Coffee and Yeasts: From Flavor to Biotechnology
Abstract
1. Introduction
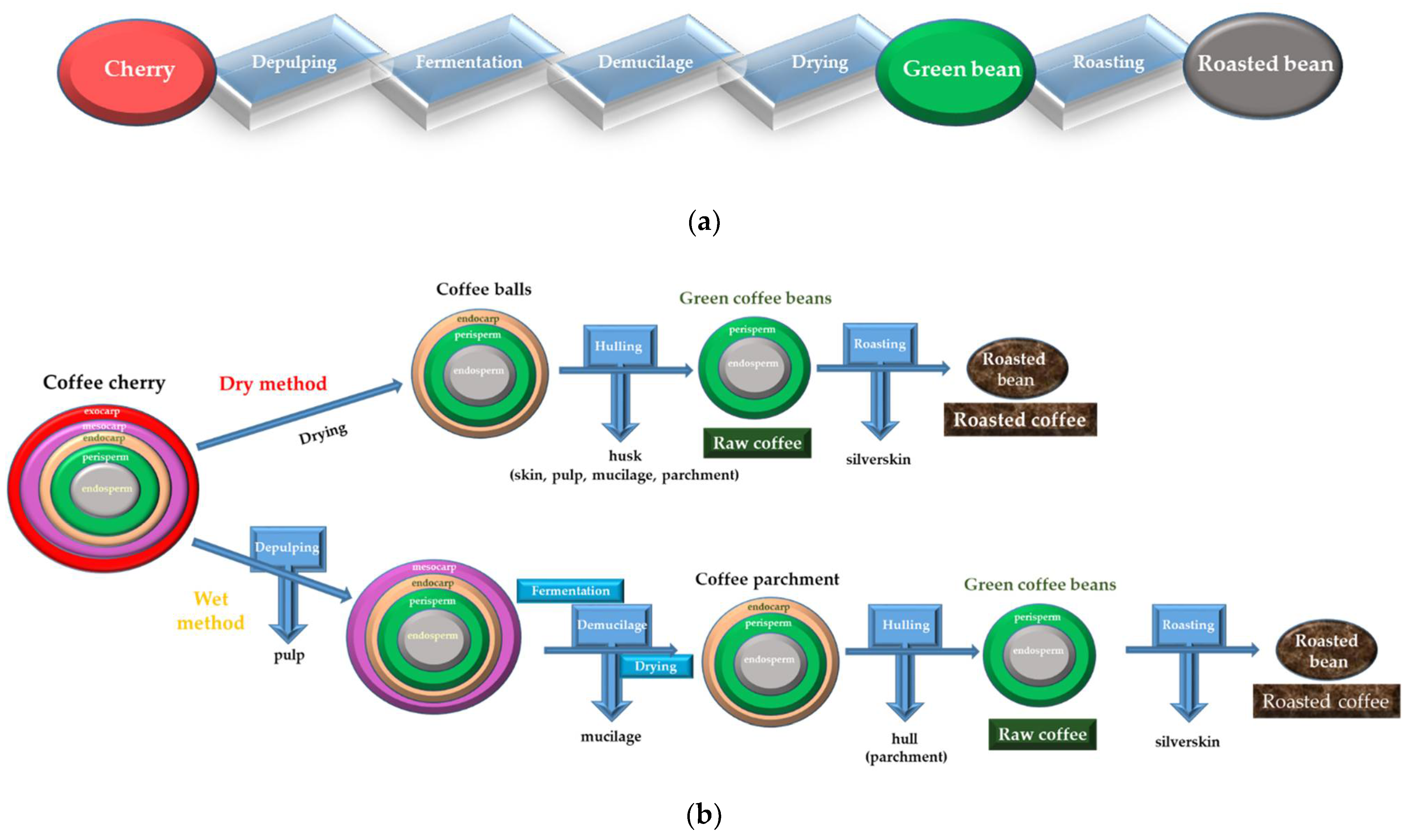
2. Yeasts and Coffee Processing
2.1. Drying Methods
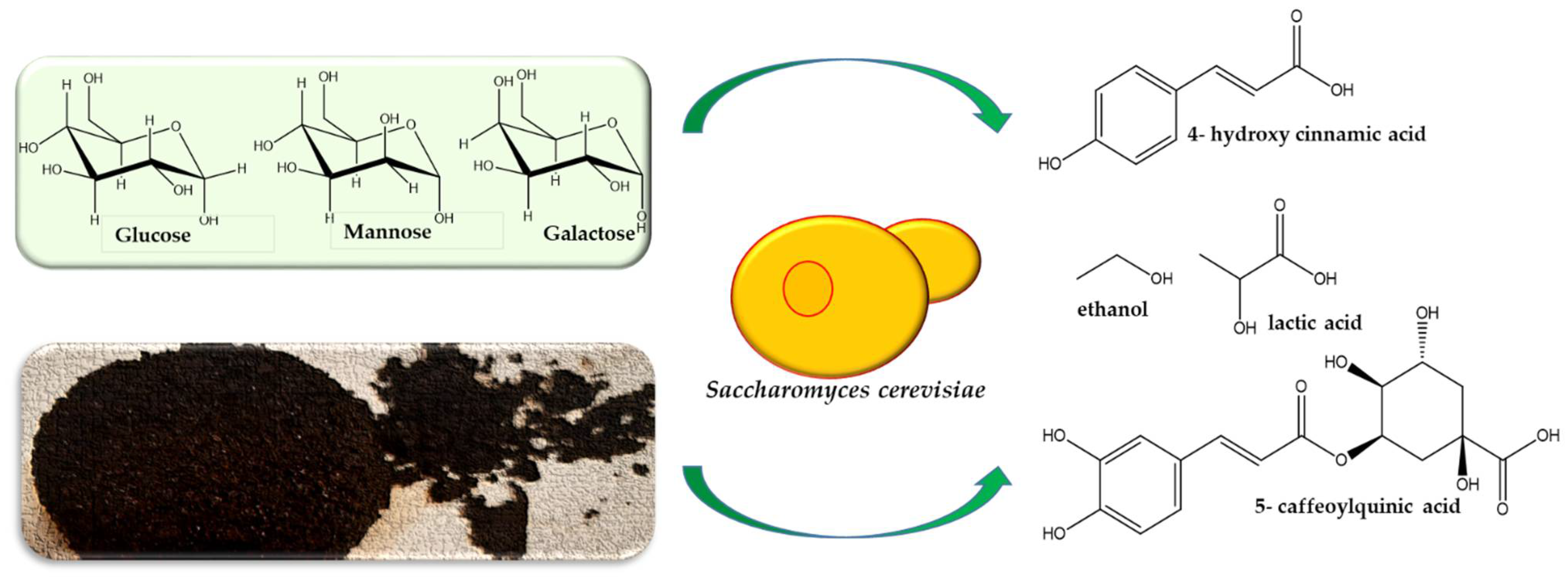
2.2. Yeast as Starter Cultures in Coffee Processing
2.3. Selection of Yeasts as Starter Cultures in Wet, Dry and Semi-Dry Processing
2.4. Role of Yeasts in Controlling the Production of Non-Desired Compounds
2.5. Role of Yeasts in Obtaining Specialty Coffees
2.6. Influence of Yeast Inoculation Methods on Chemical Composition of Coffee
2.7. Yeast, Coffee and Climate
2.8. Special Compounds in Yeast-Fermented Coffee
3. Yeasts as Toolboxes for Capitalization of Coffee Wastes
3.1. Coffee Wastes and the Environment
3.2. Spent Coffee Grounds and Biofuels
3.3. Spent Coffee Grounds and Fine Chemicals
4. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- FAO. Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org/3/a-i4985e.pdf (accessed on 17 December 2020).
- USDA. United States Department for Agriculture. Available online: https://apps.fas.usda.gov/psdonline/circulars/coffee.pdf (accessed on 17 December 2020).
- Martín, M.L.; Pablos, F.; González, A.G. Discrimination between arabica and robusta green coffee varieties according to their chemical composition. Talanta 1998, 46, 1259–1264. [Google Scholar] [CrossRef]
- Pérez, S.B.Y.; Saldaña-Trinidad, S. Chemistry and biotransformation of coffee by products to biofuels. In The Question of Caffeine; Latosinska, J.N., Latosinska, M., Eds.; InTech: London, UK, 2017; pp. 143–161. [Google Scholar] [CrossRef]
- Cheng, B.; Smyth, H.E.; Furtado, A.; Henry, R.J. Slower development of lower canopy beans produces better coffee. J. Exp. Bot. 2020, 71, 4201–4214. [Google Scholar] [CrossRef] [PubMed]
- Bressani, A.P.P.; Martinez, S.J.; Sarmento, A.B.I.; Borém, F.M.; Schwan, R.F. Organic acids produced during fermentation and sensory perception in specialty coffee using yeast starter culture. Food Res. Int. 2020, 128, 108773. [Google Scholar] [CrossRef] [PubMed]
- Sunarharum, W.B.; Williams, D.J.; Smyth, H.E. Complexity of coffee flavor: A compositional and sensory perspective. Food Res. Int. 2014, 62, 315–325. [Google Scholar] [CrossRef]
- Camarasa, C.; Sanchez, I.; Brial, P.; Bigey, F.; Dequin, S. Phenotypic landscape of Saccharomyces cerevisiae during wine fermentation: Evidence for origin-dependent metabolic traits. PLoS ONE 2011, 6, e25147. [Google Scholar] [CrossRef]
- Ruta, L.L.; Farcasanu, I.C. Anthocyanins and anthocyanin-derived products in yeast-fermented beverages. Antioxidants 2019, 8, 182. [Google Scholar] [CrossRef]
- Ludlow, C.L.; Cromie, G.A.; Garmendia-Torres, C.; Sirr, A.; Hays, A.; Field, C.; Jeffery, E.W.; Fay, J.C.; Dudle, A.D. Independent origins of yeast associated with coffee and cacao fermentation. Curr. Biol. 2016, 26, 965–971. [Google Scholar] [CrossRef]
- De Bruyn, F.; Zhang, S.J.; Pothakos, V.; Torres, J.; Lambot, C.; Moroni, A.V.; Callanan, M.; Sybesma, W.; Weckx, S.; De Vuyst, L. Exploring the impacts of postharvest processing on the microbiota and metabolite profiles during green coffee bean production. Appl. Environ. Microbiol. 2016, 83, e02398-16. [Google Scholar] [CrossRef]
- Evangelista, S.R.; Miguel, M.G.; de Souza Cordeiro, C.; Silva, C.F.; Pinheiro, A.C.; Schwan, R.F. Inoculation of starter cultures in a semi-dry coffee (Coffea arabica) fermentation process. Food Microbiol. 2014, 44, 87–95. [Google Scholar] [CrossRef]
- Vilela, D.M.; Pereira, G.V.; Silva, C.F.; Batista, L.R.; Schwan, R.F. Molecular ecology and polyphasic characterization of the microbiota associated with semi-dry processed coffee (Coffea arabica L.). Food Microbiol. 2010, 27, 1128–1135. [Google Scholar] [CrossRef]
- Silva, C.F.; Batista, L.R.; Abreu, L.M.; Dias, E.S.; Schwan, R.F. Succession of bacterial and fungal communities during natural coffee (Coffea arabica) fermentation. Food Microbiol. 2008, 25, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Vinícius de Melo Pereira, G.; Soccol, V.T.; Brar, S.K.; Neto, E.; Soccol, C.R. Microbial ecology and starter culture technology in coffee processing. Crit. Rev. Food Sci. Nutr. 2017, 57, 2775–2788. [Google Scholar] [CrossRef]
- Duarte, G.S.; Pereira, A.A.; Farah, A. Chlorogenic acids and other relevant compounds in Brazilian coffees processed by semi-dry and wet post-harvesting methods. Food Chem. 2010, 118, 851–855. [Google Scholar] [CrossRef]
- Batista, L.R.; Chalfoun, S.M.; Batista, C.F.S.; Schwan, R.F. Coffee: Types and production. In The Encyclopedia of Food and Health, 1st ed.; Caballero, B., Finglas, P., Toldrá, F., Eds.; Academic Press: Oxford, UK; Cambridge, MA, USA, 2016; p. 244. [Google Scholar]
- Silva, C.F. Microbial activity during coffee fermentation. In Cocoa and Coffee Fermentations; Schwan, R.F., Fleet, G.H., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 368–423. [Google Scholar]
- Murthy, P.S.; Naidu, M.M. Sustainable management of coffee industry by-products and value addition—A review. Resour. Conserv. Recy. 2012, 66, 45–58. [Google Scholar] [CrossRef]
- Silva, C.F.; Vilela, D.M.; de SouzaCordeiro, C.; Duarte, W.F.; Dias, D.R.; Schwan, R.F. Evaluation of a potential starter culture for enhances quality of coffee fermentation. World J. Microbiol. Biotechnol. 2013, 29, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Avallone, S.; Brillouet, J.M.; Guyot, B.; Olguin, E.; Guiraud, J.P. Involvement of pectinolytic microorganisms is coffee fermentation. Int. J. Food Sci. Technol. 2002, 37, 191–198. [Google Scholar] [CrossRef]
- Masoud, W.; Jespersen, L. Pectin degrading enzymes in yeasts involved in fermentation of Coffea arabica in East Africa. Int. J. Food Microbiol. 2006, 110, 291–296. [Google Scholar] [CrossRef]
- Ulloa Rojas, J.B.; Verreth, J.A.; Amato, S.; Huisman, E.A. Biological treatments affect the chemical composition of coffee pulp. Bioresour. Technol. 2003, 89, 267–274. [Google Scholar] [CrossRef]
- Bekalo, S.A.; Reinhardt, H. Fibers of coffee husk and hulls for the production of particleboard. Mater. Struct. 2010, 43, 1049–1060. [Google Scholar] [CrossRef]
- Aristizábal, V.M.; Chacón-Perez, Y.; Carlos, A.; Alzate, C. The biorefinery concept for the industrial valorization of coffee processing by-products. In Handbook of Coffee Processing by-Products; Galanakis, C.M., Ed.; Academic Press: Oxford, UK; Cambridge, MA, USA, 2017; pp. 63–92. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, M.K.; Lee, K.G. Effect of reversed coffee grinding and roasting process on physicochemical properties including volatile compound profiles. Innov. Food Sci. Emerg. Technol. 2017, 44, 97–102. [Google Scholar] [CrossRef]
- Martinez, S.J.; Bressani, A.P.P.; Miguel, M.G.D.C.P.; Dias, D.R.; Schwan, R.F. Different inoculation methods for semi-dry processed coffee using yeasts as starter cultures. Food Res. Int. 2017, 102, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Avallone, S.; Guyot, B.; Brillouet, J.M.; Olguin, E.; Guiraud, J.P. Microbiological and biochemical study of coffee fermentation. Curr. Microbiol. 2001, 42, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Haile, M.; Kang, W.H. The role of microbes in coffee fermentation and their impact on coffee quality. J. Food Qual. 2019, 2019, 4836709. [Google Scholar] [CrossRef]
- Haile, M.; Kang, W.H. Isolation, identification, and characterization of pectinolytic yeasts for starter culture in coffee fermentation. Microorganisms 2019, 7, 401. [Google Scholar] [CrossRef] [PubMed]
- Massawe, G.A.; Lifa, S.J. Yeasts and lactic acid bacteria coffee fermentation starter cultures. Int. J. Postharvest Technol. Innov. 2010, 2, 41–80. [Google Scholar] [CrossRef]
- Lee, L.W.; Cheong, M.W.; Curran, P.; Yu, B.; Liu, S.Q. Modulation of coffee aroma via the fermentation of green coffee beans with Rhizopusoligosporus: I. Green coffee. Food Chem. 2016, 211, 916–924. [Google Scholar] [CrossRef]
- Lee, L.W.; Cheong, M.W.; Curran, P.; Yu, B.; Liu, S.Q. Modulation of coffee aroma via the fermentation of green coffee beans with Rhizopusoligosporus: II. Effects of different roast levels. Food Chem. 2016, 211, 925–936. [Google Scholar] [CrossRef]
- Agate, A.D.; Bhat, J.V. Role of pectinolytic yeasts in the degradation of mucilage layer of Coffea robusta cherries. Appl. Microbiol. 1966, 14, 256–260. [Google Scholar] [CrossRef]
- Martins, P.M.M.; Ribeiro, L.S.; Miguel, M.G.D.C.P.; Evangelista, S.R.; Schwan, R.F. Production of coffee (Coffea arabica) inoculated with yeasts: Impact on quality. J. Sci. Food Agric. 2019, 99, 5638–5645. [Google Scholar] [CrossRef]
- Dandekar, R.; Fegade, B.; Bhaskar, V.H. GC-MS analysis of phytoconstituents in alcohol extract of Epiphyllumoxy petalum leaves. J. Pharmacogn. Phytochem. 2015, 4, 149–154. [Google Scholar]
- Martinez, S.J.; Bressani, A.P.P.; Dias, D.R.; Simão, J.B.P.; Schwan, R.F. Effect of bacterial and yeast starters on the formation of volatile and organic acid compounds in coffee beans and selection of flavors markers precursors during wet fermentation. Front. Microbiol. 2019, 10, 1287. [Google Scholar] [CrossRef] [PubMed]
- De Melo Pereira, G.V.; Soccol, V.T.; Pandey, A.; Medeiros, A.B.; Andrade Lara, J.M.; Gollo, A.L.; Soccol, C.R. Isolation, selection and evaluation of yeasts for use in fermentation of coffee beans by the wet process. Int. J. Food Microbiol. 2014, 188, 60–66. [Google Scholar] [CrossRef] [PubMed]
- De Melo Pereira, G.V.; Neto, E.; Soccol, V.T.; Medeiros, A.B.P.; Woiciechowski, A.L.; Soccol, C.R. Conducting starter culture-controlled fermentations of coffee beans during on-farm wet processing: Growth, metabolic analyses and sensorial effects. Food Res. Int. 2015, 75, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Elhalis, H.; Cox, J.; Zhao, J. Ecological diversity, evolution and metabolism of microbial communities in the wet fermentation of Australian coffee beans. Int. J. Food Microbiol. 2020, 321, 108544. [Google Scholar] [CrossRef]
- Pehl, C.; Pfeiffer, A.; Wendl, B.; Kaess, H. The effect of decaffeination of coffee on gastro-oesophageal reflux in patients with reflux disease. Aliment. Pharmacol. Ther. 1997, 11, 483–486. [Google Scholar] [CrossRef]
- Rubach, M.; Lang, R.; Skupin, C.; Hofmann, T.; Somoza, V. Activity-guided fractionation to characterize a coffee beverage that effectively down-regulates mechanisms of gastric acid secretion as compared to regular coffee. J. Agric. Food Chem. 2010, 58, 4153–4161. [Google Scholar] [CrossRef]
- Rubach, M.; Lang, R.; Seebach, E.; Somoza, M.M.; Hofmann, T.; Somoza, V. Multi-parametric approach to identify coffee components that regulate mechanisms of gastric acid secretion. Mol. Nutr. Food Res. 2012, 56, 325–335. [Google Scholar] [CrossRef]
- Tinoco, N.A.B.; Pacheco, S.; Godoy, R.L.O.; Bizzo, H.R.; de Aguiar, P.F.; Leite, S.G.F.; Rezende, C.M. Reduction of βN-alkanoyl-5-hydroxytryptamides and diterpenes by yeast supplementation to green coffee during wet processing. Food Res. Int. 2019, 115, 487–492. [Google Scholar] [CrossRef]
- Velmourougane, K.; Bhat, R.; Gopinandhan, T.N.; Panneerselvam, P. Impact of delay in processing on mold development, ochratoxin-A and cup quality in Arabica and Robusta coffee. World J. Microb. Biot. 2011, 27, 1809–1816. [Google Scholar] [CrossRef]
- Ribeiro, L.S.; Miguel, M.G.; Evangelista, S.R.; Machado Martins, P.M.; van Mullem, J.; Belizario, M.H.; Schwan, R.F. Behavior of yeast inoculated during semi-dry coffee fermentation and the effect on chemical and sensorial properties of the final beverage. Food Res. Int. 2017, 92, 26–32. [Google Scholar] [CrossRef]
- Wang, C.; Sun, J.; Lassabliere, B.; Yu, B.; Liu, S.Q. Coffee flavour modification through controlled fermentations of green coffee beans by Saccharomyces cerevisiae and Pichia kluyveri: Part I. Effects from individual yeasts. Food Res. Int. 2020, 136, 109588. [Google Scholar] [CrossRef]
- Bressani, A.P.P.; Martinez, S.J.; Evangelista, S.R.; Ribeiro, D.D.; Schwan, R.F. Characteristics of fermented coffee inoculated with yeast starter cultures using different inoculation methods. LWT Food Sci. Technol. 2018, 92, 212–219. [Google Scholar] [CrossRef]
- Pereira, L.L.; Guarçoni, R.C.; Pinheiro, P.F.; Osório, V.M.; Pinheiro, C.A.; Moreira, T.R.; Ten Caten, C.S. New propositions about coffee wet processing: Chemical and sensory perspectives. Food Chem. 2020, 310, 125943. [Google Scholar] [CrossRef] [PubMed]
- Brando, C.H.J.; Brando, M.F. Methods of coffee fermentation and drying. In Cocoa and Coffee Fermentations; Schwan, R.F., Fleet, G.H., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 367–396. [Google Scholar]
- Da Mota, M.C.B.; Batista, N.N.; Rabelo, M.H.S.; Ribeiro, D.E.; Borém, F.M.; Schwan, R.F. Influence of fermentation conditions on the sensorial quality of coffee inoculated with yeast. Food Res. Int. 2020, 136, 109482. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Zhao, M.; Liu, F.; Zeng, S.; Hu, J. Identification of 2,3-dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one as a strong antioxidant in glucose-histidine Maillard reaction products. Food Res. Int. 2013, 51, 397–403. [Google Scholar] [CrossRef]
- Cutzach, I.; Chatonnet, P.; Dubourdieu, D. Study of the formation mechanisms of some volatile compounds during the aging of sweet fortified wines. J. Agric. Food Chem. 1999, 47, 2837–2846. [Google Scholar] [CrossRef]
- Moreira, R.F.A.; Trugo, L.C.; Maria, C.A.B. Volatile components in roasted coffee. Part II. Aliphatic, alicyclic and aromatic compounds. Quím. Nova 2000, 23, 195–201. [Google Scholar] [CrossRef]
- Campos-Vega, R.; Loarca-Piña, G.; Vergara-Castañeda, H.A.; Oomah, B.D. Spent coffee grounds: A review on current research and future prospects. Trends Food Sci. Tech. 2015, 45, 24–36. [Google Scholar] [CrossRef]
- Mussatto, S.J.; Machado, E.M.S.; Martins, S.; Teixeira, J.A. Production, composition and application of coffee and its industrial residues. Food Bioprocess. Technol. 2011, 4, 661–672. [Google Scholar] [CrossRef]
- Hughes, S.R.; López-Núñez, J.C.; Jones, M.A.; Moser, B.R.; Cox, E.J.; Lindquist, M.; Galindo-Leva, L.A.; Riaño-Herrera, N.M.; Rodriguez-Valencia, N.; Gast, F.; et al. Sustainable conversion of coffee and other crop wastes to biofuels and bioproducts using coupled biochemical and thermochemical processes in a multi-stage biorefinery concept. Appl. Microbiol. Biotechnol. 2014, 98, 8413–8431. [Google Scholar] [CrossRef]
- Choi, I.S.; Wi, S.G.; Kim, S.B.; Bae, H.J. Conversion of coffee residue waste into bioethanol with using popping pretreatment. Bioresour. Technol. 2012, 125, 132–137. [Google Scholar] [CrossRef] [PubMed]
- da Silveira, J.S.; Durand, N.; Lacour, S.; Belleville, M.P.; Perez, A.; Loiseau, G.; Dornier, M. Solid-state fermentation as a sustainable method for coffee pulp treatment and production of an extract rich in chlorogenic acids. Food Bioprod. Process. 2019, 115, 175–184. [Google Scholar] [CrossRef]
- Pandey, A.; Soccol, C.R.; Nigam, P.; Brand, D.; Mohan, R.; Roussos, S. Biotechnological potential of coffee pulp and coffee husk for bioprocesses. Biochem. Eng. J. 2000, 6, 153–162. [Google Scholar] [CrossRef]
- Kim, K.; Tsao, R.; Yang, R.; Cui, S. Phenolic acid profiles and antioxidant activities of wheat bran extracts and the effect of hydrolysis conditions. Food Chem. 2006, 95, 466–473. [Google Scholar] [CrossRef]
- Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Serna-Saldívar, S.O. Bound phenolics in foods, a review. Food Chem. 2014, 152, 46–55. [Google Scholar] [CrossRef]
- Lin, C.S.K.; Pfaltzgraff, L.A.; Herrero-Davila, L.; Mubofu, E.B.; Solhy, A.; Clark, J.H.; Koutinas, A.; Kopsahelis, N.; Stamatelatou, K.; Dickson, F.; et al. Food waste as a valuable resource for the production of chemicals, materials and fuels. Current situation and global perspective. Energy Environ. Sci. 2013, 6, 426–464. [Google Scholar] [CrossRef]
- Nguyen, Q.A.; Cho, E.; Trinh, L.T.P.; Jeong, J.S.; Bae, H.J. Development of an integrated process to produce d-mannose and bioethanol from coffee residue waste. Bioresour. Technol. 2017, 244 Pt 1, 1039–1048. [Google Scholar] [CrossRef]
- Hughes, S.R.; Sterner, D.E.; Bischoff, K.M.; Hector, R.E.; Dowd, P.F.; Qureshi, N.; Bang, S.S.; Grynaviski, N.; Chakrabarty, T.; Johnson, E.T.; et al. Three-plasmid SUMO yeast vector system for automated high-level functional expression of value-added co-products in a Saccharomyces cerevisiae strain engineered for xylose utilization. Plasmid 2009, 61, 22–38. [Google Scholar] [CrossRef]
- Hughes, S.R.; Cox, E.J.; Bang, S.S.; López-Núñez, J.C.; Saha, B.C.; Qureshi, N.; Gibbons, W.R.; Fry, M.R.; Moser, B.R.; Bischoff, K.M.; et al. Process for assembly and transformation into saccharomyces cerevisiae of a synthetic yeast artificial chromosome containing a multigene cassette to express enzymes that enhance xylose utilization designed for an automated platform. J. Lab. Autom. 2015, 20, 621–635. [Google Scholar] [CrossRef][Green Version]
- Rocha, M.V.; de Matos, L.J.; Lima, L.P.; Figueiredo, P.M.; Lucena, I.L.; Fernandes, F.A.; Gonçalves, L.R. Ultrasound-assisted production of biodiesel and ethanol from spent coffee grounds. Bioresour. Technol. 2014, 167, 343–348. [Google Scholar] [CrossRef]
- Gouvea, B.M.; Torres, C.; Franca, A.S.; Oliveira, L.S.; Oliveira, E.S. Feasibility of ethanol production from coffee husks. Biotechnol. Lett. 2009, 31, 1315–1319. [Google Scholar] [CrossRef] [PubMed]
- Kwon, E.E.; Yi, H.; Jeon, Y.J. Sequential co-production of biodiesel and bioethanol with spent coffee grounds. Bioresour. Technol. 2013, 136, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, R.; Sarangapani, C.; Jaiswal, S.; Cullen, P.J.; Jaiswal, A.K. Ferric chloride assisted plasma pretreatment of lignocellulose. Bioresour. Technol. 2017, 243, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Harsono, S.S.; Fauzi, M.; Purwono, G.S.; Soemarno, D. Second generation bioethanol from Arabica coffee waste processing at small holder plantation in Ijen Plateau region of East Java. Procedia Chem. 2015, 14, 408–413. [Google Scholar] [CrossRef]
- Lee, J.W.; In, J.H.; Park, J.B.; Shin, J.; Park, J.H.; Sung, B.H.; Sohn, J.H.; Seo, J.H.; Park, J.B.; Kim, S.R.; et al. Co-expression of two heterologous lactate dehydrogenases genes in Kluyveromyces marxianus for l-lactic acid production. J. Biotechnol. 2017, 241, 81–86. [Google Scholar] [CrossRef]
- Kim, J.W.; Jang, J.H.; Yeo, H.J.; Seol, J.; Kim, S.R.; Jung, Y.H. Lactic acid production from a whole slurry of acid-pretreated spent coffee grounds by engineered Saccharomyces cerevisiae. Appl. Biochem. Biotechnol. 2019, 189, 206–216. [Google Scholar] [CrossRef]
- Menezes, E.G.; do Carmo, J.R.; Menezes, A.G.; Alves, J.G.; Pimenta, C.J.; Queiroz, F. Use of different extracts of coffee pulp for the production of bioethanol. Appl. Biochem. Biotechnol. 2013, 169, 673–687. [Google Scholar] [CrossRef]
- Bonilla-Hermosa, V.A.; Duarte, W.F.; Schwan, R.F. Utilization of coffee by-products obtained from semi-washed process for production of value-added compounds. Bioresour. Technol. 2014, 166, 142–150. [Google Scholar] [CrossRef]
- Moreira, M.D.; Melo, M.M.; Coimbra, J.M.; Reis, K.C.D.; Schwan, R.F.; Silva, C.F. Solid coffee waste as alternative to produce carotenoids with antioxidant and antimicrobial activities. Waste Manag. 2018, 82, 93–99. [Google Scholar] [CrossRef]
| Yeast | Type of Coffee | Main Acids Detected | Main Compounds Associated with Flavor Identified in Coffee Beans | General Aroma | Ref. |
|---|---|---|---|---|---|
| M. caribbica CCMA 0198 S. cerevisiae CCMA 0543 C. parapsilosis CCMA0544 T. delbrueckii CCMA 0684 | C. arabica var. Canário Amarelo Bourbon | citric, malic, succinic, lactic, oxalic, isobutyric, isovaleric | linalool, hexanal, 1-hexanol, 2-heptanol, benzyl alcohol, benzaldehyde, benzyl benzoate, ethyl hexadecanoate, methylpyrazine, 2-ethyl-5-methylpyrazine, 2- acetyl-6-methylpyrazine, ethyl phenylacetate, methyl-salicylate | sweet, fruity, and mint-like chocolate, nutty citrus flavor (orange, lemon, pineapple) | [6] |
| S. cerevisiae UFLA YCN727 S. cerevisiae UFLA YCN724 | C. arabica var. Acai | malic, lactic, acetic, butyric, propionic, citric, oxalic, succinic, isobutyric, tartaric | 1-pentanol, 2-phenylethanol, acetaldehyde, hexanal, ethyl acetate, furfuryl alcohol, furfural, 2,3-butanedione, isobutyl acetate, 3-methyl-1-butanol, | caramel flavor in the beginning and a bitter aroma at the end | [12] |
| S. cerevisiae UFLACN 727 S. cerevisiae UFLACN 724 | C. arabica, var. Acaiá | malic, isobutyrichexanoic, decanoic, nonanoic | ethanol, 2-phenylethanol, acetaldehyde, 1,2-propanediol, ethyl acetate | sweet | [20] |
| S. cerevisiae CCMA 0543 C. parapsilosis CCMA 0544 T. delbrueckii CCMA 0684 | C. arabica var. Catuaí Amarelo | citric, malic, succinic, lactic, acetic, propionic, isobutyric, chlorogenic | 2,3-butanediol, 2,5-dimethylpyrazine, 2,3-dimethylpyrazine, glycerol and ethanol, hexanal, 2-octenal, 5,9-undecadien-2-one, 6,10dimethyl-1 | fruit like, floral, sweet, caramel, nutty | [27] |
| S. cerevisiae CCMA 0200 T. delbrueckii CCMA 0684 | C. arabica var. Catai Mundo Novo | malic, lactic, acetic, butyric, propionic, citric, oxalic, succinic, tartaric | 2-furanmethanol propanoate, 2-ethyl-3,5-dimethylpyrazine | caramel, fruity, refreshing, walnut, cane molasses | [35] |
| yeasts and bacteria including S. cerevisiae CCMA 0543 | C. arabica var. Catuaí Vermelho | citric, succinic, isovaleric, isobutyric succinic citric, isobutyric, isovaleric | heptadecanol, 4-hydroxy-2-methylacetophenone, 4-ethenyl-1,2-dimethoxybenzene, phenylethyl alcohol, 1-eptadecanol, 2,3-dihydro 7-methyl-4-octanol | - | [37] |
| S. cerevisiae CCMA 0543 | C. arabica var. Mundo Novo, Ouro Amarelo | citric, malic, succinic, acetic, hexanoic | beta-linalool, 1-hexanol, nonanal, 3-methyl-benzaldehyde, 2-pentyl-furan, 3-ethyl-2-hydroxy-2-cyclopenten-1-one, furfurylmethylether, 2,5-dimethyl-4-hydroxy-3(2h)-furanone, 2,3-dimethylpyrazine | herbal or fruity caramel, musty, mushroom-like, nutty, coffee grounds-like, caramel, rasty, sweet | [46] |
| 9 Yeasts including Pichia fermentans YC5.2 Saccharomyces sp. YC9.15 | C. arabica var. Mundo Novo | acetic, latic, caprylic | isoamyl acetate and ethyl acetate acetaldehyde | pineapple-like and banana-like aroma, fruity and floral | [38] |
| Pichia fermentans YC5.2 | C. arabica var. Catuí | citric, malic, acetic, suucinic, fumaric | ethanol, acetaldehyde, ethyl acetat, isoamyl acetate | vanilla taste floral aromas | [39] |
| S. cerevisiae Pichia kluyveri | C. arabica | citric, malic, quinic, succinic, lactic, formic | 3-methylbutanol, 2-phenylethanol, 2-phenylethyl acetate, 2,3-butanedione, 4-vinylguaiacol | nutty, smoky, fruity, caramel aromas | [47] |
| S. cerevisiae CCMA 0543 C. parapsilosis CCMA 0544 T. delbrueckii CCMA 0684 | C. arabica var. Catuaí Amarelo | acetic, citric, malic, succinic, lactic, isobutyric, propionic pentanoic, butanoic | chlorogenic acids and trigonelline, benzyl alcohol and 1-nonanol, hexanal, 2-n-pentylfuran and 5,9-undecadien-2-one, 6,10-dimethyl-(Z), 1H-pyrrole, 1- (2-furanylmethyl), 2,3-pentanedione, 3-methyl- furfuryl alcohol, 2-heptanone | sweet chocolate odor creamy, caramellic notes, banana, fruity, nutty, almond, sweet | [48] |
| S. cerevisiae | C. arabica | butanoic, 3-methylbutanoic | ethyl palmitate, 2-furylmethanol octadecanal, acetyl-3-methylpyrazine, 2,3-dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one, 5-methylfurfural | sweet, spicy, sweet potato, caramel, burnt sugar, raspberries | [49] |
| S. cerevisiae CCMA 0543 T. delbrueckii CCMA 0684 | C. arabica var. Bourbon amarelo Catucaí amarelo Rubi | acetic, citric, malic, lactic, succinic | pyrrole and furan, 1-(3-methylpyrazine) ethanone, methylphenol and 2,3-dimethyl-2-cyclopenten-1-one, dihydro-3-methylene-2,5-furadione, 4-methoxy benzenamine, 2,6-dimethyl-3-octylacetate, and 1,2-epoxy-3-propyl acetate, formic acid, 1-(2-hydroxy-5-methylphenyl) ethanone, 2-furanmethanol | floral, rose-like, smoky hazelnut flavor sweet and roasted bitter, sweet, floral, and herbaceous flavors | [51] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruta, L.L.; Farcasanu, I.C. Coffee and Yeasts: From Flavor to Biotechnology. Fermentation 2021, 7, 9. https://doi.org/10.3390/fermentation7010009
Ruta LL, Farcasanu IC. Coffee and Yeasts: From Flavor to Biotechnology. Fermentation. 2021; 7(1):9. https://doi.org/10.3390/fermentation7010009
Chicago/Turabian StyleRuta, Lavinia Liliana, and Ileana Cornelia Farcasanu. 2021. "Coffee and Yeasts: From Flavor to Biotechnology" Fermentation 7, no. 1: 9. https://doi.org/10.3390/fermentation7010009
APA StyleRuta, L. L., & Farcasanu, I. C. (2021). Coffee and Yeasts: From Flavor to Biotechnology. Fermentation, 7(1), 9. https://doi.org/10.3390/fermentation7010009





