google Scholar] [CrossRef]
- Jazi, V.; Foroozandeh, A.; Toghyani, M.; Dastar, B.; Koochaksaraie, R.R. Effects of Pediococcus acidilactici, mannan-oligosaccharide, butyric acid and their combination on growth performance and intestinal health in young broiler chickens challenged with Salmonella Typhimurium. Poult. Sci. 2018, 97, 2034–2043. [Google Scholar] [CrossRef]
- Adhikari, P.; Kiess, A.; Adhikari, R.; Jha, R. An approach to alternative strategies to control avian coccidiosis and necrotic enteritis. J. Appl. Poult. Res. 2020, 29, 515–534. [Google Scholar] [CrossRef]
- Aristimunha, P.; Rosa, A.; Boemo, L.; Garcez, D.; Rosa, D.; Londero, A.; Scher, A.; Forgiarini, J. A blend of benzoic acid and essential oil compounds as an alternative to antibiotic growth promoters in broiler diets. J. Appl. Poult. Res. 2016, 25, 455–463. [Google Scholar]
- Pereira, R.; Menten, J.F.M.; Bortoluzzi, C.; Napty, G.; Longo, F.; Vittori, J.; Lourenço, M.; Santin, E. Organic acid blend in diets of broiler chickens challenged with Clostridium perfringens. J. Appl. Poult. Res. 2015, 24, 387–393. [Google Scholar] [CrossRef]
- Qing, X.; Zeng, D.; Wang, H.; Ni, X.; Liu, L.; Lai, J.; Khalique, A.; Pan, K.; Jing, B. Preventing subclinical necrotic enteritis through Lactobacillus johnsonii BS15 by ameliorating lipid metabolism and intestinal microflora in broiler chickens. AMB Express 2017, 7, 139. [Google Scholar] [CrossRef]
- He, Y.; Liu, X.; Dong, Y.; Lei, J.; Ito, K.; Zhang, B. Enterococcus faecium PNC01 isolated from the intestinal mucosa of chicken as an alternative for antibiotics to reduce feed conversion rate in broiler chickens. Microb. Cell Factories 2021, 20, 122. [Google Scholar] [CrossRef]
- Svetoch, E.A.; Eruslanov, B.V.; Perelygin, V.V.; Mitsevich, E.V.; Mitsevich, I.P.; Borzenkov, V.N.; Levchuk, V.P.; Svetoch, O.E.; Kovalev, Y.N.; Stepanshin, Y.G. Diverse antimicrobial killing by Enterococcus faecium E 50-52 bacteriocin. J. Agric. Food Chem. 2008, 56, 1942–1948. [Google Scholar] [CrossRef] [PubMed]
- Volzing, K.; Borrero, J.; Sadowsky, M.J.; Kaznessis, Y.N. Antimicrobial peptides targeting Gram-negative pathogens, produced and delivered by lactic acid bacteria. ACS Synth. Biol. 2013, 2, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jiang, Y.-L.; Liu, J.; Gao, X.; Zhang, Z.; Huang, H.-B.; Yang, W.-T.; Shi, C.-W.; Cao, X.; Yang, G.-L. Recombinant invasive Lactobacillus plantarum expressing the Eimeria tenella fusion gene TA4 and AMA1 induces protection against coccidiosis in chickens. Vet. Parasitol. 2020, 283, 109161. [Google Scholar]
| Probiotic Strains | Biological Performance | Reference |
|---|---|---|
| Bacillus amyloliquefaciencs | Improve intestinal health and growth performance | [34,35] |
| Bacillus coagulans | Enhances growth performance and gut health | [36] |
| Lactobacillus acidophillus | Improve production performance and helps the immune system and gut histomorphology | [37,38] |
| Lactobacillus bulgaricus | Enhances growth performances and improves immune functions | [39] |
| Pediococcus acidilactici | Improves laying performances and modulates intestinal microflora composition | [40,41] |
| Propionibacterium acidipropionic | Contributes to the better development of intestinal mucosa and microbiota composition | [42] |
| Saccharomyces cerevisiae | Improves growth performance and enhances laying performance | [43] |
| Streptococcus faecium | Avoided the impairment and regulated the stability of the epithelial intestine, and improves the immune functions | [44] |
| Probiotic | Biological Performance | Reference |
|---|---|---|
| Lactiplantibacillus plantrum LTC-113 | Enhances immunity against Salmonella typhimurium, and preserves intestinal epithelial barrier function | [45,97] |
| Lactobacillus johnsonii | Alleviates Salmonella sofia and Clostridium perfringens infection | [46,98] |
| Bacillus subtilis C-3102 | Decreases the number of Salmonella enterica serovars (enteritidis LM-7) | [99,100] |
| Pediococcus acidilactici | Decreases the number of Salmonella enterica serovars (Gallinarum) | [101,102] |
| Lactobacillus acidophilus | Improves body weight, decreases mortality, enhances the immune response in Escherichia coli 0157-challenged chickens | [103,104] |
| Bacillus subtilis | Increases the ratio of villus height to crypt depth, surface area available for nutrient absorption in the duodenum and ileum, the number of Blautia, Faecalibacterium and Romboutsia, and the amount of beneficial microflora (Lactobacillus, Bifidobacterium, and Enterococcus) | [105,106] |
| Bacillus subtilis PB-6 | Boosts plasma calcium and phosphorus concentrations, broiler production and welfare, increases bone mass and meat quality | [93,107] |
| Bacillus subtilis DSM29784 | Increases the number of Butyricicoccus and Faecalibacterium in the intestine, improves health, weight, and the tight junction complex in broilers suffering from necrotic enteritis | [108,109] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, R.; Yu, Y.-H.; Hsiao, F.S.-H.; Dybus, A.; Ali, I.; Hsu, H.-C.; Cheng, Y.-H. Probiotics as a Friendly Antibiotic Alternative: Assessment of Their Effects on the Health and Productive Performance of Poultry. Fermentation 2022, 8, 672. https://doi.org/10.3390/fermentation8120672
Ahmad R, Yu Y-H, Hsiao FS-H, Dybus A, Ali I, Hsu H-C, Cheng Y-H. Probiotics as a Friendly Antibiotic Alternative: Assessment of Their Effects on the Health and Productive Performance of Poultry. Fermentation. 2022; 8(12):672. https://doi.org/10.3390/fermentation8120672
Chicago/Turabian StyleAhmad, Rafiq, Yu-Hsiang Yu, Felix Shih-Hsiang Hsiao, Andrzej Dybus, Ilyas Ali, Hui-Chen Hsu, and Yeong-Hsiang Cheng. 2022. "Probiotics as a Friendly Antibiotic Alternative: Assessment of Their Effects on the Health and Productive Performance of Poultry" Fermentation 8, no. 12: 672. https://doi.org/10.3390/fermentation8120672
APA StyleAhmad, R., Yu, Y.-H., Hsiao, F. S.-H., Dybus, A., Ali, I., Hsu, H.-C., & Cheng, Y.-H. (2022). Probiotics as a Friendly Antibiotic Alternative: Assessment of Their Effects on the Health and Productive Performance of Poultry. Fermentation, 8(12), 672. https://doi.org/10.3390/fermentation8120672








