1. Introduction
Vinification by carbonic maceration (CM) involves the process whereby the whole grapes are subjected to anaerobic conditions [
1]. When the grapes are placed in a carbon dioxide (CO
2)-enriched medium, they immediately change from a respiratory metabolism to an anaerobic fermentative metabolism called intracellular fermentation (IF). For this reason, in CM the intact grape clusters, without destemming or crushing, are placed into tanks and kept under a CO
2 atmosphere. Some grapes located in the lower zone of the tank are crushed by the weight and pressure produced by the ones above and release must to the bottom of the tank. This released must is fermented by yeasts. Under anaerobic conditions, the IF is carried out by grape enzymes inside the whole berries, triggering the production of alcohol, the degradation of malic acid, pectolytic, and proteolytic phenomena, the formation of volatile compounds, and the diffusion of phenolic compounds from the skin to the pulp [
2]. As a result of these processes, the whole grapes, at a certain moment, split open and release their juice into the tank, increasing the liquid phase, which continues to fermenting due to the yeasts. Therefore, the IF of the grapes and the alcoholic fermentation (AF) of the must by yeasts occur simultaneously in the tank. After this first phase, drawing off is done by racking a free-run, partly fermented wine, and the grapes that remain whole are pressed to release a higher-density press-wine. Then, a second phase begins when both wines—mixed or separate—complete their AF by the presence of yeast and the malolactic fermentation (MLF) by lactic acid bacteria (LAB).
CM winemaking is typically used in different European regions such as Beaujolais in France, Vino novello in Italy, and Cosechero wine in La Rioja, Spain. Although CM wines are recognized as high quality young wines, recent works that describe their physicochemical and microbiological characteristics are scarce and contradictory. The contradictory results could be due to a wide range of causes such as the grape variety, the grape ripeness level, the vintage, or the winemaking conditions [
3,
4,
5]. Therefore, it is paradoxical that being a traditional processing system and widely used in many wineries, some of the phenomena that take place and the parameters that characterize them are barely known. This means that, currently, these wines are still being produced almost empirically. Consequently, to gather information about the intrinsic characteristics of CM wines and to study in depth this winemaking system, it is necessary to carry out small-scale scientific studies. However, it is possible that winemaking in small volumes sometimes does not work in the same way as in industrial cellars and results are not comparable.
This work shows the difficulties and risks of carrying out small-scale winemaking by carbonic maceration and the need to strictly control environmental conditions to achieve it.
2. Materials and Methods
2.1. Assay in Experimental Winery Scale
The study was developed in an experimental winery in 300-L tanks under conditions similar to those in the cellars. In this assay six vinifications were carried out in 300-kg tanks starting from 1800 kg of grapes of the Tempranillo variety. Three tanks were vinified by carbonic maceration (CM) and the other three by destemming and crushing (DC). The chemical composition of the initial must was: probable alcoholic strength 12.8%, pH 3.32, and malic acid 2.49 g/L.
Before the distribution of the bunches into the tanks, 250 kg of grapes were destemmed, crushed, and pressed to obtain 120 L of must that was sulfited at a dose of 40 mg/L of SO2. After this, the CM tanks were filled with industrial CO2 and eventually the whole grape bunches were carefully introduced in a proportion of 270 Kg/tank. Later, a 20-L aliquot of the must obtained previously was added to the upper part of each tank in order to simulate the breakage that occurs in industrial tanks during filling.
AF was developed spontaneously at room temperature and with no pumping over. The carbonic level inside the tanks was checked daily and if necessary, more CO2 gas was added to maintain anaerobiosis. There was also a daily control of the density and temperature of the must taken from the bottom of the tank and the temperature of the bunch mass.
The tanks were devatted when the density of the liquid was 1000, and three fractions from each tank were obtained: free-run liquid in the tank (FCM), liquid after crushing the grapes inside the tank (CCM), and liquid obtained by pressing the solid mass with a pneumatic press (PCM). Subsequently, in each fraction, the alcoholic and malolactic fermentations developed spontaneously. At the end of both processes, the wines were sulfited with 40 mg/L of SO2.
In the vinifications by DC, 270 kg of grapes were destemmed, crushed, sulfited at a dose of 40 mg/L SO2, and placed in each tank. In these processes an additional volume of 20 L of the initial must was also added to equalize volumes. AF took place spontaneously at room temperature. The density and temperature of the liquid was measured daily after pumping over. The vat time was the same as in the tanks made by CM and after devatting and pressing, MLF was occurred spontaneously. In this case, only one type of wine was obtained (DC) as a result of mixing the liquid in the tank with that obtained after pressing the grape skins.
2.2. Physicochemical Analysis
The wines were characterized by measuring the pH, acetaldehyde, and volatile acidity according to the official European Community methods [
6]. The pH was measured with a CRISON micropH pHmeter (Barcelona, Spain), the acetaldehyde was determined enzymatically by a Boehringer Manheim test (Darmstadt, Germany), and the volatile acidity was analyzed with a TDI DE2000 distiller/extractor (Barcelona, Spain) and subsequent titration with 0.1 N of NaOH in the presence of phenolphthalein. The malic, lactic, acetic, and pyruvic acids, sugars, and easily assimilable nitrogen (EAN) were determined by enzymatic methods carried out via an automated analyzer (Miura One, TDI, Barcelona, Spain), following the supplier’s instructions.
2.3. Microbiological Analysis
The microbial community present at different times during vinifications (beginning, middle, and end) was analyzed. For this purpose, serial dilutions of the must or wine samples were plated onto different culture media. Total yeasts were measured by seeding onto a GYP culture medium (20 g/L of glucose, 5 g/L of yeast extract, 5 g/L of peptone, and 100 mg/L of chloramphenicol) (Panreac, Barcelona, Spain) and incubation at 28 °C for 48 h. LAB were measured by plating onto an MRS medium (52 g/L of MRS broth, 20 g/L of agar, and 50 mg/L of pymaricine) (Scharlau, Sentmenat, Spain) and incubation at 30 °C under strict anaerobic conditions for at least ten days. Acetic acid bacteria (AAB) were determined by seeding onto a Mann culture medium (25 g/L D-mannitol, 3 g/L peptone, 5 g/L yeast extract, 20 g/L agar, 3 U/mL penicillin and 50 mg/L pymaricine) (Scharlau, Sentmenat, Spain) and incubation at 28 °C for 48 h. Once the plates were seeded and turned over, we added approximately 0.1 g/plate of biphenyl (Merck, Darmstadt, Germany) to the lid to avoid the growth of molds that make it difficult to count colonies. When the incubation periods finished, the plates were examined and colony forming units per milliliter (CFU/mL) were counted in plates with 30–300 colonies.
2.4. Statistical Analysis of the Results
Analysis of variance (ANOVA) was carried out for all the analytical results determined in wines with the IBM® SPSS® Statistic version 23. Significant differences were established by using the Tukey post hoc test (p < 0.05–0.01).
4. Discussion
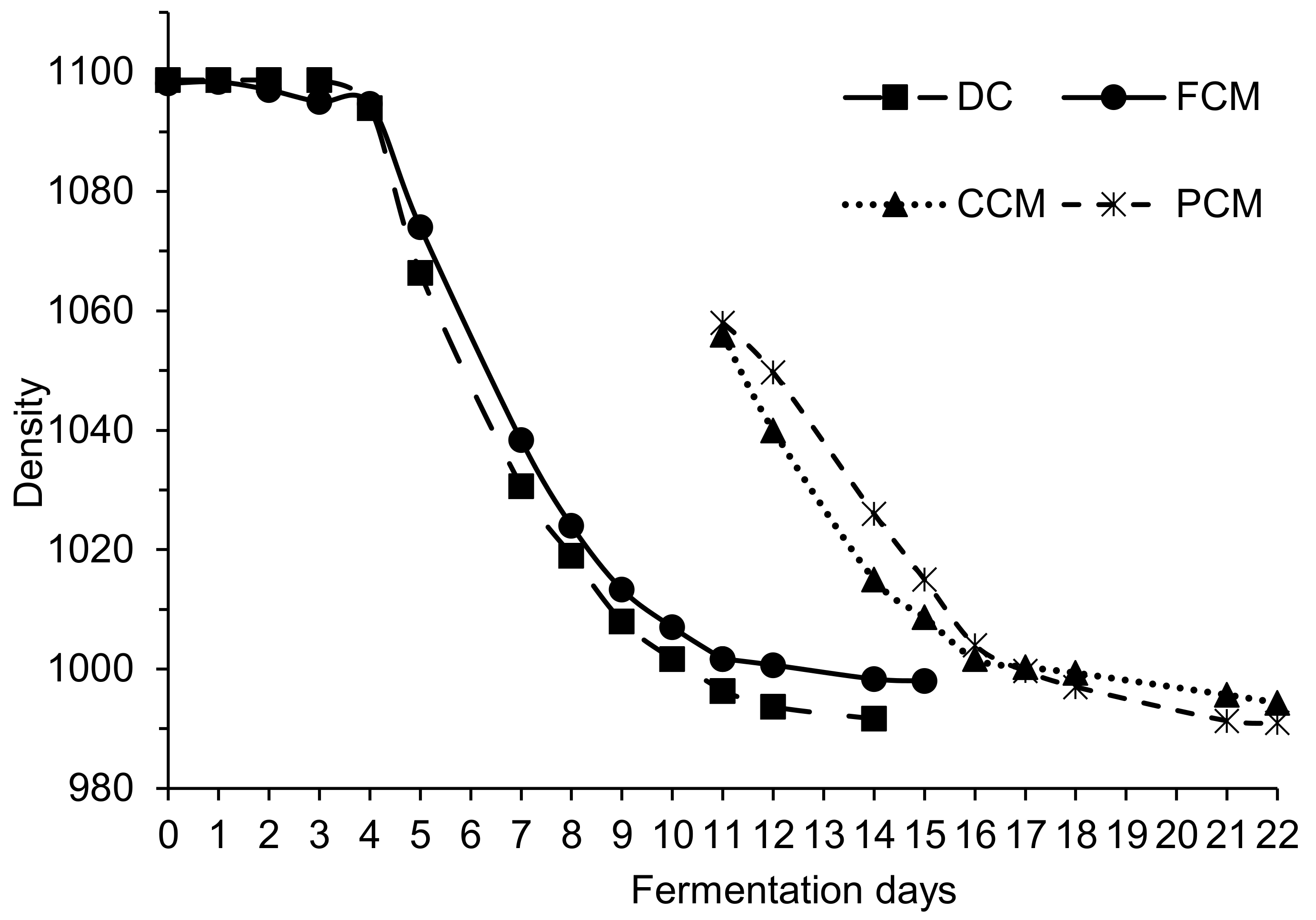
The slow progress of the alcoholic fermentation (
Figure 1) was probably due to the low temperature in the tanks. In the CM tanks the temperature of the must never exceeded 22 °C and the average temperature in the clusters throughout the period was 24 °C. In CM vinification, a temperature inside the whole grapes above 30 °C is essential for the IF to proceed optimally [
1,
2]. In our vinifications the grapes had a temperature of 17 °C when filling the tanks. During the 12 days of fermentation–maceration it was impossible to reach and maintain the optimum temperature (>30 °C). This could be due to the fact that the tanks were of small capacity and made of stainless steel. These characteristics favor the rapid release of the calories generated during fermentation to the environment, and the cooling of the tank (liquid and clusters). The traditional winemakers of the Qualified Designation of Origin (DOCa) Rioja make wines by CM in large capacity tanks made of concrete, a material that has higher thermal inertia than steel. In that case the right temperature is easily reached and maintained, and CM vinification proceeds without problems. But in order to study this vinification system in depth, it is essential to carry out tests with repetitions, and this entails the use of small tanks. In this type of studies, it is therefore necessary to ensure the right temperature to properly conduct the CM vinifications.
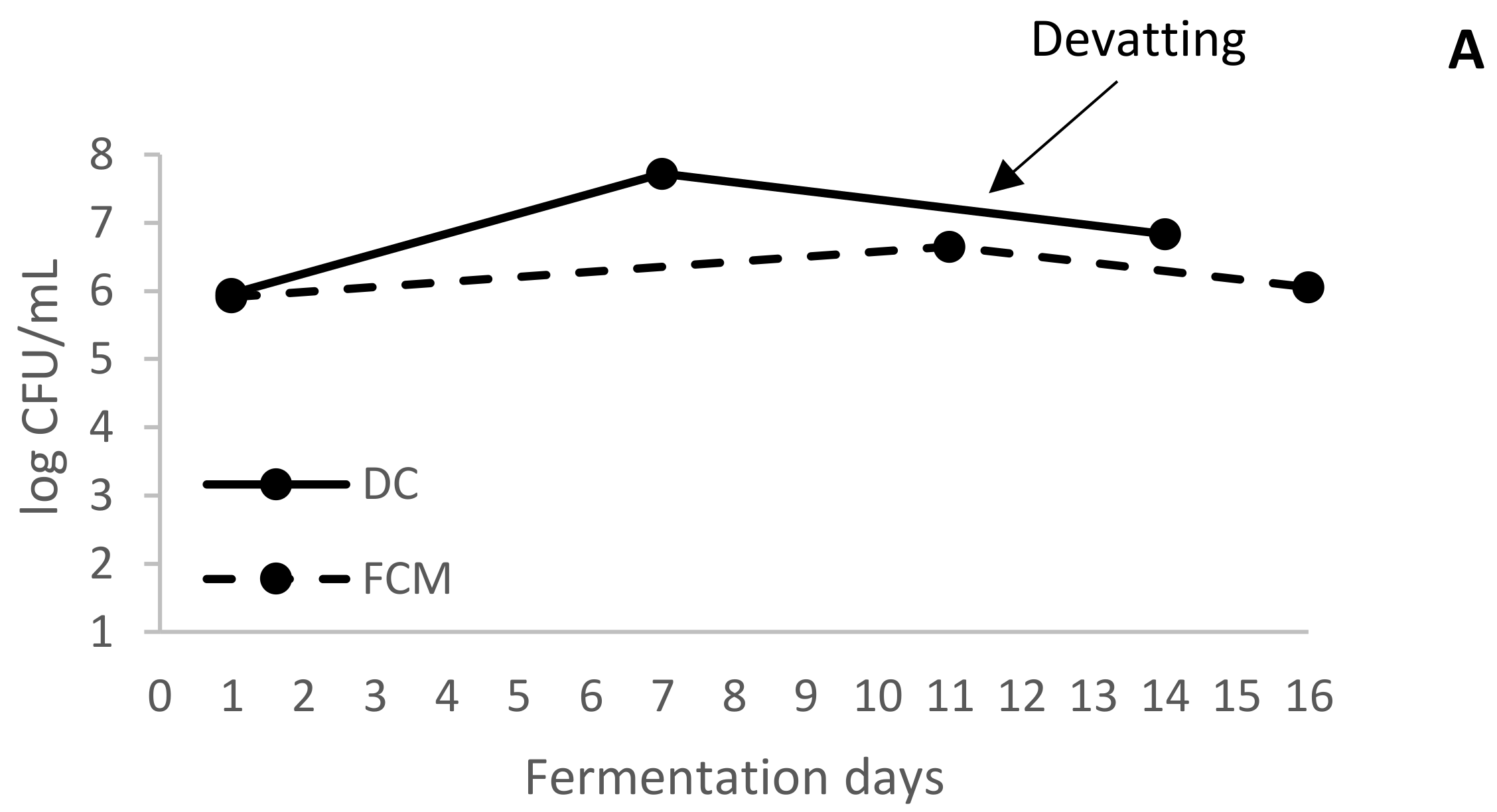
In addition, after devatting, CCM and PCM fractions took another 12 days to finish AF. In real winery conditions, the completion of alcoholic fermentation in those wines is normally achieved in 2–3 days, because at devatting time the yeast populations in the tanks are usually very high [
2]. These yeasts inoculate the musts obtained after pressing, causing rapid fermentation. The cause that can explain the delay in finishing the AF in CCM and PCM must-wines would be the low yeast population (
Figure 2A). For this reason, the yeast population that passed to CCM and PCM wines during crushing and pressing was low, slowing down the end of the alcoholic fermentations.
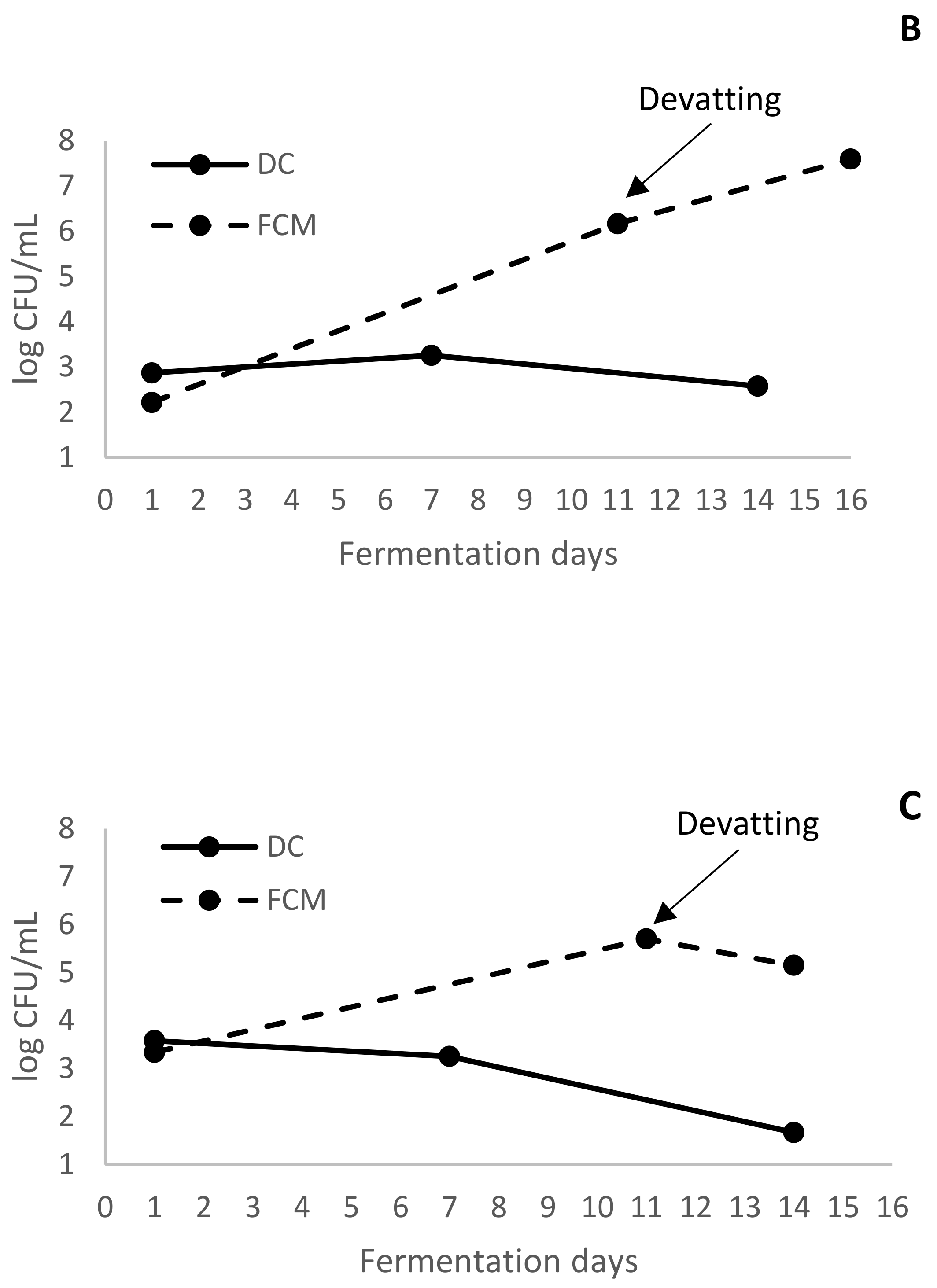
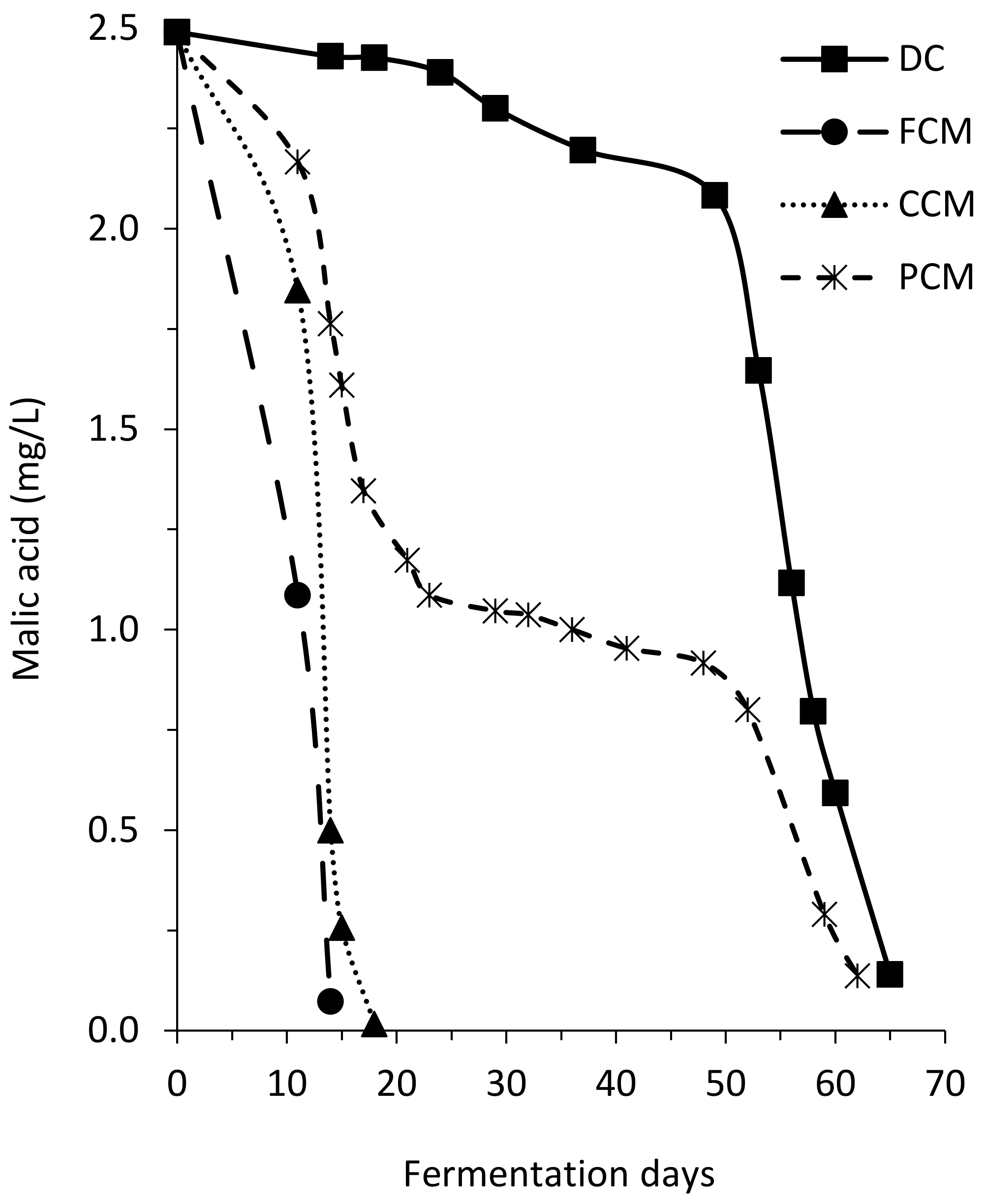
In addition to the low number of yeasts, a high population of LAB was present in the liquid of the tanks (
Figure 2B). Thus, at the devatting moment, the FCM wine had already started MLF and the completion of this process in FCM was fast (
Figure 3). During the emptying and pressing, the LAB present in the liquid contaminated the CCM and PCM must-wines, and in some fractions, there was an overlap of alcoholic and malolactic fermentations. This high presence of LAB caused a rapid development of the MLF but also an increase in volatile acidity and even the “
piqure” of these wines (
Table 2). However, in the wines made by DC the level of BAL was low, there was no overlap between the two fermentations and the volatile acidity level of the wines was correct.
Finally, there was also a great development of AAB in the deposits (
Figure 2C). These are strict aerobic microorganisms, and their presence would indicate that, despite the addition of industrial CO
2, adequate anaerobiosis was not achieved. Intact berries only shift from oxidative to fermentative metabolism under atmospheres with oxygen content lower than 1% [
2]. The morphology of the grape clusters means that air remains trapped in their structure. This air can be difficult to remove just by adding exogenous CO
2 to the tank. Perhaps only the continuous supply produced by an intense fermentation of the must at the bottom is capable of eliminating the residual air. This air could favor the development of AAB, which could also contribute to increase the volatile acidity of the wines.
In addition to liquid phase fermentations, IF was taking place inside the whole grape berries. The ability of grapes to adapt to anaerobic atmosphere and initiate IF, even the intensity of this process, depends on different factors that affect the enzymes involved in the process [
10]. The conditions that took place in the vinification tanks indicated above could affect the start-up and the development of IF inside the grape berries. The most important reaction of IF within grapes is the metabolism of malic acid that is decarboxylated into pyruvic acid, which is transformed into acetaldehyde, and this finally passes into ethanol [
11]. Pyruvic acid can also be transformed into other compounds such as glycerine and acetic, succinic, fumaric, shikimic acids. If the conditions are not suitable for the process, there could be difficulties in the route that could lead to the accumulation inside the grapes of intermediate compounds such as pyruvic, acetaldehyde or acetic acid. The composition of the three must-wine fractions obtained at the moment of devatting from the CM tanks (
Table 1), showed a higher amount of acetaldehyde and acetic acid in musts from whole grapes (CCC and PCM). These values could indicate an accumulation of intermediates from the degradation of malic acid during IF. The acetaldehyde level of the different fractions decreases during the subsequent fermentation (
Table 2), but the volatile acidity increases probably due to the additional action of the BAL and AAB, which causes abnormally high values of volatile acidity in the final wines.
All the indicated circumstances made the CM wines obtained (
Table 2) have high levels of volatile acidity and acetaldehyde. Different studies have shown that carbonic maceration vinification results in wines with slightly higher volatile acidities than destemming [
12,
13] and high amounts of acetaldehyde [
14]. Regarding the acidity parameters, there were no great differences between the wines. A higher pH should be expected in wines from CM [
11] due to the degradation of malic acid during IF. However, it is possible that the increase in pH due to this cause was compensated by the high acetic acid content of the three CM fractions.
In conclusion, in this test both lactic and acetic “piqure” occurred during the CM of grapes due to the low development of yeasts and the high populations of bacteria in the vats. There were also deviations in the IF probably because the optimal temperatures and anaerobiosis levels for this process were not reached.