Metagenomic and Metabolomic Insights into Microbial Community Dynamics and Flavor Metabolite Formation in Novel Versus Traditional Strong-Flavor Daqu
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Instrumentation and Equipment
2.3. Methods
2.3.1. Preparation and Quality Index Analysis of Novel Daqu
2.3.2. Sample Preparation and Absolute Quantitative Metagenomic Sequencing
2.3.3. Sensory Evaluation of Baijiu
2.3.4. Detection of Volatile Flavor Compounds in Daqu and Baijiu Spirits
2.3.5. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Properties of Daqu and Jiupei Before and After Pit Entry
3.2. Metagenomic Characterization of Microbial Communities in Daqu
3.2.1. Microbial Community Diversity and Compositional Discrepancy Analysis
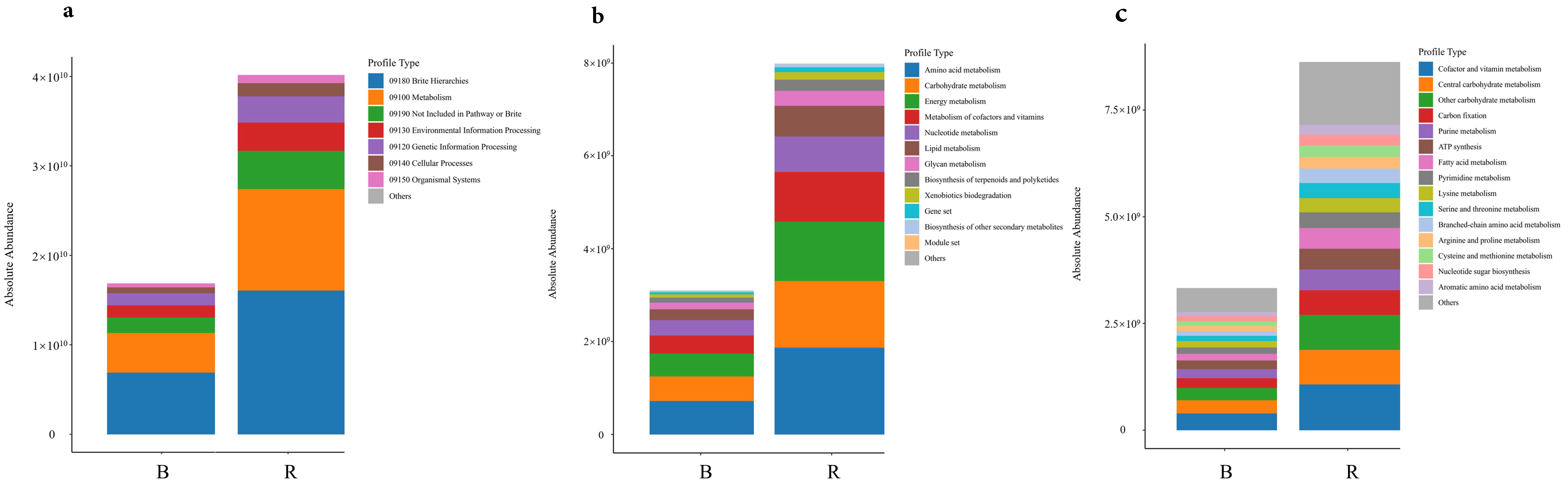
3.2.2. Functional Metabolic Prediction of Microbial Communities Based on KEGG Database
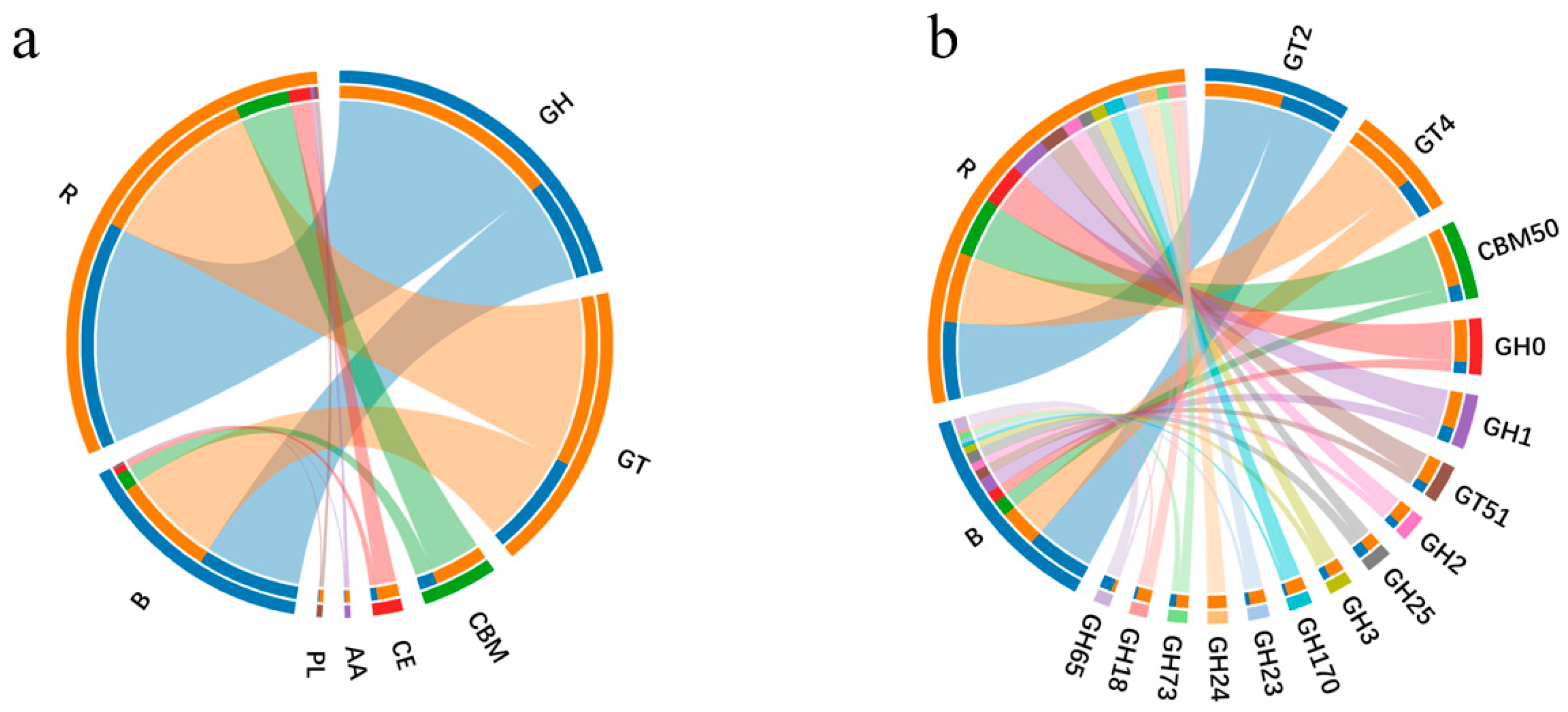
3.2.3. Carbohydrate-Active Enzymes Profiling Based on CAZy Database
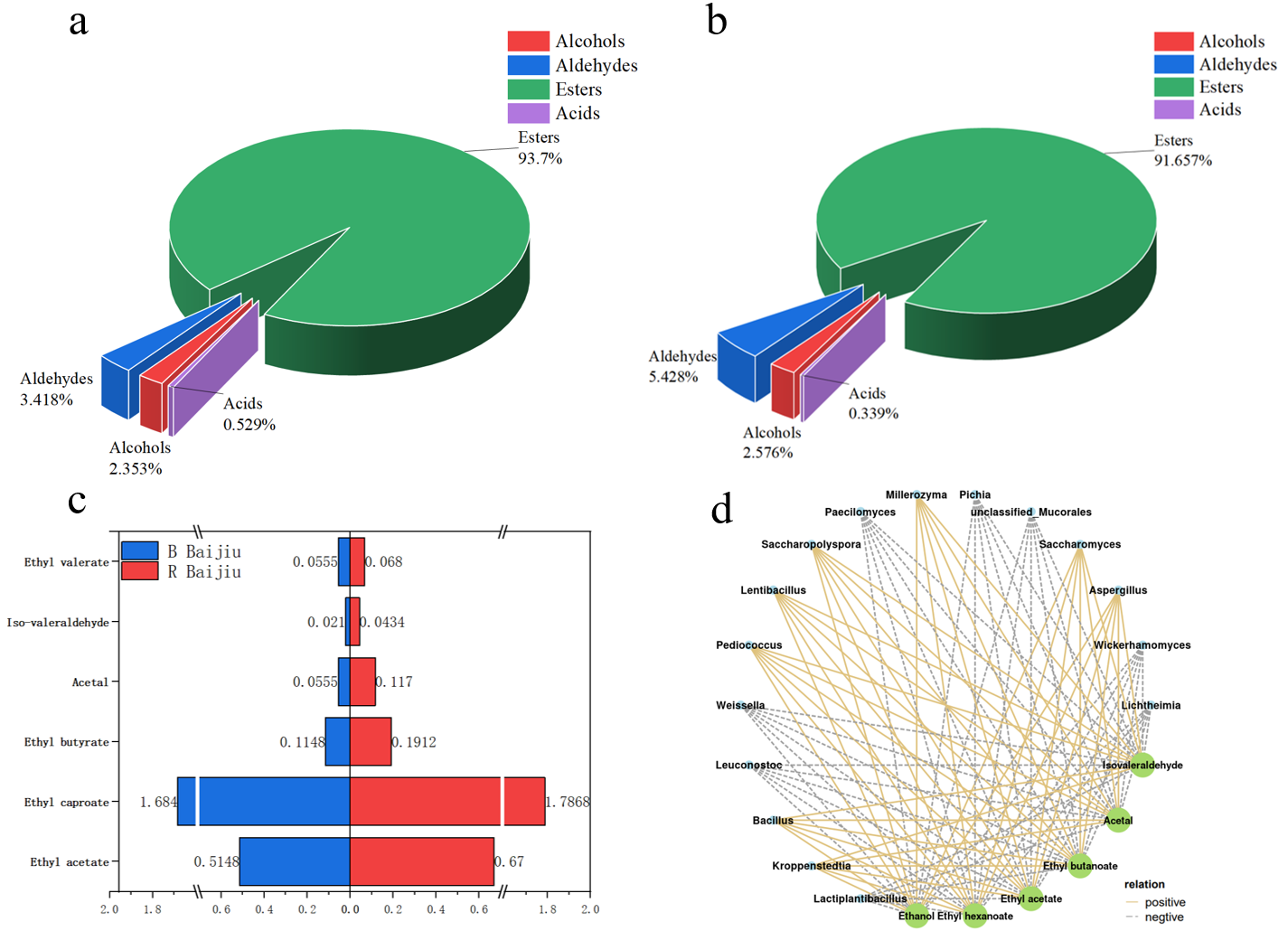
3.3. Comparative Analysis of Volatile Aroma Compounds in Daqu
3.4. Study and Analysis of Mechanism Underlying the Formation of Key Flavor Compounds in Chinese Baijiu
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HS-SPME-GC-MS | headspace solid-phase microextraction gas chromatography-mass spectrometry |
| CAZy | Carbohydrate-Active EnZyme |
| GHs | glycoside hydrolases |
| GTs | glycosyltransferases |
| GTDB | Genome Taxonomy Database |
| EI | Electron ionization |
| RDA | redundancy analysis |
| PCoA | Principal Coordinate Analysis |
| STAMP | Statistical Analysis of Metagenomic Profiles |
| LEfSe | Linear discriminant analysis effect size |
| PLs | Polysaccharide Lyases |
| AAs | Auxiliary Activities |
| CEs | Carbohydrate Esterases |
| CBMs | Carbohydrate-Binding Modules |
| EMP | Embden–Meyerhof–Parnas |
References
- Wang, R.; Lal, A.; Xin, C. Baijiu Blending Technology, 3rd ed.; Chemical Industry Press: Beijing, China, 2023. (In Chinese) [Google Scholar]
- Tang, J.; Wei, J.; Yang, Y.; Ge, X.; Zuo, Q.; Zhao, C.; Huang, Y. Spatio-Temporal Scales of the Chinese Daqu Microbiome: Heterogeneity, Niche Differentiation, and Assembly Mechanisms. Trends Food Sci. Technol. 2025, 156, 104832. [Google Scholar] [CrossRef]
- Ali, A.; Li, W.; Chen, X.; Patil, P.J.; Li, H.; Zhang, Y.; Li, X. Interdisciplinary Omics of Daqu and Baijiu: A Bibliometric Analysis (2011–2025). Food Biosci. 2025, 71, 107372. [Google Scholar] [CrossRef]
- Zhu, C.; Cheng, Y.; Zuo, Q.; Huang, Y.; Wang, L. Exploring the Impacts of Traditional Crafts on Microbial Community Succession in Jiang-Flavored Daqu. Food Res. Int. 2022, 158, 111568. [Google Scholar] [CrossRef]
- He, G.; Huang, J.; Wu, C.; Jin, Y.; Zhou, R. Bioturbation Effect of Fortified Daqu on Microbial Community and Flavor Metabolite in Chinese Strong-Flavor Liquor Brewing Microecosystem. Food Res. Int. 2020, 129, 108851. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, Z.; Qing, H.; Huang, M.; Xue, G.; Lu, X.; Che, Y.; Dong, Y.; Zhang, S.; Yu, J.; et al. Seasonal Influence on the Microbial Diversity and Flavor Substances in the Strong Flavor Daqu Fermentation. Food Chem. Mol. Sci. 2025, 11, 100332. [Google Scholar] [CrossRef]
- Wen, Z.; Wei, Y.-H.; Han, D.-Y.; Song, L.; Zhu, H.-Y.; Guo, L.-C.; Chen, S.-X.; Lin, B.; He, C.-J.; Guo, Z.-X.; et al. Deciphering the Role of Traditional Flipping Crafts in Medium-Temperature Daqu Fermentation: Microbial Succession and Metabolic Phenotypes. Curr. Res. Food Sci. 2025, 10, 101063. [Google Scholar] [CrossRef] [PubMed]
- Han, P.-J.; Luo, L.-J.; Han, Y.; Song, L.; Zhen, P.; Han, D.-Y.; Wei, Y.-H.; Zhou, X.; Wen, Z.; Qiu, J.-Z.; et al. Microbial Community Affects Daqu Quality and the Production of Ethanol and Flavor Compounds in Baijiu Fermentation. Foods 2023, 12, 2936. [Google Scholar] [CrossRef]
- Zheng, X.-W.; Han, B.-Z. Baijiu (白酒), Chinese Liquor: History, Classification and Manufacture. J. Ethn. Foods 2016, 3, 19–25. [Google Scholar] [CrossRef]
- Xu, Y.; Zhao, J.; Liu, X.; Zhang, C.; Zhao, Z.; Li, X.; Sun, B. Flavor Mystery of Chinese Traditional Fermented Baijiu: The Great Contribution of Ester Compounds. Food Chem. 2022, 369, 130920. [Google Scholar] [CrossRef]
- Wei, J.; Du, H.; Xu, Y. Revealing the Key Microorganisms Producing Higher Alcohols and their Assembly Processes during Jiang-Flavor Baijiu Fermentation. Food Biosci. 2024, 61, 104569. [Google Scholar] [CrossRef]
- Liu, W.-H.; Chai, L.-J.; Wang, H.-M.; Lu, Z.-M.; Zhang, X.-J.; Xiao, C.; Wang, S.-T.; Shen, C.-H.; Shi, J.-S.; Xu, Z.-H. Bacteria and Filamentous Fungi Running a Relay Race in Daqu Fermentation Enable Macromolecular Degradation and Flavor Substance Formation. Int. J. Food Microbiol. 2023, 390, 110118. [Google Scholar] [CrossRef]
- Yuan, L.; Li, Y.; Zheng, L.; Qin, Y.; Zhang, X.; Ma, L.; Zhang, H.; Du, L. Impact of Organic Acids on Aroma Release in Light-Flavor Baijiu: A Focus on Key Aroma-Active Compounds. Food Biosci. 2025, 65, 106071. [Google Scholar] [CrossRef]
- Yang, L.; Fan, W.; Xu, Y. Qu-Omics Elucidates the Formation and Spatio-Temporal Differentiation Mechanism Underlying the Microecology of High Temperature Daqu. Food Chem. 2024, 438, 137988. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.; Li, Q.; Li, C.; Xiao, D.; Wang, X.; Guo, X. Analysis and Formation Mechanism of Key Aroma Compounds with Ammonia-Like off-Flavors in Jiang-Flavored High-Temperature Daqu: Substances Composition and Main Microorganisms. Food Biosci. 2025, 73, 107656. [Google Scholar] [CrossRef]
- Ying, H.; Yang, J.; Yu, L.; Wei, J.; Sheng, Q.; Yuan, Y.; Yue, T. Metagenomics and GC-IMSanalyses- Reveal Microbial Community Differences and Flavor Characteristics among three Types of Feng Flavor Daqu. Food Res. Int. 2025, 221, 117551. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, L.; Li, R.; Tang, J.; Lin, B.; Qin, C.; Jiang, W.; An, L.; Zhang, F.; Shi, X.; et al. Metagenomics-Based Analysis of Microbial Community Dynamics and Flavor Compound Correlations during Rice-Flavor Baijiu Brewing. Front. Bioeng. Biotechnol. 2025, 13, 1638716. [Google Scholar] [CrossRef]
- Yang, L.; Huang, X.; Hu, J.; Deng, H.; He, J.; Zhang, C. The Spatiotemporal Heterogeneity of Microbial Community Assembly during Pit Fermentation of Soy Sauce Flavor Baijiu. Food Biosci. 2024, 61, 104438. [Google Scholar] [CrossRef]
- QB/T 4257-2011; General Methods of Analysis for Daqu. China Standards Press: Beijing, China, 2011.
- GB 5009.7-2016; Determination of Reducing Sugar in Foods. China Standards Press: Beijing, China, 2016.
- T/CBJ 004-2018; General Methods of Analysis for Solid-State Fermented Grains. China Alcoholic Drinks Association: Beijing, China, 2018.
- GB/T 33405-2016; Terminology of Sensory Evaluation of Baijiu. China Standards Press: Beijing, China, 2016.
- Xie, L.; Zhang, H.; Tian, J.; Yu, S.; Zhang, X.; Yang, H.; Hu, X.; Chen, M.; Wang, R.; Cai, A.; et al. Study on the Real-Time Online Determination of the Moisture Content of Baijiu Daqu during Fermentation: New Application of Spectral Reconstruction Technology. J. Food Compos. Anal. 2025, 146, 107904. [Google Scholar] [CrossRef]
- Ma, S.; Luo, H.; Zhao, D.; Qiao, Z.; Zheng, J.; An, M.; Huang, D. Environmental Factors and Interactions among Microorganisms Drive Microbial Community Succession during Fermentation of Nongxiangxing Daqu. Bioresour. Technol. 2022, 345, 126549. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Yan, M.; Huang, X.; Liao, H.; Bao, D.; Ge, Y.; Wang, S.; Xia, X. Temperature-Mediated Shift from Competitive to Facilitative Interactions Between Lactic Acid Bacteria and Bacillus Species in Daqu Fermentation: Insights from Metagenomics, Dual RNA-seq, and Coculture Analysis. Int. J. Food Microbiol. 2025, 442, 111352. [Google Scholar] [CrossRef]
- Wang, B.; Wu, Q.; Xu, Y.; Sun, B. Synergistic Effect of Multiple Saccharifying Enzymes on Alcoholic Fermentation for Chinese Baijiu Production. Appl. Environ. Microbiol. 2020, 86, e00013-20. [Google Scholar] [CrossRef]
- Chen, L.; Peng, Q.; Chen, Y.; Che, F.; Chen, Z.; Feng, S. Analysis of Dominant Microorganisms and Core Enzymes in Qingke Baijiu Daqu by High-Throughput Sequencing and Proteomics. Food Res. Int. 2025, 204, 115941. [Google Scholar] [CrossRef]
- Zhang, Z.; Ran, X.; Guo, Z.; Hou, Q.; Qu, D.; Wang, C.; Xu, Y.; Wang, Y. Microbial Diversity, Functional Properties, and Flavor Characteristics of High-Temperature Daqu With Different Colors. Food Res. Int. 2025, 212, 116406. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Chen, X.; Niu, J.; Li, J.; Li, W.; Zhu, H.; Li, X.; Sun, B. Storage Time Drives Divergent Microbial Functions and Flavor Metabolism in High-Temperature Daqu. Food Res. Int. 2026, 228, 118363. [Google Scholar] [CrossRef]
- Tu, H.; Zhou, G.; Lu, J.; Tan, F.; Han, Y.; Yang, F.; Du, H.; Liu, Y.; Li, J.; Wang, L.; et al. Cooperative Interaction Between Pediococcus and Bacillus Communities as a Key Factor in the High-Temperature Thermal Differentiation of Daqu. Food Biosci. 2024, 62, 105457. [Google Scholar] [CrossRef]
- Zhu, Q.; Chen, L.; Peng, Z.; Zhang, Q.; Huang, W.; Yang, F.; Du, G.; Zhang, J.; Wang, L. The Differences in Carbohydrate Utilization Ability between Six Rounds of Sauce-Flavor Daqu. Food Res. Int. 2023, 163, 112184. [Google Scholar] [CrossRef]
- Wei, Y.; Han, D.; Song, L.; Guo, L.; Bai, M.; Han, P.; Zhu, H.; Wen, Z. Integrative Analysis of Fungal Communities in Three Types of Baijiu Daqu Using Third-Generation Sequencing and Culturomics. Front. Microbiol. 2026, 16, 1748163. [Google Scholar] [CrossRef] [PubMed]
- Tie, Y.; Wang, L.; Ding, B.; Deng, Z.; Yang, Q.; Zhu, M.; Wu, Z.; Tang, L.; Suyama, T.; Zhang, W. Investigating the Main Contributors to Esterification Activity and Identifying the Aqueous-Phase Ester Synthases in Daqu. Food Biosci. 2025, 66, 106227. [Google Scholar] [CrossRef]
- Wang, Y.; Yun, J.; Zhou, M.; Wang, Z.; Li, D.; Jia, X.; Gao, Q.; Chen, X.; Xie, G.; Wu, H.; et al. Exploration and Application of Saccharomyces cerevisiae NJ002 to Improve the Fermentative Capacity of Medium-High Temperature Daqu. Bioresour. Technol. Rep. 2023, 23, 101571. [Google Scholar] [CrossRef]
- Niu, M.; Ma, L.; Yang, Y.; Jia, J.; Xiang, L.; Tu, T.; Zhao, X.; Shen, C.; Wang, S. Uncovering the Key Volatile Compounds Affecting the Roasted Aroma in Sauce-Flavor Baijiu and their Perceptual Interactions. LWT 2025, 237, 118705. [Google Scholar] [CrossRef]
- Chen, H.; Zhu, Y.; Xie, Y.; Long, W.; Lan, W.; She, Y.; Fu, H. Rapid Identification of High-Temperature Daqu Baijiu with the Same Aroma Type Through the Excitation Emission Matrix Fluorescence of Maillard Reaction Products. Food Control 2023, 153, 109938. [Google Scholar] [CrossRef]
- Ali, A.; Wu, Y.; Li, W.; Duan, Z.; Zhang, R.; Liu, J.; Patil, P.J.; Shah, H.; Li, X. Insight into Microorganisms and Flavor Substances in Traditional Chinese Fermented Food Starter: Daqu. Process Biochem. 2024, 146, 433–450. [Google Scholar] [CrossRef]
- Zhang, Q.; Huo, N.; Wang, Y.; Zhang, Y.; Wang, R.; Hou, H. Aroma-Enhancing Role of Pichia manshurica Isolated from Daqu in the Brewing of Shanxi Aged Vinegar. Int. J. Food Prop. 2017, 20, 2169–2179. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, H.; Xiong, A.; Liu, L.; Tong, R.; Pei, W.; Yang, L.; Wu, X.; Wang, Z. Baijiu: Chemical Composition, Biological Functions, and Endogenous Metabolic Profiles. Food Res. Int. 2025, 219, 116895. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhang, J.; Zeng, R.; Lin, Y.; Duan, Y.; Song, C.; Zhang, S.; Zuo, Y. Distribution Characteristic, Synthesis Mechanism and Regulation Measures of Ethyl Acetate in Strong Flavor Baijiu: A Review. Food Biosci. 2025, 73, 107681. [Google Scholar] [CrossRef]
- Qiu, F.; Du, B.; Zhang, C.; Zhu, L.; Yan, Y.; Li, W.; Leng, W.; Pang, Z.; Li, X.; Sun, B. Effects of Saccharomyces cerevisiae on Microbial Community and Flavor Metabolites in Solid-State Fermentation of Strong-Flavor Baijiu. Food Biosci. 2024, 59, 103925. [Google Scholar] [CrossRef]
- Zhao, H.; Xu, J.; Wang, R.; Liu, X.; Peng, X.; Guo, S. Succession and Diversity of Microbial Flora during the Fermentation of Douchi and their Effects on the Formation of Characteristic Aroma. Foods 2023, 12, 329. [Google Scholar] [CrossRef]
- Norouzian, D.; Akbarzadeh, A.; Scharer, J.M.; Moo Young, M. Fungal Glucoamylases. Biotechnol. Adv. 2006, 24, 80–85. [Google Scholar] [CrossRef]
- Liu, Y.; Li, H.; Liu, W.; Ren, K.; Li, X.; Zhang, Z.; Huang, R.; Han, S.; Hou, J.; Pan, C. Bioturbation Analysis of Microbial Communities and Flavor Metabolism in a High-Yielding Cellulase Bacillus subtilis Biofortified Daqu. Food Chem. X 2024, 22, 101382. [Google Scholar] [CrossRef]
- Huang, Q.; Liu, Y.; Tian, L.; Xiong, F.; He, Z.; Zhao, Y.; Xiang, S.; Qiu, X.; Yu, J.; Guan, T. Effects of Storage Time on Flavor Characteristics of Bran-Free Fermented Baijiu by Using Electronic Sensory, Descriptive Sensory Analysis, GC× GC–MS, and ICP-MS. Food Chem. X 2024, 23, 101667. [Google Scholar] [CrossRef]
- Dai, W.; Tan, W.; Chen, X.; Wang, J.; Yu, Y.; Zheng, Q. Why Aging Matters: Acetaldehyde-Acetal Interconversion in Baijiu. J. Food Compos. Anal. 2025, 148, 108222. [Google Scholar] [CrossRef]
- Wei, Y.; Zou, W.; Shen, C.; Yang, J. Basic Flavor Types and Component Characteristics of Chinese Traditional Liquors: A Review. J. Food Sci. 2020, 85, 4096–4107. [Google Scholar] [CrossRef]
- Li, C.; Yang, F.; Han, Y.; Yang, C.; Qin, X.; Zheng, H.; Chen, L.; Lu, J.; Zhang, C.; Lu, F.; et al. Aldehyde Metabolism in Maotai-Flavor Baijiu: Insights from Integrated Metagenomic and Metaproteomic Analyses. Food Res. Int. 2025, 221, 117518. [Google Scholar] [CrossRef]
- Xiao, Z.; Shen, Y.; Tu, Z.; Yang, X.; Zhang, Y.; Luo, Y. Enhanced Phenyllactic Acid Production From Grass Carp Viscera Hydrolysate Using Engineered Pediococcus acidilactici. Bioresour. Technol. 2026, 440, 133460. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Fan, W.; Xu, Y.; Cheng, P.; Sun, Y.; Zhu, X.; Hu, J. Characterization of Volatile Bitter off-Taste Compounds in Maotai-Flavor Baijiu (Chinese Liquor). LWT 2025, 217, 117363. [Google Scholar] [CrossRef]
- Wang, Y.; Li, M.; Wang, Z.; Liu, S.; O’Young, L. Furfural Production: A Review on Reaction Mechanism and Conventional Production Process. Ind. Crops Prod. 2025, 230, 121103. [Google Scholar] [CrossRef]
- Tu, H.; Zhou, G.; Tan, F.; Yang, F.; Han, Y.; Li, J.; Liu, Y.; Wu, Y.; Chen, J.; Wang, L. Temperature-Driven Deterministic-Dominated Microbial Assembly Underpins Seasonal Divergence in Baijiu High-Temperature Daqu. Food Biosci. 2025, 74, 107985. [Google Scholar] [CrossRef]
- Wu, S.; Du, H.; Xu, Y. Daqu Microbiota Adaptability to Altered Temperature Determines the Formation of Characteristic Compounds. Int. J. Food Microbiol. 2023, 385, 109995. [Google Scholar] [CrossRef]
- Du, J.; Zhang, Y.; Ning, Z.; Jiang, S.; Liu, J.; Ni, X.; Li, Q.; Yang, Q. Comparative Analysis of Pyrazine Compounds in Soy Sauce Aroma Baijiu from Different Regions and their Impact on Flavor Profiles. J. Food Compos. Anal. 2025, 148, 108447. [Google Scholar] [CrossRef]





| B-Daqu | R-Daqu | |
|---|---|---|
| Moisture Content (%) | 9.32 ± 0.35 b | 22.92 ± 0.87 a |
| Saccharifying Power (U/g) | 431 ± 28 b | 968 ± 45 a |
| Liquefying Power (U/g) | 1.1 ± 0.3 b | 10.7 ± 1.1 a |
| Fermenting Power (U/g) | 0.8 ± 0.3 b | 10.2 ± 0.9 a |
| Acidity (mmol/10 g) | 1.5 ± 0.1 b | 3.4 ± 0.3 a |
| Reducing Sugar Content (glucose %) | 2.07 ± 0.15 b | 2.42 ± 0.18 a |
| Free Amino Acid Content (%) | 0.14 ± 0.02 a | 0.16 ± 0.03 a |
| B-Jiupei | R-Jiupei | |||||
|---|---|---|---|---|---|---|
| Pit a | Pit b | Pit c | Pit d | Pit e | Pit f | |
| Alcohol content (% vol) | 5 ± 0.6 | 5.1 ± 0.7 | 3.8 ± 0.5 | 4.7 ± 0.6 | 4.4 ± 0.7 | 5.7 ± 0.5 |
| Total acidity (mmol/10 g) | 2.8 ± 0.3 | 2.6 ± 0.2 | 3 ± 0.4 | 3.3 ± 0.4 | 3.9 ± 0.5 | 3.7 ± 0.4 |
| Moisture content (%) | 57.23 ± 1.65 | 57.49 ± 1.88 | 58.30 ± 2.05 | 59.23 ± 1.85 | 59.71 ± 2.12 | 60.41 ± 1.94 |
| Starch content (%) | 10.9 ± 1.2 | 11.7 ± 1.3 | 11.9 ± 1.1 | 10.8 ± 1.1 | 8.8 ± 0.9 | 8.5 ± 0.8 |
| Reducing sugar content (%) | 0.93 ± 0.39 | 0.55 ± 0.22 | 1.01 ± 0.41 | 0.51 ± 0.22 | 0.73 ± 0.38 | 0.49 ± 0.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Jiao, G.; Tian, H.; Wang, J.; Li, N.; Liu, K.; Li, P.; Lu, F.; Wang, Q.; Wang, R.; Du, P. Metagenomic and Metabolomic Insights into Microbial Community Dynamics and Flavor Metabolite Formation in Novel Versus Traditional Strong-Flavor Daqu. Fermentation 2026, 12, 235. https://doi.org/10.3390/fermentation12050235
Jiao G, Tian H, Wang J, Li N, Liu K, Li P, Lu F, Wang Q, Wang R, Du P. Metagenomic and Metabolomic Insights into Microbial Community Dynamics and Flavor Metabolite Formation in Novel Versus Traditional Strong-Flavor Daqu. Fermentation. 2026; 12(5):235. https://doi.org/10.3390/fermentation12050235
Chicago/Turabian StyleJiao, Guanhua, Haoyu Tian, Junqing Wang, Nan Li, Kaiquan Liu, Piwu Li, Fengyong Lu, Qi Wang, Ruiming Wang, and Peng Du. 2026. "Metagenomic and Metabolomic Insights into Microbial Community Dynamics and Flavor Metabolite Formation in Novel Versus Traditional Strong-Flavor Daqu" Fermentation 12, no. 5: 235. https://doi.org/10.3390/fermentation12050235
APA StyleJiao, G., Tian, H., Wang, J., Li, N., Liu, K., Li, P., Lu, F., Wang, Q., Wang, R., & Du, P. (2026). Metagenomic and Metabolomic Insights into Microbial Community Dynamics and Flavor Metabolite Formation in Novel Versus Traditional Strong-Flavor Daqu. Fermentation, 12(5), 235. https://doi.org/10.3390/fermentation12050235






