Silage Fermentation: A Potential Microbial Approach for the Forage Utilization of Cyperus esculentus L. By-Product
Abstract
:1. Introduction
2. Materials and Methods
2.1. CLL Harvest and Silage Making
2.2. Analysis of Culture-Based Microbial
2.3. Fermentation Quality Analysis
2.4. Chemical Composition Analysis
2.5. Bacterial Diversity Sequenced by High-Throughput Sequencing
2.6. Metabolomics Analysis Using LC-MS
2.7. Calculation of Relative Feed Value and Fermentation Coefficient
2.8. Statistical Analysis
3. Results
3.1. Characteristics of Fresh CLL
3.2. Effect of Additives on Silage Quality and Microbial Population of CLL Silages
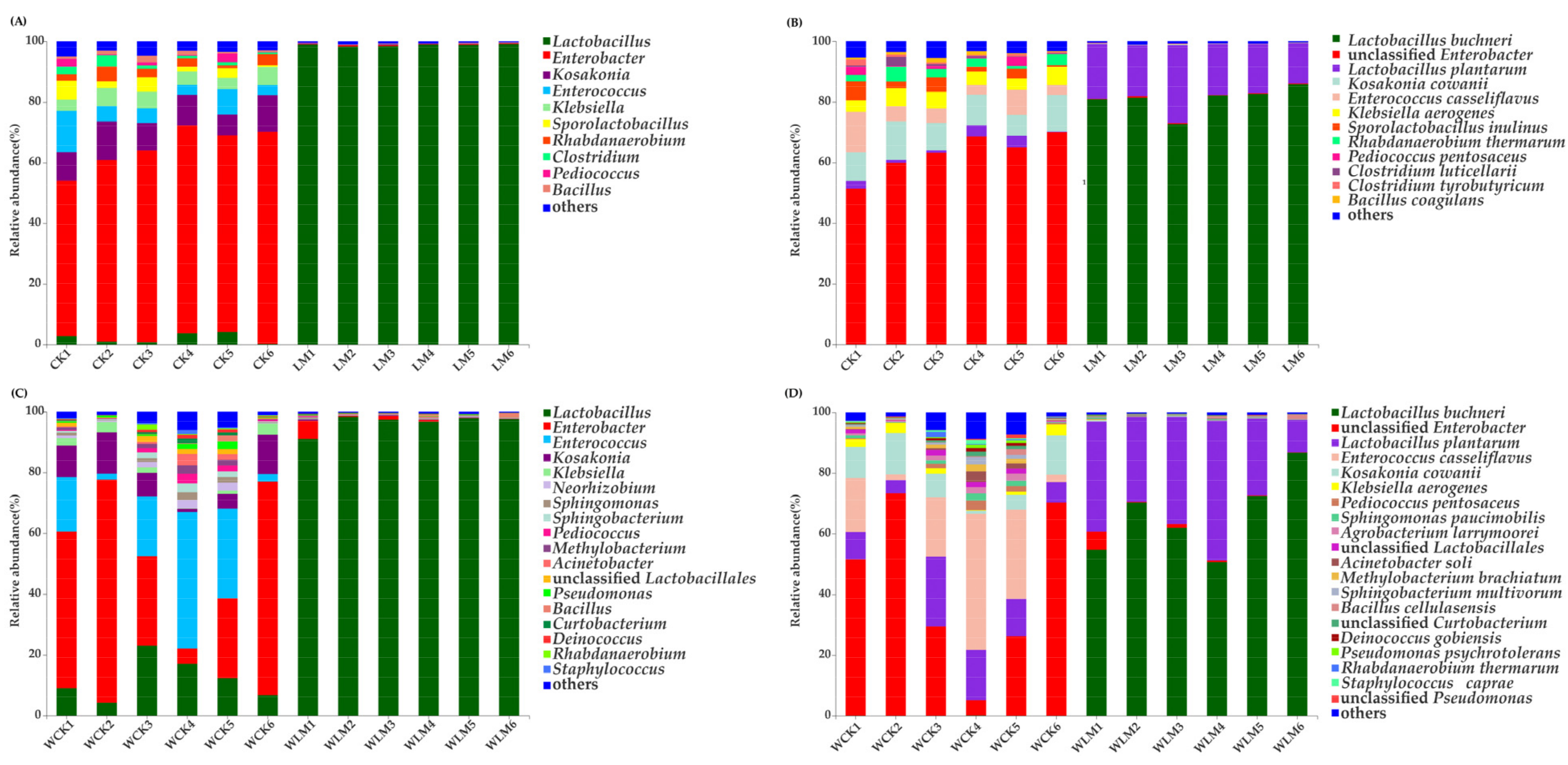
3.3. Effects of the Additives on the Microbial Communities of CLL Silages
3.4. Metabolomics Profiles of CLL Silages Ensiling for 60 Days
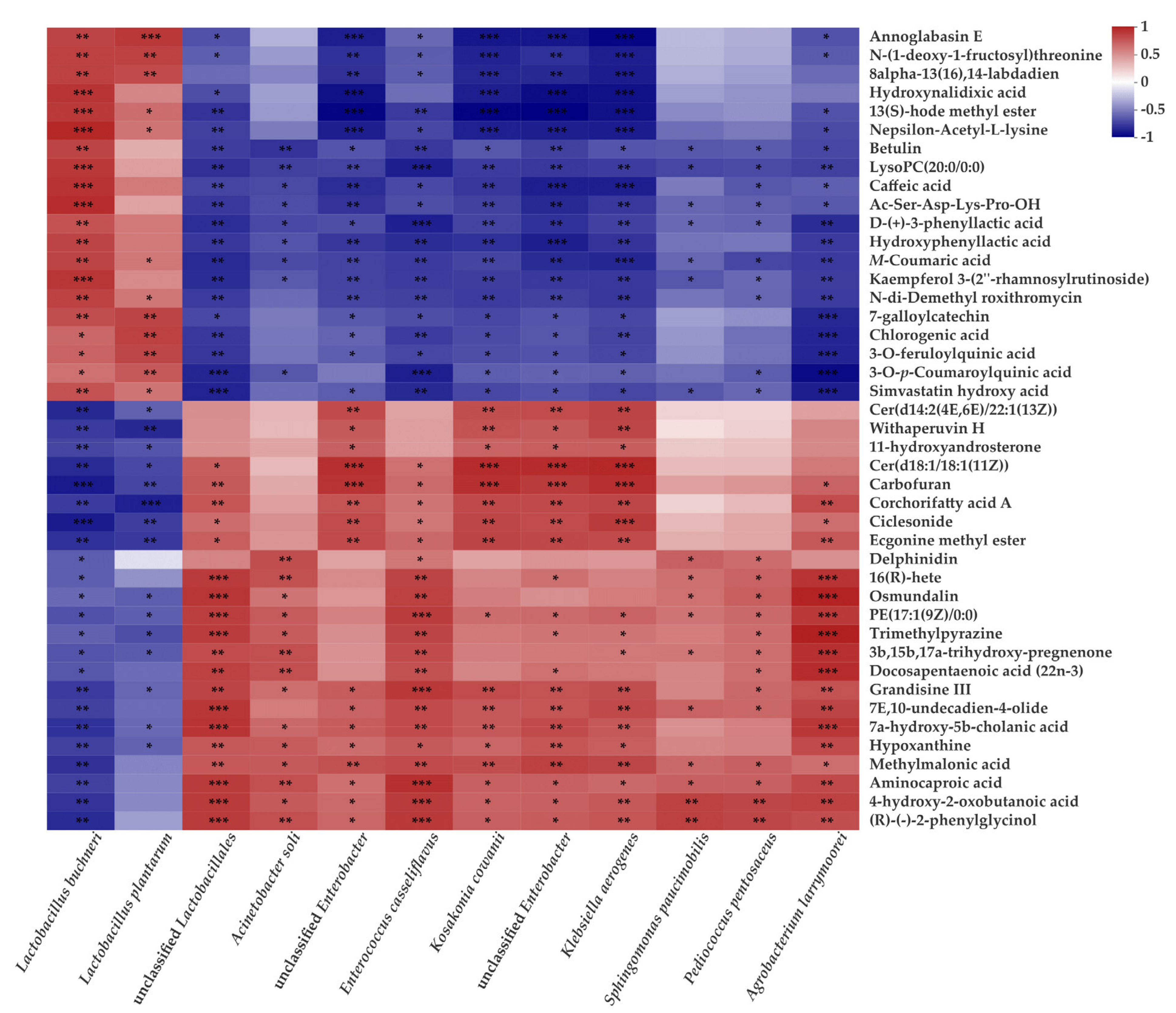
3.5. Correlations between Main Microorganisms and Metabolites in CLL Silage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Castro, O.; Gargiulo, R.; Del Guacchio, E.; Caputo, P.; De Luca, P. A molecular survey concerning the origin of Cyperus esculentus (Cyperaceae, Poales): Two sides of the same coin (weed vs. crop). Ann. Bot. 2015, 115, 733–745. [Google Scholar] [CrossRef] [Green Version]
- Ayeh-Kumi, P.F.; Tetteh-Quarcoo, P.B.; Duedu, K.O.; Obeng, A.S.; Addo-Osafo, K.; Mortu, S.; Asmah, R.H. A survey of pathogens associated with Cyperus esculentus L. (tiger nuts) tubers sold in a Ghanaian city. BMC Res. Notes 2014, 7, 343. [Google Scholar] [CrossRef] [Green Version]
- Cortes, C.E.M.; Frigola, A. Quality characteristics of horchata (a Spanish vegetable beverage) treated with pulsed electric field during shelf life. Food Chem. 2005, 91, 319–325. [Google Scholar] [CrossRef]
- Qu, P.; Cheng, Z.; Long, C.; Su, M.; Yang, D. Comprehensive development of chufa (Cyperuseslent L. varsatius) s. China Oils Fats 2007, 61–63. (In Chinese) [Google Scholar]
- Jing, S.; Wang, S.; Zhong, R.; Zhang, J.; Wu, J.; Tu, Y.; Pu, Y.; Yan, L. Neuroprotection of Cyperus esculentus L. orientin against cerebral ischemia/reperfusion induced brain injury. Neural Regen. Res. 2020, 15, 548–556. [Google Scholar] [CrossRef]
- Coskuner, Y.; Ercan, R.; Karababa, E.; Nazlican, A.N. Physical and chemical properties of chufa (Cyperus esculentus L.) tubers grown in the climate region of Turkey. J. Sci. Food Agric. 2002, 82, 625–631. [Google Scholar] [CrossRef]
- Roselló-Soto, E.; Martí-Quijal, F.J.; Cilla, A.; Munekata, P.E.S.; Lorenzo, J.M.; Remize, F.; Barba, F.J. Influence of temperature, solvent and pH on the selective extraction of phenolic compounds from tiger nuts by-products: Triple-TOF-LC-MS-MS characterization. Molecules 2019, 24, 797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, G.; Ju, Z.; Chai, J.; Jiao, T.; Jia, Z.; Casper, D.; Zeng, L.; Wu, J. Effects of silage additives and varieties on fermentation quality, aerobic stability, and nutritive value of oat silage. J. Anim. Sci. 2018, 96, 3151–3160. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, X.; Dai, J.; Meng, X.; Zhao, Y.; Li, Z.; Hu, Y.; Cui, Z. Construction and effect of lactic acid bacteria in oat silage and mechanism at low temperature. Trans. CSAE 2019, 35, 308–314. (In Chinese) [Google Scholar]
- Wang, Y.; McAllister, T.A.; Acharya, S. Condensed tannins in sainfoin: Composition, concentration, and effects on nutritive and feeding value of sainfoin forage. Crop Sci. 2015, 55, 13–22. [Google Scholar] [CrossRef]
- Tao, Y.; Sun, Q.; Li, F.; Xu, C.; Cai, Y. Comparative analysis of ensiling characteristics and protein degradation of alfalfa silage prepared with corn or sweet sorghum in semiarid region of Inner Mongolia. Anim. Sci. J. 2020, 91, 13321. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, C.; Zhou, W.; Yang, F.; Chen, X.; Zhang, Q. Effects of wilting and Lactobacillus plantarum addition on the fermentation quality and microbial community of Moringa oleifera leaf silage. Front. Microbiol. 2018, 9, 1817. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ni, K.; Wang, F.; Zhu, B.; Yang, J.; Zhou, G.; Pan, Y.; Tao, Y.; Zhong, J. Effects of lactic acid bacteria and molasses additives on the microbial community and fermentation quality of soybean silage. Bioresour. Technol. 2017, 238, 706–715. [Google Scholar] [CrossRef]
- Su, R.; Ni, K.; Wang, T.; Yang, X.; Zhang, J.; Liu, Y.; Shi, W.; Yan, L.; Jie, C.; Zhong, J. Effects of ferulic acid esterase-producing Lactobacillus fermentum and cellulase additives on the fermentation quality and microbial community of alfalfa silage. PeerJ 2019, 7, 7712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, H.; Feng, Y.; Pei, J.; Li, J.; Wang, Z.; Fu, S.; Zheng, Y.; Li, Z.; Peng, Z. Effects of Lactobacillus plantarum additive and temperature on the ensiling quality and microbial community dynamics of cauliflower leaf silages. Bioresour. Technol. 2020, 307, 123238. [Google Scholar] [CrossRef] [PubMed]
- Broderick, G.A.; Kang, J.H. Automated simultaneous determination of ammonia and total amino acids in ruminal fluid and In vitro media. J. Dairy Sci. 1980, 63, 64–75. [Google Scholar] [CrossRef]
- Playne, M.; McDonald, P. The buffering constituents of herbage and of silage. J. Sci. Food Agric. 1966, 17, 264–268. [Google Scholar] [CrossRef]
- Wang, T.; Teng, K.; Cao, Y.; Shi, W.; Xuan, Z.; Zhou, J.; Zhang, J.; Zhong, J. Effects of Lactobacillus hilgardii 60TS-2, with or without homofermentative Lactobacillus plantarum B90, on the aerobic stability, fermentation quality and microbial community dynamics in sugarcane top silage. Bioresour. Technol. 2020, 312, 123600. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.P. A method for the extraction of plant samples and the determination of total soluble carbohydrates. J. Sci. Food Agric. 1958, 9, 714–717. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists. Official Methods of Analysis; Association of Official Analytical Chemists: Arlington, VA, USA, 1990. [Google Scholar]
- VanSoest, P.H.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Ni, K.; Zhao, J.; Zhu, B.; Su, R.; Pan, Y.; Ma, J.; Zhou, G.; Tao, Y.; Liu, X.; Zhong, J. Assessing the fermentation quality and microbial community of the mixed silage of forage soybean with crop corn or sorghum. Bioresour. Technol. 2018, 265, 563–567. [Google Scholar] [CrossRef]
- Wang, X.; Sun, G.; Feng, T.; Zhang, J.; Huang, X.; Wang, T.; Xie, Z.; Chu, X.; Yang, J.; Wang, H. Sodium oligomannate therapeutically remodels gut microbiota and suppresses gut bacterial amino acids-shaped neuroinflammation to inhibit Alzheimer’s disease progression. Cell Res. 2019, 29, 787–803. [Google Scholar] [CrossRef]
- Xie, Z.; Zhang, T.; Chen, X.; Li, G.; Zhang, J. Effects of maturity stages on the nutritive composition and silage quality of whole crop wheat. Asian-Australas. J. Anim. Sci. 2012, 25, 1374–1380. [Google Scholar] [CrossRef] [PubMed]
- Weissbach, F.; Honig, H. On the anticipation and control of the run of fermentation silage making from extensively grown forages. Landbauforchung Völkenrode 1996, 46, 10–17, (In German, English Summary). [Google Scholar]
- Guo, X.; Ke, W.; Ding, W.; Ding, L.; Xu, D.; Wang, W.; Zhang, P.; Yang, F. Profiling of metabolome and bacterial community dynamics in ensiled Medicago sativa inoculated without or with Lactobacillus plantarum or Lactobacillus buchneri. Sci. Rep. 2018, 8, 357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Broderick, G.A.; Walgenbach, R.P.; Sterrenburg, E. Performance of lactating dairy cows fed alfalfa or red clover silage as the sole forage. J. Dairy Sci. 2000, 83, 1543–1551. [Google Scholar] [CrossRef]
- Cai, Y.; Benno, Y.; Ogawa, M.; Ohmomo, S.; Kumai, S.; Nakase, T. Influence of Lactobacillus spp. from an inoculant and of Weissella and Leuconostoc spp. from forage crops on silage fermentation. Appl. Environ. Microbiol. 1998, 64, 2982–2987. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, A.S.; Weinberg, Z.G.; Ogunade, I.M.; Cervantes, A.A.P.; Arriola, K.G.; Jiang, Y.; Kim, D.; Li, X.; Gonçalves, M.C.M.; Vyas, D.; et al. Meta-analysis of effects of inoculation with homofermentative and facultative heterofermentative lactic acid bacteria on silage fermentation, aerobic stability, and the performance of dairy cows. J. Dairy Sci. 2017, 100, 4587–4603. [Google Scholar] [CrossRef] [Green Version]
- Ávila, C.L.S.; Carvalho, B.F. Silage fermentation-updates focusing on the performance of microorganisms. J. Appl. Microbiol. 2020, 128, 966–984. [Google Scholar] [CrossRef] [Green Version]
- Kung, L., Jr.; Ranjit, N.K. The effect of Lactobacillus buchneri and other additives on the fermentation and aerobic stability of barley silage. J. Dairy Sci. 2001, 84, 1149–1155. [Google Scholar] [CrossRef]
- Zhang, Q.; Wu, B.; Nishino, N.; Wang, X.; Yu, Z. Fermentation and microbial population dynamics during the ensiling of native grass and subsequent exposure to air. Anim. Sci. J. 2016, 87, 389–397. [Google Scholar] [CrossRef]
- Li, X.; Chen, F.; Wang, X.; Sun, L.; Guo, L.; Xiong, Y.; Wang, Y.; Zhou, H.; Jia, S.; Yang, F. Impacts of low temperature and ensiling period on the bacterial bommunity of oat silage by SMRT. Microorganisms 2021, 9, 274. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Li, X.; Guan, H.; Huang, L.; Ma, X.; Peng, Y.; Li, Z.; Nie, G.; Zhou, J.; Yang, W.; et al. Microbial community and fermentation characteristic of Italian ryegrass silage prepared with corn stover and lactic acid bacteria. Bioresour. Technol. 2019, 279, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, C.; FitzGerald, J.; O’Shea, R.; Xia, A.; O’Kiely, P.; Murphy, J.D. Ensiling of seaweed for a seaweed biofuel industry. Bioresour. Technol. 2015, 196, 301–313. [Google Scholar] [CrossRef]
- Borreani, G.; Tabacco, E.; Schmidt, R.J.; Holmes, B.J.; Muck, R.E. Silage review: Factors affecting dry matter and quality losses in silages. J. Dairy Sci. 2018, 101, 3952–3979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ertekin, İ.; Kızılşimşek, M. Effects of lactic acid bacteria inoculation in pre-harvesting period on fermentation and feed quality properties of alfalfa silage. Asian-Australas. J. Anim. Sci. 2019, 33, 245–253. [Google Scholar] [CrossRef] [Green Version]
- Hackmann, T.J.; Sampson, J.D.; Spain, J.N. Comparing relative feed value with degradation parameters of grass and legume forages. J. Anim. Sci. 2008, 86, 2344–2356. [Google Scholar] [CrossRef] [Green Version]
- Xu, D.; Ding, Z.; Wang, M.; Bai, J.; Ke, W.; Zhang, Y.; Guo, X. Characterization of the microbial community, metabolome and biotransformation of phenolic compounds of sainfoin (Onobrychis viciifolia) silage ensiled with or without inoculation of Lactobacillus plantarum. Bioresour. Technol. 2020, 316, 123910. [Google Scholar] [CrossRef]
- Xu, D.; Ding, W.; Ke, W.; Li, F.; Zhang, P.; Guo, X. Modulation of metabolome and bacterial community in whole crop Corn silage by inoculating homofermentative Lactobacillus plantarum and heterofermentative Lactobacillus buchneri. Front. Microbiol. 2018, 9, 3299. [Google Scholar] [CrossRef] [Green Version]
- Heinl, S.; Wibberg, D.; Eikmeyer, F.; Szczepanowski, R.; Blom, J.; Linke, B.; Goesmann, A.; Grabherr, R.; Schwab, H.; Pühler, A.; et al. Insights into the completely annotated genome of Lactobacillus buchneri CD034, a strain isolated from stable grass silage. J. Biotechnol. 2012, 161, 153–166. [Google Scholar] [CrossRef]
- Pei, K.; Ou, J.; Huang, J.; Ou, S. p-Coumaric acid and its conjugates: Dietary sources, pharmacokinetic properties and biological activities. J. Sci. Food Agric. 2016, 96, 2952–2962. [Google Scholar] [CrossRef]
- Li, W.; Yuan, S.; Sun, J.; Li, Q.; Jiang, W.; Cao, J. Ethyl p-Coumarate exerts antifungal activity in vitro and in vivo against fruit Alternaria alternata via membrane-targeted mechanism. Int. J. Food Microbiol. 2018, 278, 26–35. [Google Scholar] [CrossRef]
- Brown, G.K.; Cromby, C.H.; Manning, N.J.; Pollitt, R.J. Urinary organic acids in succinic semialdehyde dehydrogenase deficiency: Evidence of alpha-oxidation of 4-hydroxybutyric acid, interaction of succinic semialdehyde with pyruvate dehydrogenase and possible secondary inhibition of mitochondrial beta-oxidation. J. Inherit. Metab. Dis. 1987, 10, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Jiang, H.; Yang, J.; Jin, M.; Du, Y.; Sun, Q.; Cao, L.; Xu, H. Safety assessment and antioxidant evaluation of betulin by LC-MS combined with free radical assays. Anal. Biochem. 2019, 587, 113460. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Y.Z.; Xu, W.C.; Chen, W.J.; Wu, S.; Huang, Y.Y. Metabolite and microbiome profilings of Pickled Tea elucidate the role of anaerobic fermentation in promoting high levels of gallic acid accumulation. J. Agric. Food Chem. 2020, 68, 13751–13759. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Zhang, W.; Lin, Z.; Pang, S.; Huang, Y.; Bhatt, P.; Chen, S. Carbofuran toxicity and its microbial degradation in contaminated environments. Chemosphere 2020, 259, 127419. [Google Scholar] [CrossRef] [PubMed]

| Item | CLL | Canola Straw | S1 | S2 |
|---|---|---|---|---|
| Chemical composition | ||||
| DM% | 19.90 | 90.78 | 37.20 | 36.40 |
| CP (%DM) | 11.56 | 5.21 | 5.17 | 11.96 |
| NDF (%DM) | 56.08 | 62.40 | 61.40 | 55.10 |
| ADF (%DM) | 28.92 | 50.51 | 40.55 | 29.40 |
| ADL (%DM) | 9.54 | 3.53 | 4.54 | 8.40 |
| WSC (%DM) | 2.52 | 8.45 | 6.46 | 3.20 |
| Cultivable microbial population (log10 CFU/g FM) | ||||
| Yeast | 6.71 | 5.16 | / | / |
| Aerobes | 6.92 | 6.14 | / | / |
| LAB | 4.00 | 4.70 | / | / |
| Item | S1 Group | S2 Group | ||||
|---|---|---|---|---|---|---|
| CK | LM | p Value | WCK | WLM | p Value | |
| The fermentation quality | ||||||
| pH value | 5.66 ± 0.18 a | 4.09 ± 0.06 b | <0.01 | 5.84 ± 0.02 a | 3.91 ± 0.01 b | <0.01 |
| Lactic acid (g/kg DM) | ND | 97.78 ± 24.05 | <0.01 | ND | 146.33 ± 8.98 | <0.01 |
| Acetic acid (g/kg DM) | 14.00 ± 2.10 b | 146.34 ± 13.74 a | <0.01 | 18.67 ± 1.97 b | 215.00 ± 2.83 a | <0.01 |
| Ethanol (g/kg DM) | 17.00 ± 1.63 a | 10.17 ± 1.94 b | <0.01 | 19.67 ± 5.35 | ND | <0.01 |
| Ammonia-N (g/kg DM) | 2.26 ± 0.12 a | 1.30 ± 0.20 b | <0.01 | 5.66 ± 0.49 a | 1.63 ± 0.32 b | <0.01 |
| BC (meq/100 g DM) | 19.79 ± 1.29 | 22.83 ± 1.84 | 0.19 | 21.55 ± 0.06 | 18.45 ± 0.56 | 0.02 |
| FC | 36.01 ± 1.30 | 36.83 ± 0.27 | 0.48 | 31.94 ± 2.74 | 37.61 ± 2.61 | 0.17 |
| Chemical composition | ||||||
| DM% | 36.88 ± 0.02 | 37.89 ± 0.02 | 0.31 | 32.74 ± 0.01 b | 37.20 ± 0.03 a | <0.01 |
| CP (%DM) | 8.21 ± 0.38 b | 9.60 ± 0.10 a | <0.01 | 12.17 ± 0.57 b | 13.35 ± 0.17 a | <0.01 |
| NDF (%DM) | 63.68 ± 1.89 | 61.78 ± 1.93 | 0.21 | 55.75 ± 1.06 a | 51.70 ± 1.88 b | 0.01 |
| ADF (%DM) | 43.95 ± 1.06 | 41.82 ± 2.51 | 0.17 | 30.68 ± 0.48 a | 28.69 ± 0.32 b | <0.01 |
| ADL (%DM) | 8.95 ± 0.75 | 8.62 ± 0.84 | 0.57 | 3.89 ± 0.37 | 3.63 ± 0.28 | 0.30 |
| WSC (%DM) | 0.39 ± 0.05 b | 0.71 ± 0.04 a | <0.01 | 0.63 ± 0.20 b | 1.01 ± 0.08 a | 0.01 |
| RFV | 79.93 ± 3.61 | 84.92 ± 5.39 | 0.17 | 108.5 ± 2.51 b | 119.87 ± 4.94 a | <0.01 |
| Cultivable microbial population (log10 CFU/g FM) | ||||||
| LAB | 3.95 ± 2.21 b | 5.54 ± 0.15 a | <0.01 | 2.04 ± 2.36 b | 5.38 ± 0.14 a | <0.01 |
| Aerobes | 4.39 ± 0.12 a | 2.37 ± 1.84 b | <0.01 | 4.22 ± 0.03 a | 1.17 ± 1.61 b | <0.01 |
| Item | S1 Group | S2 Group | ||||
|---|---|---|---|---|---|---|
| CK | LM | p Value | WCK | WLM | p Value | |
| Shannon | 1.99 ± 0.08 a | 0.58 ± 0.03 b | <0.01 | 2.00 ± 0.24 a | 0.83 ± 0.08 b | <0.01 |
| Simpson | 0.25 ± 0.02 b | 0.69 ± 0.02 a | <0.01 | 0.29 ± 0.06 b | 0.55 ± 0.05 a | 0.02 |
| ACE | 240.46 ± 11.46 | 209.68 ± 16.93 | 0.13 | 207.95 ± 21.69 | 159.66 ± 10.51 | 0.13 |
| Chao 1 | 234.94 ± 11.42 | 177.19 ± 18.94 | 0.03 | 190.38 ± 17.50 | 136.69 ± 10.24 | 0.07 |
| Coverage | 0.99 | 0.99 | 0.18 | 0.99 | 0.99 | 0.24 |
| Metabolite Name | S1 Group | S2 Group | |||||
|---|---|---|---|---|---|---|---|
| CK | LM | Fold Change | WCK | WLM | Fold Change | ||
| Benzene and substituted derivatives | Syringic acid | 12.42 ± 0.18 b | 14.29 ± 0.55 a | 1.87 ± 0.53 | 13.28 ± 0.30 b | 15.3 ± 0.13 a | 2.02 ± 0.31 |
| Carboxylic acids and derivatives | L-Tyrosine | 6.52 ± 0.83 b | 9.89 ± 0.14 a | 3.37 ± 0.88 | ND | ND | ND |
| Valyl-Isoleucine | ND | ND | ND | 2.92 ± 0.65 b | 9.87 ± 0.15 a | 3.50 ± 0.61 | |
| Threoninyl-Isoleucine | ND | ND | ND | 1.55 ± 2.33 b | 9.63 ± 0.15 a | 8.08 ± 2.34 | |
| Glycyl-Isoleucine | ND | ND | ND | 5.18 ± 2.18 b | 10.85 ± 0.11 a | 5.66 ± 2.14 | |
| Asparaginyl-Isoleucine | ND | ND | ND | 6.29 ± 0.65 b | 10.50 ± 0.12 a | 4.20 ± 0.74 | |
| Cinnamic acids and derivatives | Sinapic acid | 9.53 ± 1.29 | 11.09 ± 0.70 | 1.56 ± 1.86 | 2.92 ± 0.65 b | 9.87 ± 0.15 a | 6.96 ± 0.77 |
| Caffeic acid | 10.04 ± 0.11 | 11.81 ± 0.79 | 1.78 ± 0.82 | 10.60 ± 0.49 b | 13.53 ± 0.12 a | 2.93 ± 0.48 | |
| 2-hydroxycinnamic acid | 12.25 ± 0.90 b | 14.76 ± 0.12 a | 2.51 ± 0.90 | ND | ND | ND | |
| M-Coumaric acid | 8.18 ± 0.80 b | 11.61 ± 0.29 a | 3.42 ± 0.94 | 9.27 ± 0.76 b | 12.64 ± 0.08 a | 3.38 ± 0.73 | |
| Punicic acid | Punicic acid | 14.93 ± 0.28 | 15.25 ± 0.11 | 0.32 ± 0.27 | ND | ND | ND |
| Flavonoids | Quercetin | 12.21 ± 1.40 | 14.14 ± 0.63 | 1.94 ± 1.68 | ND | ND | ND |
| Theaflavic acid | 14.73 ± 0.29 | 15.27 ± 0.20 | 0.54 ± 0.38 | ND | ND | ND | |
| Isorhamnetin | 14.21 ± 1.13 | 15.52 ± 0.31 | 1.31 ± 1.21 | ND | ND | ND | |
| Isovitexin | 14.27 ± 0.33 | 14.76 ± 0.13 | 0.49 ± 0.40 | ND | ND | ND | |
| 7-galloylcatechin | ND | ND | ND | 2.96 ± 1.54 b | 10.24 ± 0.41 a | 7.27 ± 1.76 | |
| Kaempferol 3-(2″-rhamnosylrutinoside) | ND | ND | ND | 9.31 ± 0.79 b | 13.64 ± 0.29 a | 4.32 ± 0.67 | |
| Imidazopyrimidines | Hypoxanthine | 12.34 ± 0.28 | 10.95 ± 0.15 | −1.39 ± 0.37 | 12.50 ± 0.39 a | 10.09 ± 0.20 b | −2.40 ± 0.49 |
| Macrolides and analogues | Zeranol | 8.84 ± 0.53 b | 10.48 ± 0.78 a | 1.64 ± 0.84 | ND | ND | ND |
| Organooxygen compounds | 1,3-dicaffeoylquinic acid | ND | ND | ND | 11.39 ± 0.94 | 12.35 ± 0.36 | 0.96 ± 0.92 |
| 3-O-p-Coumaroylquinic acid | ND | ND | ND | 10.07 ± 0.24 b | 12.43 ± 0.54 a | 2.36 ± 0.43 | |
| Chlorogenic acid | ND | ND | ND | 11.73 ± 0.54 b | 14.67 ± 0.57 a | 2.95 ± 0.72 | |
| N-di-Demethyl roxithromycin | ND | ND | ND | 9.52 ± 1.47 b | 11.59 ± 0.58 a | 2.07 ± 1.87 | |
| 3-O-feruloylquinic acid | ND | ND | ND | 7.17 ± 1.38 b | 10.71 ± 0.75 a | 3.54 ± 1.51 | |
| Phenylpropanoic acids | Hydroxyphenyllactic acid | ND | ND | ND | 10.34 ± 0.42 b | 12.18 ± 0.14 a | 1.84 ± 0.45 |
| Prenol lipids | Annoglabasin E | 8.02 ± 0.94 | 9.96 ± 0.33 | 1.94 ± 1.12 | 6.36 ± 0.38 b | 10.56 ± 0.55 a | 4.20 ± 0.40 |
| Betulin | ND | ND | ND | 7.08 ± 1.61 b | 11.02 ± 0.92 a | 4.02 ± 1.82 | |
| Piperidine | Piperidine | ND | ND | ND | 14.64 ± 0.21 | 14.39 ± 0.10 | −0.25 ± 0.18 |
| Steroids and steroid derivatives | 7a-hydroxy-5b-cholanic acid | 13.19 ± 0.66 | 11.87 ± 0.97 | −1.32 ± 1.41 | 3.97 ± 0.20 | 3.57 ± 0.30 | −0.40 ± 0.10 |
| Quinolines and derivatives | Grandisine III | ND | ND | ND | 11.94 ± 0.74 | 10.05 ± 0.54 | −1.88 ± 0.91 |
| Others | Ferulic acid | 11.47 ± 0.69 | 13.31 ± 0.20 | 1.84 ± 0.63 | 12.26 ± 0.63 | 13.09 ± 0.14 | 0.83 ± 0.71 |
| D-(+)-3-phenyllactic acid | 13.66 ± 0.21 | 14.71 ± 0.06 | 1.05 ± 0.26 | 12.67 ± 0.52 b | 14.86 ± 0.06 a | 2.19 ± 0.53 | |
| 7-epi-Jasmonic acid | 12.15 ± 1.06 a | 8.46 ± 0.48 b | −3.69 ± 1.42 | 12.77 ± 3.24 a | 2.42 ± 1.23 b | −10.36 ± 3.24 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Wang, T.; Huang, F.; Liu, Y.; Shi, W.; Ma, C.; Zhong, J. Silage Fermentation: A Potential Microbial Approach for the Forage Utilization of Cyperus esculentus L. By-Product. Fermentation 2021, 7, 273. https://doi.org/10.3390/fermentation7040273
Sun J, Wang T, Huang F, Liu Y, Shi W, Ma C, Zhong J. Silage Fermentation: A Potential Microbial Approach for the Forage Utilization of Cyperus esculentus L. By-Product. Fermentation. 2021; 7(4):273. https://doi.org/10.3390/fermentation7040273
Chicago/Turabian StyleSun, Jiahao, Tianwei Wang, Fuqing Huang, Yayong Liu, Weixiong Shi, Cui Ma, and Jin Zhong. 2021. "Silage Fermentation: A Potential Microbial Approach for the Forage Utilization of Cyperus esculentus L. By-Product" Fermentation 7, no. 4: 273. https://doi.org/10.3390/fermentation7040273
APA StyleSun, J., Wang, T., Huang, F., Liu, Y., Shi, W., Ma, C., & Zhong, J. (2021). Silage Fermentation: A Potential Microbial Approach for the Forage Utilization of Cyperus esculentus L. By-Product. Fermentation, 7(4), 273. https://doi.org/10.3390/fermentation7040273





