Effects of Ultrasound on Fermentation of Glucose to Ethanol by Saccharomyces cerevisiae
Abstract
1. Introduction
2. Materials and Methods
2.1. Inoculum and Medium Preparation
2.2. Fermentation
2.3. Ultrasound
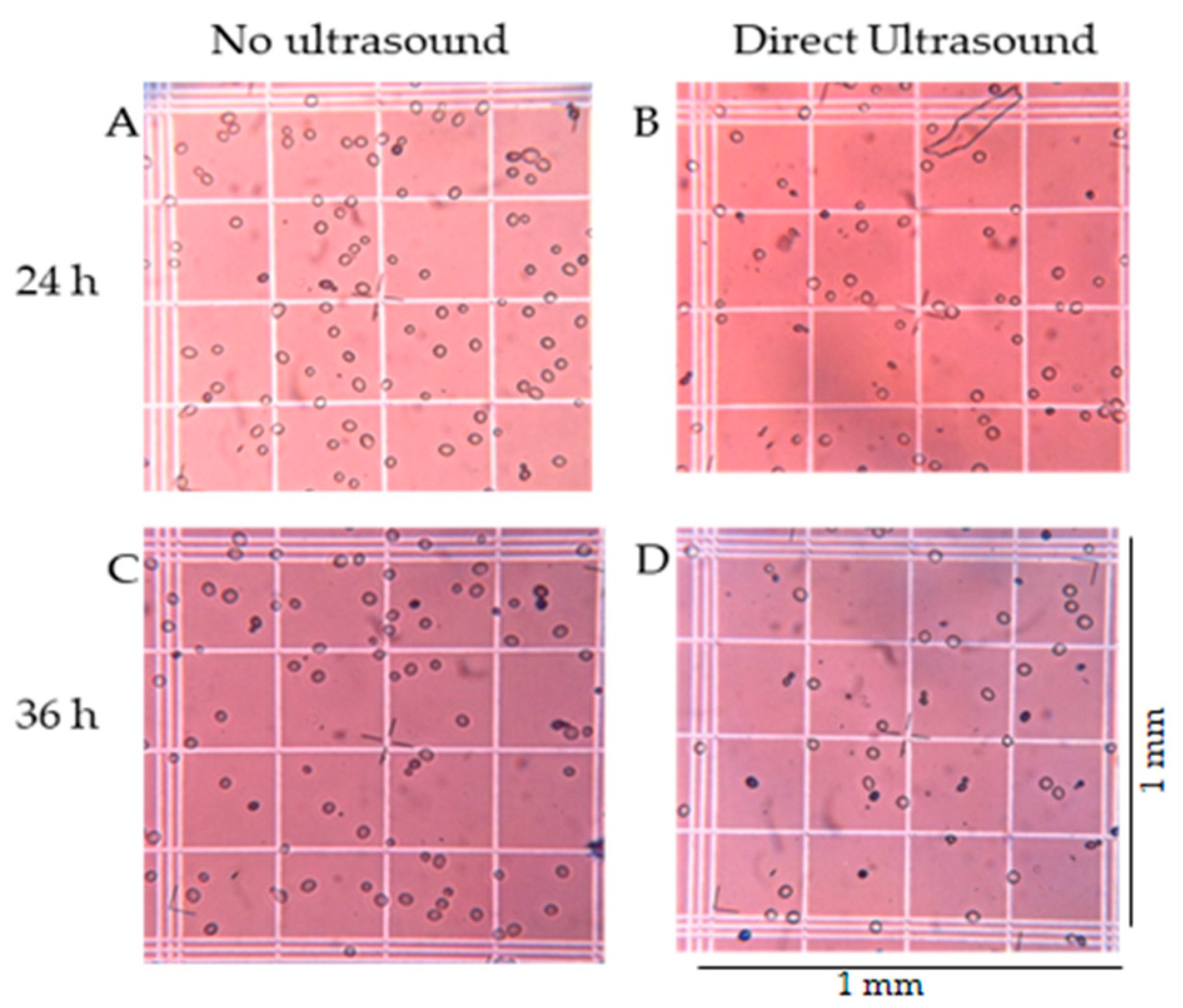
2.4. Sampling and Measurements
2.5. Experimental Design
2.6. Mass Transfer Limitation
2.7. Statistical Analysis
3. Results and Discussion
3.1. Mass Transfer Limitation
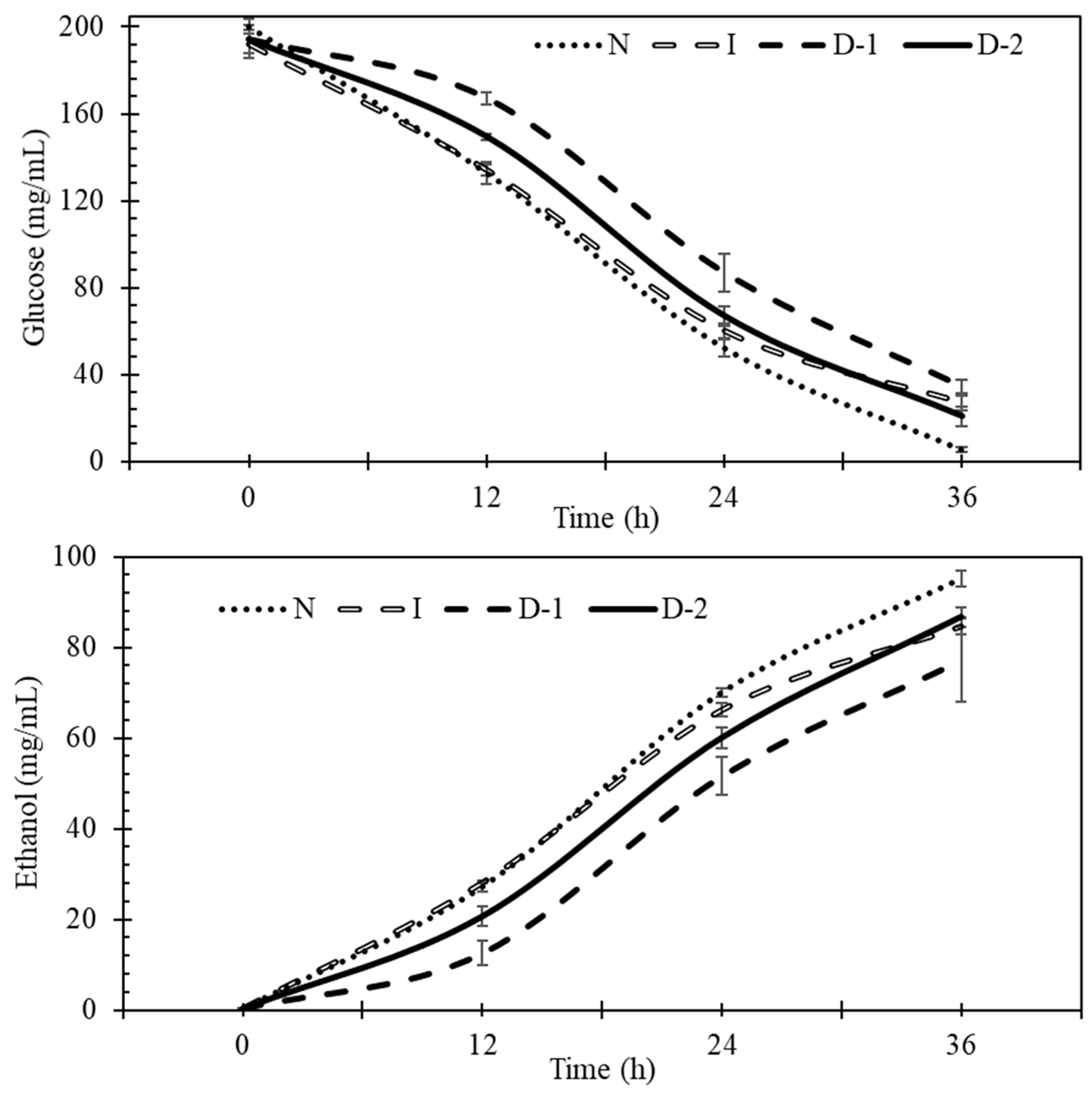
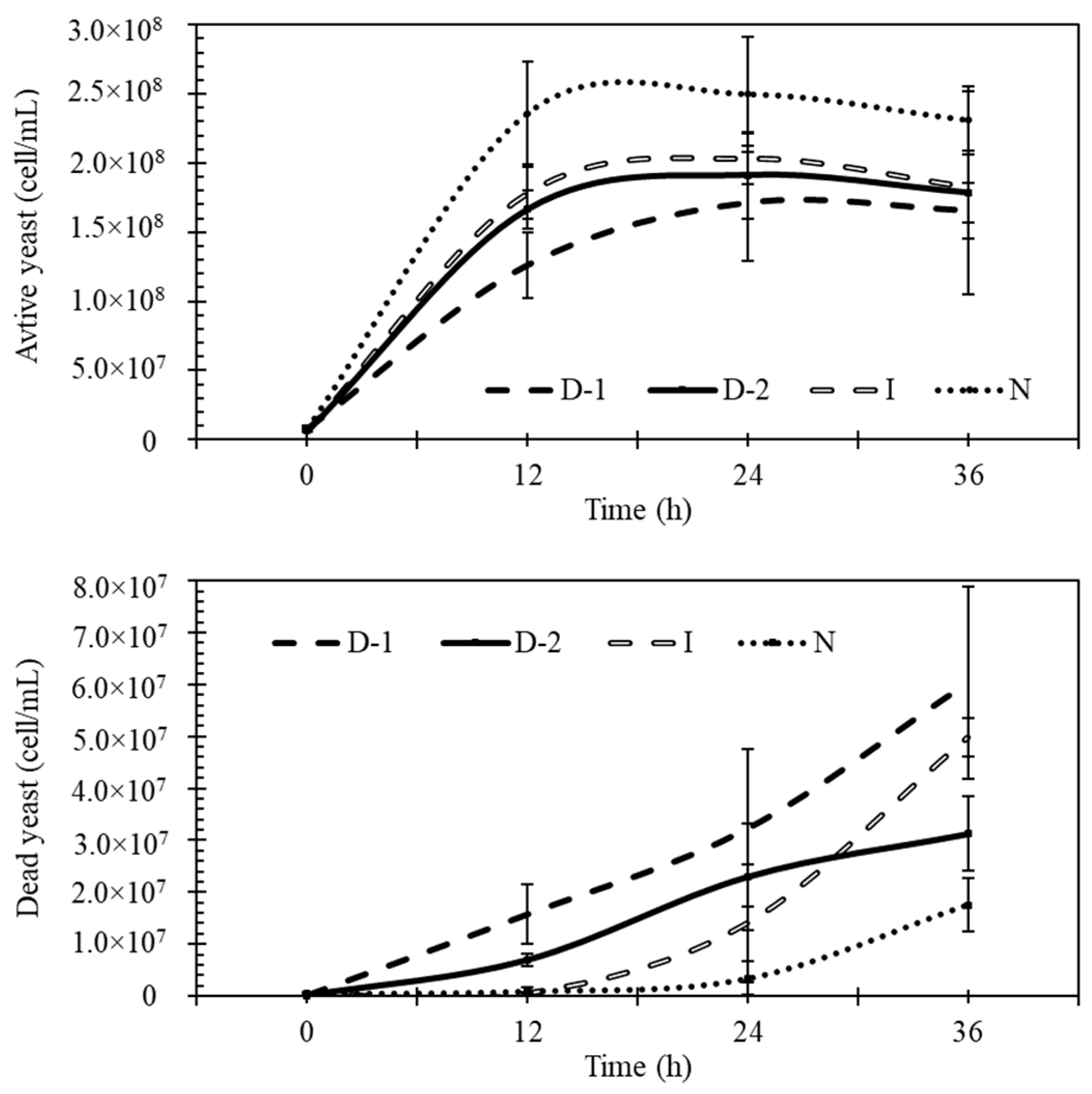
3.2. Fermentation
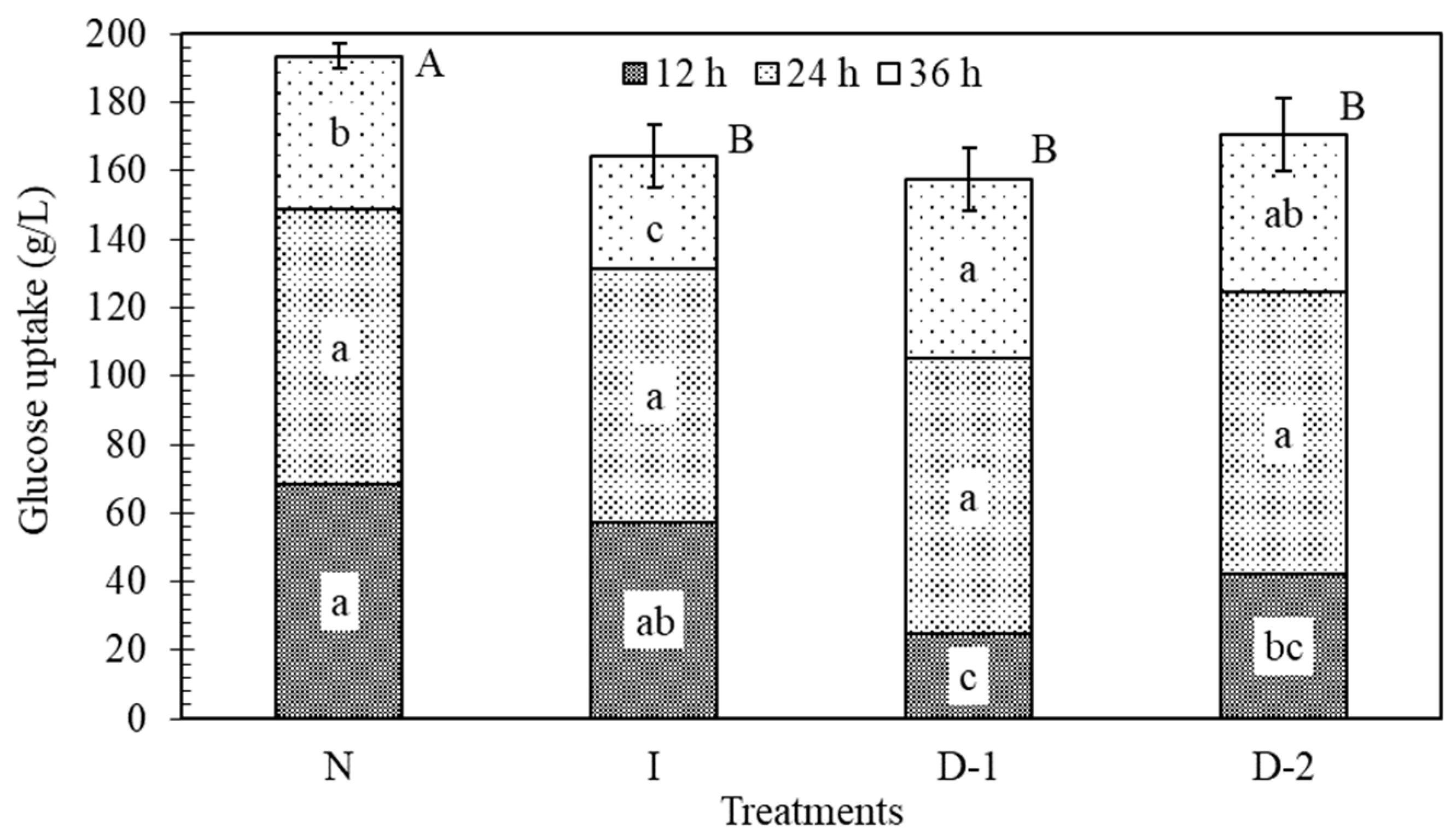
3.3. Glucose Uptake
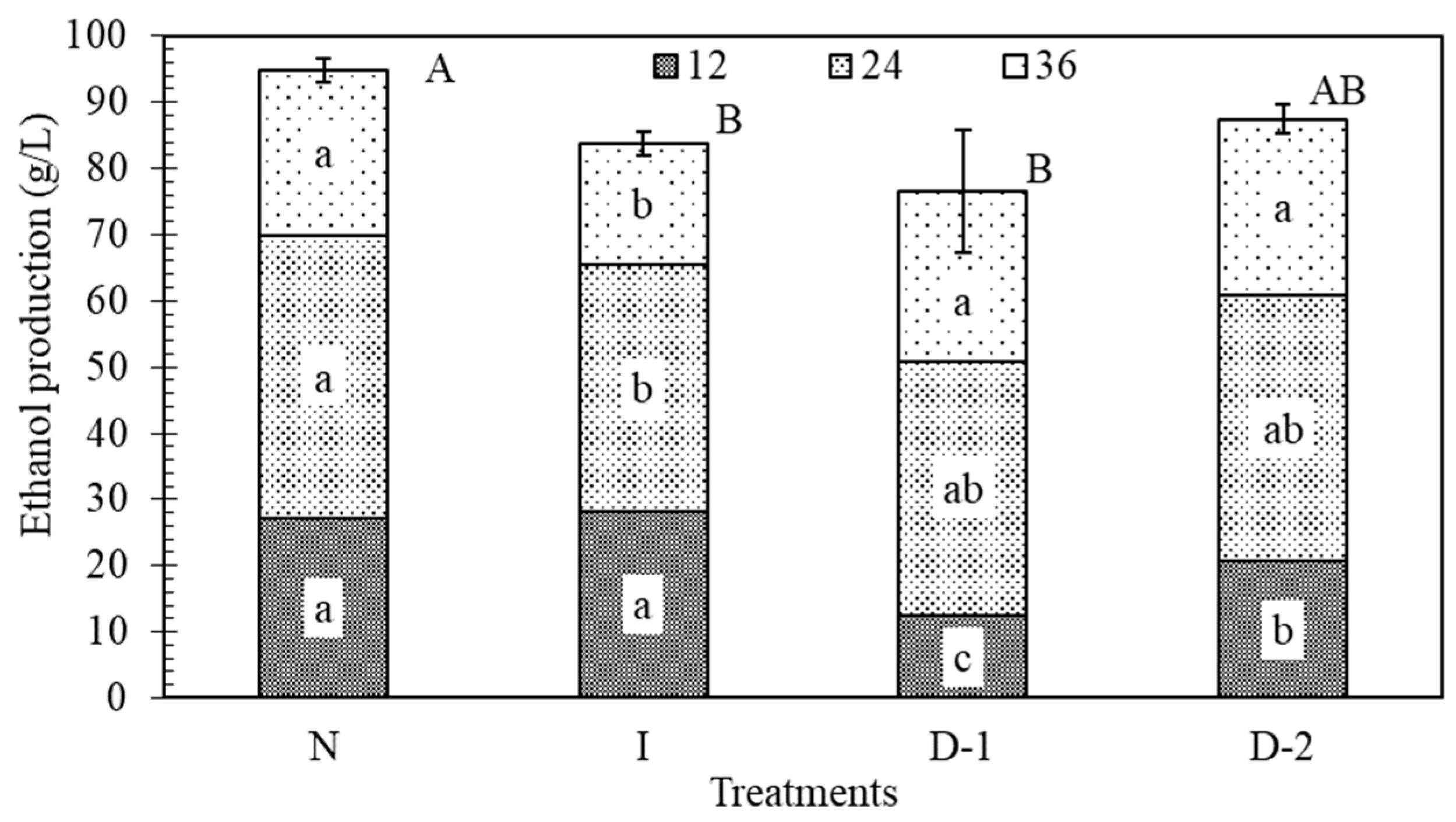
3.4. Ethanol Production
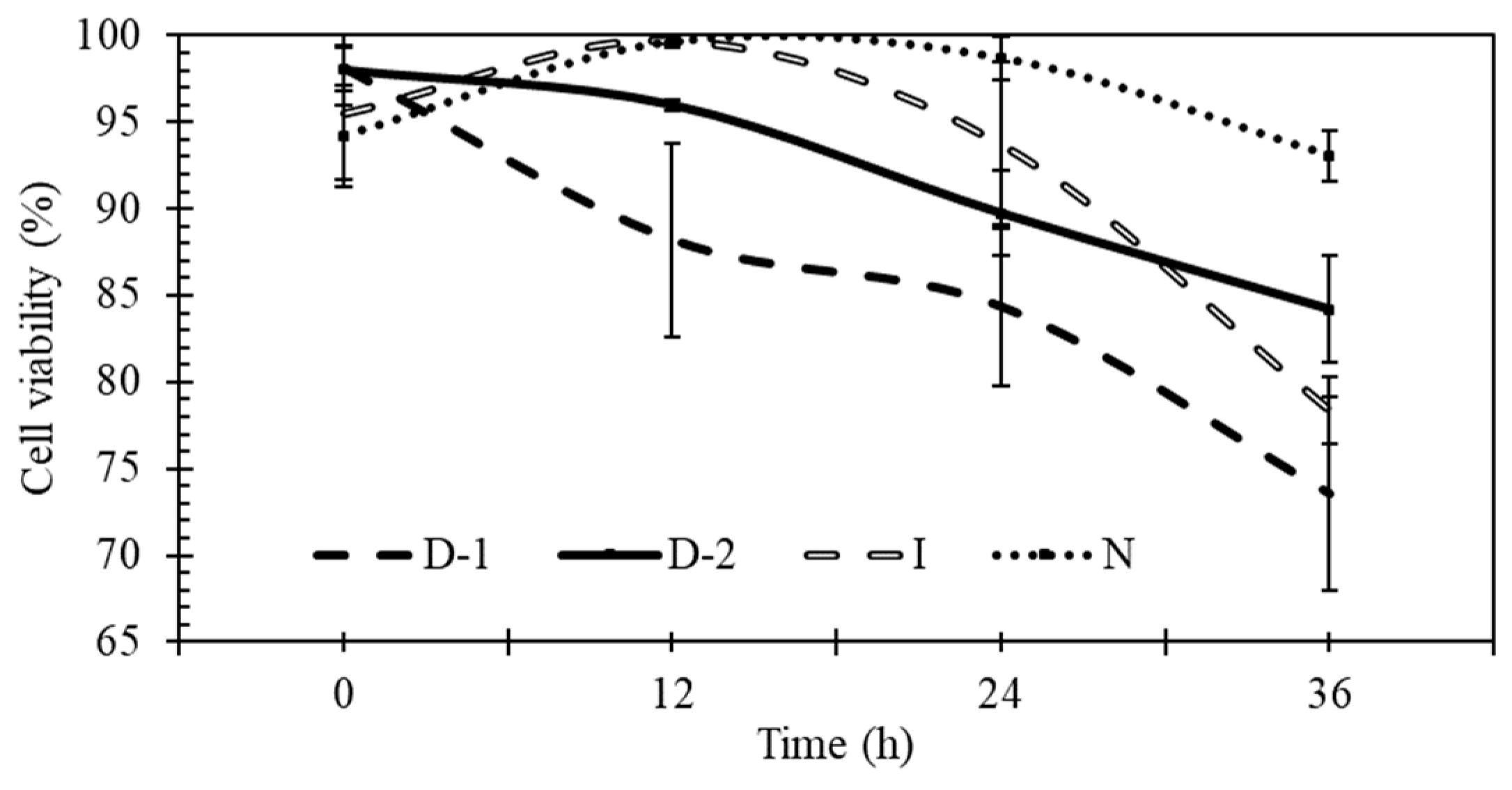
3.5. Yeast Viability
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Renewable Fuel Association World Fuel Ethanol Production. [Internet]. (2016). Available online: http://ethanolrfa.org/resources/industry/statistics/#1454098996479-8715d404-e546 (accessed on 27 December 2016).
- Aristidou, A.; Baghaei, N.; Javed, M.; Hartley, B. Conversion of Renewable Resources to Biofuel and Fine Chemicals: Current Trends and Future Prospects. In Fermentation Microbiology and Biotechnology; El-Mansi, E., Bryce, C., Dahhou, B., Sanchez, S., Demain, A., Allman, A., Eds.; CRC Press: New York, NY, USA, 2012; pp. 222–262. [Google Scholar]
- Fleet, G. The microorganisms of wine making—Isolation, enumeration and identification. In Wine Microbiology and Beotechnology; CRC Press: Boca Raton, FL, USA, 1993; pp. 1–25. [Google Scholar]
- Dequin, S. The potential of genetic engineering for improving brewing, wine-making and baking yeasts. Appl. Microbiol. Biotechnol. 2001, 56, 577–588. [Google Scholar] [CrossRef]
- Ramirez-cadavid, D.A.; Kozyuk, O.; Lyle, P.; Michel, F.C. Effects of hydrodynamic cavitation on dry mill corn ethanol production. Process Biochem. 2016, 51, 500–508. [Google Scholar] [CrossRef]
- Ramirez, D.; Kozyuk, O.; Michel, F. Improvement in commercial scale dry mill corn ethanol production using controlled flow cavitation and cellulose hydrolysis. Biomass Conver. Biorefinery 2013, 211–224. [Google Scholar]
- Dhanalakshmi, N.P.; Nagarajan, R. Ultrasonic Intensification of the Chemical Degradation of Methyl Violet: An experimental Study. Int. J. Chem. Mol. Nucl. Mater. Metallurg. Eng. 2011, 5, 537–542. [Google Scholar]
- Suslick, K.S.; Didenko, Y.; Fang, M.M.; Hyeon, T.; Kolbeck, K.J.; McNamara, W.B., III; Mdleleni, M.M.; Wong, M. Acoustic cavitation and its chemical consequences. Philos. Trans. R. Soc. A 1999, 357, 335–353. [Google Scholar] [CrossRef]
- Michel, F.; Kozyuk, O. Hydrodynamic Cavitation Processing. In Chemical Processes for a Sustainable Future; Letcher, T., Scott, J., Patterson, D., Eds.; The Royal Society of Chemistry: London, UK, 2015; pp. 84–142. [Google Scholar]
- Khanal, S.K.; Montalbo, M.; Van Leeuwen, J.H.; Srinivasan, G.; Grewell, D. Ultrasound Enhanced Glucose Release From Corn in Ethanol Plants. Biotechnol. Bioeng. 2007, 98, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Nitayavardhana, S.; Shrestha, P.; Rasmussen, M.L.; Lamsal, B.P.; van Leeuwen, J.; Khanal, S.K. Ultrasound improved ethanol fermentation from cassava chips in cassava-based ethanol plants. Bioresour. Technol. 2010, 101, 2741–2747. [Google Scholar] [CrossRef] [PubMed]
- Nikoli, S.; Mojovi, L.; Rakin, M.; Pejin, D.; Pejin, J. Ultrasound-assisted production of bioethanol by simultaneous saccharification and fermentation of corn meal. Food Chem. 2010, 122, 216–222. [Google Scholar] [CrossRef]
- Bussemaker, M.; Zhang, D. Effect of Ultrasound on Lignocellulosic Biomass as a Pretreatment for Biorefinery and Biofuel Applications. Ind. Eng. Chem. Res. 2013, 52, 3563–3580. [Google Scholar] [CrossRef]
- Sulaiman, A.Z.; Ajit, A.; Yunus, R.M.; Chisti, Y. Ultrasound-assisted fermentation enhances bioethanol productivity. Biochem. Eng. J. 2011, 54, 141–150. [Google Scholar] [CrossRef]
- Ramirez, D. Improvement of Ethanol Production on Dry-Mill Process Using Hydrodynamic Cavitation Pretreatment; The Ohio State University: Columbus, OH, USA, 2012. [Google Scholar]
- Feng, X.; Deng, J.; Lei, H.; Bai, T.; Fan, Q.; Li, Z. Dewaterability of waste activated sludge with ultrasound conditioning. Bioresour. Technol. 2009, 100, 1074–1081. [Google Scholar] [CrossRef]
- Entezari, M.; Petrier, C. A combination of ultrasound and oxidative enzyme: Sono-biodegradation of substituted phenols. Ultrasound Sonochem. 2003, 10, 241–246. [Google Scholar] [CrossRef]
- Khanal, S.K.; Grewell, D.; Sung, S.; Van Leeuwen, J. Ultrasound Applications in Wastewater Sludge Pretreatment: A Review. Crit. Rev. Environ. Sci. Technol. 2007, 37, 277–313. [Google Scholar] [CrossRef]
- Anderson, J.M. Effects of Ultrasonic Radiation on Growth and Fermentation in the Yeast, Saccharomyces cerevisiae. Biochim. Biophys. Acta 1953, 11, 122–137. [Google Scholar] [CrossRef]
- Radel, S.; McLoughlin, A.J.; Gherardini, L.; Doblhoff-Dier, O.; Benes, E. Viability of yeast cells in well controlled propagating and standing ultrasonic plane waves. Ultrasonics 2000, 38, 633–637. [Google Scholar] [CrossRef]
- Guerrero, S.; Tognon, M.; Alzamora, S.M. Response of Saccharomyces cerevisiae to the combined action of ultrasound and low weight chitosan. Food Control 2005, 16, 131–139. [Google Scholar] [CrossRef]
- Tsukamoto, I.; Constantinoiu, E.; Furuta, M.; Nishimura, R.; Maeda, Y. Inactivation effect of sonication and chlorination on Saccharomyces cerevisiae. Calorimetric Analysis. Ultrason. Sonochem. 2004, 11, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, I.; Yim, B.; Stavarache, C.E.; Furuta, M.; Hashiba, K.; Maeda, Y. Inactivation of Saccharomyces cerevisiae by ultrasonic irradiation. Ultrason. Sonochem. 2004, 11, 61–65. [Google Scholar] [CrossRef]
- Ma, Y.Q.; Chen, J.C.; Liu, D.H.; Ye, X.Q. Effect of ultrasonic treatment on the total phenolic and antioxidant activity of extracts from citrus peel. J. Food Sci. 2008, 73, T115–T120. [Google Scholar] [CrossRef]
- Liu, D.; Zeng, X.A.; Sun, D.W.; Han, Z. Disruption and protein release by ultrasonication of yeast cells. Innov. Food Sci. Emerg. Technol. 2013, 18, 132–137. [Google Scholar] [CrossRef]
- Iida, Y.; Tuziuti, T.; Yasui, K.; Kozuka, T.; Towata, A. Protein release from yeast cells as an evaluation method of physical effects in ultrasonic field. Ultrason. Sonochem. 2008, 15, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Mason, T.J.; Paniwnyk, L.; Lorimer, J.P. The uses of ultrasound in food technology. Ultrason. Sonochem. 1996, 3, S253–S260. [Google Scholar] [CrossRef]
- Vivek, K.; Subbarao, K.V.; Srivastava, B. Optimization of postharvest ultrasonic treatment of kiwifruit using RSM. Ultrason. Sonochem. 2016, 32, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Wood, B.E.; Aldrich, H.C.; Ingram, L.O. Ultrasound Stimulates Ethanol Production during the Simultaneous Saccharification and Fermentation of Mixed Waste Office Paper. Biotechnol. Prog. 1997, 13, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Chuanyun, D.; Bochu, W.; Chuanren, D.; Sakanishi, A. Low ultrasonic stimulates fermentation of riboflavin producing strain Ecemothecium ashbyii. Colloids Surf. B Biointerfaces 2003, 30, 37–41. [Google Scholar] [CrossRef]
- Matsuura, K.; Hirotsune, M.; Nunokawa, Y.; Satoh, M.; Honda, K. Acceleration of Cell Growth and Ester Formation by Ultrasonic Wave Irradiation. J. Ferment. Bioeng. 1994, 77, 36–40. [Google Scholar] [CrossRef]
- Schläfer, O.; Sievers, M.; Klotzbücher, H.; Onyeche, T.I. Improvement of biological activity by low energy ultrasound assisted bioreactors. Ultrasonics 2000, 38, 711–716. [Google Scholar] [CrossRef]
- Choi, E.J.; Ahn, H.; Kim, M.; Han, H.; Kim, W.J. Effect of ultrasonication on fermentation kinetics of beer using six-row barley cultivated in Korea. J. Inst. Brew. 2015, 121, 510–517. [Google Scholar] [CrossRef]
- Chisti, Y.; Moo-Young, M. Disruption of microbial cells for intracellular products. Enzym. Microb. Technol. 1986, 8, 194–204. [Google Scholar] [CrossRef]
- Neel, P.; Gedanken, A.; Schwarz, R.; Sendersky, E. Mild sonication accelerates ethanol production by yeast fermentation. Energy Fuels 2012, 26, 2352–2356. [Google Scholar] [CrossRef]
- Lanchun, S.; Bochu, W.; Zhiming, L.; Chuanren, D.; Chuanyun, D.; Sakanishi, A. The research into the influence of low-intensity ultrasonic on the growth of S. cerevisiaes. Colloids Surf. B Biointerfaces 2003, 30, 43–49. [Google Scholar] [CrossRef]
- Michel, F.C.; Grulke, E.A. A Kinetic Model for the Fungal Pellet Lifecycle. AIChE J. 1992, 38, 1449–1460. [Google Scholar] [CrossRef]
- Dowe, N.; McMillan, J. SSF Experimental Protocols—Lignocellulosic Biomass Hydrolysis and Fermentation; Laboratory Analytical Procedure: Washington, DC, USA, 2008.
- Montalbo-lomboy, M.T. Ultrasonic Pretreatment for Enhanced Saccharification and Fermentation of Ethanol Production from Corn; Iowa State University: Ames, IA, USA, 2008. [Google Scholar]
- Doran, P.M. Heterogeneous Reactions. In Bioprocess Engineering Principles; All Rights Reserved; Elsevier Ltd.: Amsterdam, The Netherlands, 2013; ISBN 9780471186588. [Google Scholar]
- Kraakman, N.J.R.; Rocha-Rios, J.; Van Loosdrecht, M.C.M. Review of mass transfer aspects for biological gas treatment. Appl. Microbiol. Biotechnol. 2011, 91, 873–886. [Google Scholar] [CrossRef] [PubMed]
- Gutenwik, J.; Nilsson, B.; Axelsson, A. Mass transfer effects on the reaction rate for heterogeneously distributed immobilized yeast cells. Biotechnol. Bioeng. 2002, 79, 664–673. [Google Scholar] [CrossRef] [PubMed]
- Chaix, E.; Guillaume, C.; Guillard, V. Oxygen and Carbon Dioxide Solubility and Diffusivity in Solid Food Matrices: A Review of Past and Current Knowledge. Compr. Rev. Food Sci. Food Saf. 2014, 13, 261–286. [Google Scholar] [CrossRef]
- Bang, J.H.; Suslick, K.S. Applications of ultrasound to the synthesis of nanostructured materials. Adv. Mater. 2010, 22, 1039–1059. [Google Scholar] [CrossRef] [PubMed]
- Moulin, G.; Boze, H.; Galzy, P. Inhibition of Alcoholic Fermentation. Biotechnol. Genet. Eng. 1984, 2, 365–382. [Google Scholar] [CrossRef]
- Maiorella, B.; Blanch, H.W.; Wilke, C.R. By-product inhibition effects on ethanolic fermentation by Saccharomyces cerevisiae. Biotechnol. Bioeng. 1983, 25, 103–121. [Google Scholar] [CrossRef]
- Lanchun, S.; Bochu, W.; Liancai, Z.; Jie, L.; Yanhong, Y.; Chuanren, D. The influence of low-intensity ultrasonic on some physiological characteristics of Saccharomyces cerevisiae. Colloids Surf. B Biointerfaces 2003, 30, 61–66. [Google Scholar] [CrossRef]
- Rosa, M.F.; Sá-Correia, I. Intracellular acidification does not account for inhibition of Saccharomyces cerevisiae growth in the presence of ethanol. FEMS Microbiol. Lett. 1996, 135, 271–274. [Google Scholar] [CrossRef]
- Lucero, P.; Penalver, E.; Moreno, E.; Lagunas, R. Internal trehalose protects endocytosis from inhibition by ethanol in Saccharomyces cerevisiae. Appl. Environ. Microbiol. 2000, 66, 4456–4461. [Google Scholar] [CrossRef] [PubMed]
- Crowe, L.M.; Mouradian, R.; Crowe, J.H.; Jackson, S.A.; Womersley, C. Effects of carbohydrates on membrane stability at low water activities. Biochim. Biophys. Acta BBA Biomembr. 1984, 769, 141–150. [Google Scholar] [CrossRef]
- Liu, Z.; Li, X. The kinetics of ethanol fermentation based on adsorption processes. Kem. Ind. J. Chem. Chem. Eng. 2014, 63, 259–264. [Google Scholar]
- Doblhoff-Dier, O.; Gaida, T.; Katinger, H.; Burger, W.; Gröschl, M.; Benes, E. A novel ultrasonic resonance field device for the retention of animal cells. Biotechnol. Prog. 1994, 10, 428–432. [Google Scholar] [CrossRef] [PubMed]

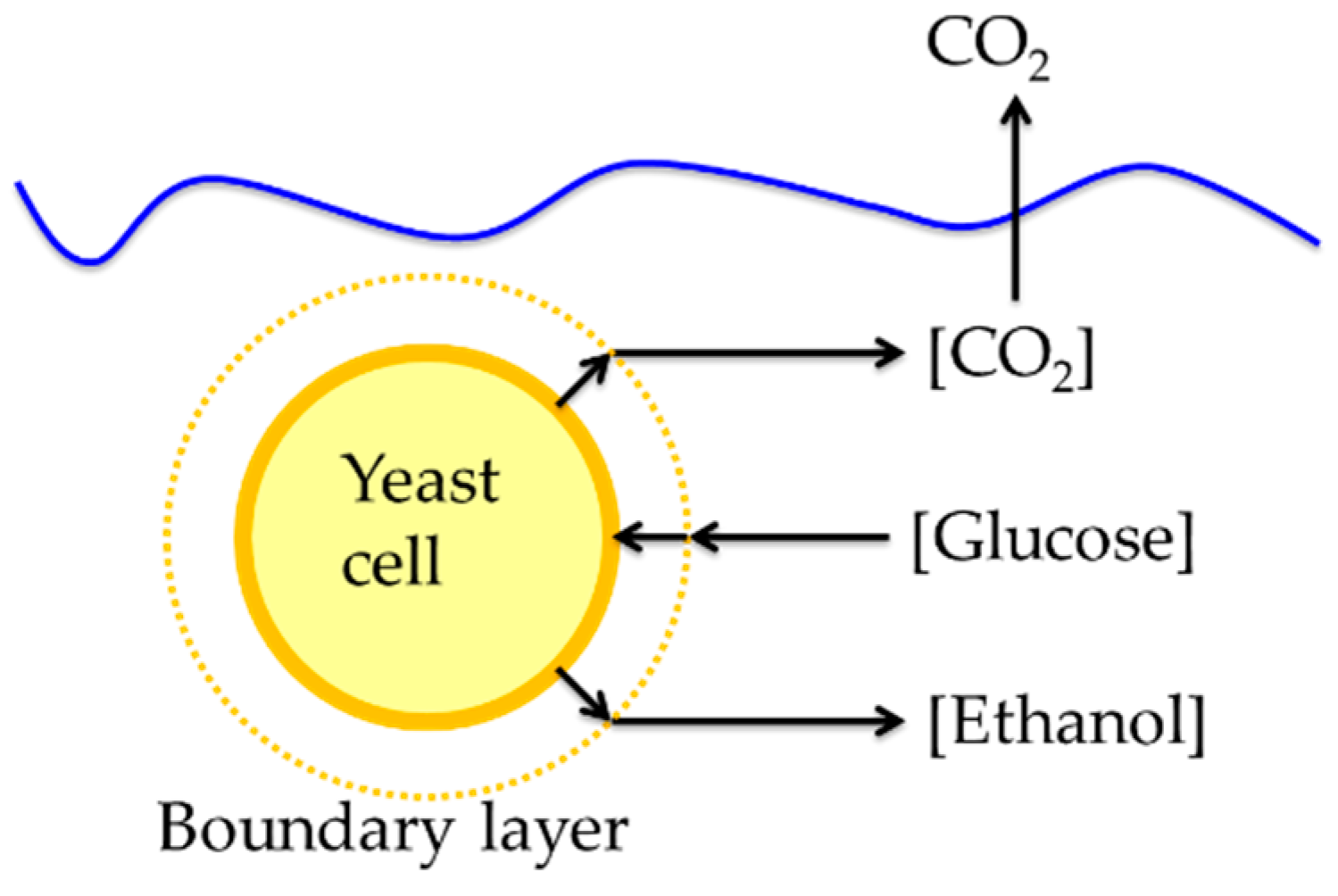
| Yeast Radii (µm) | Compounds | Diffusivity (m2/s) | Concentrations (g/L) | Rates (g/L)/12 h |
|---|---|---|---|---|
| 1 and 5 | Glucose | 7.3 × 10−10 | 200 and 20 | 25 and 75 |
| Ethanol | 1.4 × 10−9 | 10 and 100 | 15 and 50 | |
| CO2 | 2.1 × 10−9 | 1.6 × 10−2 M | 15 and 50 |
| External Mass Transfer Modulus, Ω | ||||
| Yeast Radius (m) | ||||
| 1.50 × 10−6 | 5.00 × 10−6 | |||
| Glucose (g/m3) | Rate ((g/L)/12 h) | |||
| 25 | 75 | 25 | 75 | |
| 200,000 | 3.33 × 10−9 | 9.99 × 10−9 | 1.11 × 10−8 | 3.33 × 10−8 |
| 20,000 | 3.33 × 10−8 | 9.99 × 10−8 | 1.11 × 10−7 | 3.33 × 10−7 |
| Ethanol (g/m3) | Rate ((g/L)/12 h) | |||
| 15 | 50 | 15 | 50 | |
| 10,000 | 2.01 × 10−8 | 6.69 × 10−8 | 6.69 × 10−8 | 2.23 × 10−7 |
| 100,000 | 2.01 × 10−9 | 6.69 × 10−9 | 6.69 × 10−9 | 2.23 × 10−8 |
| CO2 (g/m3) | Rate ((g/L)/12 h) | |||
| 15 | 50 | 15 | 50 | |
| 704 | 1.87 × 10−7 | 6.22 × 10−7 | 6.22 × 10−7 | 2.07 × 10−6 |
| Intraparticle Thiele Observable Modulus, Φ | ||||
| Yeast Radius (m) | ||||
| 1.50 × 10−6 | 5.00 × 10−6 | |||
| Glucose (g/m3) | Rate ((g/L)/12 h) | |||
| 25 | 75 | 25 | 75 | |
| 200,000 | 1.03 ×10−9 | 3.09 × 10−9 | 1.14 × 10−8 | 3.43 × 10−8 |
| 20,000 | 1.03 × 10−8 | 3.09 × 10−8 | 1.14 × 10−7 | 3.43 × 10−7 |
| Ethanol (g/m3) | Rate ((g/L)/12 h) | |||
| 15 | 50 | 15 | 50 | |
| 10,000 | 6.17 × 10−9 | 2.06 × 10−8 | 6.85 × 10−8 | 2.28 × 10−7 |
| 100,000 | 6.17 × 10−10 | 2.06 × 10−9 | 6.85 × 10−9 | 2.28 × 10−8 |
| CO2 (g/m3) | Rate ((g/L)/12 h) | |||
| 15 | 50 | 15 | 50 | |
| 704 | 5.76 × 10−8 | 1.92 × 10−7 | 6.40 × 10−7 | 2.13 × 10−6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huezo, L.; Shah, A.; Michel Jr., F.C. Effects of Ultrasound on Fermentation of Glucose to Ethanol by Saccharomyces cerevisiae. Fermentation 2019, 5, 16. https://doi.org/10.3390/fermentation5010016
Huezo L, Shah A, Michel Jr. FC. Effects of Ultrasound on Fermentation of Glucose to Ethanol by Saccharomyces cerevisiae. Fermentation. 2019; 5(1):16. https://doi.org/10.3390/fermentation5010016
Chicago/Turabian StyleHuezo, Luis, Ajay Shah, and Frederick C. Michel Jr. 2019. "Effects of Ultrasound on Fermentation of Glucose to Ethanol by Saccharomyces cerevisiae" Fermentation 5, no. 1: 16. https://doi.org/10.3390/fermentation5010016
APA StyleHuezo, L., Shah, A., & Michel Jr., F. C. (2019). Effects of Ultrasound on Fermentation of Glucose to Ethanol by Saccharomyces cerevisiae. Fermentation, 5(1), 16. https://doi.org/10.3390/fermentation5010016





