Intensive Production of Carboxylic Acids Using C. butyricum in a Membrane Bioreactor (MBR)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Chemicals
2.1.2. Inoculum Source
2.2. Methods
2.2.1. Experimental
Preservation of Microorganism
Inoculum Preparation
Measurement of Cellular Growth and Biomass
Analysis of End Products Using Gas Chromatography
Carbohydrate Consumption Rate Determination
Purity of Cultures
Continuously Stirred Tank Reactor (CSTR) Unit Design
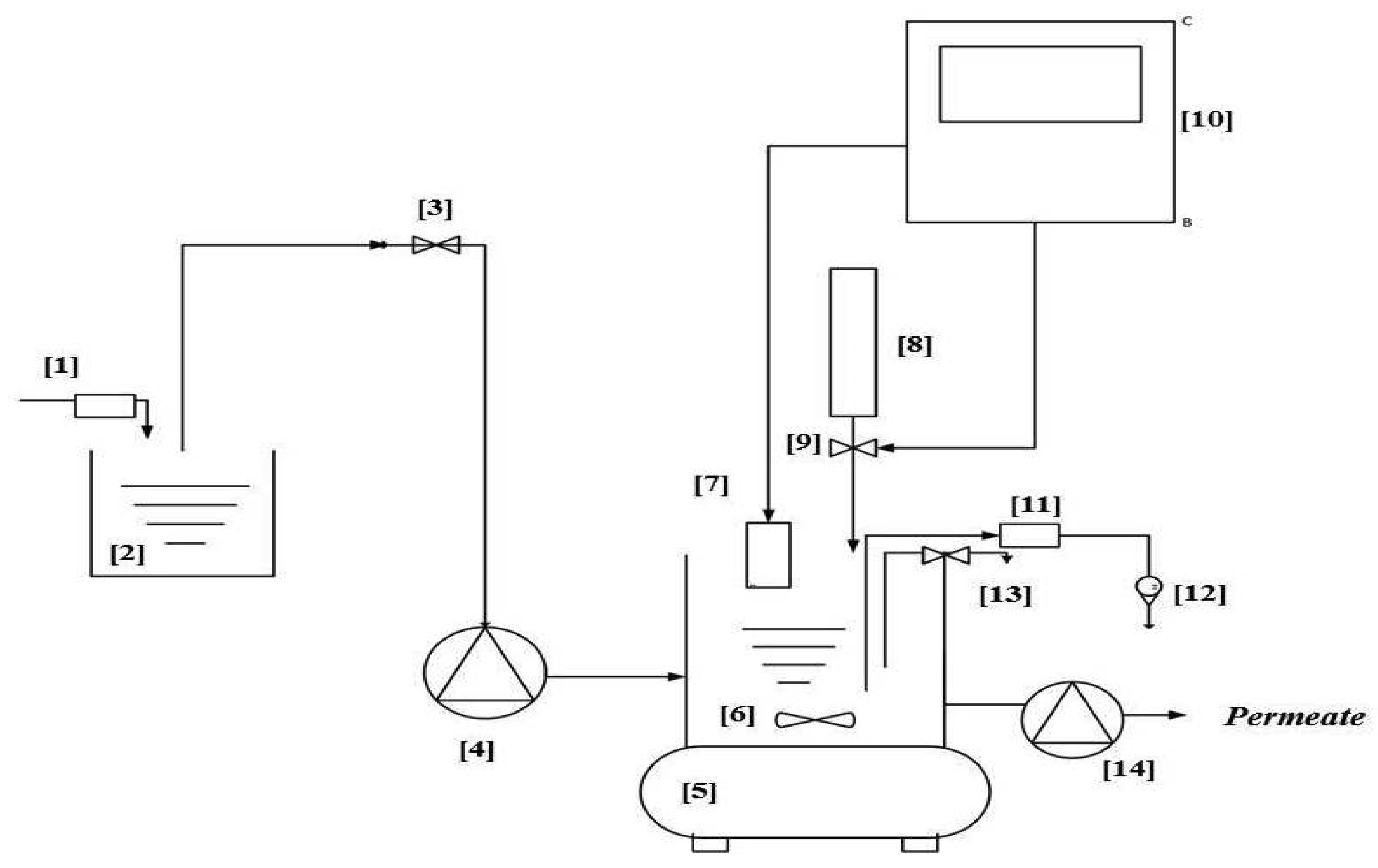
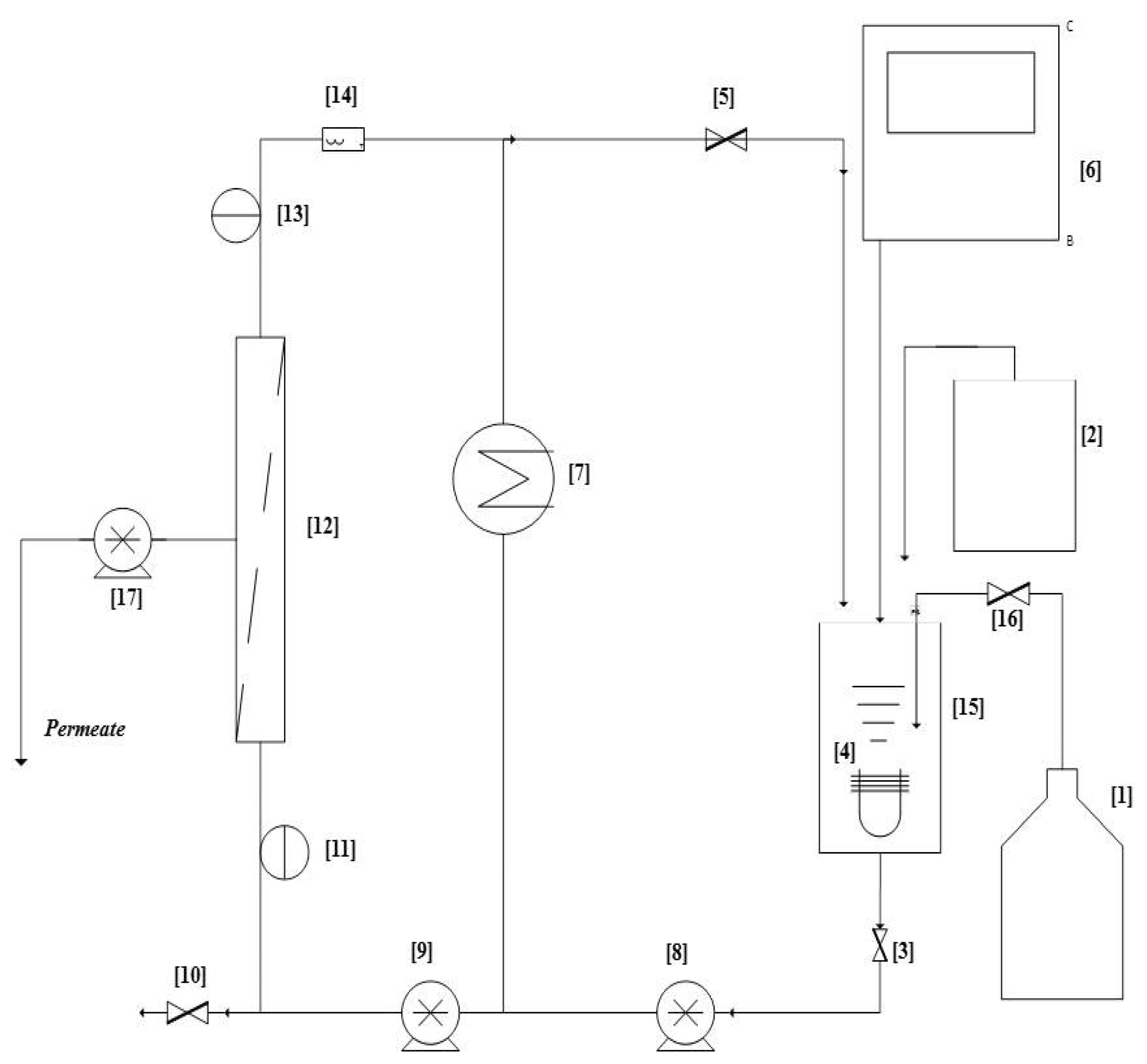
Membrane Bioreactor (MBR) Unit Design
2.2.2. Statistical Analysis
3. Theoretical
Determination of Kinetic Parameters
4. Cost Estimation
4.1. Process Description
4.2. Design and Cost of the Units
4.3. General Economic Parameters
5. Results and Discussion
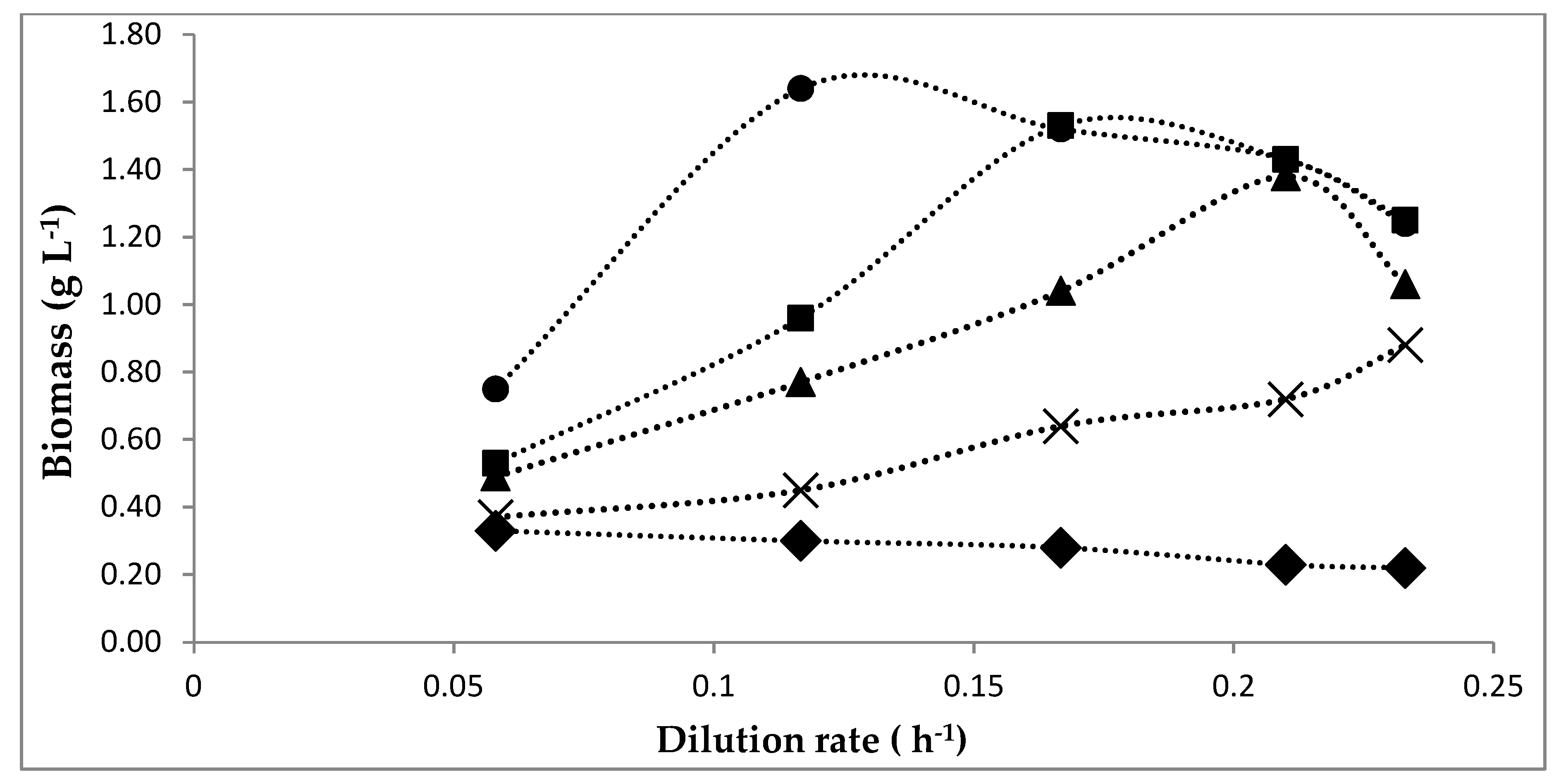
5.1. Growth on a CSTR on Continuous Mode
5.2. Product Formation on a CSTR on Continuous Mode
5.3. Growth on a Membrane Bioreactor (MBR) on Fed-Batch Mode with Varying Permeation Rates
5.4. Product Formation on a Membrane Bioreactor (MBR) on Fed-Batch Mode with Varying Permeation Rates
5.5. Cost Estimation
6. Conclusions
- MBR is the most productive system in terms of cellular biomass and carboxylic acid of the 2 (MBR, continuous reactor).
- MBR is a truly beneficial system for intensive microbial culturing due to the ability to work around different modes and culture various microorganisms.
- Depending on culturing conditions and nutrient composition, different acid productivities were observed on the 2 systems.
- Depending on culturing conditions/nutrient composition, different acid ratios were observed, but overall acetic acid productions was favored.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- DECC (Department of Energy and Climate Change Website). Increasing the Use of Low Carbon Technologies. 2014. Available online: https://www.gov.uk/government/policies/increasing-the-use-of-low-carbon-technologies (accessed on 8 August 2018).
- Zacharof, M.-P.; Lovitt, R.W. Complex effluent streams as a potential source of volatile fatty acids. Waste Biomass Valor. 2013, 4, 4557–4581. [Google Scholar] [CrossRef]
- Elliott, D.C.; Orth, R.; Gao, J.; Werpy, T.; Eakin, D.; Schmidt, A.; Neuenschwander, G.; Murry, J.; Flagg, A.; Lahman, L. Biorefinery concept development based on wheat flour milling. Fuel Chem. Div. Prepr. 2002, 47, 361–362. [Google Scholar]
- Rostkowski, K.H.; Criddle, C.S.; Lepech, M.D. Cradle-to-gate life cycle assessment for a cradle-to-cradle cycle: Biogas-to-bioplastic (and back). Environ. Sci. Technol. 2012, 46, 9822–9829. [Google Scholar] [CrossRef] [PubMed]
- Zacharof, M.-P. Grape winery waste as feedstock for bioconversions: Applying the biorefinery concept. Waste Biomass Valor. 2016, 8, 1011–1025. [Google Scholar] [CrossRef]
- Bioref-Integ Project. 2008. Available online: http://www.bioref-integ.eu (accessed on 8 August 2018).
- Yang, S.T.; Yu, M.; Chang, W.L.; Tang, I.C. Anaerobic Fermentations for the Production of Acetic and Butyric Acids. In Bioprocessing Technologies in Biorefinery for Sustainable Production of Fuels, Chemicals, and Polymers; Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Chen, S.D.; Lee, K.S.; Lo, Y.C.; Chen, W.M.; Wu, J.F.; Lin, C.Y.; Chang, J.S. Batch and continuous biohydrogen production from starch hydrolysate by clostridium species. Int. J. Hydrogen Energy 2008, 33, 1803–1812. [Google Scholar] [CrossRef]
- Guan, W.; Shi, S.; Tu, M.; Lee, Y.Y. Acetone–butanol–ethanol production from Kraft paper mill sludge by simultaneous saccharification and fermentation. Bioresour. Technol. 2016, 200, 713–721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Credence Research, Market Research Reports. Available online: https://www.credenceresearch.com/press/global-butyric-acid-market (accessed on 8 August 2018).
- Global Market, Insights to Innovation. Available online: https://www.gminsights.com/industry-analysis/acetic-acid-market (accessed on 8 August 2018).
- Murali, N.; Srinivas, K.; Ahring, B.K. Biochemical Production and Separation of Carboxylic Acids for Biorefinery Applications. Fermentation 2017, 3, 22. [Google Scholar] [CrossRef]
- Cision PR Newswire. Available online: https://www.prnewswire.com/news-releases/butyric-acid-market-by-application-by-type-by-geography---global-analysis-and-forecast-to-2020-for-the-289-million-industry-300142162.html (accessed on 8 August 2018).
- Yoki, H.; Maki, R.; Hirose, J.; Hayashi, S. Microbial production of hydrogen from starch-manufacturing wastes. Biomass Bioenergy 2002, 22, 389–395. [Google Scholar] [CrossRef]
- Yoki, H.; Mori, S.; Hirose, J.; Hayashi, S.; Takasoki, Y. H2 production from starch by a mixed culture of Clostridium butyricum and rhodobacter spp. M-19. Biotechnol. Lett. 1998, 20, 895–899. [Google Scholar] [CrossRef]
- Miyake, J.; Masato, M.; Yasuo, A. Biotechnological hydrogen production: Research for efficient light energy conversion. J. Biotechnol. 1999, 70, 89–101. [Google Scholar] [CrossRef]
- Abbad-Andaloussi, S.; Amine, J.; Gerard, P.; Petitdemange, H. Effect of glucose on glycerol metabolism by Clostridium butyricum DSM 5431. J. Appl. Microbiol. 1998, 84, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jin, B. Process optimization of biological hydrogen production from molasses by a newly isolated Clostridium butyricum. J. Biosci. Bioeng. 2009, 107, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Jung, I.S.; Lovitt, R.W. A comparative study of the growth of lactic acid bacteria in a pilot scale membrane bioreactor. J. Chem. Technol. Biotechnol. 2010, 85, 1250–1259. [Google Scholar] [CrossRef]
- Jung, I.S.; Lovitt, R.W. A comparative study of an intensive malolactic transformation of cider using Lactobacillus brevis and Oenococcus oeni in a membrane bioreactor. J. Ind. Microbiol. Biotechnol. 2010, 37, 727–740. [Google Scholar] [CrossRef] [PubMed]
- Tajaruddin, H.A.; Lovitt, R.W.; Nur, K.T.; Azwa, N.M.B. A Study of Fatty Acid Production in the Batch Reactor via the Carbohydrate Fermentation by C. butyricum. Int. J. Biol. Biomol. Agric. Food Biotechnol. Eng. 2013, 7, 828–833. [Google Scholar]
- Lovitt, R.; Jung, I.; Jones, M. The performance of a membrane bioreactor for the malolactic fermentation of media containing ethanol. Desalination 2006, 199, 24–28. [Google Scholar] [CrossRef]
- Zacharof, M.P.; Lovitt, R.W. Modelling and simulation of cell growth dynamics, substrate consumption, and lactic acid production kinetics of Lactococcus lactis. Biotechnol. Bioprocess Eng. 2013, 18, 52–64. [Google Scholar] [CrossRef]
- Kirsop, B.E.; Siell, J.J.S. Maintenance of Microorganisms: A Manual of Laboratory Methods, 1st ed.; Academic Press Ltd.: Cambridge, MA, USA, 1984. [Google Scholar]
- Demain, L.A.; Davies, J.E. Manual of Industrial Microbiology and Biotechnology; ASM Press: Washington, DC, USA, 1999; pp. 29–45, 49–60, 49–60, 61–75, 80–93, 183–147, 151–163, 165–179. [Google Scholar]
- Willis, P.A. Anaerobic Bacteriology, Clinical and Laboratory Practise, 3rd ed.; Butterworth and Co.: London, UK, 1977. [Google Scholar]
- Gerhard, G. Bacterial Metabolism, 1st ed.; Springer: New York, NY, USA, 1979. [Google Scholar]
- Bu’lock, J.D.; Kristiansen, B. Basic Biotechnology; Academic Press: New York, NY, USA; London, UK, 1978; pp. 75–132, 133–172, 371–374. [Google Scholar]
- McKane, L.; Kandel, J. Microbiology Essentials and Applications, 1st ed.; McGraw Hill Inc.: New York, NY, USA, 1996. [Google Scholar]
- Malek, I.; Beran, K.; Hospodka, J. Continuous Cultivation of Microorganisms, 1st ed.; Academic Press Ltd.: Cambridge, MA, USA, 1969. [Google Scholar]
- Gruger, A.; Gruger, W. Biotechnology: A Textbook of Industrial Microbiology, 1st ed.; Mass Sinauer Associates: Sunderland, MA, USA, 1989. [Google Scholar]
- Gerrard, A.M. Guide to Capital Cost Estimating; Institution of Chemical Engineers (IChemE): Rugby, UK, 2000. [Google Scholar]
- Towler, G.; Sinnott, R. Chemical Engineering Design Principles, Practice and Economics of Plant and Process Design, 2nd ed.; Elsevier: Oxford, UK, 2013. [Google Scholar]
- TAMI Industries INSIDE CeRAM. Available online: http://www.tami-industries.com/INSIDECeRAMTM.109þM54a708de802.0.html (accessed on 8 August 2018).
- Pall Corporation Membralox® Ceramic Membrane Products. Available online: http://www.pall.com/main/food-and-beverage/product.page?id¼41052 (accessed on 8 August 2018).
- Atkinson, B.; Mavituna, F. Biochemical Engineering and Biotechnology Handbook, 2nd ed.; Stockton Pr: New York, NY, USA, 1991. [Google Scholar]
- Zigová, J.; Sturdik, E. Advances in biotechnological production of butyric acid. J. Ind. Microbiol. Biotechnol. 2000, 24, 153–160. [Google Scholar] [CrossRef]
- Antoni, D.; Zverlov, V.V.; Schwarz, W.H. Biofuels from microbes. Appl. Microbiol. Biotechnol. 2007, 77, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Zacharof, M.-P.; Vouzelaud, C.; Mandale, S.J.; Lovitt, R.W. Valorization of spent anaerobic digester effulents through production of platform chemicals using C. butyricum. Biomass Bioenergy 2015, 81, 294–303. [Google Scholar] [CrossRef]
- Yokoi, H.; Tokushige, T.; Hirose, J.; Hayashi, S.; Takasaki, Y. H2 production from starch by a mixed culture of Clostridium butyricum and Enterobacter aerogenes. Biotechnol. Lett. 1998, 20, 143–147. [Google Scholar] [CrossRef]
- Fröstl, J.M.; Seifritz, C.; Drake, H.L. Effect of nitrate on the autotrophic metabolism of the acetogens Clostridium thermoautotrophicum and Clostridium thermoaceticum. J. Bacteriol. 1996, 178, 4597–4603. [Google Scholar] [CrossRef] [PubMed]
- González-Pajuelo, M.; Meynial-Salles, I.; Mendes, F.; Andrade, J.C.; Vasconcelos, I.; Soucaille, P. Metabolic engineering of Clostridium acetobutylicum for the industrial production of 1,3-propanediol from glycerol. Metab. Eng. 2005, 7, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Canganella, F.; Kuk, S.-U.; Morgan, H.; Wiegel, J. Clostridium thermobutyricum: Growth studies and stimulation of butyrate formation by acetate supplementation. Microbiol. Res. 2002, 157, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Biebl, H. Fermentation of glycerol by Clostridium pasteurianum—Batch and continuous culture studies. J. Ind. Technol. Biotechnol. 2001, 27, 18–26. [Google Scholar] [CrossRef]
- Milic, T.V.; Rakin, M.; Siler-Marinkovic, S. Utilization of baker’s yeast (Saccharomyces cerevisiae) for the production of yeast extract: Effects of different enzymatic treatments on solid, protein and carbohydrate recovery. J. Serb. Chem. Soc. 2007, 72, 451–457. [Google Scholar] [CrossRef]
- Gomez-Pastor, R.; Perez-Torrado, R.; Garre, E.; Matallana, E. Recent advances in yeast biomass production. In Biomass Detection, Production and Usage; Matovic, D., Ed.; InTech: London, UK, 2011. [Google Scholar]
- Cote, P.; Buisson, H.; Pound, C.; Arakaki, G. Immersed membrane activated sludge for the reuse of municipal wastewater. Desalination 1997, 113, 189–196. [Google Scholar] [CrossRef]
- Buisson, H.; Cote, P.; Praderie, M.; Paillard, H. The use of immersed membranes for upgrading wastewater treatment plants. Water Sci. Technol. 1998, 37, 89–95. [Google Scholar] [CrossRef]
- Wisniewski, C.; Cruz, A.L.; Grasmick, A. Kinetics of organic carbon removal by a mixed culture in a membrane bioreactor. Biochem. Eng. J. 1999, 3, 61–69. [Google Scholar] [CrossRef]
- Du, J.; McGraw, A.; Lorenz, N.; Beitle, R.R.; Clausen, E.C.; Hestekin, J.A. Continuous fermentation of Clostridium tyrobutyricum with partial cell recycle as a long-term strategy for butyric acid production. Energies 2012, 5, 2835–2848. [Google Scholar] [CrossRef]
- LabM Limited Price Catalogue. Available online: http://www.labm.com/ (accessed on 8 August 2018).
- Neogen Corporation. Acumedia Manufacturing. Available online: http://www.neogen.com/Acumedia/ (accessed on 8 August 2018).
- Chegini, S. Removal of Potential Inhibitors from Hemicellulose Hydrolysate by Membrane Filtration. Master’s Thesis, École Polytechnique de Montréal, Montreal, QC, Canada, 2014. Available online: https://publications.polymtl.ca/1495/ (accessed on 8 August 2018).
- Choi, J.-I.; Lee, S.Y. Process analysis and economic evaluation for poly (3-hydroxybutyrate) production by fermentation. Bioprocess Eng. 1997, 17, 335–342. [Google Scholar] [CrossRef]
- Van Wegen, R.J.; Ling, Y.; Middelberg, A.P.J. Industrial production of polydroxyalkanoates using Escherichia coli: An economic analysis. Trans. IChemE 1998, 76, 417–426. [Google Scholar] [CrossRef]
- Naranjo, J.M.; Posada, J.A.; Higuita, J.C.; Cardona, C.A. Valorization of glycerol through the production of biopolymers: The PHB case using Bacillus megaterium. Bioresour. Technol. 2013, 133, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Efremenko, E.N.; Nikolskaya, A.B.; Lyagin, I.V.; Senko, O.V.; Makhlis, T.A.; Stepanov, N.A.; Maslova, O.V.; Mamedova, F.; Varfolomeev, S.D. Production of biofuels from pretreated microalgae biomass by anaerobic fermentation with immobilized Clostridium acetobutylicum cells. Bioresour. Technol. 2012, 114, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Petitdemange, H.; Cherrier, C.; Bengone, J.M.; Bray, R. Etude des activites NADH et NADPH-ferredoxine oxydoreductasiques chez Clostridium acetobutylicum. Can. J. Microbiol. 1977, 23, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Karjalainen, P.P. Bin the Carbon Footprint of the Finnish Beverage Industry for Years 2000–2012 as Calculated with CCaLC. Master’s Thesis, University of Helsinki, Helsinki, Finland, 2013. [Google Scholar]
- Dwidar, M.; Park, J.-Y.; Mitchell, R.J.; Sang, B.-I. The Future of Butyric Acid in Industry. Sci. World J. 2012, 2012, 471417. [Google Scholar] [CrossRef] [PubMed]
- Vandak, D.; Zigová, J.; Šturdık, E.; Schlosser, S. Evaluation of solvent and pH for extractive fermentation of butyric acid. Process Biochem. 1997, 32, 245–251. [Google Scholar] [CrossRef]
- Melford Chemical and Biochemical Manufacturing Price Catalogue. Available online: http://melford.co.uk/ (accessed on 8 August 2018).
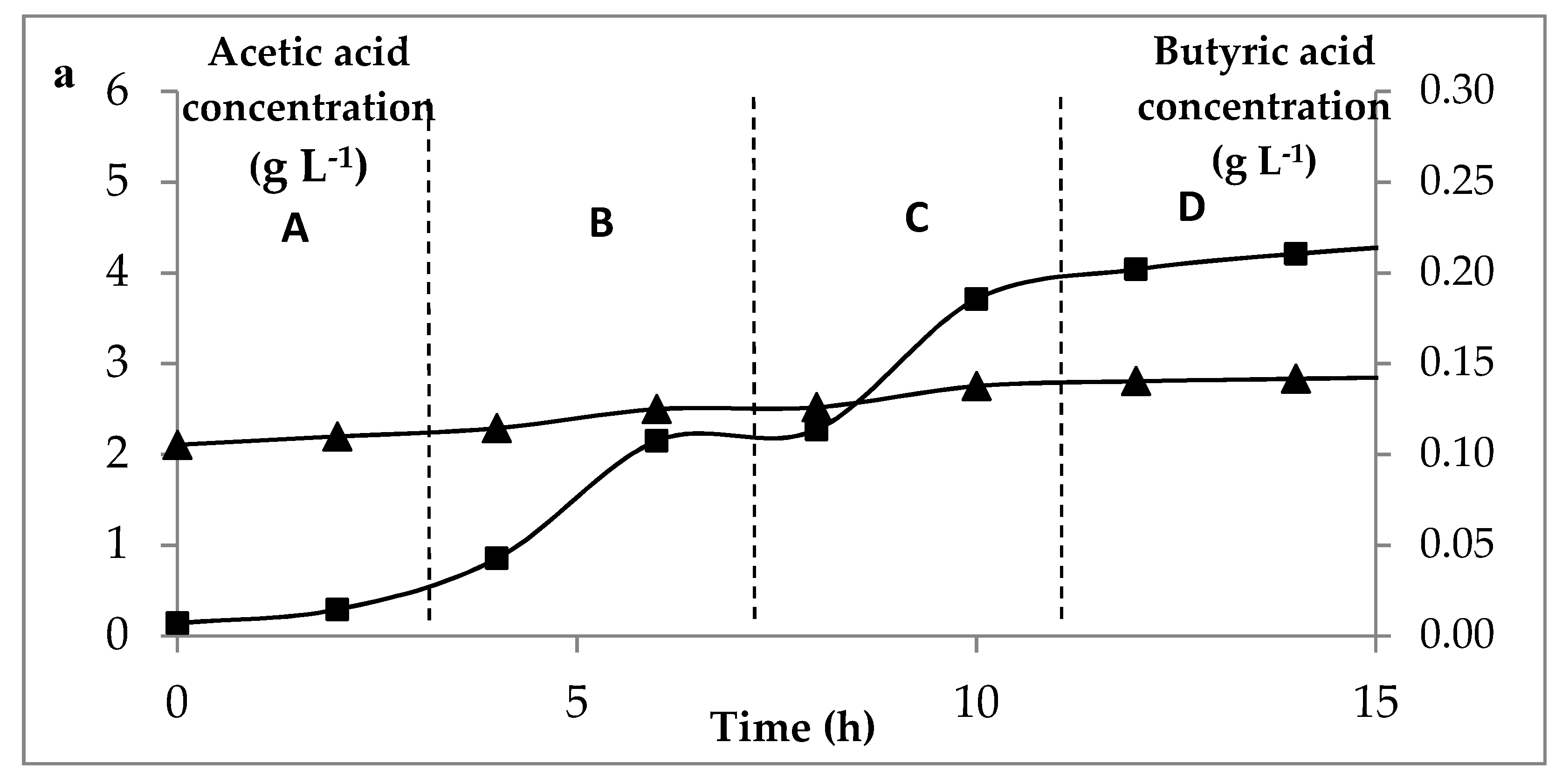
 ) of C. butyricum on glucose media at three concentrations of glucose. (a) for 5 g L−1 feed, (b) 10 g L−1 feed and (c) 15 g L−1, at permeation rates: A: 4 L h−1, B: 8 L h−1, C: 16 L h−1, D: 32 L.
) of C. butyricum on glucose media at three concentrations of glucose. (a) for 5 g L−1 feed, (b) 10 g L−1 feed and (c) 15 g L−1, at permeation rates: A: 4 L h−1, B: 8 L h−1, C: 16 L h−1, D: 32 L.
 ) of C. butyricum on glucose media at three concentrations of glucose. (a) for 5 g L−1 feed, (b) 10 g L−1 feed and (c) 15 g L−1, at permeation rates: A: 4 L h−1, B: 8 L h−1, C: 16 L h−1, D: 32 L.
) of C. butyricum on glucose media at three concentrations of glucose. (a) for 5 g L−1 feed, (b) 10 g L−1 feed and (c) 15 g L−1, at permeation rates: A: 4 L h−1, B: 8 L h−1, C: 16 L h−1, D: 32 L.


| Initial Glucose Concentration (Si, g L−1) | Dilution Rate (D, h−1) | Glucose Spent (ΔS, g L−1) | Biomass (X, g L−1) | Carboxylic Acids Concentration (g L−1) | Carboxylic Acids Volumetric Productivity (g−1 L−1 h−1) | Carboxylic Acids Specific Cell Productivity (g−1 g−1 h−1) | Biomass Yield (Yx/s) | Carboxylic Acid Yield (Yp/s) | Total Carbon Recovery (%) | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetic Acid | Butyric Acid | Acetic Acid | Butyric Acid | Acetic Acid | Butyric Acid | |||||||
| 5 | 0.058 | 1.4 | 0.34 | 3.49 | 0.18 | 0.2 | 0.01 | 0.59 | 0.03 | 0.24 | 0.73 | 33.70 |
| 0.117 | 1.2 | 0.3 | 3.13 | 0.18 | 0.37 | 0.02 | 1.23 | 0.07 | 0.25 | 0.66 | 35.32 | |
| 0.167 | 1.1 | 0.29 | 2.83 | 0.18 | 0.47 | 0.03 | 1.62 | 0.10 | 0.26 | 0.60 | 36.87 | |
| 0.21 | 1.0 | 0.23 | 2.71 | 0.18 | 0.57 | 0.04 | 2.48 | 0.17 | 0.23 | 0.58 | 40.11 | |
| 0.23 | 0.8 | 0.22 | 2.65 | 0.09 | 0.61 | 0.02 | 2.77 | 0.09 | 0.28 | 0.55 | 43.74 | |
| 10 | 0.058 | 5.1 | 0.48 | 5.87 | 2.05 | 0.34 | 0.12 | 0.71 | 0.25 | 0.09 | 0.79 | 77.16 |
| 0.117 | 5.58 | 0.56 | 6.02 | 2.62 | 0.7 | 0.31 | 1.25 | 0.55 | 0.1 | 0.86 | 83.45 | |
| 0.167 | 7.19 | 0.9 | 6.36 | 2.91 | 1.06 | 0.49 | 1.18 | 0.54 | 0.13 | 0.93 | 83.46 | |
| 0.21 | 7.86 | 1.08 | 6.93 | 3.05 | 1.46 | 0.64 | 1.35 | 0.59 | 0.14 | 1.00 | 86.78 | |
| 0.23 | 8.1 | 1.25 | 7.7 | 3.5 | 1.77 | 0.81 | 1.42 | 0.65 | 0.15 | 1.12 | 96.91 | |
| 15 | 0.058 | 8.41 | 0.5 | 6.16 | 2.09 | 0.36 | 0.12 | 0.72 | 0.24 | 0.06 | 0.55 | 68.06 |
| 0.117 | 8.96 | 0.78 | 6.78 | 2.93 | 0.79 | 0.34 | 1.01 | 0.44 | 0.09 | 0.65 | 80.42 | |
| 0.167 | 9.45 | 1.06 | 7.08 | 3.61 | 1.18 | 0.6 | 1.11 | 0.57 | 0.11 | 0.71 | 88.64 | |
| 0.21 | 10.2 | 1.4 | 7.78 | 4.46 | 1.63 | 0.94 | 1.16 | 0.67 | 0.14 | 0.82 | 99.87 | |
| 0.23 | 10.0 | 1.05 | 7.39 | 3.78 | 1.7 | 0.87 | 1.62 | 0.83 | 0.11 | 0.74 | 90.18 | |
| 20 | 0.058 | 13.5 | 0.54 | 7.04 | 4.67 | 0.41 | 0.27 | 0.76 | 0.50 | 0.04 | 0.59 | 80.40 |
| 0.117 | 13.79 | 0.98 | 8.24 | 5.2 | 0.96 | 0.61 | 0.98 | 0.62 | 0.07 | 0.67 | 92.61 | |
| 0.167 | 14.04 | 1.56 | 5.66 | 3.33 | 0.95 | 0.56 | 0.61 | 0.36 | 0.11 | 0.45 | 65.24 | |
| 0.21 | 13.68 | 1.45 | 4.47 | 2.31 | 0.94 | 0.49 | 0.65 | 0.34 | 0.11 | 0.34 | 50.80 | |
| 0.23 | 13.32 | 1.27 | 3.77 | 1.92 | 0.87 | 0.44 | 0.69 | 0.35 | 0.1 | 0.28 | 43.44 | |
| 28 | 0.058 | 17.65 | 0.77 | 7.76 | 5.11 | 0.45 | 0.3 | 0.58 | 0.39 | 0.04 | 0.46 | 80.00 |
| 0.117 | 14.49 | 1.67 | 6.98 | 5.03 | 0.82 | 0.59 | 0.49 | 0.35 | 0.12 | 0.43 | 78.25 | |
| 0.167 | 13.05 | 1.55 | 5.23 | 3.53 | 0.87 | 0.59 | 0.56 | 0.38 | 0.12 | 0.31 | 61.93 | |
| 0.21 | 12.49 | 1.46 | 4.81 | 2.82 | 1.01 | 0.59 | 0.69 | 0.40 | 0.12 | 0.27 | 55.07 | |
| 0.23 | 11.03 | 1.27 | 3.85 | 1.94 | 0.89 | 0.45 | 0.70 | 0.35 | 0.12 | 0.21 | 41.39 | |
| Initial Glucose Concentration (Si, g L−1) | Flow Rate (L h−1) | Glucose Spent (ΔS, g L−1) | µmax (h−1) | Doubling Time (h) | Acetic Acid Volumetric Productivity (g−1 L −1 h−1) | Butyric Acid Volumetric Productivity (g−1 L −1 h−1) | Biomass (X, g L−1) | Acetic Acid Specific Cell Productivity (g−1 g−1 h−1) | Butyric Acid Specific Cell Productivity (g−1 g−1 h−1) | Total Carbon Recovery (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| 5 | 4 | 2.26 | 0.07 | 13.56 | 1.65 | 0.09 | 0.74 | 2.23 | 0.12 | 31.29 |
| 8 | 4.17 | 0.18 | 5.51 | 6.26 | 0.29 | 2.25 | 2.78 | 0.13 | 58.04 | |
| 16 | 4.72 | 0.22 | 4.56 | 14.33 | 0.63 | 4.23 | 3.39 | 0.15 | 77.65 | |
| 32 | 4.85 | 0.16 | 6.42 | 29.68 | 0.85 | 5.68 | 5.23 | 0.15 | 84.31 | |
| 10 | 4 | 5.32 | 0.18 | 5.56 | 3.56 | 0.92 | 0.83 | 4.29 | 1.11 | 61.69 |
| 8 | 8.88 | 0.52 | 2.03 | 10.42 | 3.14 | 2.62 | 3.98 | 1.2 | 84.31 | |
| 16 | 9.35 | 0.50 | 1.94 | 18.68 | 6.92 | 4.33 | 4.31 | 1.6 | 90.03 | |
| 32 | 9.81 | 0.40 | 2.54 | 37.88 | 14.34 | 5.76 | 6.46 | 2.45 | 99.46 | |
| 15 | 4 | 10.59 | 0.25 | 4.10 | 4.16 | 1.58 | 1.35 | 4.48 | 1.17 | 64.23 |
| 8 | 14.45 | 0.75 | 1.33 | 14.37 | 3.21 | 2.62 | 5.48 | 1.23 | 84.53 | |
| 16 | 14.74 | 0.55 | 1.83 | 28.29 | 6.49 | 4.17 | 6.78 | 1.56 | 90.11 | |
| 32 | 14.97 | 0.43 | 2.35 | 57.19 | 14.51 | 5.88 | 9.73 | 2.47 | 99.71 |
| Unit | Element | Type | Surface Area (m2) | Material | Total Cost (GBP, £) | Power Usage (MJ) | Cooling Water (kg/day) |
|---|---|---|---|---|---|---|---|
| MBR | Tank | Processing | 37.3 | Stainless steel Type 304 | 74,500 | - | 17,000 |
| Collection | 37.3 | - | |||||
| Pumps | Feed | - | Plastic/Metal | 2200 | 37.15 | ||
| Recirculation | - | ||||||
| Membrane | Hollow fiber | 180 | Polymeric (PVDF) | 5680 | - | ||
| Heat Exchanger | Shell and tube | 173.2 | Stainless steel Type 304 | 52,000 | - | ||
| Raw materials | Powdered chemicals i.e., yeast extract, glucose, sodium chloride etc. and tap water | - | Powder or liquid | 317,034 |
| Fixed Capital Estimate Summary | MBR |
|---|---|
| Total Plant Direct Cost (TPDC, £) (Physical Cost) | |
| Equipment erection | 0.4 |
| Piping | 0.7 |
| Instrumentation | 0.2 |
| Electricals | 0.1 |
| Buildings | none required |
| Utilities | not applicable |
| Storage | provided in PCE |
| Site development | not applicable |
| Ancillary buildings | none required |
| Design and Engineering | 0.3 |
| Variable Costs | |
| Raw materials | £317,034.00 |
| Miscellaneous materials | £1316.92 |
| Utilities Cost | |
| Cooling water | £294.81 |
| Power | £2227.57 |
| Water | £5500 |
| Shipping and Packaging | not applicable |
| Fixed Costs | |
| Maintenance | £13,169.24 |
| Operating labor | £54,000 |
| Plant overheads | £27,000.00 |
| Capital charges | £34,240.02 |
| Insurance | £2633.85 |
| Local taxes | not applicable |
| Royalties | none required |
| Sales expenses | not applicable |
| General overheads | not applicable |
| R&D | not applicable |
| Total annual production rate(rounded) | £457,416.41 |
| Reactor System | Initial Glucose Concentration (g L−1) | Acetic Acid Volumetric Productivity (g−1 L −1 h−1) | Butyric Acid Volumetric Productivity (g−1 L −1 h−1) | Acetic Acid Productivity Ratio | Butyric Acid Productivity Ratio |
|---|---|---|---|---|---|
| Batch | 10 | 0.95 | 0.15 | 1.0 | 1.0 |
| Continuous | 4.41 | 1.27 | 4.6 | 8.7 | |
| MBR | 37.88 | 14.44 | 39.9 | 96.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tajarudin, H.A.; Zacharof, M.-P.; Ratanapongleka, K.; Williams, P.M.; Lovitt, R.W. Intensive Production of Carboxylic Acids Using C. butyricum in a Membrane Bioreactor (MBR). Fermentation 2018, 4, 81. https://doi.org/10.3390/fermentation4040081
Tajarudin HA, Zacharof M-P, Ratanapongleka K, Williams PM, Lovitt RW. Intensive Production of Carboxylic Acids Using C. butyricum in a Membrane Bioreactor (MBR). Fermentation. 2018; 4(4):81. https://doi.org/10.3390/fermentation4040081
Chicago/Turabian StyleTajarudin, Husnul Azan, Myrto-Panagiota Zacharof, Karnika Ratanapongleka, Paul M. Williams, and Robert W. Lovitt. 2018. "Intensive Production of Carboxylic Acids Using C. butyricum in a Membrane Bioreactor (MBR)" Fermentation 4, no. 4: 81. https://doi.org/10.3390/fermentation4040081
APA StyleTajarudin, H. A., Zacharof, M.-P., Ratanapongleka, K., Williams, P. M., & Lovitt, R. W. (2018). Intensive Production of Carboxylic Acids Using C. butyricum in a Membrane Bioreactor (MBR). Fermentation, 4(4), 81. https://doi.org/10.3390/fermentation4040081