Effects of Energy Cane (Saccharum spp.) Juice on Corn Ethanol (Zea mays) Fermentation Efficiency: Integration towards a More Sustainable Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Design
2.3. Analytical Methods
2.3.1. Yeast Viability
2.3.2. Anions, Glucose, Fructose and Sucrose Content
2.3.3. Bacterial Contamination
2.3.4. Starch Content in the Corn
2.3.5. Alcohol Content
2.4. Fermentation Yield and Efficiency
2.5. Statistics
3. Results
3.1. Water and Corn Use
3.2. Final Yeast Viability, Residual Sugars, and Contamination
3.3. Final Alcohol Content and Fermentation Yield and Efficiency
3.4. Anions Content in the Fermented Juice
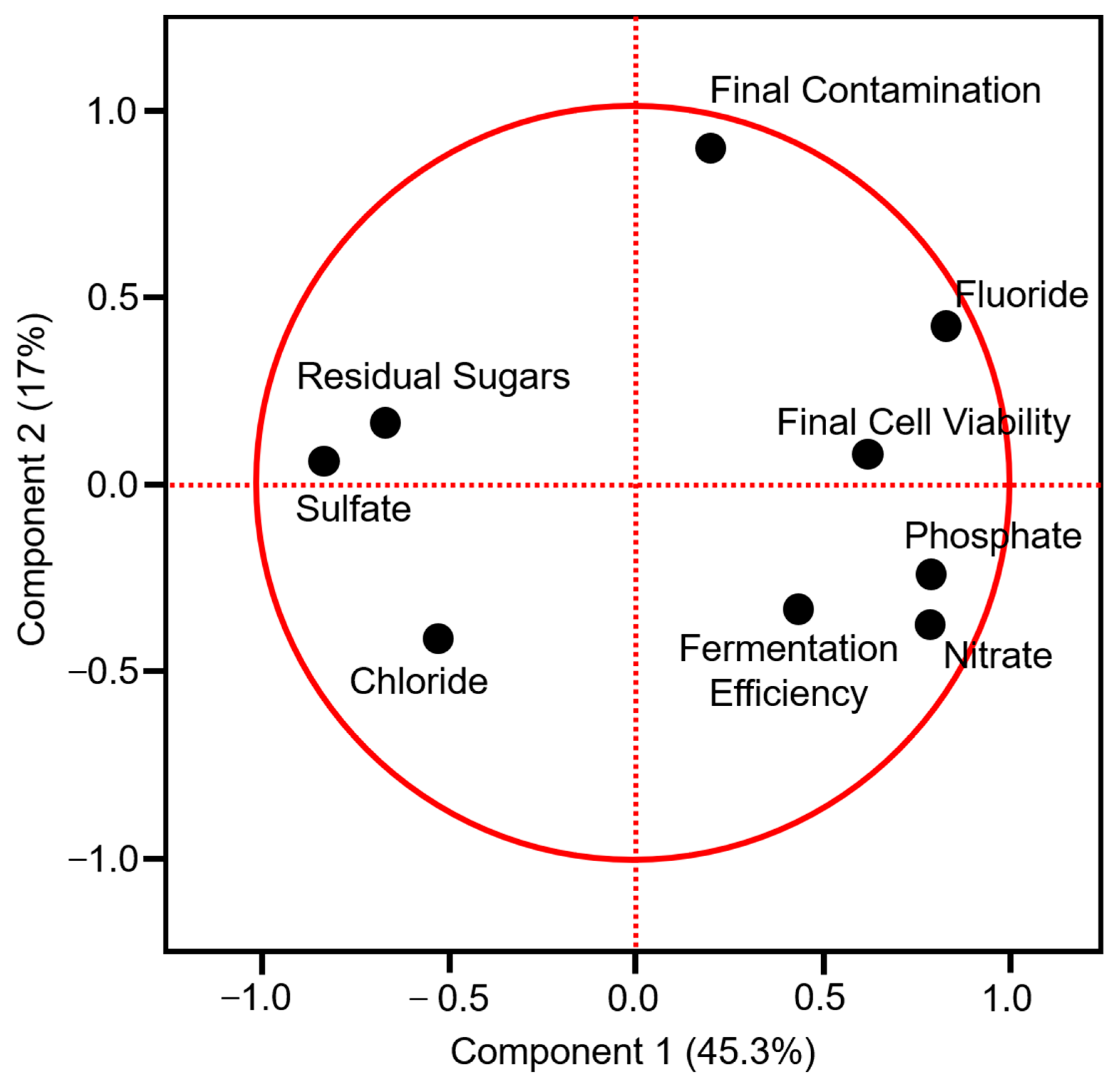
3.5. Principal Component Analysis (PCA) and Correlations between CT and PT
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- UNICA (Brazilian Sugarcane Industry Association—Sugarcane Observatory). Available online: https://www.observatoriodacana.com.br (accessed on 10 November 2019).
- EIA U.S. Energy Information Administration: Total Energy Monthly Data. Available online: https://www.eia.gov/totalenergy/data/monthly/#renewable (accessed on 10 November 2019).
- RFA (Renewable Fuels Association) 2020 Ethanol Industry Outlook. Available online: www.eenergyadams.com (accessed on 7 February 2021).
- U.S. Government. Energy Policy Act of 2005-Public Law 109-58; U.S. Government: Washington, DC, USA, 2005.
- U.S. Government. Energy Independence and Security Act of 2007-Public Law 110-140; U.S. Government: Washington, DC, USA, 2007.
- EPA Renewable Identification Numbers (RINs) under the Renewable Fuel Standard Program. Available online: https://www.epa.gov/renewable-fuel-standard-program/renewable-identification-numbers-rins-under-renewable-fuel-standard (accessed on 7 February 2021).
- Woody, K. Brazil: Grain and Feed Annual; USDA: Brasilia, Brazil, 2019.
- ANP-Brazilian National Agency of Petroleum, Natural Gas, and B. RenovaCalc. Available online: http://www.anp.gov.br/producao-de-biocombustiveis/renovabio/renovacalc (accessed on 10 November 2019).
- Grassi, M.C.B.; Pereira, G.A.G. Energy-cane and RenovaBio: Brazilian vectors to boost the development of Biofuels. Ind. Crop Prod. 2019, 129, 201–205. [Google Scholar] [CrossRef]
- UNICA. Cane’s Bioelectricity in Numbers-January 2017; UNICA: Sao Paulo, Brazil, 2017. (In Portuguese) [Google Scholar]
- Goldemberg, J. Ethanol for a sustainable energy future. Science 2007, 315, 808–810. [Google Scholar] [CrossRef]
- Macedo, I.C.; Seabra, J.E.A.; Silva, J.E.A.R. Green house gases emissions in the production and use of ethanol from sugarcane in Brazil: The 2005/2006 averages and a prediction for 2020. Biomass Bioenergy 2008, 32, 582–595. [Google Scholar] [CrossRef]
- Hill, J. Environmental costs and benefits of transportation biofuel production from food- and lignocellulose-based energy crops. A review. Agron. Sustain. Dev. 2007, 27, 1–12. [Google Scholar] [CrossRef]
- FAO. Crop Prospects and Food Situation. Available online: http://www.fao.org/3/ca5327en/ca5327en.pdf (accessed on 10 November 2019).
- USDA Brazil: Biofuels Annual. Available online: https://apps.fas.usda.gov/newgainapi/api/report/downloadreportbyfilename?filename=BiofuelsAnnual_SaoPauloATO_Brazil_8-10-2018.pdf (accessed on 10 November 2019).
- Kurambhatti, C.; Kumar, D.; Rausch, K.; Tumbleson, M.; Singh, V. Ethanol Production from Corn Fiber Separated after Liquefaction in the Dry Grind Process. Energies 2018, 11, 2921. [Google Scholar] [CrossRef]
- Devantier, R.; Pedersen, S.; Olsson, L. Characterization of very high gravity ethanol fermentation of corn mash. Effect of glucoamylase dosage, pre-saccharification and yeast strain. Appl. Microbiol. Biot. 2005, 68, 622–629. [Google Scholar] [CrossRef]
- Thomas, K.C.; Hynes, S.H.; Ingledew, W.M. Effect of lactobacilli on yeast growth, viability and batch and semi-continuous alcoholic fermentation of corn mash. J. Appl. Microbiol. 2001, 90, 819–828. [Google Scholar] [CrossRef]
- Alexander, A.G. The Energy Cane Alternative; Elsevier Scince Ltd.: Amsterdam, The Netherlands, 1985. [Google Scholar]
- Matsuoka, S.; Kennedy, A.J.; Santos, E.G.; Tomazela, A.L.; Rubio, L.C. Energy Cane: Its Concept, Development, Characteristics, and Prospects. Adv. Bot. 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- FAO FAOSTAT. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 10 November 2019).
- Matsuoka, S.; Bressiani, J.; Maccheroni, W.; Fouto, I. Sugarcane bioenergy. In Sugarcane: Agricultural Production, Bioenergy and Ethanol; Elsevier Inc.: Amsterdam, The Netherlands, 2015; pp. 383–405. ISBN 9780128022399. [Google Scholar]
- Sica, P. Sugarcane Breeding for Enhanced Fiber and Its Impacts on Industrial Processes. In Sugarcane; Khan, M.S., Ed.; IntechOpen: London, UK, 2021. [Google Scholar]
- Carvalho-Netto, O.V.; Bressiani, J.A.; Soriano, H.L.; Fiori, C.S.; Santos, J.M.; Barbosa, G.V.; Xavier, M.A.; Landell, M.G.; Pereira, G.A. The Potential of the Energy Cane as the Main Biomass Crop for the Cellulosic Industry; Springer International Publishing: Berlin/Heidelberg, Germany, 2014; Volume 1, p. 20. [Google Scholar]
- de Souza, R.B.; de Menezes, J.A.S.; de Souza, R.F.R.; Dutra, E.D.; de Morais, M.A. Mineral Composition of the Sugarcane Juice and Its Influence on the Ethanol Fermentation. Appl. Biochem. Biotechnol. 2015, 175, 209–222. [Google Scholar] [CrossRef]
- Ceccato-Antonini, S.R.; Bassi, A.P.G.; Paraluppi, A.L.; dos Santos, E.G.D.; Matsuoka, S. Deterioration and fermentability of energy cane juice. Cienc. Rural. 2017, 47. [Google Scholar] [CrossRef]
- Barros, S.; Woody, K. Corn Ethanol Production Booms in Brazil; USDA: Brasilia, Brazil, 2020.
- Braga, V.S. The Influence of Temperature in Conducting Two Fermentation Processes for the Production of Cachaça; University of Sao Paulo: Piracicaba, Brazil, 2006. (In Portuguese) [Google Scholar]
- Pierce, J.S. Institute of Brewing: Analysis committee measurement of yeast viability. J. Inst. Brew. 1970, 76, 442–443. [Google Scholar] [CrossRef]
- Oliveira, A.; Gallo, C.; Alcarde, V.; Godoy, A.; Amorim, H. Methods for Microbiological Control in Sugar and Alcohol Production (in Portuguese); FERMENTEC/ESALQ/FEALQ: Piracicaba, Brazil, 1996. [Google Scholar]
- Eith, C.; Kolb, M.; Seubert, A.; Viehweger, K.H. Practical Ion Chromatography: An Introduction; Methrom: Herisau, Switzerland, 2002. [Google Scholar]
- Corbacho, I.; Teixidó, F.; Velázquez, R.; Hernández, L.M.; Olivero, I. Standard YPD, even supplemented with extra nutrients, does not always compensate growth defects of Saccharomyces cerevisiae auxotrophic strains. Anton. Leeuw. Int. J. G. 2011, 99, 591–600. [Google Scholar] [CrossRef]
- PubChem Hexanamide|C6H13NO-PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/hexanamide#section=GHS-Classification (accessed on 10 November 2019).
- Rickard, J.E.; Behn, K.R. Evaluation of acid and Enzyme hydrolytic methods for the determination of cassava starch. J. Sci. Food Agric. 1987, 41, 373–379. [Google Scholar] [CrossRef]
- Basso, L.C.; De Amorim, H.V.; De Oliveira, A.J.; Lopes, M.L. Yeast Selection for Fuel Ethanol Production in Brazil. FEMS Yeast Res. 2008, 8, 1155–1163. [Google Scholar] [CrossRef]
- Silva, L.F.L.F.; Bernardino, C.D.; Ré, F.E.; Furtado, C.H.F.; Amorim, H.V. Analytical Methods for the Control of Alcohol and Sugar Production; Fermentec: Piracicaba, Brazil, 2003. [Google Scholar]
- Alterthum, F.; Cruz, M.R.M.; Vairo, M.L.R.; Gambassi, D.M. Effect of contaminating microorganisms of alcoholic fermentation on microdistilleries (in Portuguese). STAB Açúcar Álcool Subprodutos 1984, 3, 42–49. [Google Scholar]
- Bai, F.W.; Chen, L.J.; Zhang, Z.; Anderson, W.A.; Moo-Young, M. Continuous ethanol production and evaluation of yeast cell lysis and viability loss under very high gravity medium conditions. J. Biotechnol. 2004, 110, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Petrovska, B.; Winkelhausen, E.; Kuzmanova, S. Glycerol Production by Yeasts under Osmotic and Sulfite Stress. Can. J. Microbiol. 1999, 45, 695–699. [Google Scholar] [CrossRef]
- Prado, R.D.M.; Caione, G.; Campos, C.N.S. Filter Cake and Vinasse as Fertilizers Contributing to Conservation Agriculture. Appl. Environ. Soil Sci. 2013, 2013. [Google Scholar] [CrossRef]
- Moreira, B.R.A.; Viana, R.S.; Lisboa, L.A.M.; Lopes, P.R.M.; Figueiredo, P.A.M.; Ramos, S.B.; Bonini, C.S.B.; Trindade, V.D.R.; Andrade, M.G.O.; May, A. Classifying Hybrids of Energy Cane for Production of Bioethanol and Cogeneration of Biomass-Based Electricity by Principal Component Analysis-Linked Fuzzy C-Means Clustering Algorithm. J. Agric. Sci. 2019, 11, 246–253. [Google Scholar] [CrossRef]
- Viana, R.D.; Moreira, B.D.; May, A.; Miasaki, C.T.; Caraschi, J.C.; Andrade, M.D. Juice technological quality, lignocellulosic physical-chemical attributes and biomass yield from energy cane clones. Aust. J. Crop Sci. 2019, 13, 746–752. [Google Scholar] [CrossRef]
- Knoll, J.E.; Anderson, W.F.; Richard, E.P.; Doran-Peterson, J.; Baldwin, B.; Hale, A.L.; Viator, R.P. Harvest date effects on biomass quality and ethanol yield of new energycane (Saccharum hyb.) genotypes in the Southeast USA. Biomass Bioenergy 2013, 56, 147–156. [Google Scholar] [CrossRef]
- Li, Z.; Wang, D.; Shi, Y.C. Effects of nitrogen source on ethanol production in very high gravity fermentation of corn starch. J. Taiwan Inst. Chem. Eng. 2017, 70, 229–235. [Google Scholar] [CrossRef]
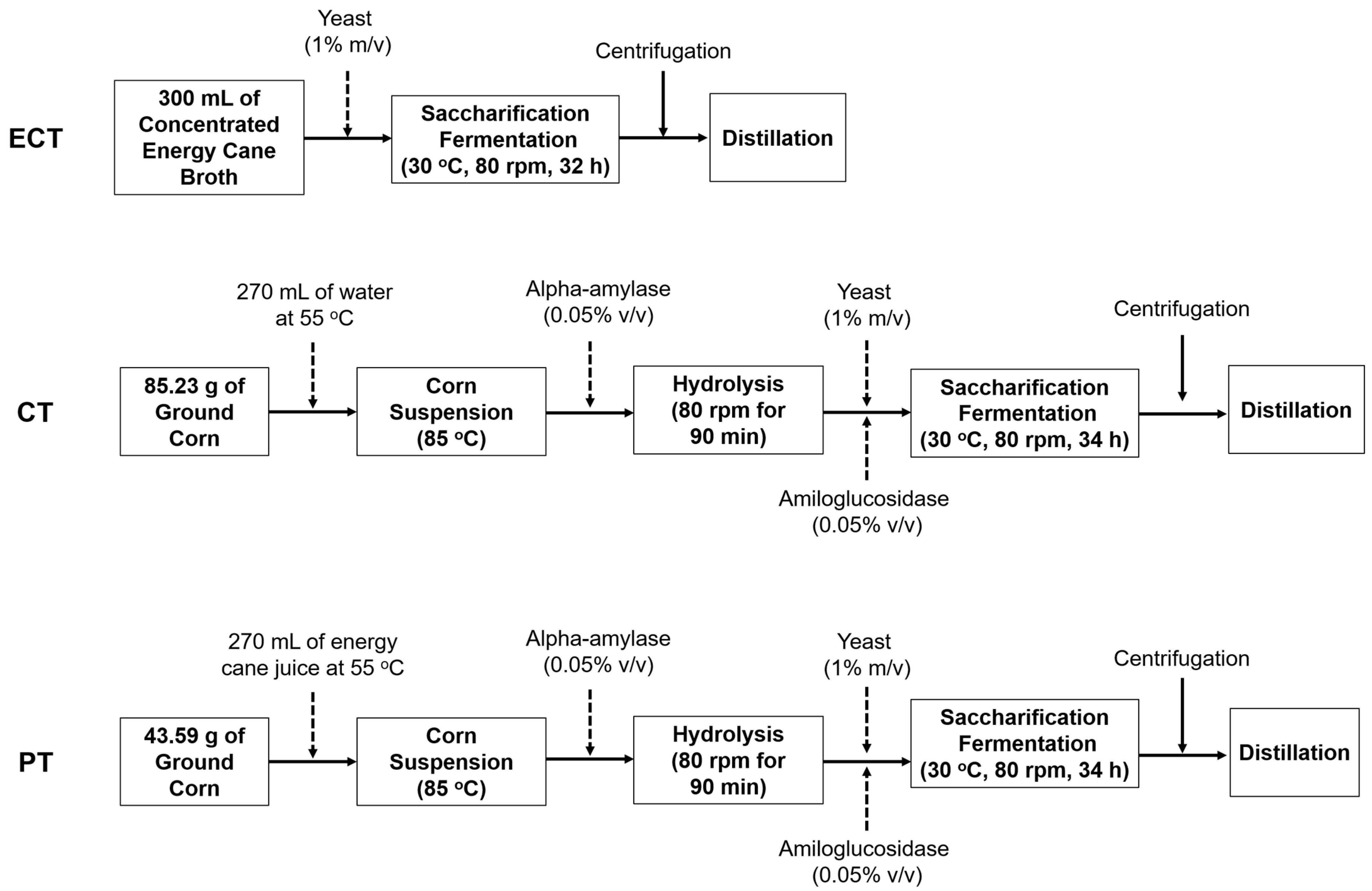
| Water Added in Each Reactor (mL) | Corn Used in Each Reactor (g) | g of Corn per mL of Ethanol | |
|---|---|---|---|
| CT | 270 | 85.23 | 2.53 |
| PT | 0 | 43.59 | 1.21 |
| Treatment | Yeast Viability (%) | Residual Sugars (%) | CFU/mL (103) |
|---|---|---|---|
| ECT | 91.91 ± 3.56 a | 0.27 ± 0.04 b | 7.69 ± 6.03 |
| CT | 88.26 ± 4.81 ab | 0.55 ± 0.16 a | 6.18 ± 4.38 |
| PT | 85.94 ± 5.49 b | 0.33 ± 0.16 ab | 6.04 ± 4.58 |
| Treatment | Alcohol Content (v·v−1) | Yield (mL100·g−1) | Efficiency (%) |
|---|---|---|---|
| ECT | 10.5 ± 0.22 c | 52.2 ± 1.08 c | 80.6 ± 1.67 c |
| CT | 11.2 ± 0.47 b | 55.9 ± 2.34 b | 86.4 ± 3.61 b |
| PT | 12.0 ± 0.39 a | 60.2 ± 1.97 a | 90.8 ± 3.04 a |
| Fluoride | Chloride | Sulfate | Nitrate | Phosphate | |
|---|---|---|---|---|---|
| ECT | 116 ± 3 a | 1969 ± 71 a | 1244 ± 32 a | 3 ± 1 b | 3547 ± 95 a |
| CT | 10 ± 1 c | 318 ± 15 c | 461 ± 6 b | 0 ± 0 c | 347 ± 59 c |
| PT | 71 ± 1 b | 1011 ± 28 b | 192 ± 10 c | 17 ± 1 a | 1948 ± 157 b |
| Effic. | R.S. | Contam. | Viab. | Fluor. | Chlor. | Sulf. | Nitr. | Phos. | |
|---|---|---|---|---|---|---|---|---|---|
| Effic. | 1 | ||||||||
| R. S. | −0.34 * | 1 | |||||||
| Contam. | −0.03 | −0.07 | 1 | ||||||
| Viab. | −0.15 | −0.06 | −0.2 | 1 | |||||
| Fluor. | 0.73 * | −0.97 * | 0.41 | 0.63 | 1 | ||||
| Chlor. | −0.24 | 0.34 | −0.12 | −0.31 | −0.60 * | 1 | |||
| Sulf. | −0.66 * | 0.88 * | −0.25 | −0.37 | −0.98 * | 0.23 | 1 | ||
| Nitr. | 0.80 * | −0.82 * | −0.17 | 0.85 * | 0.80 * | −0.26 | −0.88 * | 1 | |
| Phos. | 0.99 * | −0.97 * | −0.01 | 0.33 | 0.99 * | −0.35 | −0.95 * | -0.86 * | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sica, P.; Prado, L.M.L.M.; Granja, P.; Carvalho, E.M.d.; Mattos, E.d.C.; Calegari, R.P.; Silverio, M.; Martins, B.C.; Baptista, A.S. Effects of Energy Cane (Saccharum spp.) Juice on Corn Ethanol (Zea mays) Fermentation Efficiency: Integration towards a More Sustainable Production. Fermentation 2021, 7, 30. https://doi.org/10.3390/fermentation7010030
Sica P, Prado LMLM, Granja P, Carvalho EMd, Mattos EdC, Calegari RP, Silverio M, Martins BC, Baptista AS. Effects of Energy Cane (Saccharum spp.) Juice on Corn Ethanol (Zea mays) Fermentation Efficiency: Integration towards a More Sustainable Production. Fermentation. 2021; 7(1):30. https://doi.org/10.3390/fermentation7010030
Chicago/Turabian StyleSica, Pietro, Laysa Maciel Lewandowski Meira Prado, Pedro Granja, Elias Miguel de Carvalho, Eduardo de Castro Mattos, Rubens Perez Calegari, Manuella Silverio, Bianca Chaves Martins, and Antonio Sampaio Baptista. 2021. "Effects of Energy Cane (Saccharum spp.) Juice on Corn Ethanol (Zea mays) Fermentation Efficiency: Integration towards a More Sustainable Production" Fermentation 7, no. 1: 30. https://doi.org/10.3390/fermentation7010030
APA StyleSica, P., Prado, L. M. L. M., Granja, P., Carvalho, E. M. d., Mattos, E. d. C., Calegari, R. P., Silverio, M., Martins, B. C., & Baptista, A. S. (2021). Effects of Energy Cane (Saccharum spp.) Juice on Corn Ethanol (Zea mays) Fermentation Efficiency: Integration towards a More Sustainable Production. Fermentation, 7(1), 30. https://doi.org/10.3390/fermentation7010030






