Abstract
Carboxylic acids are traditionally produced from fossil fuels and have significant applications in the chemical, pharmaceutical, food, and fuel industries. Significant progress has been made in replacing such fossil fuel sources used for production of carboxylic acids with sustainable and renewable biomass resources. However, the merits and demerits of each carboxylic acid processing platform are dependent on the application of the final product in the industry. There are a number of studies that indicate that separation processes account for over 30% of the total processing costs in such processes. This review focuses on the sustainable processing of biomass resources to produce carboxylic acids. The primary focus of the review will be on a discussion of and comparison between existing biochemical processes for producing lower-chain fatty acids such as acetic-, propionic-, butyric-, and lactic acids. The significance of these acids stems from the recent progress in catalytic upgrading to produce biofuels apart from the current applications of the carboxylic acids in the food, pharmaceutical, and plastics sectors. A significant part of the review will discuss current state-of-art of techniques for separation and purification of these acids from fermentation broths for further downstream processing to produce high-value products.
1. Introduction
There is significant interest among industrial and academic researchers around the world in replacing feedstocks for fuel and chemical production with sustainable biomass resources to supply the increasing population while using cutting-edge technologies to counteract environmental problems such as global warming. Biorefineries, in principle, work similarly to petrochemical refineries in that lignocellulosic biomass, which is a complex mixture containing sugars and aromatic components, are broken down into several high-value products [1]. Studies have also shown that certain functional groups added to naphtha in petrochemical refineries for the production of chemicals are usually naturally present in lignocellulosic biomass [2]. Also, the catalytic processing of petrochemical derivatives such as naphtha and syngas traditionally used for producing carboxylic acids involves high temperature and pressure conditions that result in high energy inputs [3].
The current North American acetic acid market was valued at $2.3 billion in 2014 and was expected to increase to $2.8 billion by 2019 [4]. Other reports have stated that the industry for biomass-based renewable chemicals was expected to increase at a calculated annual growth rate of 7.7%, resulting in a net market of $83.4 billion by 2018 [5]. Reports from the American Chemical Council have found that the global chemical production volume increased by approximately 10% between 2012 and 2016 [6]. This shows that the application of carboxylic acids, as chemicals or solvents, in industrial production has a large volume, as will be further discussed in Section 2. It should be noted that the current world production of lignocellulosic biomass is estimated at between 3 and 5 gigatons per year [1]. In fact, reports from a 2004 study indicated that existing forestry reserves around the world had the capacity to supply up to 9.2 billion tons of oil equivalents, which is enough to supply around 82% of the global energy demand [7]. Several routes have been developed for the conversion of lignocellulosic biomass to biofuels such as pyrolysis [8], gasification [9], liquefaction [10], and a combination of comparatively low-severity thermochemical pretreatment followed by concerted action of enzymes and microorganisms [11]. While each of these technologies has different merits and problems, as discussed in several reviews on their respective fields, the current state of the art in the latter platform is usually considered more specific and cost-efficient and results in high-value byproduct streams [2]. The biomass sugars obtained after thermochemical and enzymatic pretreatment have previously usually been converted to alcohols such as ethanol or butanol [12] and to a lesser degree anaerobically fermented to produce carboxylic acids for further catalytic upgrading to biofuels [13]. It, therefore, seems timely to update the current state of the art of the developments for biomass-based carboxylic acid production.
2. Carboxylic Acid: Formation and Applications
2.1. Current State of the Art
Carboxylic acids or short-chain fatty acids are, by definition, a group of aliphatic mono- and di-carboxylic acids [14] and include organic acids such as formic, acetic, propionic, iso-butytric, butyric, iso-valeric, valeric, iso-caproic, caproic, oxalic, lactic, succinic, malic, fumaric, itaconic, levulinic, citric, gluconic, ascorbic, etc. Another study describes carboxylic acids as “dissociated organic acids that are characterized by the presence of at least one carboxyl group” [15]. Each of the aforementioned organic acids has specific production pathways and applications in the market. For example, acetic acid is produced through carbonylation of methanol and its current prices are primarily controlled by natural gas markets [16]. Studies have indicated that the methanol carbonylation methods for producing acetic acid are accompanied by several drawbacks such as catalyst solubility limitations and the loss of expensive noble metal catalyst during separation steps [17]. Acetic acid has significant applications in the food industry as vinegar [18] and in several food preparations [19]. Acetic acid is also used in the production of vinyl acetate monomers for further polymerization to produce polyvinyl acetate or PVA, which is used in several plastics, and in the production of terephthalic acid and ethyl acetate, which replaces several industrial solvents as a “green” solvent [4]. Other applications for acetic acid include use as an etching agent [20], as a component in the manufacturing of hydrophobic and lipophobic papers in polymer industries [21], in production of cellulose acetate [22], etc.
Propionic acid is usually synthesized by hydrocarboxylation of ethylene in the presence of a catalyst such as nickel carbonyl or rhodium [23]. This study also indicated that this synthesis route has accounted for about 93,000 of the 102,000 ton capacity in the USA in 1991. However, it was found that the production capacity for propionic acid decreased to around 55,000 tons in the USA by 1998. Propionic acid has several uses but its primary application is in the preservation of food grains and animal fodder [24]. Its other applications include the manufacturing of esters [25], herbicides [26], and pharmaceuticals [27]. Moreover, it was found that propionic acid was a major byproduct in the oxidation of light distillate fuel to produce acetic acid in the petrochemical industry [23]. Since the shift in propionic acid production in the USA during the late 1990s, there has been a significant increase in the demand for propionic acid and its derivatives with the growth of the food industry. Market research has estimated the current worth of the propionic acid market to be $935.7 million in 2012, with an estimated increase of 7.8% until 2018 [28].
Similar to propionic acid, butyric acid has also been produced through an oxo-synthesis process, from the oxidation of butyraldehyde [29]. Butyric acid has significant applications in food and flavorings due to its butter-like taste and texture [30], in pharmaceuticals as a component in several anti-cancer drugs and other therapeutic treatments [31], and in perfumes in the form of esters due to its fruity fragrance [32]. Market research valued the butyric acid market at $124.6 million in 2014 with an estimated growth rate as high as 15.1% (significantly higher than any other bio-based chemical) by 2020 due to its varied applications [33].
Collectively, C1–C7 acids are referred to as “volatile fatty acids” (VFAs) due to their relatively high volatility and low vapor pressure compared to other carboxylic acids. However, the focus of the manuscript will be primarily on the short-chain carboxylic acids such as acetic, propionic, butyric (referred to as “VFAs” in this manuscript), and lactic acids, which are predominantly produced as reaction intermediates in the microbial conversion of sugars, as will be discussed in detail in the manuscript. As indicated previously, drawbacks related to poor efficiencies, expensive catalysts, and extreme reaction conditions involved in the current petro-catalysis methods for producing these organic acids are shifting the focus onto alternative biochemical methods.
An example of a carboxylic acid that is commercially produced from optimized biochemical methods, using corn sugars as a substrate, is lactic acid. Chemical synthesis of lactic acid was based on strong acid hydrolysis of lactonitrile, which is a minor byproduct of petrochemical processing [34,35]. This was followed by base-catalyzed degradation of sugars, oxidation of propylene glycol, and high-temperature and high-pressure catalytic reactions between acetaldehyde, carbon monoxide and water [36]. Apart from the obvious drawbacks of these chemical synthesis methods such as high energy and cost requirements, these processes were also plagued by the production of a mixture of L- and D- lactic acid isomers, which affected the further processing of lactic acid for its varied applications. Some of the applications of lactic acid include the production of chemicals such as pyruvic acid, acrylic acid, 1,2-propane diol and ethyl lactate, which are currently replacing several toxic organic solvents in industries as “green” alternatives; and the polymerization to poly-lactic acid (PLA), which is used as a biodegradable plastic [37]. Currently, complete commercial production of lactic acid from pure sugar substrates is being done using biochemical methods, with an annual production capacity reported to be around 800,000 tons in 2013 [38].
2.2. Challenges and Considerations for the Production of Carboxylic Acids from Lignocellulosic Biomass
In order to better understand the steps required for developing cost-effective methods for producing organic acids from biomass instead of pure sugar substrates, it is important to understand the complex structure of the lignocellulosic biomass. The converted and unconverted components of the lignocellulosic biomass at different steps in a biorefinery will help engineers and scientists to develop optimal conversion techniques resulting in pure carboxylic acids that can be further converted to chemicals, polymers, or fuels.
Lignocellulosic biomass contains cellulose, hemicellulose, and lignin as its primary structural components. Studies have shown that the primary component of biomass is 35–50% cellulose along with 20–35% hemicellulose and 10–25% lignin [39]. Cellulose is a high molecular weight linear homo-polymer of repeated units of glucose held together by β-1, 4 glycosidic linkages and is the primary source of glucose or C–6 sugar for carboxylic acid production [40]. Hemicellulose is a linear and branched heterogeneous polymer containing different C5 and C6 sugars as well as other components such as ferulic, acetic, and glucuronic units, which are used for the linkage between sugar units or with lignin and cellulose [40]. While hemicellulose contains a mixture of C5 and C6 sugars and is amorphous in nature, it is easily hydrolysable compared to cellulose. While not the main carbon source for several microorganisms, studies have been done to engineer microbes to also take sugars developed from hemicellulose as the substrate for carboxylic acid production and, thereby, increase the biomass conversion efficiency to produce carboxylic acids. Unlike cellulose and hemicellulose, lignin is a complex structure constructed using phenyl–propane units and is bonded closely with cellulose and hemicellulose to provide rigidity and cohesion to the structure of the cell wall [41]. Lignin mainly consists of aromatic compounds and, due to its complex structure, is highly resistant to enzymatic and microbial action. However, recent studies have shown anaerobic digestibility of wet exploded lignin, resulting in increased methane production through conversion of around 44.4 wt % lignin in the pretreated material [42]. This study also found production of several phenolic compounds and fatty acids in the effluent after anaerobic digestion, which is an indication of the complexities of the fermentation broth, while designing separation processes fitted to produce carboxylic acid as the final product.
3. Biochemical Routes to Carboxylic Acid Production
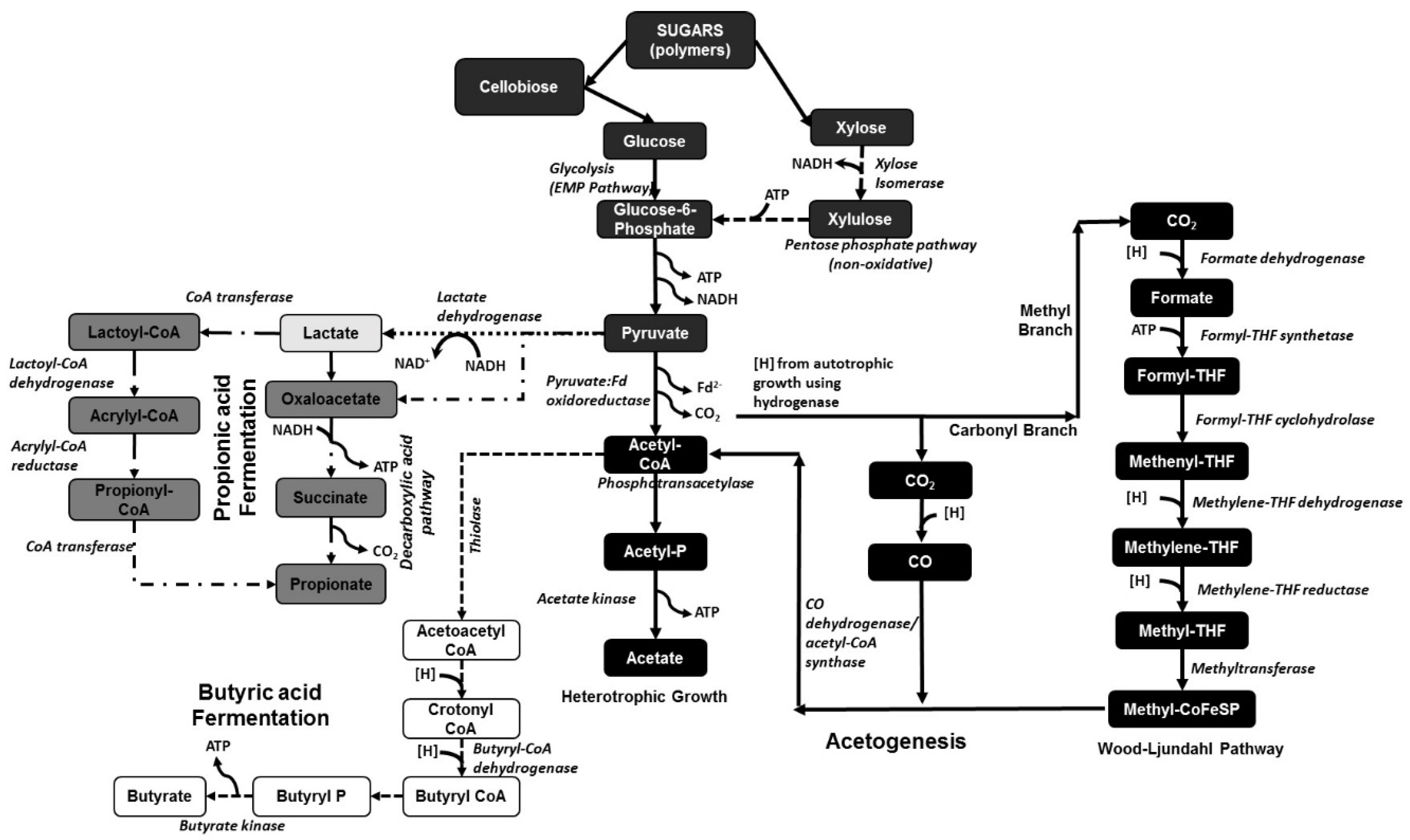
The biochemical routes for producing carboxylic acids from sugar substrates are well known. However, these biochemical pathways are based on using glucose (or fructose) as the substrate and not on the use of a complex sugar mixture derived from biomass after pretreatment, which again will create many other components. The biochemical pathway for converting sugar substrates to different carboxylic acids is shown in Figure 1.
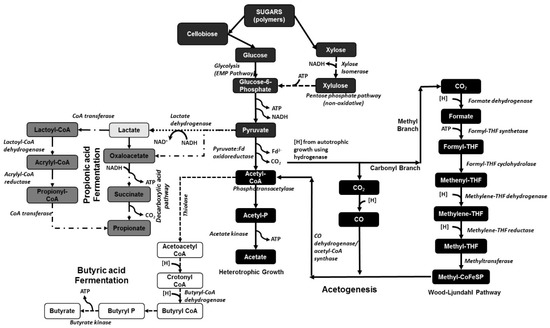
Figure 1.
Different biochemical pathways in different microorganisms for the production of acetic, lactic, propionic, and butyric acid. The different pathways are represented using different line forms: (─) acetogenesis; (···) lactate pathway; (─·) propionate pathway; and (- -) butyrate pathway. [THF: tetrahydrofolate; P: phosphate; H: hydrogen; CoA: coenzyme A; ATP: adenosine triphosphate; CO2: carbon dioxide; Fd2−: ferredoxin ion; NADH: nicotinamide adenine dinucleotide].
3.1. Microbes Used for Acetic Acid Production
Homoacetogens are a group of bacteria that selectively produce acetic acid from organic matter. These homoacetogens or ‘carbon dioxide reducing acetogens’ are obligate anaerobes that use the Wood–Ljungdahl or reductive acetyl CoA pathway as their main mechanism for the synthesis of acetic acid [43]. One molecule of glucose is converted to two molecules of pyruvate using the Embden–Meyerhof–Parnas (EMP) pathway, which in turn is decarboxylated to two molecules of acetyl-CoA and finally into two molecules of acetate (see Figure 1) [44]. The acetogens convert the available H2 and CO2 to acetic acid through the Wood–Ljungdahl pathway [45], which has two branches, the carbonyl and the methyl branches, with CO and/or CO2 substrates. The carbonyl branch reduces CO/CO2 through cobalamine and tetrahydrofolate compounds into a methyl group, which binds to Coenzyme A to form acetyl CoA [43]. The net reaction is as follows:
Most of the known homoacetogens belong to the genera Clostridium and Acetobacetrium. Over 100 acetogens have been isolated and, out of this pool, genomic sequencing data for around 22 acetogens have been published [43]. Some of the most common and most studied acetogens include Clostridium thermoaceticum (or Moorella thermoacetica), Acetobacterium woodii, and Clostridium ljungdahlii. The biochemical pathways relating to these acetogenic bacteria, including their thermodynamics, have been intensively reviewed by Schuchmann and Muller [46]. Moorella thermoacetica, also known as Clostridium thermoaceticum ATCC 49707, is a gram-positive, endospore-forming, and strictly anaerobic thermophilic bacteria with an optimal growth temperature between 55 and 60 °C. There are several studies describing metabolic models of this strain’s acetogenesis pathways [46,47]. The ability of M. thermoacetica to use a wide variety of substrates including sugars, gases such as CO2 & H2, other organic acids, methoxylated aromatic compounds, etc. has increased interest in studying and engineering the bacteria not only for research but also for industrial applications [46,48]. Previous studies have indicated that, with a complex mixture of sugars, Moorella thermoacetica (M. thermoacetica) initially consumes xylose followed by fructose and then glucose, making it a unique microbial strain [49]. Batch fermentation studies operated with 20 g/L xylose showed a maximum acetic acid yield using M. thermoacetica of 0.76 g acid/g xylose [50]. Other studies using pure glucose as a substrate have shown a conversion efficiency of only around 0.65 g acetic acid/g glucose. Very few studies have been done with lignocellulosic biomass substrates where acetic acid yield from an agricultural raw material such as sugarcane bagasse was found to be around 0.71 g acid/g biomass sugars while that from a forestry residue was found to not exceed 0.35 g acid/g biomass sugars [51]. These studies indicate that while the thermal degradation products of sugars and lignin obtained during thermochemical pretreatment (in this case, steam explosion) did not significantly inhibit M. thermoacetica, the primary source of inhibition was the glucose/xylose ratio and the inability of the strain to ferment other sugars such as arabinose, mannose, and galactose with high efficiency. The metabolic function of M. thermoacetica with a xylose substrate was studied using an in situ Nuclear Magnetic Resonance (NMR) bioreactor by Xue et al. [49], where around 12 metabolites were identified and quantified using in situ NMR capabilities and more than 40 metabolites were identified using ex situ HR-NMR (after sample preparation to remove spectral inhibition by cells and media). Real-time NMR analysis also showed production of formate, ethanol, and methanol by M. thermoacetica using xylose as a substrate [49].
C. ljundahlii (ATCC 49587) is similar in structure to M. thermoacetica but lacks the cytochromes and quinones that would normally result in salt imbalances in the microorganisms [46]. However, similar to M. thermoacetica, C. ljundahlii does not require Na+ for growth and optimally grows between pH 5 and 7 under thermophilic conditions. Studies have indicated that this strain favors the production of ethanol over acetic acid at a pH below 4.5 and can produce a spectrum of products including acetic and butyric acids at higher pH [52]. However, most of the reported data on C. ljundahlii for the production of acetic acid focus on gaseous substrates such as synthesis gas (CO, CO2 & H2) and very few studies have been done with sugar substrates [53].
Unlike M. thermoacetica and C. ljundhalii, A. woodii is a non-spore-forming mesophilic bacterium that grows robustly on H2 and CO2 to produce predominantly acetate and has also been used in varying conditions to produce small amounts of other chemicals such as formate, methanol, ethanol, 1,2-propanediol, 2,3-butanediol, ethylene glycol, lactate, etc. [46]. Being mesophilic, A. woodii is significantly affected by contamination and the absence of cytochromes and quinones makes the strain dependent on Na+ [54,55]. Metabolic modeling of the acetogenesis in A. woodii has shown that, during heterotrophic growth, 4 moles of hydrogen are produced and further used during autotrophic growth (producing 2 moles CO2), resulting in the production of 1 mole of acetate and 0.3 moles of ATP [56].
Studies have also been done to understand the biochemical and energy profile of reductive acetogenesis in combination with the Wood–Ljundahl pathway for acetic acid production [57]. Theoretical models have shown that ATP is formed during acetate kinase reaction and during pyruvate formation but an equal amount of ATP is consumed for conversion of ATP to acetyl-CoA and during the formyl-THF synthase reaction (in the Wood–Ljundahl pathway). Hence, membrane-bound complexes are required to be coupled with the pathways to control the energetics resulting in active generation of ATP. Some metabolic modeling studies have reported that hexose fermentation in acetogens uses low-molecular-weight, iron–sulfur center-containing proteins known as ferredoxin for the conversion of pyruvate to acetyl-CoA [46,58] (shown in Figure 1). These metabolic studies hypothesized that this reaction through ferredoxin as an electron acceptor has a standard redox potential that is much more negative compared to the NAD+/NADH electron transport, resulting in an acceptable energy balance within the biochemical pathways of the acetogens. However, other studies [59] hypothesized another possible route through a reaction using formate-hydrogen lyase and methylene-tetrahydrofolate reductase where the reaction resulted in the transport of two protons out of the cell membrane, resulting in a proton gradient to facilitate electron transport in the acetogens. This model was investigated for M. thermoacetica iAI558 using CO2 and H2 as substrates to produce acetic acid and the metabolic studies indicated that both the routes (as described above) did not individually show a complete energy balance during the conversion of pyruvate to acetyl-coA in an acetogen. However, these studies concluded that the combination of both these routes could explain the increased efficiency of acetogens such as M. thermoacetica due to their ability to generate energy through thermodynamically infeasible cycles without a specific growth substrate [47]. Due to the absence of cytochromes in C. ljundahlii, a proton gradient facilitating electron transport and energy balance was hypothesized to be achieved through membrane-bound Rnf complexes [60]. Unlike thermophilic bacteria, energy balance analysis through metabolic modeling of mesophilic bacteria such as A. woodii indicated that the Na+ gradient was used by the membrane-bound ATP synthase to produce ATP [56]. These studies also indicated that the acetogenesis began with H2 being oxidized by an electron-bifurcating hydrogenase that coupled the reduction of ferredoxin with that of NAD to promote acetogenesis. Such metabolic information not only helps validate the biochemical pathway found by a pure microbe to consume a specific carbon substrate to produce a specific carboxylic acid, but also provides information regarding the energetics of specific parts of the pathway that will help us understand a mixed microbial consortium that contains one or more such microbes.
3.2. Microbes Used for Butyric Acid Production
Similar to acetogens, butyric acid production through fermentation is one of the oldest and most-studied techniques since the First World War. Several reviews have been published on different strains used for butyric acid production and seven different genera have been investigated for their potential industrial applications [56,61]. Some of the most commonly studied and industrially used strains includes C. tyrobutyricum [62,63], C. acetobutylicum [64,65], and C. thermobutyricum [66,67]. A major drawback of using clostridial strains for butyric acid production is the formation of other side products such as acetic acid, ethanol, butanol, acetaldehyde, and lactate, as well as gases such as CO2 and H2. Butyric acid production starts with the metabolism of glucose to pyruvate through the Embden–Mayerhof–Parnas (EMP) pathway followed by conversion to acetyl-CoA and then to acetoacetyl-CoA through a thiolase reaction (as shown in Figure 1). The butyryl CoA produced from acetoacetyl-CoA is then converted to butyryl phosphate by phosphotransbutyrylase and then converted to butyrate by butyrate kinase [62]. Metabolic studies showed that C. tyrobutyricum, C. thermobutyricum, and C. acetobutylicum followed different pathways with different efficiency, as shown in Equations (2)–(4):
Similar to the acetogenic strains discussed in Section 3.1, the clostridial strains targeted for butyrate production usually do not possess cellulolytic enzymes and have a lesser ability to use cellulose as substrate; hence, they require extensive pretreatment and enzymatic hydrolysis to deconstruct biomass into simple sugars for carboxylic acid production. While some studies have indicated that certain clostridial species have cellulases encoding for genes allowing them to utilize polymers such as cellulose, most of these studies also indicated that these strains employ carbon catabolite repression (CCR), whereby preferred carbon substrates such as glucose will suppress the utilization of other sugars [56]. As can be seen from Equations (2) and (3), clostridial strains for butyrate production are also significantly pH-sensitive. Studies have indicated that at pH > 5 and with iron limitations, lactate can replace butyrate as the major product of fermentation in C. butyricum [68]. Other studies showed the highest butyrate production in C. tyrobutyricum between pH 6 and 6.7 [62]. These studies indicated that the shift in metabolic flux as a function of pH is related to the difference in activity of lactate dehydrogenase, phosphotransbutyrylase, and phosphotransacetylase.
Significant metabolic studies have been done on butyric acid production to understand the pathways leading to butanol production, especially after inhibiting hydrogen production by controlling the ferredoxin in the pathway (ferredoxin is produced during conversion of pyruvate to acetyl-CoA, as shown in Figure 1) [64]. These studies also showed that a proper energy (ATP) balance in the butyrate pathway (shown in Figure 1) could not be established for butyric-acid-producing strains such as C. acetobutylicum due to the absence of Rnf membranes or a similar membrane-bound electron transport system existing in its pathways. Other studies found that, in C. butyricum, the lower partial pressure of H2 decreased the acetate/butyrate ratio, followed by an increased ATP yield, resulting in an energetically favorable pathway that would take up the produced acetate and convert it back to butyrate [69]. Such energetically favorable reversible reactions have been found to be of importance, especially in mixed microbial cultures compared to pure cultures.
3.3. Microbes Used for Propionic Acid Production
Paludibacter and Propionibacterium produce propionic acid as the major end product of fermentation. For example, Paludibacter propionicigenes produces propionate and acetate as major products when fermenting glucose [70]. Paludibacter belongs to the phylum Bacteroidetes and is one of the most abundant species in wastewater treatment plants [71]. Paludibacter jiangxiensis is another bacterium that utilizes sugars to produce propionate as the major product. However, compared with P. propionicigenes, it was found that yeast extract was not required for the growth of the bacterium [72]. Propionibacteria selectively produce propionic acid from organic matter and make up 1.4% of the ruminal microflora [73]. Propionate production from lactate occurs either via the ′direct reduction′ pathway or the ′decarboxylic acid′ pathway. In the decarboxylic acid pathway, succinate is initially formed (see Figure 1) from heterotrophic fixation of carbon dioxide, which is then decarboxylated to propionate [74].
Another bacterium that has the ability to produce propionic acid from sugar substrates following an alternate “acrylate pathway” is Clostridium propionicum (see Figure 1) [75,76]. Other studies have indicated that certain cellulolytic bacteria such as C. saccharolyticum and C. thermocelluum also follow a similar pathway during the production of propionic acid [56]. In this acrylate pathway, the sugars are initially converted to lactate, which is then converted to lactoyl-CoA through the action of CoA synthase, followed by dehydrogenase action leading towards acrylic acid and then propionic acid. Studies have indicated that, following the decarboxylic acid pathway, propionic acid production is hindered due to the production of lactic and succinic acid as byproducts [77]. Another study using glycerol as a substrate with Propionibacterium acidipropionici and Propionibacterium freudenreichii ssp. shermanii bacteria resulted in the production of 0.79 mol of propionic acid/mol substrate consumed at a productivity of around 0.42 g/L/h at optimized conditions, with acetic acid produced as the main byproduct along with lactic acid and succinic acid [78]. These studies also showed that when glycerol was replaced with glucose as the carbon substrate, a higher amount of acetic acid was produced as a byproduct, followed by a decrease in the propionate yield. Metabolic studies on the different biochemical pathways for propionic acid production showed that the optimized pathway had acrylic acid as an intermediate [79]. Metabolic studies using Biological Network Integrated Computational Explorer (BNICE) were one of the first successful attempts at using pure computational modeling to study around 16 different biochemical pathways towards propionate production in different propionate bacteria. These studies predicted that the rate-limiting step for propionate production was the reduction of acrylic acid to propionic acid through the “acrylate” pathway. Such computational methods, blended with actual metabolic data on the different biochemical pathways, allow for accurate prediction of the enzymatic activities of the different steps in a specific pathway.
3.4. Microbes Used for Lactic Acid Production
Lactic acid bacteria have been studied for several years, especially related to their use in the food industry. Taxonomically, lactic acid bacteria belong to two distinct phyla, Firmicutes (with Lactobacillus and Lactococcus as the most-studied genera) and Actinobacteria (with Bifidobacterium as the most-studied genus) [80]. Lactobacillus has been studied for biorefinery applications as they are facultative anaerobes and can also live under microaerophilic conditions in nature with the production of different fermentation end-products including lactic acid and sometimes further formic acid, acetic acid, ethanol, and carbon dioxide [81]. The lactate production follows glycolysis through the EMP pathway as shown in Figure 1, followed by the action of lactate dehydrogenase on pyruvate to produce lactate. The major advantage of the bacterium is its tolerance to low pH when compared to other carboxylic-acid-producing strains with optimal pH over 5 [82]. Another advantage of some Lactobacillus strains is their non-sporulating nature, which will be further discussed in Section 3.5. Based on the nature of the fermentation, lactic acid bacteria in general have been classified as homo-fermentative, resulting primarily in L-lactic acid as the only product, and hetero-fermentative, which produce small amounts of acetic acid and ethanol as byproducts. Major homo-fermentative products includes Lactococcus lactic [83], Lactobacillus delbrueckii [84]; Lactobacillus helveticus [85]; Lactobacillus casei [86], Bacillus subtilis [87], and Bacillus coagulans [88,89,90]. Similar to the VFA-producing bacteria, the Lactobacillus or Bacillus strains are rod-shaped bacteria that have a tendency to sporulate [80]. A high lactate-producing bacterial strain such as B. coagulans showed as much as 92% lactic acid yield from sucrose [88]. Continuous fermentation of lignocellulosic biomass using an isolated B. coagulans strain resulted in lactic acid yield of 0.95 g/g biomass sugars with a productivity as high as 3.69 g/L/h at the optimum pH of 6 with a residence time as low as 6 h [90]. Similar lactic acid yields were obtained with batch fermentation of pure glucose using the B. coagulans WCP10–4 strain with a productivity as high as 3.5 g/L/h [91]. These studies show that lactic acid bacteria are severely inhibited by product concentrations above a certain range and, hence, it would be beneficial to remove the product immediately to increase the efficiency of the fermentation.
3.5. Disadvantages of Pure Microbial Cultures in Biorefineries
The previous sections discussed several pure bacterial strains and pathways for the conversion of sugar substrates to different carboxylic acids. The advantages of using such pure strains include complete knowledge and control of the biochemical pathways towards carboxylic acid production from sugars, lesser contamination from other strains under sterile conditions, and easy genetic engineering of the strains to add other functionalities such as cellulose degradation (which allows the pure strains to digest cellulose to produce carboxylic acids instead of just simple sugars). However, there are several disadvantages related to using pure cultures, especially due to the risk of contamination from other strains that are more energetically favorable, resulting in the formation of other less-desired products instead of carboxylic acids. Also, in biorefineries, complex substrates such as lignocellulosic biomass have several components other than sugars such as aromatic and phenolic compounds coming from the lignin, which have antimicrobial properties and can inhibit some pure microbial strains more than others. Clostridial strains, as mentioned previously, have been indicated to suffer from low growth rates when lignocellulosic biomass is used as the substrate [56].
Another disadvantage with an acetogen such as M. thermoacetica is its significant dependence on pH. Studies have shown that the growth of M. thermoacetica (or C. thermoaceticum) strains stopped when the pH of the anaerobic glucose fermentation dropped under 5 [92]. Other studies indicated that M. thermoacetica strains survived well in a pH range of 6 to 7 with acetic acid yield between 0.8 and 0.95 g acid/g glucose but that the growth of M. thermoacetica decreased significantly with an increase in acetic acid concentration in the fermentation broth [93]. The pH sensitivity of butyrate producing strains has already been discussed in Section 3.3. Apart from pH sensitivity, these strains were also found to be sensitive to other factors such as the H2 partial pressures, resulting in a significant effect on the product distribution. The sensitivity of such pure microbial strains to acid concentrations, system pH, gas partial pressures, and contamination from other microbes altering the end product of the fermentation point to the use of mixed microbial consortia specifically grown towards target acid product and the development and optimization of in situ separation techniques to remove acids from the fermentation broth as part of its production.
Another disadvantage of clostridial strains, in general, for biorefinery applications is the tendency of the strains to sporulate as a form of asymmetric cell division [94]. Studies have indicated that such spores tend to become metabolically inactive [95]. Studies on C. acetobutylicum have been done to change the sporulating pattern of this strain by inactivating sporulation-related sigma factors, resulting in mutants without the ability to sporulate at the stationary phase, leading to efficient acidogenesis and, hence, butyric acid production [96].
Other common engineered strains for biorefinery applications include E. coli, S. cerevisiae, and other yeasts. Several reviews and metabolic studies have been published on these mutants [97] but most of these studies indicated that such engineered strains were significantly limited by substrate utilization, especially the C6 and C5 sugars present in lignocellulosic biomass [98]. Mixed microbial consortia such as those present in the rumen, on the other hand, can combine both substrate utilization and targeted product formation through optimizing pathways and fermentation conditions, especially related to high carboxylic acid yields in biorefineries.
3.6. Mixed Bacterial Consortia for Carboxylic Acid Production
Ruminants are the most populous group of animals in the world, with more than 3.5 billion on each continent [99]. Ruminants (e.g. cows) are known to produce VFAs, especially acetic, propionic, and butyric acids, from organic matter [100]. They have the ability to digest cellulose and break it down into simple sugars, which can be attributed to the presence of a unique microflora in their rumen, including cellulolytic microorganisms [101] and consisting of about 1010 to 1011 bacterial cells per milliliter and 103 to 105 zoospores per milliliter, in addition to protozoans and bacteriophages. Many groups of microbes including bacteria, fungi, and archaea are involved in the production of carboxylic acids; of these the most important group of bacteria are the acetogens.
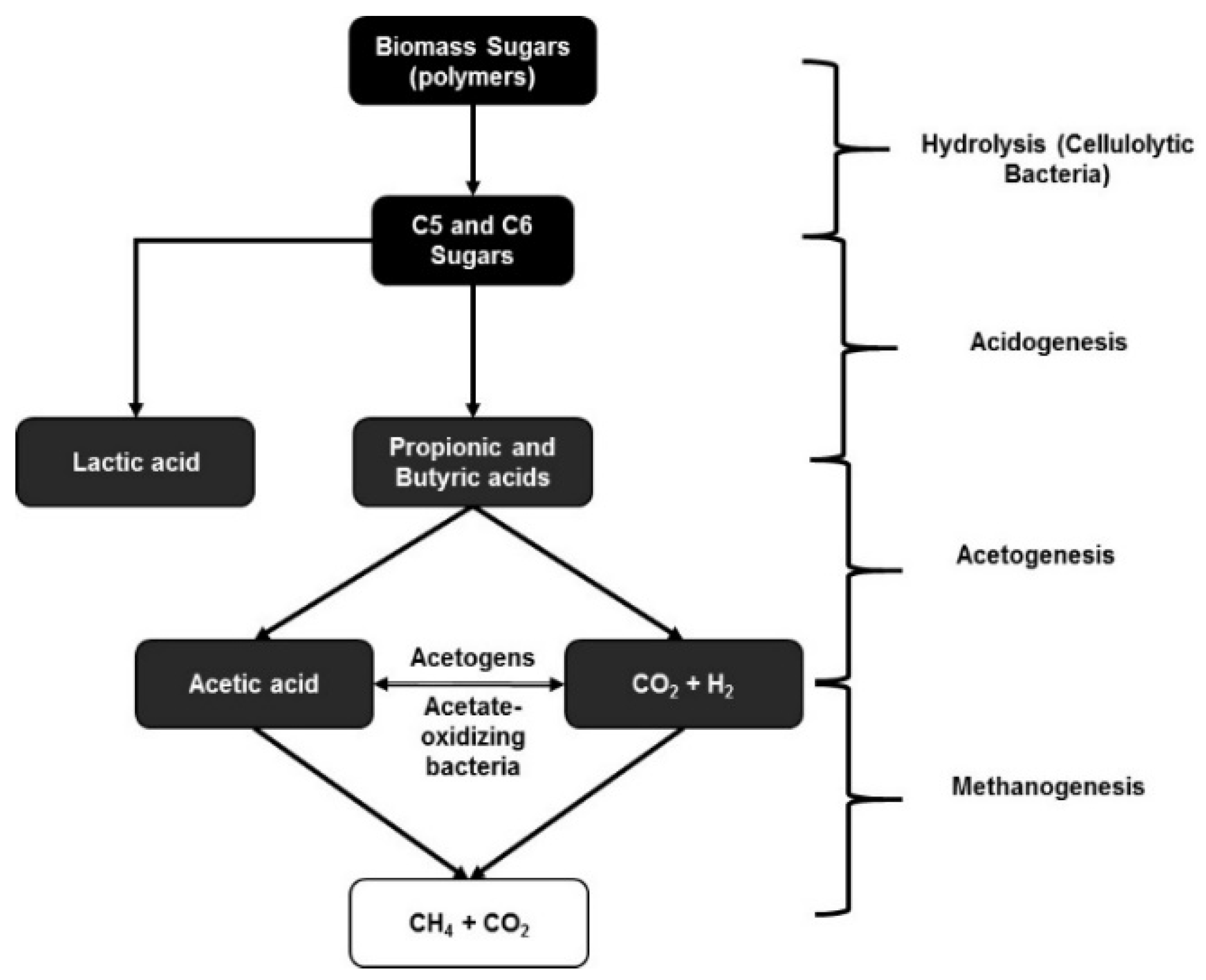
Ruminal fermentation of organic matter requires interaction between three groups of anaerobic microbes (Figure 2): fermentative bacteria, which degrade organic matter to hydrogen, carbon dioxide, and VFAs; acetogenic bacteria, which convert hydrogen, carbon dioxide, and acetate; and methanogenic bacteria, which reduce hydrogen, carbon dioxide, and formate to methane [102]. In the bovine rumen, methane production is primarily from the reduction of carbon dioxide with hydrogen as the electron donor [103]. Studies have indicated that although methanogenesis represents a 2–15% energy loss in cattle [104], a low concentration of hydrogen is required for complete fermentation of the organic matter, maximum yield of ATP, and optimal microbial growth in the rumen [105]. If methanogenesis in an artificial rumen reactor is inhibited, hydrogen accumulation increases through the inhibition of oxidation of the reduced NADH [106] and, in turn, VFA production including acetate and propionate production will further increase [107]. It has also been shown that high levels of propionic and butyric acids can inhibit methanogenesis [108]. In other words, it is ideal to prevent methanogenesis in the rumen microflora in order to increase the VFA production through anaerobic ruminal fermentation.
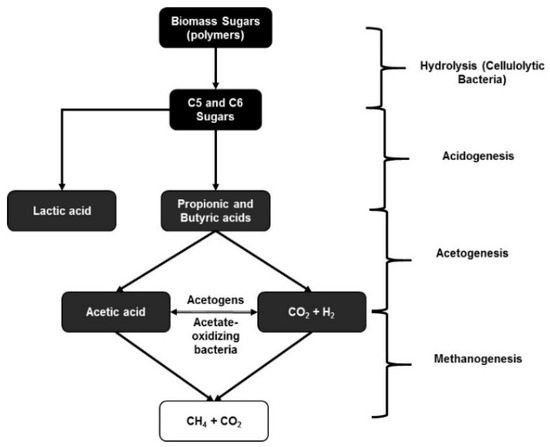
Figure 2.
Different stages of biomass conversion through fermentation using a mixed microbial consortia such as rumen microflora.
It is common knowledge from several in situ fermentation studies that the rumen microflora consists of several cellulose-degrading, acidogenic, acetogenic, and methanogenic bacteria that are necessary for the optimal function of the rumen in a ruminant animal [99,100,101,102,103]. Several studies have been done to identify and understand the microbial population of the ruminal microflora, especially related to biorefinery applications. One such study identified around 27,755 putative carbohydrate-active genes that accounted for as much as 57% of the cellulose-degrading enzymatic activity in the rumen microflora [109]. These metagenomic studies done with cellulose and switchgrass as substrates showed the presence of endoglucanases, β-glucosidases, and cellobiohydrolases in the ruminal microflora. Some of these cellulolytic strains present in the rumen include Fibrobacter succinogenes, Ruminococcus flavefaciens, and Ruminococcus albus [110]. Unlike pure microbial cultures used for carboxylic acid production, which would require genetic alterations using certain cellulolytic bacteria such as C. thermocellum (thermophilic) or C. cellulolyticum (mesophilic) [111], mixed microbial consortia have the advantage of the collaboration of several different cellulolytic bacteria that together produce a more diverse and optimal cellulolytic enzyme mixture for the conversion of cellulose in lignocellulosic materials. This internal enzyme capability is of major importance, since biorefineries are affected by the high enzyme costs required to completely convert the sugars present in pretreated biomass into C5 and C6 sugars for uptake by pure microbial cultures. Using a mixed rumen microflora, it is possible to significantly reduce the cost of the production process by harvesting the intrinsic capability for producing a complex enzyme mixture along with fermenting the resulting sugars into carboxylic acids.
Apart from cellulolytic bacteria and methanogenic archaea present in the rumen, a significant portion of the rumen microflora is composed of acidogenic bacteria. Studies have indicated that Acetitomaculum ruminis is the most predominant acetogen present in the ruminal microflora [112]. Other acetogens present in the rumen include Eubacterium limosum, Ruminococcus productus [113], and obligate hydrogen-producing acetogens such as Syntrophomonas wolfeii and Syntrophobacter wolinii [114]. Ruminococcus schinkii, which is phytogenically similar to the Clostridial species, has been isolated from the rumen in suckling lambs and has a greater propensity towards acetogenesis even when using O-methylated aromatic compounds [115]. The advantage of A. ruminis present in the rumen versus the other acetogens is its ability to produce acetate via heterotrophic growth (see Figure 1), while those acetogens that use autotrophic growth compete with methanogens for H2 [116]. Reductive acetogenesis by acetogens following autotrophic growth in rumen microflora is usually similar to that shown in Equation (5), unlike the traditional acetogenic reaction shown in Equation (1):
Davidson and Rehberger [117] showed that ruminal populations of Propionibacteria ranged from 103 to 104 cfu/mL and, out of 132 isolates studied, 126 were identified as P. acidipropionici, which is also capable of nitrate reduction. Initial studies have indicated that rumen microflora from a ruminant animal that has a high forage diet usually have Streptococcus bovis as the primary lactic-acid-producing bacteria [118]. However, other studies showed the metabolic shift in the lactate-producing ruminal population toward Lactobacillus when there was an increase in sugar substrates in the ruminant diet [119]. Hence, the Lactobacillus bacteria present in rumen microflora are essential to understand when using mixed microbial consortia for biorefinery applications. Studies have identified that Lactobacillus ruminis and Lactobacillus vitulinus are two prominent lactic acid bacteria in the rumen when feeding a high carbohydrate content in the diet [120]. Other studies isolated around 36 different lactate-producing bacteria from the rumen including B. licheniformis, B. coagulans, B. circulans, B. laterosporous, B. pumilis, etc. [121]. Even with the presence of such lactate-producing bacteria, one of the advantages (and, in certain cases, disadvantages) of using rumen mixed cultures for biorefinery applications is that the cellulolytic bacteria and the lactic acid bacteria work at different optimal pH ranges. It is commonly known that cellulolytic bacteria function optimally at a pH range between 6 and 7, while several studies found the optimal performance of lactic acid bacteria at a pH below 6. The advantage of this discrepancy is the ease of guiding mixed culture fermentation of biomass towards VFAs with negligible effect from the lactic acid bacteria present in the rumen microflora, simply by maintaining a pH of over 6. It is properties such as this that have led to the interpretations of ruminal mixed cultures as “habitat-simulated” mixed cultures [122], which can be selectively targeted towards specific carboxylic acid produced under specific controlled experimental conditions. Studies into isolating lactic acid bacteria from rumen microflora also resulted in isolating Sphaerophorus prevot species, which had the ability to produce butyric acid as a minor byproduct [123]. There is no specific literature on predominantly butyric-acid-producing bacteria in the rumen microflora, but butyric acid is produced as a minor byproduct by several acetogens and propionate-producing bacteria present in the rumen. These microbes include Bacteriodes amylogenes [124] and Butyrivibrio fibisolvens [125]. Also of interest to biorefinery applications is the effect of xylans as a substrate for rumen bacteria. Studies aimed at characterizing bovine ruminal bacteria isolated from the microflora after growth in a xylan-supplemented medium showed the presence of strains such as Butyrivibrio fibrisolvens and Bacteriodes ruminicola, with propionic acid as primary products [126]. The study found that there was a significant reduction in cellulolytic bacteria in the rumen microflora grown selectively on a xylan medium, and while the actual microbial population did not change significantly, the transition of the carbon substrate for growth from xylan to glucose resulted in a decrease in propionic acid production. Murali et al. [13] have successfully produced volatile fatty acids (acetic, propionic, and butyric acid) from pretreated corn stover with increased conversion of corn stover when solids loading was increased from 2.5% to 5%. This study showed good performance of the ruminal bacteria towards VFA production with a decrease in biomass carbohydrates (sugar polymers) due to the presence of cellulolytic bacteria in the rumen that can depolymerize cellulose and hemicellulose into sugars followed by acetogenesis. Such a microbial system can effectively reduce the costs of producing VFA by eliminating the need for external lignocellulolytic enzymes such as cellulases and hemicellulases.
In vitro studies were done in an anaerobic fermenter to study the dynamic response of the system to pulses of volatile fatty acids input into the system [127]. These studies found that the addition of butyrate and valerate to the anaerobic fermenter led to an increase in acetic acid production, with propionic acid as a minor byproduct. However, pulsed input of propionic acid to the anaerobic fermenter destabilized the system, resulting in a reduction in acetogenesis. Such effects of process imbalances have also been studied in batch cultures from anaerobic reactor systems [128,129,130,131]. Such studies are, again, sufficient evidence for the need for in situ product separation to control as well as prevent system instability during the anaerobic fermentation of lignocellulosic biomass to produce carboxylic acids.
4. Product Separation and Purification
As indicated earlier in this manuscript, separation processes are required in a biorefinery at different stages primarily to (a) separate and purify the product/intermediate for the next stage of processing and (b) remove biomass components that are inhibitory at a particular stage of processing. However, here we will primarily focus on the significance of in situ product recovery from biochemical fermentation in biorefineries that produce carboxylic acid as a product or an intermediate for fuels and/or chemicals. Mixed acid fermentation is usually inhibited by the product/products produced, i.e., in situ product recovery during fermentation will sufficiently remove the inhibitory effects of carboxylic acids produced during fermentation. For example, studies done by Garrett et al. [82] have shown that fermentation productivity using B. coagulans for the production of lactic acid was significantly inhibited above a lactate concentration of 20 g/L in the fermentation broth at 50 °C and pH 5.5. These studies using Amberlite IRA-67 for in situ extraction of lactic acid showed an increase in microbial productivity by at least 1.4-fold when compared to fed-batch fermentation with conventional salt precipitation. Several similar studies done on in situ carboxylic acid recovery from fermentation broths are shown in Table 1.

Table 1.
Existing literature data on in situ carboxylic acid separation during fermentation.
In situ or on-line product recovery of carboxylic acids from fermentation broths using dialysis, distillation, adsorption, and extraction have been attempted [146]. Adsorption and extraction are two fairly common techniques that have been used in the continuous acid recovery from anaerobic fermentation.
4.1. Separation Using Ion Exchange Resins
Adsorption with conventional adsorbents such as activated carbon usually results in contamination of the product, especially in the case of biomass fermentations due to the presence of unconverted biomass components that have an affinity towards activated carbon [146]. For example, polymeric adsorbent XAD-4 was used to adsorb furfural from acid-catalyzed biomass hydrolysate before ethanol fermentation for removal of inhibitory contaminant [147]. This study indicated that such polymeric adsorbents are a very good source of detoxification of the biomass hydrolysate before anaerobic fermentation. However, in the context of product separation and purification, adsorption by ion exchange seems to be far more efficient. Ion exchange resins are usually polymeric resins with a linked cation or anion exchange group [148]. For carboxylic acid separation, predominantly used resins are strong or weak base resins, which have tertiary or quaternary amines as the ion exchange group [149,150]. The carboxylic acids are usually recovered from the ion exchange resin through caustic elution and can be concentrated through evaporation and hydrolyzed to give a pure acid for further processing. As can be seen from Table 1, there are a number of studies on in situ separation of lactic acid from the fermentation broth. This is primarily because of the dependence of efficiency of ion exchange resin-mediated separation of carboxylic acid on the fermentation pH. The pKa of acetic acid is 4.76, while that of lactic acid is 3.86. As discussed previously in the manuscript, anaerobic fermentation to produce acetic acid and other VFAs is usually optimal at a pH between 6 and 7, while that to produce lactic acid can function optimally at pH < 6. Hence, under these conditions, ion exchange resins will not function optimally for in situ VFA recovery from fermentation broth without the addition of an acidification column before the ion exchange resin. Studies have indicated, at its pKa, that the maximum adsorption efficiency of Amberlite IRA-67 for acetic acid is around 61.36% at 25 °C, with acid loading on the resin as high as 33 g acetic acid/g resin [151]. Other studies showed maximum propionic acid loading onto Amberlite IRA-67 resin of around 36 g/g resin [152]. Currently, the only study on using ion exchange resin such as Amberlite IRA-67 for in situ product recovery was done by Garrett et al. [82] for anaerobic fermentation of corn stover using B. coagulans at a pH of 5.5 (well above the pKa of 3.86) and a temperature of 50 °C. The resin loading for acetic acid and lactic acid were determined to be 3.81 mg/g resin and 170.2 mg/g resin, respectively. However, through the use of a continuous product recovery loop controlled by fermentation pH, the lactic acid produced during fermentation could be continuously separated by the resin and this process removed the necessity of a separate pH control for the fermentation, while also removing product inhibition due to lactate accumulation. However, if an acidification resin such as Amberlite IR-120 was connected to the product recovery loop before the ion exchange resin to decrease the pH of the effluent, the acid loading onto the weak/strong base resin could be sufficiently increased. Studies done by Bishai et al. [144] and Monteagudo & Aldavero [143], as shown in Table 1, have attempted this with strong and weak base resins, respectively, connected to lactate fermentation, resulting in increased product recovery and acid loading on the resin.
4.2. Separation Using Solvent Extraction
One of the primary disadvantages with using solvent extraction for in situ product recovery during fermentation is the difficulty of finding a common biocompatible solvent that has a high extraction coefficient for the product. Another problem is the ability of the solvents to function at the fermentation pH (ranging between 5.5 and 7), which is the interesting range for carboxylic acid production [153]. The primary problem is related to phase separation and, hence, different tertiary and quaternary amines need to be used for reactive extraction of the carboxylic acids from the fermentation broth. Unlike solvent extraction, in reactive extraction, the acid is extracted onto an organic phase and complexed with a carrier (additive) or complexing agent, resulting in higher extraction efficiencies. Several studies using amines with oleyl alcohol, iso-decanol, and kerosene as diluents or additives to the amines for effective in situ extraction of different carboxylic acids from fermentation broth are shown in Table 1. The most commonly used amines were Alamine-336 and Hostarex-A327, while other studies examined other amines and found their performance in carboxylic acid extraction to be comparatively low. Studies have further been done using tri-n-octylphosphine dissolved in methyl isobutyl ketone, which showed higher distribution coefficients for propionic and butyric acids when compared to these amines for reactive extraction from fermentation broth [154]. These studies also showed that the phase separation during reactive extraction could be increased by increasing the contact of the acids with the amines using membranes such as hollow fibers (see Table 1). While the reactive extraction using amines had similar efficiency when compared to ion exchange resins, there were problems related to the complete recovery of the amines during back-extraction [140] and separation of the diluent resulted in the need for additional separation steps during downstream processing. The loss of cells also presented a problem and can be controlled by immobilization, which can negatively affect the process operation costs.
Due to the volatile nature of the VFAs, reactive extraction of acids from fermentation broth has further been tested using pervaporation or supercritical fluid extraction. In pervaporation, the fermentation effluent containing two or more miscible components contacted a non-porous/molecularly porous membrane with a vacuum applied on the other side to facilitate extraction [155]. In supercritical fluid extraction, supercritical fluids such as carbon dioxide were pressurized, heated, and allowed to diffuse through the fermentation effluent with the conditions controlled to selectively and efficiently remove VFAs [156]. Both these techniques are energy-intensive and, due to the harmful nature of pressure and temperature on the microbes, require an extra filtration step to prevent any contact with the microbes. However, these methods were found to have very high fluxes and were capable of achieving high yields. Pertraction was used with 20 wt % Hostarex in oleyl alcohol as the extractant, resulting in an increased butyric acid titer during fermentation from 7.3 g/L at control to as high as 20 g/L [136]. This study showed that in the absence of pertraction but with amines included, the extraction efficiency only increased by 1.4-fold, while with pertraction the increase was significantly higher. In another study with in situ separation using pertraction, butyric acid and hexanoic acid productivity was found to increase by at least 3-fold while using pertractive fermentation when compared to batch fermentation [141]. These studies indicated that oleyl alcohol with trioctylamine as the extractant was found to be less inhibitory to the microbial culture during pertractive fermentation when compared to octanol-trihexylamine extractant. There have been relatively fewer studies on the supercritical fluid extraction of carboxylic acids from the fermentation broths, which was found to work better at acidic pH [156]. Garrett et al. [157] used supercritical carbon dioxide to remove acetic acid from model fermentation broths, resulting in 93% acid recovery at optimized conditions of 2150 psi, 45 °C, and 5 h extraction time.
4.3. Effect of Product Concentration on Separation Processes
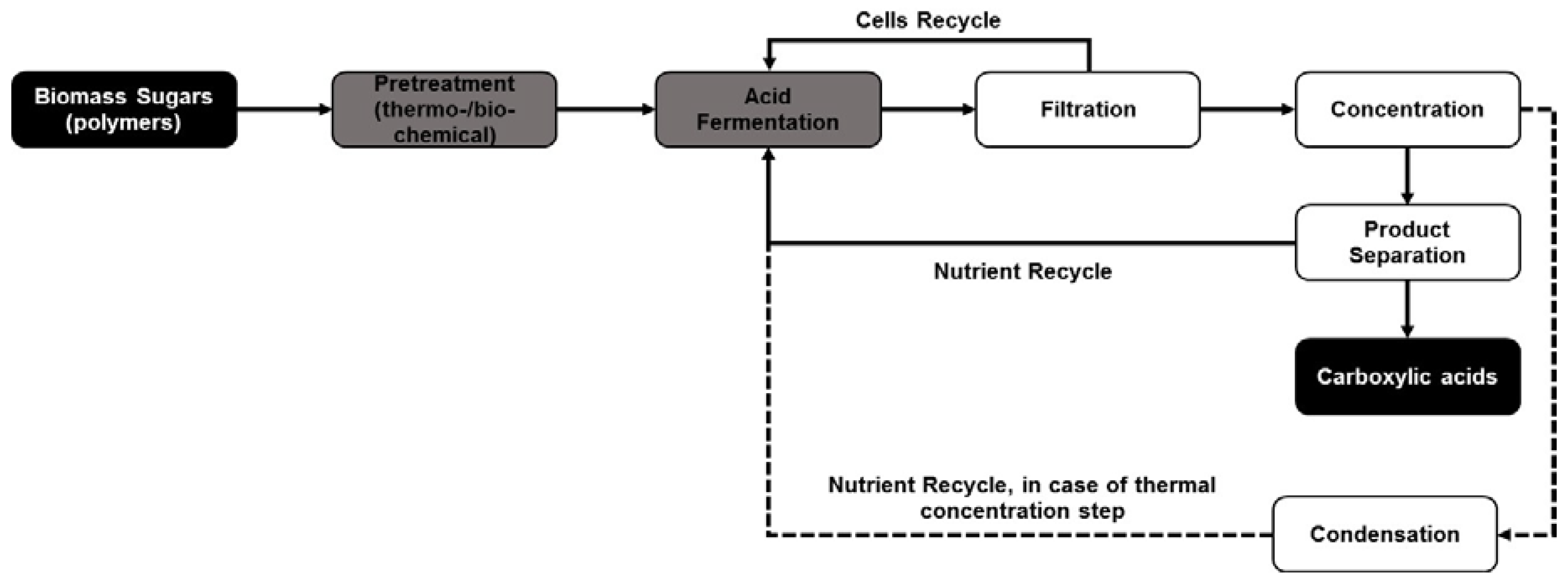
It should be noted that these separation procedures are significantly limited by concentration, i.e., the higher the concentration of the acid in the solution the higher the extraction efficiency at optimized conditions. Studies done by Garrett et al. [157] showed that the acetic acid extraction from fermentation broth decreased from 93% at an initial acid concentration of 92.4 g/L to around 81% at an initial concentration of 10 g/L. These studies showed that there was a significant effect of initial acid concentration on the amount of carbon dioxide used for extraction. The studies also showed that the carboxylic acid yield as a function of amount of CO2 used for extraction decreased from around 6.5 mg acetic acid extracted per g CO2 used when the initial acetic acid concentration was 92.4 g/L to as low as 0.5 mg acetic acid extracted per g CO2 used at an initial acetic acid concentration of 10 g/L. The concentration effect is also applicable, in a slightly different manner, in the case of ion exchange resin-mediated separation. The concentration of acid ions in the fermentation broth is a function of the pH of the fermentation and with an increased fermentation pH above the pKa of the carboxylic acid, there is a decreased concentration of the acids in the form of ions versus that in the form of inactive salts. For example, at pH 5.5, approximately 95% of the lactic acid produced through fermentation exists in the form of sodium lactate, which has a lower tendency to bind to the ion exchange resin, whereas only 5% exists as lactate ions that can be extracted using ion exchange resin. This usually results in the requirement of an extra acidification step in the product recovery loop to extract all the lactic acid from the fermentation broth. Hence, a concentration step is necessary before product separation (as shown in Figure 3), which can vary between acidification in the case of ion exchange-mediated separation and evaporation or membranes in the case of reactive extraction or precipitation (an example of a membrane is the hollow fibers used concurrently with pertraction for separation from fermentation broths).
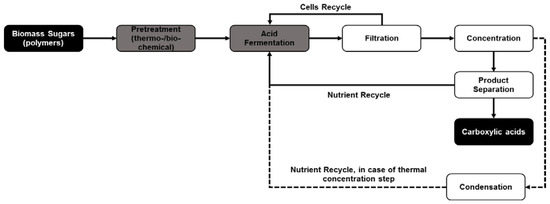
Figure 3.
Efficient processing pathway outlining different stages in biomass conversion with fermentation and in situ product separation for biorefinery applications. (─ shows that processing pathway that uses direct nutrient cycle due to non-thermal separation processes while - - shows the nutrient recycle loop in processing pathways that uses thermal separation processes such as evaporation or distillation requiring an extra processing step before nutrient recycle).
4.4. Separation Using Electrodialysis
Another important technology for in situ product recovery of carboxylic acids is electrodialysis using an electrical gradient to facilitate ionic transport towards electrodes for optimal acid removal. Several electrodialysis processes have been tested for acid recovery including conventional electrodialysis: electro-metathesis, electro-ion substitution, electro-deionization, bipolar membrane electrodialysis, etc. [158]. While industries currently use crystallization and distillation techniques for product recovery, which are inefficient and can be energy-intensive and in some cases result in product loss or transformation [159], electrodialysis can result in high acid purity, as shown in the studies elucidated in Table 1. However, electrodialysis is also concentration-limiting but not in a similar manner to the other techniques discussed above. Higher acid and cell loading in the fermenter effluent can result in concentration polarization during electrodialysis after a certain time (depending on the current density), resulting in inefficient separation performance [158]. Hence, the concentration step from Figure 3 would be ill-suited to using electrodialysis for acid separation. One of the primary disadvantages of electrodialysis is the electricity requirements, which can result in high processing costs. For example, studies have indicated that the power requirements for lactic acid separation using electrodialysis can vary from 0.21 to 0.71 kWh/kg acid produced under optimized conditions for maximum lactate recovery [160,161]. However, even with the membrane fouling problems and electricity requirements, electrodialysis has shown significant promise (through improvements in membrane quality) in in situ extraction of carboxylic acid from fermentation broths [133,159]. An extension of electrodialysis known as reverse electro-enhanced electrodialysis (REED) has been successfully used to minimize fouling problems during the in situ extraction of butyric acid [132] and lactic acid [162]. As indicated previously, both these carboxylic acids have a significant inhibitory effect on the microbial productivity during fermentation at high concentrations and require in situ removal. The REED process is equipped with a mechanism that allows for polarity reversal during times of imposed electrical potential gradient that will help clear concentration polarization during continuous operation, allowing for Donnan dialysis operation [163]. Such systems can significantly reduce the electricity costs during operation since they reduce the time of less-than optimal performance of the electrodialytic separation during continuous operation. However, studies are currently being done to further reduce processing costs by developing low-cost, robust materials to be used as electrodes and membranes during electrodialysis.
5. Conclusions and Future Perspectives
In this review we have discussed different microbes and in situ product recovery processes for carboxylic acid production from lignocellulosic biomass for biorefinery applications. Only limited focus was devoted to the different types of fermentation processes used since a number of studies have concluded that, for most biochemical processes, continuous fermentation (by controlling feeding rate and retention time) has significant advantages over fed-batch or batch fermentations [90,164]. The production of specific carboxylic acids usually necessitates specific pure cultures. Pure cultures are, however, easily affected by microbial contamination, resulting in a requirement for high sterility, which can add to the production costs. Using a mixed microbial consortium such as a rumen to develop a selective culture for the production of carboxylic acids works well without the need for strain engineering of specific pure microbes. The presence of cellulose- and hemicellulose-degrading strains in mixed cultures such as a mixed rumen culture also serves as an advantage, resulting in minimizing or even eliminating the need for external enzymes in biorefineries, which can further reduce processing costs [13]. Carbohydrate and protein metabolism in the rumen during cellulose fermentation was studied to understand the effect of fermentation conditions on the sugar-degrading bacteria; such studies indicated an increase in the concentration of cellulolytic bacteria when the system was left unperturbed [165]. However, further studies are required to fully understand the nature of the enzymes and microbial populations active during the anaerobic fermentation of biomass using mixed microbial cultures. Such studies can provide scientific input for increasing validation and data reproducibility and can pave the way for the application of mixed culture fermentations in biorefineries.
Apart from optimization of the microbial strains (or consortia) used during fermentation, further improvements are required to facilitate in situ product recovery during fermentation. We have discussed the advantages and disadvantages of different separation processes for the in situ separation of carboxylic acids from fermentation effluent. The ideal separation process will sufficiently and selectively separate the carboxylic acid while having little or no effect on the fermentation process itself. There is sufficient evidence that in situ product recovery has a positive effect on both microbial productivity (through the elimination or minimization of product inhibition) and product purity (through optimization for selective removal of the desired carboxylic acid product), thereby producing a carboxylic acid intermediate that can be directly upgraded to fuels and/or used as chemicals. However, it is important to gain further knowledge on how to integrate the product recovery with the fermentation processes, especially related to the effects of product concentrations and membrane fouling. The development and optimization of such in situ product separation processes can enhance the quality of the product produced from the biorefineries while reducing the overall cost to the biorefineries.
Acknowledgments
This work was done with support from the United States Department of Agriculture (USDA)-National Institute of Food and Agriculture (NIFA) under the Northwest Advanced Renewables Alliance (NARA) (Grant# 2011–68005–30416) and Denmark’s Energinet program ForskEL-funded XEL2Gas project (#12437).
Author Contributions
All the authors contributed equally to the review article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lange, J.-P. Lignocellulose conversion: An introduction to chemistry, process and economics. Biofuels Bioprod. Bioref 2007, 1, 39–48. [Google Scholar] [CrossRef]
- Baumann, I.; Westermann, P. Microbial production of short chain fatty acids from lignocellulosic biomass: Current processes and market. BioMed Res. Intl. 2016, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Worrell, E.; Phylipsen, D.; Einstein, D.; Martin, N. Energy Use and Energy Intensity of the US Chemical Industry. US DOE Report LBNL-44314; 2000. Available online: http://ateam.lbl.gov/PUBS/doc/LBNL-44314.pdf (accessed on 8 November 2016).
- Micromarket monitor. North America Acetic Acid Market by Application (Vinyl Acetate Monomer (VAM), Purified Terephthalic Acid (PTA), Acetic Anhydride, Ester Solvents & Others) & by Country–Trends & Forecasts to 2019. Report Code AC 1086. 2015. Available online: http://www.micromarketmonitor.com/market/north-america-acetic-acid-9541396535.html?utm_source=NL-NAAAM&utm_medium=NL-NAAAM&utm_campaign=NL-NAAAM (accessed on 25 May 2015).
- Renewable Chemicals Market–Alcohols (Ethanol, Methanol), Biopolymers (Starch Blends, Regenerated Cellulose, PBS, bio-PET, PLA, PHA, bio-PE, and Others), Platform Chemicals & Others-Global Trends & Forecast to 2020. Report Code CH 2063. 2015. Available online: http://www.marketsandmarkets.com/Market-Reports/renewable-chemical-274.html (accessed on 1 October 2015).
- Brindle, F. American chemistry council: Global chemical production looks positive. Energy Glob. 2016. Available online: https://www.energyglobal.com/downstream/petrochemicals/13042016/american-chemistry-council-positive-growth-cpri-global-chemical-production-3023/ (accessed on 13 April 2016).
- Metzger, J.O.; Huttermann, A. Sustainable global energy supply based on lignocellulosic biomass from afforestation of degraded areas. Naturwissenschaften 2009, 96, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Kan, T.; Strezov, V.; Evans, T.J. Lignocellulosic biomass pyrolysis: a review of product properties and effects of pyrolysis parameters. Ren. Sust. Energy Rev. 2016, 57, 1126–1140. [Google Scholar] [CrossRef]
- Kumar, A.; Jones, D.D.; Hanna, M.A. Thermochemical biomass gasification: A review of the current status of the technology. Energies 2009, 2, 556–581. [Google Scholar] [CrossRef]
- Elliott, D.C.; Biller, P.; Ross, A.B.; Schmidt, A.J.; Jones, S.B. Hydrothermal liquefaction of biomass: Developments from batch to continuous process. Bioresour. Technol. 2015, 178, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, D.; Dasgupta, D.; Agrawal, D.; Kaul, S.; Adhikari, D.K.; Kurmi, A.K.; Arya, P.K.; Bangwal, D.; Negi, M.S. Fuels and chemicals from lignocellulosic biomass: An integrated biorefinery approach. Energy Fuels 2015, 29, 3149–3157. [Google Scholar] [CrossRef]
- Liao, J.C.; Mi, L.; Pontrelli, S.; Luo, S. Fuelling the future: Microbial engineering for the production of sustainable biofuels. Nat. Rev. Microbiol. 2016, 14, 288–304. [Google Scholar] [CrossRef] [PubMed]
- Murali, N.; Fernandez, S.; Ahring, B.K. Fermentation of wet-exploded corn stover for the production of volatile fatty acids. Bioresour. Technol. 2017, 227, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Raposo, F.; Borja, R.; Cacho, J.A.; Mumme, J.; Orupold, K.; Esteves, S.; Nogruel-Arias, J.; Picard, S.; Nielfa, A.; Scherer, P.; et al. First international comparative study of volatile fatty acids in aqueous samples by chromatographic techniques: Evaluating sources of error. Trends Anal. Chem. 2013, 51, 127–143. [Google Scholar] [CrossRef]
- Agler, M.T.; Wrenn, B.A.; Zinder, S.H.; Angenent, L.T. Waste to bioproducts conversion with undefined mixed cultures: the carboxylate platform. Trends Biotechnol. 2011, 29, 70–78. [Google Scholar] [CrossRef] [PubMed]
- GBIR Research. Acetic Acid Global Market to 2020-Surge in VAM and PTA Sectors in Asia Pacific to Drive Global demand. Report Code GBICH0082MR. 2013. Available online: http://www.gbiresearch.com/report-store/market-reports/archive/acetic-acid-global-market-to-2020-surge-in-vam-and-pta-sectors-in-asia-pacific-to-drive-global-demand (accessed on 1 February 2013).
- Yoneda, N.; Kusano, S.; Yasui, M.; Pujado, P.; Wilcher, S. Recent advances in processes and catalysts for the production of acetic acid. Appl. Catal. A 2001, 221, 253–265. [Google Scholar] [CrossRef]
- Wood, B.J.B. Microbiology of Fermented Foods, 2nd ed.; Springer: New York, NY, USA, 1998. [Google Scholar]
- Sengun, I.Y.; Karabiyikli, S. Importance of acetic acid bacteria in food industry. Food Control 2011, 22, 647–656. [Google Scholar] [CrossRef]
- Chavez, K.L.; Hess, D.W. A novel method of etching copper oxide using acetic acid. J. Electrochem. Soc. 2001, 148, G640–G643. [Google Scholar] [CrossRef]
- Gandini, A. Polymers from renewable resources: a challenge for the future of macromolecular materials. Macromolecules 2008, 41, 9491–9504. [Google Scholar] [CrossRef]
- Ganster, J.; Fink, H.-P. Cellulose and cellulose acetate. In Bio-Based Plastics: Materials and Applications, 1st ed.; Kabasci, S., Ed.; Wiley: West Sussex, UK, 2014; pp. 35–62. [Google Scholar]
- Weissermel, K.; Arpe, H.-J. Industrial Organic Chemistry, 4th ed.; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Luck, E.; Jager, M. Propionic acid. In Antimicrobial Food Additives, 2nd ed.; Luck, E., Jager, M., Eds.; Springer: Berlin, Germany, 1997; pp. 145–151. [Google Scholar]
- Ali, S.H.; Tarakmah, A.; Merchant, S.Q.; Al-Sahhaf, T. Synthesis of esters: Development of the rate expression for the Dowex 50 Wx8-400 catalyzed esterification of propionic acid with 1-propanol. Chem. Eng. Sci. 2007, 62, 3197–3217. [Google Scholar] [CrossRef]
- El-Shahawy, T.A.E.-G. Chemicals with a natural reference for controlling water hyacinth, Eichhornia crassipes (Mart.) Solms. J. Plant Protec. Res. 2015, 55, 294–300. [Google Scholar] [CrossRef]
- Ihre, H.; Hult, A.; Soderlind, E. Synthesis, characterization and 1H NMR self-diffusion studies of dendritic aliphatic polyesters based on 2,2-bis (hydroxymethyl) propionic acid and 1,1,1-Tris(hydroxyphenyl)ethane. J. Am. Chem. Soc. 1996, 118, 6388–6395. [Google Scholar] [CrossRef]
- Propionic Acid & Derivatives Market by Applications (Animal Feed & Grain Preservatives, Food Preservatives, Herbicides, Cellulose Acetate Propionate) & Geography–Global Trends & Forecasts to 2018. Report Code CH 1533. 2013. Available online: http://www.marketsandmarkets.com/PressReleases/propionic-acid-derivatives.asp (accessed on 1 July 2013).
- Playne, M.J. Propionic and butyric acids. In Comprehensive Biotechnology; Moo-Young, M., Ed.; Pergamon Press: Oxford, UK, 1985. [Google Scholar]
- Jha, A.K.; Li, J.; Yuan, Y.; Baral, N.; Ai, B. A review on bio-butyric acid production and its optimization. Int. J. Agric. Biol. 2014, 16, 1019–1024. [Google Scholar]
- Entin-Meer, M.; Rephaeli, A.; Yang, X.; Nudelman, A.; VandenBerg, S.R.; Haas-Kogan, D.A. Butyric acid prodrugs are histone deacetylase inhibtiors that show antineoplastic activity and radiosensitizing capacity in the treatment of malignant gliomas. Mol. Cancer Ther. 2005, 4, 1952–1961. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.W.; Yamazaki, H. Natural flavours production: A biotechnological approach. Trends Biotechnol. 1986, 4, 264–268. [Google Scholar] [CrossRef]
- Butyric Acid Market by Application (Animal Feed, Chemical Intermediate, Foods & Flavors, Pharmaceuticals, Perfumes, Others), by Type (Synthetic Butyric Acid, Renewable Butyric Acd) by Geography (APAC, North America, Europe, ROW)—Global Analysis and Forecast to 2020. Report Code CH 3662. 2015. Available online: http://www.marketsandmarkets.com/Market-Reports/butyric-acid-market-76962011.html (accessed on 8 January 2015).
- Holten, C.H.; Muller, A.; Rehbinder, D. Lactic Acid; International Research Association; Verlag Chemie: Copenhagen, Denmark, 1971. [Google Scholar]
- John, R.P.; Nampoothiri, K.M.; Pandey, A. Fermentative production of lactic acid from biomass: An overview on process developments and future perspectives. Appl. Microb. Biotechnol. 2007, 74, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Datta, R.; Tsai, S.-P.; Bonsignore, P.; Moon, S.-H.; Frank, J.R. Technological and economical of poly (lactic acid) and lactic acid derivatives. FEMS Mircob. Rev. 1995, 16, 221–231. [Google Scholar] [CrossRef]
- Datta, R.; Henry, M. Lactic acid: Recent advances in products, processes and technologies—A review. J. Chem. Technol. Biotechnol. 2006, 81, 1119–1129. [Google Scholar] [CrossRef]
- Lactic Acid Market by Application (Biodegradable Polymer, Food & Beverage, Personal Care & Pharmaceutical) & Polylactic Acid Market by Application (Packaging, Agriculture, Automobile, Electronics, Textile), & by Geography-Global Trends & Forecasts to 2020. 2015. Available online: http://www.rnrmarketresearch.com/lactic-acid-market-by-application-biodegradable-polymer-food-beverage-personal-care-pharmaceutical-polylactic-acid-market-by-application-packaging-agriculture-automobile-electronics-tex-market-report.html (accessed on 12 March 2015).
- Dence, C.W.; Lin, S.Y. General structural features of lignin. In Methods in Lignin Chemisry; Lin, S.Y., Dence, C.W., Eds.; Springer: Berlin/Heidelberg, German, 1992. [Google Scholar]
- Cao, S.; Pu, Y.; Studer, M.; Wyman, C.; Ragauskas, A.J. Chemical transformations of Populus trichocarpa during dilute acid pretreatment. RSC Adv. 2012, 2, 10925–10936. [Google Scholar] [CrossRef]
- Tejado, A.; Pena, C.; Labidi, J.; Echeverria, J.M.; Mondragon, I. Physico-chemical characterization of lignins from different sources for use in phenol-formaldehyde resin synthesis. Bioresour. Technol. 2007, 98, 1655–1663. [Google Scholar] [CrossRef] [PubMed]
- Ahring, B.K.; Biswas, R.; Ahamed, A.; Teller, P.J.; Uellendahl, H. Making lignin accessible for anaerobic digestion by wet-explosion pretreatment. Bioresour. Technol. 2015, 175, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Schiel-Bengelsdorf, B.; Durre, P. Pathway engineering and synthetic biology using acetogens. FEBS Lett. 2012, 586, 2191–2198. [Google Scholar] [CrossRef] [PubMed]
- Ragsdale, S.W.; Pierce, E. Acetogenesis and the Wood-Ljundahl pathway of CO2 fixation. Biochim. Biophys. Acta. 2008, 1784, 1873–1898. [Google Scholar] [CrossRef] [PubMed]
- Ni, B.J.; Liu, H.; Nie, Y.Q.; Zeng, R.J.; Du, G.C.; Chen, J.; Yu, H.Q. Coupling glucose fermentation and homoacetogenesis for elevated acetate production: experimental and mathematical approaches. Biotechnol. Bioeng. 2010, 108, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Schuchmann, K.; Muller, V. Autotrophy at the thermodynamic limit of life a model for energy conservation in acetogenic bacteria. Natur. Rev. Microbiol. 2014, 12, 809–821. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.A.; Zengler, K.; Edwards, E.A.; Mahadevan, R.; Stephanopoulos, G. Investigating Moorella thermoacetica metabolism with a genome-scale constraint-based metabolic model. Integr. Biol. 2015, 7, 869–882. [Google Scholar] [CrossRef] [PubMed]
- Drake, H.L.; Daniel, S.L. Physiology of the thermophilic acetogen Moorella thermoacetica. Res. Microbiol. 2004, 155, 869–883. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Isern, N.G.; Ewing, R.J.; Liyu, A.V.; Sears, J.A.; Knapp, H.; Iversen, J.; Sisk, D.R.; Ahring, B.K.; Majors, P.D. New generation NMR bioreactor coupled with high-resolution NMR spectroscopy leads to novel discoveries in Moorella thermoacetica metabolic profiles. Envrion. Biotechnol. 2014, 98, 8367–8375. [Google Scholar] [CrossRef] [PubMed]
- Balasubramaniam, N.; Kim, J.S.; Lee, Y.Y. Fermentation of xylose into acetic acid by Clostridium thermoaceticum. J. Appl. Biochem. Biotechnol. 2001, 367–376. [Google Scholar] [CrossRef]
- Ehsanipour, M.; Suko, A.V.; Bura, R. Fermentation of lignocellulosic sugars to acetic acid by Moorella thermoacetica. J. Ind. Microbiol. Biotechnol. 2016, 43, 807–816. [Google Scholar] [CrossRef] [PubMed]
- Gaddy, J.L.; Clausen, E.C. Clostridium ljungdahlii, an anaerobic ethanol and acetate producing microorganism. U.S. Patent 5,173,429, 1992. [Google Scholar]
- Phillips, J.R.; Clausen, E.C.; Gaddy, J.L. Synthesis gas as substrate for the biological production of fuels and chemicals. Appl. Biochem. Biotechnol. 1994, 45, 145–157. [Google Scholar] [CrossRef]
- Heise, R.; Muller, V.; Gottschalk, G. Sodium dependence of acetate formation by the acetogenic bacterium Acetobacterium wooddii. J. Bacteriol. 1989, 171, 5473–5478. [Google Scholar] [CrossRef] [PubMed]
- Muller, V.; Bowien, S. Differential effects of sodium ions on motility in the homoacetogeniic bacteria Acetobacterium wooddii and Sporomusa sphaeroides. Arch. Microbiol. 1995, 164, 363–369. [Google Scholar] [CrossRef]
- Tracy, B.P.; Jones, S.W.; Fast, A.G.; Indurthi, D.C.; Papoutsakis, E.T. Clostridia: The importance of their exceptional substrate and metabolite diversity for biofuel and biorefinery applications. Curr. Opin. Biotechnol. 2012, 23, 364–381. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Ljungdahl, L.G. Electron-transport system in acetogens. In Biochemistry and Physiology of Anaerobic Bacteria; Ljungdahl, L.G., Adams, M.W., Barton, L., Ferry, J.G., Johnson, M.K., Eds.; Springer: New York, NY, USA, 2003. [Google Scholar]
- Huang, H.; Wang, S.; Moll, J.; Thauer, R.K. Electron bifurcation involved in the energy metabolism of the acetoggenic bacterium Moorella thermoacetica growing on glucose or H2 plus CO2. J. Bacteriol. 2012, 194, 3689–3699. [Google Scholar] [CrossRef] [PubMed]
- Mock, J.; Wang, S.; Huang, H.; Kahnt, J.; Thauer, R.K. Evidence for a hexaheterometric methylenetetrahydrofolate reductase in Moorella thermoacetica. J. Bacteriol. 2014, 196, 3303–3314. [Google Scholar] [CrossRef] [PubMed]
- Kopke, M.; Held, C.; Hujer, S.; Liesegang, H.; Wiezer, A.; Wollherr, A.; Ehrenreich, A.; Liebl, W.; Gottschalk, G.; Durre, P. Clostridium ljungdahlii represents a microbial production platform based on syngas. Proc. Natl. Acad. Sci. USA 2010, 107, 13087–13092. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yang, H.; Yang, F.; Ma, Y. Current progress on butyric acid production by fermentation. Curr. Microbiol. 2009, 59, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Yang, S.T. Effects of pH on metabolic pathway shift in fermentation of xylose by Clostridium tyrobutyricum. J. Biotechnol. 2004, 110, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhu, Y.; Yang, S.T. Construction and characterization of ack deleted mutant of Clostridium tyrobutyricum for enhanced butyric acid and hydrogen production. Biotechnol. Prog. 2006, 22, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Lutke-Eversloh, T.; Bahl, H. Metabolic engineering of Clostridium acetobutylicum: Recent advances to improve butanol production. Curr. Opin. Biotechnol. 2011, 22, 634–647. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jang, Y.S.; Choi, S.J.; Im, J.A.; Song, H.; Cho, J.H.; Seung, D.Y.; Papoutsakis, T.; Bennett, G.N.; Lee, S.Y. Metabolic engineering of Clostridium acetobutylicum ATCC 824 for isopropanol-butanol-ethanol fermentation. App. Environ. Microbiol. 2012, 78, 1416–1423. [Google Scholar] [CrossRef] [PubMed]
- Wiegel, J.; Kuk, S.U.; Kohring, G.W. Clostridium thermobutyricum sp. nov., a moderate thermophile isolated from a cellulolytic culture, that produces butyrate as the major product. Int. J. Sys. Bacteriol. 1989, 39, 199–204. [Google Scholar] [CrossRef]
- Zhang, C.H.; Ma, Y.J.; Yang, F.X.; Liu, W.; Zhang, Y.-D. Optimization of medium composition for butyric acid production by Clostridium thermobutyricum using response surface methodology. Bioresour. Technol. 2009, 100, 4284–4288. [Google Scholar] [CrossRef] [PubMed]
- Bahl, H.; Gottschalk, G. Parameters affecting solvent production by Clostridium acetobutylicum in continuous culture. Biotechnol. Bioeng. Symp. 1984, 14, 215–233. [Google Scholar]
- Vandak, D.; Tomaska, M.; Zigova, J.; Sturdik, E. Effect of growth supplements and whey pretreatment on butyric-acid production by Clostridium butyricum. World J. Microbiol. Biotechnol. 1995, 11, 363. [Google Scholar] [CrossRef] [PubMed]
- Ueki, A.; Akasaka, H.; Suzuki, D.; Ueki, K. Paludibacter propionicigenes gen. nov., sp. nov., a novel strictly anaerobic, gram-negative, propionate-producing bacterium isolated from plant residue in irrigated rice-field soil in Japan. Int. J. Sys. Evol. Microbiol. 2006, 56, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Narihiro, T.; Kim, N.K.; Mei, R.; Nobu, M.K.; Liu, W.-T. Microbial community analysis of anaerobic reactors treating soft drink wastewater. PLoS ONE 2015, 10, e0119131. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.L.; Kuang, X.Z.; Shi, X.S.; Yuan, X.Z.; Guo, R.B. Paludibacter jiangxiensis sp. nov., a strictly anaerobic, propionate-producing bacterium isolated from rice paddy field. Arch. Microbiol. 2014, 196, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Oshio, S.; Tahata, I.; Minato, H. Effect of diets differing in ratios of roughage to concentrate on microflora in the rumen of heifers. J. Gen. Appl. Microbiol. 1987, 33, 99–111. [Google Scholar] [CrossRef]
- Paynter, M.J.; Elsdent, S.R. Mechanism of propionate formation by Selenomonas ruminatium, a rumen microorganism. Microbiology 1970, 61, 1–7. [Google Scholar]
- Akedo, M.; Cooney, C.L.; Sinskey, A.J. Direct demonstration of lactate-acrylate interconversion in Clostridium propionicum. Natur. Biotechnol. 1983, 1, 791–794. [Google Scholar] [CrossRef]
- Johns, A.T. The mechanism of propionic acid formation by Clostridium propionicum. J. Gen. Microbiol. 1952, 6, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, B.A.; Stowers, C.C.; Pham, V.; Cox, B.M. The production of propionic acid, propanol and propylene via sugar fermentation: an industrial perspective on the progress, technical challenges and future outlook. Green Chem. 2014, 16, 1066–1076. [Google Scholar] [CrossRef]
- Himmi, E.H.; Bories, A.; Boussaid, A.; Hassani, L. Propionic acid fermentation of glycerol and glucose by Propionibacterium acidipropionici and Propionibacterium freudenreichii ssp. shermanii. Appl. Microbiol. Biotechnol. 2000, 53, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Stine, A.; Zhang, M.; Ro, S.; Clendennen, S.; Shelton, M.C.; Tyo, K.E.J.; Broadbelt, L.J. Exploring de novo metabolic pathways from pyruvate to propionic acid. Biotechnol. Prog. 2016, 32, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Holzapfel, W.H.; Wood, B.J.B. Lactic Acid Bacteria—Biodiversity and Taxonomy; John Wiley & Sons, Wiley Blackwell Publishing: Oxford, UK, 2014. [Google Scholar]
- Claesson, M.J.; van Sinderen, D.; O’Toole, P.W. The genus Lactobacillus–a genomic basis for understanding its diversity. FEMS Microbiol. Lett. 2007, 269, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Garrett, B.G.; Srinivas, K.; Ahring, B.K. Performance and stability of Amberlite IRA-67 ion exchange resin for product extraction and pH control during homolactic fermentation of corn stover sugars. Biochem. Eng. J. 2015, 94, 1–8. [Google Scholar] [CrossRef]
- Nolasco-Hipolito, C.; Matsunaka, T.; Kobayashi, G.; Sonomoto, K.; Ishizaki, A. Synchronized fresh cell bioreactor system for continuous l-(+)-lactic acid production using Lactococcus lactis IO-1 in hydrolysed sago starch. J. Biosci. Bioeng. 2002, 93, 281–287. [Google Scholar] [CrossRef]
- Kadam, S.R.; Patil, S.S.; Bastawde, K.B.; Khire, J.M.; Gokhale, D.V. Strain improvement of Lactobacillus delbruecki NCIM 2365 for lactic acid production. Proc. Biochem. 2006, 41, 120–126. [Google Scholar] [CrossRef]
- Tango, M.; Ghaly, A. A continuous lactic acid production system using an immobilized packed bed of Lactobacillus helveticus. Appl. Microbiol. Biotechnol. 2002, 58, 712–720. [Google Scholar] [PubMed]
- Hujanen, M.; Linko, S.; Linko, Y.-Y.; Leisola, M. Optimization of media and cultivation conditions for L (+)(S)-lactic acid production by Lactobacillus casei NRRL B-441. Appl. Microbiol. Biotechnol. 2001, 56, 126–130. [Google Scholar] [CrossRef] [PubMed]
- Romero-Garcia, S.; Hernandez-Bustos, C.; Merino, E.; Gosset, G.; Martinez, A. Homolactic fermentation from glucose and cellobiose using Bacillus subtilis. Microb. Cell Fac. 2009, 8, 23. [Google Scholar] [CrossRef] [PubMed]
- Payot, T.; Chemaly, Z.; Fick, M. Lactic acid production by Bacillus coagulans–kinetic studies and optimization of culture medium for batch and continuous fermentations. Enzym. Microb. Technol. 1999, 24, 191–199. [Google Scholar] [CrossRef]
- Pol, E.C.; Eggink, G.; Weusthuis, R.A. Production of l(+)-lactic acid from acid pretreated sugarcane bagasse using Bacillus coagulans DSM2314 in a simultaneous saccharification and fermentation strategy. Biotechnol. Biofuels 2016, 9, 248. [Google Scholar] [CrossRef] [PubMed]
- Ahring, B.K.; Traverso, J.J.; Murali, N.; Srinivas, K. Continuous fermentation of clarified corn stover hydrolysate for the production of lactic acid at high yield and productivity. Biochem. Engg. J. 2016, 109, 162–169. [Google Scholar] [CrossRef]
- Zhou, X.; Ye, L.; Wu, J.C. Efficient production of L-lactic acid by newly isolated thermophilic Bacillus coagulans WCP 10–4 with high glucose tolerance. Appl. Microbiol. Biotechnol. 2013, 97, 4309–4314. [Google Scholar] [CrossRef] [PubMed]
- Baranofsky, J.J.; Schreurs, W.J.; Kashket, E.R. Uncoupling by acetic acid limits growth of and acetogenesis by Clostridium thermoaceticum. Appl. Environ. Microbiol. 1984, 48, 1134–1139. [Google Scholar]
- Schwartz, R.D.; Keller, F.A. Isolation of a strain of Clostridium thermoaceticum capable of growth and acetic acid production at pH 4.5. Appl. Environ. Microbiol. 1982, 43, 117–123. [Google Scholar] [PubMed]
- Paredes, C.J.; Alsaker, K.V.; Papoutsakis, E.T. A comparative genomic view of clostridial sporulation and physiology. Nat. Rev. Microbiol. 2005, 3, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.S.; Bumbaca, D.; Kosman, J.; Setlow, P.; Jedrzejas, M.J. Structure of a protein-DNA complex essential for DNS protection in spores of Bacillus spores. Proc. Nat. Acad. Sci. USA 2008, 105, 2806–2811. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Zheng, H.; Gu, Y.; Zhao, J.; Zhang, W.; Yang, Y.; Wang, S.; Zhao, G.; Yang, S.; Jiang, W. Comparative genomic and transcriptomic analysis revealed genetic characteristics related to solvent formation and xylose utilization in Clostridium acetobutylicum EA 2018. BMC Genom. 2011, 12, 93. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Jarboe, L.R. Metabolic engineering of biocatalysts for carboxylic acids production. Comput. Struct. Biotechnol. J. 2012, 3, e201210011. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Chokhawala, H.A.; Nadler, D.C.; Blanch, H.W.; Clark, D.S. Binding modules alter the activity of chimeric cellulases: Effects of biomass pretreatment and enzyme source. Biotechnol. Bioeng. 2010, 107, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Hackmann, T.J.; Spain, J.N. Invited review: Ruminant ecology and evolution: Perspectives useful to ruminant livestock research and production. J. Dairy Sci. 2010, 93, 1320–1324. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, J.; Forbes, J.M.; France, J. Quantitative Aspects of Ruminant Digestion and Metabolism; CABI Publishing: Cambridge, MA, USA, 1994. [Google Scholar]
- Saleem, F.; Bouatra, S.; Guo, A.C.; Psychogios, N.; Mandal, R.; Dunn, S.M.; Ametaj, B.N.; Wishart, D.S. The bovine ruminal fluid metabolome. Metabolomics 2013, 9, 360–378. [Google Scholar] [CrossRef]
- Ahring, B.K. Perspectives for anaerobic digestion. Adv. Biochem. Eng. Biotechnol. 2003, 81, 1–30. [Google Scholar] [PubMed]
- Mah, R.A.; Ward, D.M.; Baresi, L.; Glass, T.L. Biogenesis of methane. Annu. Rev. Microbiol. 1977, 31, 309–341. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.A.; Johnson, D.E. Methane emissions from cattle. J. Anim. Sci. 1995, 73, 2483–2492. [Google Scholar] [CrossRef] [PubMed]
- Schulman, M.D.; Valentino, D. Factors influencing rumen fermentation: Effect of hydrogen on formation of propionate. J. Dairy Sci. 1976, 59, 1444–1451. [Google Scholar] [CrossRef]
- Miller, T.L.; Wolin, M.J. Inhibition of growth of methane-producing bacteria of the ruminant forestomach by hydroxymethylglutaryl~ SCoA reductase inhibitors. J. Dairy Sci. 2001, 84, 1445–1448. [Google Scholar] [CrossRef]
- Van Kessel, J.A.S.; Russell, J.B. The effect of pH on ruminal methanogenesis. FEMS Microbiol. Ecol. 1996, 20, 205–210. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Wang, J.; Meng, L. Effects of volatile fatty acid concentrations on methane yield and methanogenic bacteria. Biomass Bioenergy 2009, 33, 848–853. [Google Scholar] [CrossRef]
- Hess, M.; Sczyrba, A.; Egan, R.; Kim, T.W.; Chokhawala, H.; Schroth, G.; Luo, S.; Clark, D.S.; Chen, F.; Zhang, T.; et al. Metagenomic discovery of biomass-degrading genes and genomes from cow rumen. Science 2011, 331, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Weimer, P.J. Manipulating ruminal fermentation: a microbial ecological perspective. J. Anim. Sci. 1998, 76, 3114–3122. [Google Scholar] [CrossRef] [PubMed]
- Perret, S.; Casalot, L.; Fierobe, H.-P.; Tardif, C.; Sabathe, F.; Belaich, J.-P.; Belaich, A. Production of heterologous and chimeric scaffoldins by Clostridium acetobutylicum ATCC 824. J. Bacteriol. 2004, 186, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Le Van, T.D.; Robinson, J.A.; Ralph, J.; Greening, R.C.; Smolenski, W.J.; Leedle, J.A.; Schaefer, D.M. Assessment of reductive acetogenesis with indigenous ruminal bacterium populations and Acetitomaculum ruminis. Appl. Environ. Microbiol. 1998, 64, 3429–3436. [Google Scholar] [PubMed]
- Lopez, S.; Valdes, C.; Newbold, C.J.; Wallace, R.J. Influence of sodium fumarate addition on rumen fermentation in vitro. Br. J. Nutr. 1999, 81, 59–64. [Google Scholar] [PubMed]
- Christy, P.M.; Gopinath, L.R.; Divya, D. A review on anaerobic decomposition and enhancement of biogas production through enzymes and microorganisms. Ren. Sus. Energ. Rev. 2014, 34, 167–173. [Google Scholar] [CrossRef]
- Rieu-Lesme, F.; Morva, B.; Collins, M.D.; Fonty, G.; Willems, A. A new H2/CO2-using acetogenic bacterium from the rumen: Description of Ruminococcus schinkii sp. nov. FEMS Microbiol. Lett. 2006, 140, 281–286. [Google Scholar]
- Leedle, J.A.; Greening, R.C. Postprandial changes in methanogenic and acidogenic bacteria in the rumens of steers fed high- or low-forage diets once daily. Appl. Environ. Microbiol. 1988, 54, 502–506. [Google Scholar] [PubMed]
- Davidson, C.A.; Rehberger, T.G. Characterization of Propionibacterium isolated from rumen of lactating dairy cows. Proc. Am. Soc. Microbiol. 1995, 1, 6. [Google Scholar]
- Russell, J.R.; Hino, T. Regulation of lactate production in Streptococcus bovis: A spiraling effect that contributes to rumen acidosis. J. Dairy Sci. 1985, 68, 1712–1721. [Google Scholar] [CrossRef]
- Wells, J.E.; Krause, D.O.; Callaway, T.R.; Russell, J.B. A bacteriocin-mediated antagonism by ruminal lactobacilli against Streptococcus bovis. FEMS Microbiol. Ecol. 1997, 22, 237–243. [Google Scholar] [CrossRef]
- Sharpe, M.E.; Latham, M.J.; Garvie, E.I.; Zirngibl, J.; Kandler, O. Two new species of Lactobacillus isolated from the bovine rumen, Lactobacillus ruminis sp. nov. and Lactobacillus vitulinus sp. nov. J. Gen. Microbiol. 1973, 77, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.G.; Withers, S.E. Changes in the rumen microbial population and its activities during the refaunation period after the reintroduction of ciliate protozoa into the rumen of defaunated sheep. Can. J. Microbiol. 1993, 39, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Hobson, P.N.; Stewart, C.S. The rumen microbial ecosystem; Blackie academic & professional, Chapman & Hall: London, UK, 1997. [Google Scholar]
- Bryant, M.P. Bacterial species of the rumen. Bacteriol. Rev. 1959, 23, 125–153. [Google Scholar] [PubMed]
- Doetsch, R.N.; Howard, B.H.; Mann, S.O. Physiological factors in the production of an iodophilic polysaccharide from pentose by a sheep rumen bacterium. J. Gen. Microbiol. 1957, 16, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Bryant, M.P.; Small, N. Characteristics of two new genera of anaerobic curved rods isolated from the rumen of cattle. J. Bacteriol. 1956, 72, 22–26. [Google Scholar] [PubMed]
- Dehority, B.A. Characterization of several bovine rumen bacteria isolated with a xylan medium. J. Bacteriol. 1966, 91, 1724–1729. [Google Scholar] [PubMed]
- Pind, P.F.; Angelidaki, I.; Ahring, B.K. Dynamics of the anaerobic process: effects of volatile fatty acids. Biotechnol. Bioeng. 2003, 82, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Ahring, B.K.; Sandberg, M.; Angelidaki, I. Volatile fatty acids as indicators of process imbalance in anaerobic digestors. Appl. Microbiol. Biotechnol. 1995, 43, 559–565. [Google Scholar] [CrossRef]
- Ahring, B.K. Status on science and application of thermophilic anaerobic digestion. Water Sci. Technol. 1994, 30, 241–249. [Google Scholar]
- Ahring, B.K.; Wstermann, P. Kinetics of butyrate, acetate, and hydrogen metabolism in a thermophilic, anaerobic, butyrate-degrading triculture. Appl. Environ. Microbiol. 1987, 53, 434–439. [Google Scholar] [PubMed]
- Angelidaki, I.; Ahring, B.K. Establishment and characterization of an anaerobic thermophilic 55 (deg) C enrichment culture degrading long-chain fatty acids. Appl. Environ. Microbiol. 1995, 61, 2442–2445. [Google Scholar] [PubMed]
- Baroi, G.N.; Skiadas, I.V.; Westermann, P.; Gavala, H.N. Continuous fermentation of wheat straw hydrolysate by Clostridium tyrobutyircum with in-situ acids removal. Wast. Biomass Valor. 2015, 6, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Y.; Zhang, X.; Feng, H.; Xu, T. In-situ combination of fermentation and electrodialysis with bipolar membranes for the production of lactic acid: continuous operation. Bioresour. Technol. 2013, 147, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.T.; Matsuoka, H.; Toda, K. Production and recovery of propionic and acetic acids in electrodialysis culture of Propionibacterium shermanii. J. Ferm. Bioeng. 1993, 75, 276–282. [Google Scholar] [CrossRef]
- Jones, R.J.; Massanet-Nicolau, J.; Guwy, A.; Premier, G.C.; Dinsdale, R.M.; Reilly, M. Removal and recovery of inhibitory volatile fatty acids from mixed acid fermentations by conventional electrodialysis. Bioresour. Technol. 2015, 189, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Zigova, J.; Sturdik, E.; Vandak, D.; Schlosser, S. Butyric acid production by Clostridium butyricum with integrated extraction and pertraction. Proc. Biochem. 1999, 34, 835–843. [Google Scholar] [CrossRef]
- Tik, N.; Bayraktar, E.; Mehmetoglu, U. In situ reactive extraction of lactic acid from fermentation media. J. Chem. Technol. Biotechnol. 2001, 76, 764–768. [Google Scholar] [CrossRef]
- Jin, Z.; Yang, S.-T. Extractive fermentation for enhanced propionic acid production from lactose by Propionibacterium acidipropionici. Biotechnol. Prog. 1998, 14, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Lee, Y.Y. Membrane-mediated extractive fermentation for lactic acid production from cellulosic biomass. Appl. Biochem. Biotechnol. 1997, 63, 435. [Google Scholar] [CrossRef] [PubMed]
- Von Frieling, P.; Schugerl, K. Recovery of lactic acid from aqueous model solutions and fermentation broths. Proc. Biochem. 1999, 34, 685–696. [Google Scholar] [CrossRef]
- Nelson, R.S.; Peterson, D.J.; Karp, E.M.; Beckham, G.T.; Salvachua, D. Mixed carboxylic acid production by Megasphaera elsdenii from glucose and lignocellulosic hydrolysate. Fermentation 2017, 3, 10. [Google Scholar] [CrossRef]
- Ataei, S.A.; Vasheghani-Farahani, E.J. In situ separation of lactic acid from fermentation broth using ion exchange resins. J. Ind. Microbiol. Biotechnol. 2008, 35, 1229–1233. [Google Scholar] [CrossRef] [PubMed]
- Monteagudo, J.M.; Aldavero, M. Production of L-lactic acid by Lactobacillus delbrueckii in chemostat culture using an ion exchange resins system. J. Chem. Technol. Biotechnol. 1999, 74, 627–634. [Google Scholar] [CrossRef]
- Bishai, M.; de, S.; Adhikari, B.; Banerjee, R. A platform technology of recovery of lactic acid from a fermentation broth of novel substrate Zizyphus oenophlia. Biotech 2015, 5, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Yousuf, A.; Bonk, F.; Bastidas-Oyandel, J.-R.; Schmidt, J.E. Recovery of carboxylic acids produced during dark fermentation of food waste by adsorption on Amberlite IRA-67 and activated carbon. Bioresour. Technol. 2016, 217, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Freeman, A.; Woodley, J.M.; Lilly, M.D. In-situ product removal as a tool for bioprocessing. Biotechnol. 1993, 11, 1007–1012. [Google Scholar] [CrossRef]
- Weil, J.R.; Dien, B.; Bothast, R.; Hendrickson, R.; Mosier, N.S.; Ladisch, M.R. Removal of fermentation inhibitors formed during pretreatment of biomass by polymeric adsorbents. Ind. Eng. Chem. Res. 2002, 41, 6132–6138. [Google Scholar] [CrossRef]
- Xu, T. Ion exchange membranes: State of their development and perspective. J. Membr. Sci. 2005, 263, 1–29. [Google Scholar] [CrossRef]
- Gonzalez, M.I.; Alvarez, S.; Riera, F.A.; Alvarez, R. Purification of lactic acid from fermentation broths by ion-exchange resins. Ind. Eng. Chem. Res. 2006, 45, 3242–3247. [Google Scholar] [CrossRef]
- Dethe, M.J.; Marathe, K.V.; Gaikar, V.G. Adsorption of lactic acid on weak base polymeric resins. J. Separ. Sci. Technol. 2006, 41, 2947–2971. [Google Scholar] [CrossRef]
- Uslu, H.; Inci, I.; Bayazit, S.S. Adsorption equilibrium data for acetic acid and glycolic acid onto Amberlite IRA-67. J. Chem. Eng. Data 2010, 55, 1295–1299. [Google Scholar] [CrossRef]
- Uslu, H.; Inci, I.; Bayazit, S.S.; Demir, G. Comparison of solid-liquid equilibrium data for the adsorption of propionic acid and tartaric acid from aqueous solution onto Amberlite IRA-67. Ind. Eng. Chem. Res. 2009, 48, 7767–7772. [Google Scholar] [CrossRef]
- Yang, S.T.; White, S.A.; Hsu, S.T. Extraction of carboxylic acids with tertiary and quarternary amines: effect of pH. Ind. Eng. Chem. Res. 1991, 30, 1335–1342. [Google Scholar] [CrossRef]
- Keshav, A.; Wasewar, K.L.; Chand, S. Reactive extraction of propionic acid using tri-n-octylamine, tri-n-butyl phosphate and aliquat 336 in sunflower oil as diluent. J. Chem. Technol. Biotechnol. 2009, 84, 484–489. [Google Scholar] [CrossRef]
- Vane, L.M. A review of pervaporation of product recovery from biomass fermentation processes. J. Chem. Technol. Biotechnol. 2005, 80, 603–629. [Google Scholar] [CrossRef]
- Cockrem, M.C.M. Process for Recovering Organic Acids From Aqueous Salt Solutions. U.S. Patent 5,522,995, 1996. [Google Scholar]
- Garrett, B.G.; Srinivas, K.; Ahring, B.K. Design and optimization of a semi-continuous high pressure carbon dioxide extraction system for acetic acid. J. Supercrit. Fluids 2014, 95, 243–251. [Google Scholar] [CrossRef]
- Huang, C.; Xu, T.; Zhang, Y.; Xue, Y.; Chen, G. Application of electrodialysis to the production of organic acids: state-of-the-art and recent developments. J. Membr. Sci. 2007, 288, 1–12. [Google Scholar] [CrossRef]
- Akerberg, C.; Zacchi, G. An economic evaluation of the fermentative production of lactic acid from wheat flour. Bioresour. Technol. 2000, 75, 119–126. [Google Scholar] [CrossRef]
- Habova, V.; Melzoch, K.; Rychtera, M.; Sekavova, B. Electrodialysis as a useful technique for lactic acid separation from a model solution and a fermentation broth. Desal. 2004, 162, 361–372. [Google Scholar] [CrossRef]
- Lee, E.G.; Moon, S.H.; Chang, Y.K.; Yoo, I.-K.; Chang, H.N. Lactic acid recovery using two-stage electrodialysis and its modelling. J. Membr. Sci. 1998, 145, 53–66. [Google Scholar]
- Prado-Rubio, O.A.; Jorgensen, S.B.; Jonsson, G. Reverse electro-enhanced dialysis for lactate recovery from a fermentation broth. J. Membr. Sci. 2011, 374, 20–32. [Google Scholar] [CrossRef]
- Garde, A. Production of Lactic Acid From Renewable Resources Using Electrodialysis for Product Recovery. Ph.D. Thesis, Technical University of Denmark, Copenhagen, Denmark, 2002. [Google Scholar]
- Dwidar, M.; Lee, S.; Mitchell, R.J. The production of biofuels from carbonated beverages. Appl. Energy 2012, 100, 47–51. [Google Scholar] [CrossRef]
- Boaro, A.A.; Kim, Y.M.; Konopka, A.E.; Callister, S.J.; Ahring, B.K. Integrated omics analysis for studying the microbial community response to a pH perturbation of a cellulose-degrading bioreactor culture. FEMS Microbiol. Ecol. 2014, 90, 802–815. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).