The Effect of Salt and Temperature on the Growth of Fresco Culture
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microorganisms
2.2. Inoculation and Cultivation Conditions
2.3. The Effect of NaCl Addition on the Growth of Fresco Culture
2.4. Total Counts of Fresco Culture in Growth Media
2.5. Fitting the Growth Curves and Calculating the Growth Parameters
2.6. Secondary Models
2.7. Validation of the Models
3. Results
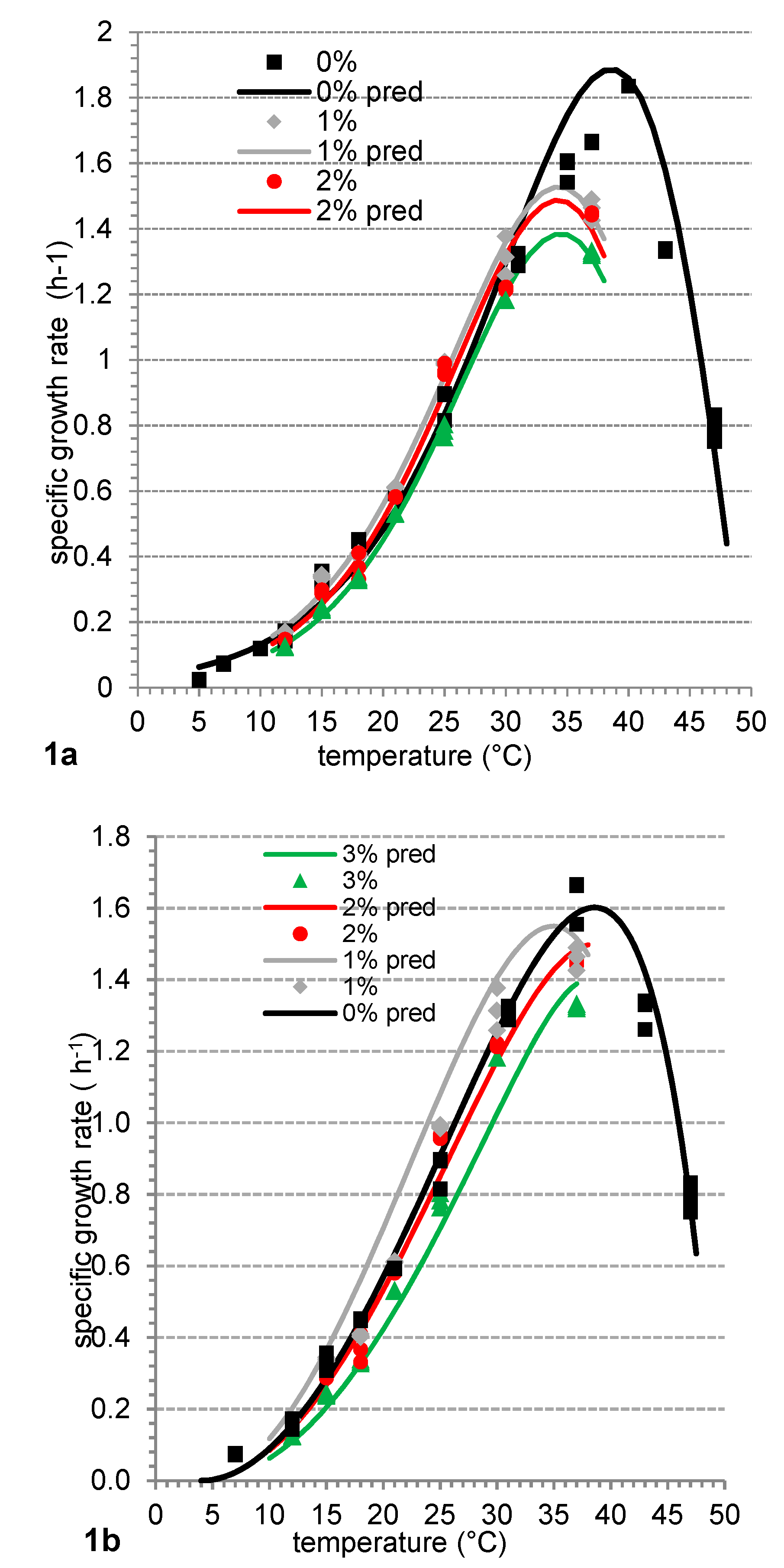
3.1. The Effect of Incubation Temperature on Fresco Culture Growth Rate
3.2. The Effect of NaCl on the Growth Dynamics of Fresco Culture
3.3. Validation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ross, T.; McMeekin, T.A. Predictive microbiology. Int. J. Food Microbiol. 1994, 23, 241–264. [Google Scholar] [CrossRef]
- Aghababaie, M.; Khanahmadi, M.; Beheshti, M. Developing a detailed kinetic model for the production of yogurt starter bacteria in single strain cultures. Food Bioprod. Process. 2014, 94, 657–667. [Google Scholar] [CrossRef]
- Alvarez, M.M.; Aguirre-Ezkauriatza, E.J.; Ramírez-Medrano, A.; Rodríguez-Sánchez, Á. Kinetic analysis and mathematical modeling of growth and lactic acid production of Lactobacillus casei var. rhamnosus in milk whey. J. Dairy Sci. 2010, 93, 5552–5560. [Google Scholar] [CrossRef] [PubMed]
- Lahtinen, S.; Salminen, S.; Von Wright, A.; Ouwehand, A.C. Lactic Acid Bacteria: Microbiological and Functional Aspects, 4th ed.; CRC Press: Boca Raton, FL, USA, 2011; 779p, ISBN 978-1139836781. [Google Scholar]
- Ljungh, A.; Wadström, T. Lactobacillus Molecular Biology: From Genomics to Probiotics, 1st ed.; Horizon Scientific Press: Norfolk, FL, USA, 2009; 205p, ISBN 978-1904455417. [Google Scholar]
- Marth, E.C.; Steel, J.L. Applied Dairy Microbiology, 2nd ed.; Marcel Dekker, Inc.: New York, NY, USA, 2001; 736p, ISBN 978-0824705367. [Google Scholar]
- Kavitake, D.; Kandasamy, S.; Devi, P.B.; Shetty, P.H. Recent developments on encapsulation of lactic acid bacteria as potential starter culture in fermented foods. Food Biosci. 2018, 21, 34–44. [Google Scholar] [CrossRef]
- Mancuskova, T.; Medved’ova, A.; Ozbolt, M. The medical functions of probiotics and their role in clinical nutrition. Curr. Nutr. Food Sci. 2017, 13, 1–8. [Google Scholar] [CrossRef]
- Sasaki, Y.; Horiuchi, H.; Kawashima, H.; Mukai, T.; Yamamoto, Y. NADH oxidase of S. thermophilus 1131 is required for the effective yogurt fermentation with L. delbrueckii subsp. bulgaricus 2038. Biosci. Microbiota Food Health 2014, 33, 31–40. [Google Scholar] [CrossRef]
- Wang, T.; Xu, Z.; Lu, S.; Xin, M.; Kong, J. Effects of glutathione on acid stress resistance and symbiosis between S. thermophilus and L. delbrueckii subsp. bulgaricus. Int. Dairy J. 2016, 61, 22–28. [Google Scholar] [CrossRef]
- Neysens, P.; Messens, W.; De Wuyst, L. Effect of NaCl on growth and bacteriocin production by L. amylovorus DCE 471. Int. J. Food Microbiol. 2003, 88, 29–39. [Google Scholar] [CrossRef]
- Fresco DVS 1010 Culture. Available online: www.chr-hansen.com/en (accessed on 25 May 2018).
- Ramchandran, L.; Sanciolo, P.; Vasiljevic, T.; Broome, M.; Powell, I.; Diuke, M. Improving cell yield and lactic acid production of L. lactis ssp. cremoris by a novel submerged membrane fermentation process. J. Membr. Sci. 2012, 403, 179–187. [Google Scholar] [CrossRef]
- EN ISO 4833-1:2013. In Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 1: Colony Count at 30 °C by the Pour Plate Technique; International Organization for Standardization: Geneva, Switzerland, 2013; 13p.
- Baranyi, J.; Roberts, T.A. A dynamic approach to predicting bacterial growth in food. Int. J. Food Microbiol. 1994, 23, 277–294. [Google Scholar] [CrossRef]
- Rosso, L.; Lobry, J.; Flanders, J. Unexpected correlation between temperatures of microbial growth highlighted by new model. J. Theor. Biol. 1993, 162, 447–463. [Google Scholar] [CrossRef]
- Gibson, A.; Baranyi, J.; Pitt, J.I.; Eyles, M.J.; Roberts, T.A. Predicting fungal growth—The effect of water activity on A. flavus and related species. Int. J. Food Microbiol. 1994, 23, 419–431. [Google Scholar] [CrossRef]
- Ratkowsky, D.A.; Lowry, R.K.; McMeekin, T.A.; Stokes, A.N.; Chandler, R.E. Model for bacterial culture growth rate throughout the entire biokinetic temperature range. J. Bacteriol. 1983, 154, 1222–1226. [Google Scholar]
- Daughtry, B.J.; Davey, K.R.; King, K.D. Temperature dependence of growth kinetics of food bacteria. Food Microbiol. 1997, 14, 21–30. [Google Scholar] [CrossRef]
- TeGiffel, M.C.; Zwietering, M.H. Validation of predictive models describing the growth of L. monocytogenes. Int. J. Food Microbiol. 1999, 46, 135–149. [Google Scholar] [CrossRef]
- Zwietering, M.H.; De Koos, J.T.; Hasenack, B.E.; De Wit, J.C.; Riet, K. Modelling of bacterial growth as a function of temperature. Appl. Environ. Microbiol. 1991, 57, 1094–1101. [Google Scholar] [PubMed]
- Zurera-Cosano, G.; Garciá-Gimeno, R.M.; Rodríguez-Pérez, R.; Hervás-Martínez, C. Performance of response surface model for predition of L. mesenteroides growth parameters under different experimental conditions. Food Control 2006, 17, 429–438. [Google Scholar] [CrossRef]
- Baranyi, J.; Pin, C.; Ross, T. Validating and comparing predictive models. Int. J. Food Microbiol. 1999, 48, 159–166. [Google Scholar] [CrossRef]
- MedveĎová, A.; Liptáková, D.; Valík, L. Characterization of the growth of L. rhamnosus GG in milk at suboptimal temperature. J. Nutr. Food Res. 2008, 47, 60–67. [Google Scholar]
- Medvedova, A.; Mancuskova, T.; Valik, L. Growth of L. acidophilus NCFM in dependence on temperature. Acta Aliment. 2016, 45, 104–111. [Google Scholar] [CrossRef]
- Matejčeková, Z.; Liptáková, D.; Spodniaková, S.; Valík, Ľ. Characterisazion of the growth of L. plantarum in milk in dependence on temperature. Acta Chim. 2016, 9, 104–108. [Google Scholar] [CrossRef]
- Champagne, C.P.; Gagnon, D.; St. Gelais, D.; Vullemard, J.C. Interactions between L. lactis and S. thermophilus strains in Cheddar cheese processing conditions. Int. Dairy J. 2009, 19, 669–674. [Google Scholar] [CrossRef]
- Valík, L.; Ačai, P.; Medved’ová, A. Application of competitive models in predicting the simultaneous growth of S. aureus and lactic acid bacteria in milk. Food Control 2018, 87, 145–152. [Google Scholar] [CrossRef]
- Ačai, P.; Valík, L.; Medved’ová, A.; Rosskopf, F. Modelling and predicting the simultaneous growth of E. coli and lactic acid bacteria in milk. Food Sci. Technol. Int. 2016, 22, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Šípková, A.; Valík, L.; Liptáková, D.; Pelikánová, J. Effect of lactic acid bacteria on the growth dynamics of Geotrichum candidum in fresh cheeses during storage. J. Food Nutr. Res. 2014, 53, 224–231. [Google Scholar]
- Liu, S.Q.; Asmundson, V.; Gopal, P.K.; Holland, R.; Crow, V.L. Influence of reduced water activity on lactose metabolism by Lactococcus lactis ssp. cremoris at different pH values. Appl. Environ. Microbiol. 1998, 64, 2111–2116. [Google Scholar] [PubMed]
- Woollard, D.C.; Indyk, H.E.; Woollard, G.A. Carnitine in milk: A survey of content, distribution and temporal variation. Food Chem. 1999, 66, 121–127. [Google Scholar] [CrossRef]
- Uguen, P.; Hamelin, J.; Le Pennec, J.P.; Blanco, C. Influence of osmolarity and the presence of an osmoprotectant on Lactococcus lactis growth and bacteriocin production. Appl. Environ. Microbiol. 1999, 65, 291–293. [Google Scholar]
- Larsen, R.F.; Añón, M.C. Effect of water activity of milk on acid production by Streptococcus thermophilus and Lactobacillus bulgaricus. J. Food Sci. 1989, 54, 917–921. [Google Scholar] [CrossRef]
- Ross, T.; Dalgaard, P.; Tienungon, S. Predictive modelling of the growth and survival of Listeria in fishery products. Int. J. Food Microbiol. 2000, 62, 231–246. [Google Scholar] [CrossRef]
- Valík, L.; Medved’ová, A.; Čižniar, M.; Liptáková, D. Evaluation of temperature effect on growth rate of Lactobacillus rhamnosus GG in milk using secondary models. Chem. Pap. 2013, 67, 737–742. [Google Scholar] [CrossRef]
| T | Gr | λ | Nmax | rpH | lagpH |
|---|---|---|---|---|---|
| 5 | 0.010 ± 0.001 | 146.8 ± 8.96 | 8.94 ± 0.06 | −0.033 | 547.0 |
| 7 | 0.032 ± 0.001 | 43.0 ± 4.44 | 8.17 ± 0.02 | −0.004 | 130.0 |
| 12 | 0.071 ± 0.007 | 3.4 ± 0.17 | 8.87 ± 0.09 | −0.053 | 54.3 |
| 15 | 0.140 ± 0.011 | 6.7 ± 1.80 | 9.26 ± 0.05 | −0.048 | 24.1 |
| 18 | 0.196 ± 0.001 | 4.2 ± 0.10 | 8.97 ± 0.03 | −0.087 | 19.9 |
| 21 | 0.257 ± 0.000 | - | 9.33 ± 0.02 | −0.308 | 17.3 |
| 25 | 0.390 ± 0.008 | 1.2 ± 0.02 | 9.27 ± 0.14 | −0.324 | 7.9 |
| 31 | 0.567 ± 0.008 | 2.2 ± 0.04 | 9.35 ± 0.05 | −0.612 | 9.7 |
| 37 | 0.722 ± 0.027 | 0.5 ± 0.05 | 8.59 ± 0.05 | −0.451 | 4.5 |
| 40 | 0.961 ± 0.217 | 1.87 ± 0.54 | 8.68 ± 0.07 | −0.234 | 12.3 |
| 43 | 0.582 ± 0.019 | 0.8 ± 0.08 | 8.40 ± 0.21 | −0.381 | 7.5 |
| 47 | 0.349 ± 0.018 | - | 7.90 ± 0.21 | −0.070 | - |
| T | aw | Gr | λ | Nmax | rpH | lagpH |
|---|---|---|---|---|---|---|
| 12 | 0.991 | 0.074 ± 0.000 | 3.8 ± 0.1 | 9.36 ± 0.01 | −0.175 | 101.2 |
| 0.987 | 0.064 ± 0.000 | 7.7 ± 0.2 | 9.11 ± 0.04 | −0.101 | 112.2 | |
| 0.981 | 0.054 ± 0.001 | 14.0 ± 1.4 | 9.13 ± 0.13 | −0.040 | 116.3 | |
| 15 | 0.993 | 0.147 ± 0.002 | 3.5 ± 0.3 | 9.27 ± 0.03 | −0.126 | 42.0 |
| 0.988 | 0.127 ± 0.003 | 4.1 ± 0.2 | 9.21 ± 0.08 | −0.113 | 51.9 | |
| 0.981 | 0.105 ± 0.002 | 6.6 ± 0.0 | 9.15 ± 0.04 | −0.075 | 63.4 | |
| 18 | 0.993 | 0.175 ± 0.002 | 0.2 ± 0.5 | 9.12 ± 0.06 | −0.086 | 23.5 |
| 0.989 | 0.159 ± 0.017 | 2.0 ± 0.6 | 9.01 ± 0.06 | −0.098 | 32.3 | |
| 0.981 | 0.145 ± 0.002 | 4.6 ± 0.6 | 8.61 ± 0.08 | −0.056 | 38.7 | |
| 21 | 0.992 | 0.265 ± 0.001 | 0.7 ± 0.1 | 9.33 ± 0.02 | −0.209 | 21.2 |
| 0.989 | 0.253 ± 0.001 | 1.0 ± 0.0 | 9.60 ± 0.01 | −0.155 | 21.2 | |
| 0.977 | 0.231 ± 0.000 | 2.1 ± 0.0 | 9.09 ± 0.06 | −0.358 | 32.4 | |
| 25 | 0.992 | 0.430 ± 0.002 | 1.2 ± 0.0 | 9.28 ± 0.00 | −0.176 | 12.3 |
| 0.988 | 0.420 ± 0.008 | 1.8 ± 0.2 | 9.32 ± 0.00 | −0.172 | 12.7 | |
| 0.977 | 0.342 ± 0.009 | 1.9 ± 0.1 | 8.91 ± 0.06 | −0.132 | 18.4 | |
| 30 | 0.990 | 0.570 ± 0.019 | 0.4 ± 0.2 | 9.14 ± 0.11 | −0.181 | 9.8 |
| 0.981 | 0.527 ± 0.011 | 0.1 ± 0.3 | 9.08 ± 0.26 | −0.168 | 9.0 | |
| 0.977 | 0.514 ± 0.010 | 0.7 ± 0.3 | 9.17 ± 0.16 | −0.182 | 11.2 | |
| 37 | 0.992 | 0.634 ± 0.014 | 0.3 ± 0.0 | 8.98 ± 0.02 | −0.297 | 7.0 |
| 0.986 | 0.629 ± 0.003 | 0.6 ± 0.0 | 9.38 ± 0.02 | −0.231 | 8.6 | |
| 0.980 | 0.576 ± 0.003 | 1.2 ± 0.0 | 8.72 ± 0.04 | −0.178 | 12.1 |
| Equation/Coefficients of the Equation | ||||||||
|---|---|---|---|---|---|---|---|---|
| Af | Bf | %Df | R2 | %V | RSS | RMSE | %SEP | |
| CTMI model | ||||||||
| 0 % | 1.070 | 0.995 | 7.0 | 0.987 | 98.3 | 0.069 | 0.051 | 6.1 |
| 1 % | 1.193 | 1.156 | 19.3 | 0.949 | 93.3 | 0.231 | 0.093 | 12.3 |
| 2 % | 1.090 | 0.987 | 9.0 | 0.985 | 98.0 | 0.067 | 0.049 | 6.9 |
| 3 % | 1.134 | 0.904 | 13.4 | 0.949 | 93.1 | 0.185 | 0.083 | 13.3 |
| 0 % | 1.146 | 1.010 | 14.6 | 0.984 | 98.3 | 0.129 | 0.058 | 16.1 |
| 1 % | 1.082 | 0.999 | 8.2 | 0.988 | 98.6 | 0.032 | 0.039 | 5.2 |
| 2 % | 1.090 | 0.999 | 9.0 | 0.987 | 98.5 | 0.060 | 0.055 | 8.0 |
| 3 % | 1.055 | 0.999 | 5.5 | 0.995 | 99.5 | 0.008 | 0.020 | 3.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Medveďová, A.; Šipošová, P.; Mančušková, T.; Valík, Ľ. The Effect of Salt and Temperature on the Growth of Fresco Culture. Fermentation 2019, 5, 2. https://doi.org/10.3390/fermentation5010002
Medveďová A, Šipošová P, Mančušková T, Valík Ľ. The Effect of Salt and Temperature on the Growth of Fresco Culture. Fermentation. 2019; 5(1):2. https://doi.org/10.3390/fermentation5010002
Chicago/Turabian StyleMedveďová, Alžbeta, Petra Šipošová, Tatiana Mančušková, and Ľubomír Valík. 2019. "The Effect of Salt and Temperature on the Growth of Fresco Culture" Fermentation 5, no. 1: 2. https://doi.org/10.3390/fermentation5010002
APA StyleMedveďová, A., Šipošová, P., Mančušková, T., & Valík, Ľ. (2019). The Effect of Salt and Temperature on the Growth of Fresco Culture. Fermentation, 5(1), 2. https://doi.org/10.3390/fermentation5010002







