Fermented Beverages, Ethanol and Health: A Critical Appraisal of Meta-Analytical Studies
Abstract
1. Introduction
2. Understanding the Basic Theoretical Background of Meta-Analysis
2.1. Conceptual Definitions and Production Pipeline
2.2. The Significance of Effects in Meta-Analytical Determinations
- (i)
- Large studies are produced (over several thousand subjects) over small studies (100-fold smaller);
- (ii)
- Large effects are obtained (relative risks 3–20) compared to small effects (relative risks 1.1–1.5);
- (iii)
- Smaller numbers of relationships are used, which allows the greater the selection of tested relationships;
- (iv)
- There is smaller the flexibility in designs, definitions, outcomes, and analytical models;
- (v)
- Financial and other interests and prejudices in a scientific field are minimized, including expert opinion.
3. The Financial Biases and Societal Implications
3.1. The Power to Influence Institutional and Public Opinions
The Quick Propagation Among Popular Media
3.2. Examples of Contradictory Health Studies with Societal Impact
4. Interim Summary
5. Understanding Meta-Analytic Outcomes
5.1. The Heterogeneity of Outcomes in WHO Studies
5.1.1. True Effects and Prediction Intervals
5.1.2. The Enhanced WHO Reports
5.2. EHN Studies on Cardiovascular Health
5.2.1. The Debate on J-Shaped Associations and Confounding Factors
5.2.2. How to Make the J-Shape Disappear
5.2.3. The Persistence of J-Shaped Relationships
6. Recent Studies in Alcohol Related Meta-Analysis
- (a)
- Occasional drinkers as reference category
- (b)
- Self-reported levels and patterns of consumption
- (c)
- The mandatory inclusion of a wide variety of confounding factors
Mendelian Randomisation: The Ultimate Approach
7. Limitations and Future Prospects
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Caffrey, E.; Perelman, D.; Ward, C.; Sonnenburg, E.; Gardner, C.; Sonnenburg, J. Unpacking Food Fermentation: Clinically Relevant Tools for Fermented Food Identification and Consumption. Adv. Nutr. 2025, 16, 412. [Google Scholar] [CrossRef]
- Avîrvarei, A.C.; Salanță, L.C.; Pop, C.R.; Mudura, E.; Pasqualone, A.; Anjos, O.; Barboza, N.; Usaga, J.; Dărab, C.P.; Burja-Udrea, C.; et al. Fruit-Based Fermented Beverages: Contamination Sources and Emerging Technologies Applied to Assure Their Safety. Foods 2023, 12, 838. [Google Scholar] [CrossRef]
- Cuamatzin-García, L.; Rodríguez-Rugarcía, P.; El-Kassis, E.G.; Galicia, G.; Meza-Jiménez, M.L.; Baños-Lara, M.D.; Zaragoza-Maldonado, D.S.; Pérez-Armendáriz, B. Traditional Fermented Foods and Beverages from around the World and Their Health Benefits. Microorganisms 2022, 10, 1151. [Google Scholar] [CrossRef] [PubMed]
- Bonaccio, M.; Iacoviello, L.; Donati, M.B.; de Gaetano, G. The tenth anniversary as a UNESCO world cultural heritage: An unmissable opportunity to get back to the cultural roots of the Mediterranean diet. Eur. J. Clin. Nutr. 2022, 76, 179–183. [Google Scholar] [CrossRef] [PubMed]
- WHO. No Level of Alcohol Consumption Is Safe for Our Health. 2023. Available online: https://www.who.int/europe/news/item/04-01-2023-no-level-of-alcohol-consumption-is-safe-for-our-health (accessed on 29 October 2025).
- EHN. EHN Position Paper on the Impact of Alcohol Consumption on Cardiovascular Disease. 2025. Available online: https://ehnheart.org/wp-content/uploads/2025/06/07324-CVD-and-Alcohol_web.pdf (accessed on 30 November 2025).
- Trichopoulou, A. Mediterranean diet as intangible heritage of humanity: 10 years on. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 1943–1948. [Google Scholar] [CrossRef]
- Baschali, A.; Tsakalidou, E.; Kyriacou, A.; Karavasiloglou, N.; Matalas, A.-L. Traditional Low-Alcoholic and Non-alcoholic Fermented Beverages Consumed in European Countries: A Neglected Food Group. Nutr. Res. Rev. 2017, 30, 1–24. [Google Scholar] [CrossRef]
- Bensi, P.; Divakar, S.; Merrylin, J. Exploring the rich heritage and health benefits of diverse fruit wines and their production. J. Food Sci. Technol. 2025, 62, 999–1006. [Google Scholar] [CrossRef]
- Morris, J.; Boness, C.L.; Hartwell, M. Key Terms and Concepts in Alcohol Use and Problems: A Critical Evaluation. Subst. Use Res. Treat. 2025, 19, 29768357241312555. [Google Scholar] [CrossRef]
- Herdiana, Y. Alcohol in Daily Products: Health Risks, Cultural Considerations, and Economic Impacts. Risk Manag. Health Policy 2025, 18, 217–237. [Google Scholar] [CrossRef] [PubMed]
- Ntais, C.; Talias, M.A. Unveiling the Value of Meta-Analysis in Disease Prevention and Control: A Comprehensive Review. Medicina 2024, 60, 1629. [Google Scholar] [CrossRef]
- Halevi, G.; Pinotti, R. Systematic Reviews: Characteristics and Impact. Publ. Res. Q. 2020, 36, 523–537. [Google Scholar] [CrossRef]
- Hoffmann, F.; Allers, K.; Rombey, T.; Helbach, J.; Hoffmann, A.; Mathes, T.; Pieper, D. Nearly 80 systematic reviews were published each day: Observational study on trends in epidemiology and reporting over the years 2000–2019. J. Clin. Epidemiol. 2021, 138, 1–11. [Google Scholar] [CrossRef]
- Ioannidis, J. The mass production of redundant, misleading, and conflicted systematic reviews and meta-analyses. Milbank Q. 2016, 94, 485–514. [Google Scholar] [CrossRef]
- LeLorier, J.; Grégoire, G.; Benhaddad, A.; Lapierre, J.; Derderian, F. Discrepancies between meta-analyses and subsequent large randomized, controlled trials. N. Engl. J. Med. 1997, 337, 536–542. [Google Scholar] [CrossRef]
- Bigby, M. Understanding and Evaluating Systematic Reviews and Meta-analyses. Indian J. Dermatol. 2014, 59, 134–139. [Google Scholar] [CrossRef]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. (Eds.) Sub-group analysis. In Introduction to Meta-Analysis; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2009; Chapter 19. [Google Scholar] [CrossRef]
- Estruch, R.; Henk, F.; Hendriks, J. Associations between Low to Moderate Consumption of Alcoholic Beverage Types and Health Outcomes: A Systematic Review. Alcohol Alcohol. 2022, 57, 176–184. [Google Scholar] [CrossRef]
- Israel, H.; Richter, R. A Guide to Understanding Meta-analysis. J. Orthop. Sports Phys. Ther. 2011, 41, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, J. Integration of evidence from multiple meta-analyses: A primer on umbrella reviews, treatment networks and multiple treatments meta-analyses. Can. Med. Assoc. J. 2009, 181, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Clephas, P.; Heesen, M. Interpretation of meta-analyses. Interv. Pain Med. 2022, 1, 100120. [Google Scholar] [CrossRef]
- Wallach, J.D. Meta-analysis Metastasis. JAMA Intern. Med. 2019, 179, 1594–1595. [Google Scholar] [CrossRef]
- Ioannidis, J. Meta-research: The art of getting it wrong. Res. Synth. Methods 2010, 1, 169–184. [Google Scholar] [CrossRef]
- Deeks, J.J.; Higgins, J.P.; Altman, D.G.; McKenzie, J.E.; Veroniki, A. Analysing data and undertaking meta-analyses. In Cochrane Handbook for Systematic Reviews of Interventions, (version 6.5) [last updated November 2024]; Higgins, J.P., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane: London, UK, 2024; Chapter 10; Available online: www.cochrane.org/handbook (accessed on 30 November 2025).
- Neyeloff, J.L.; Fuchs, S.C.; Moreira, L.B. Meta-analyses and Forest plots using a microsoft excel spreadsheet: Step-by-step guide focusing on descriptive data analysis. BMC Res. Notes 2012, 5, 52. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions, version 6.5 (updated August 2024); Cochrane: London, UK, 2024; Available online: www.cochrane.org/handbook (accessed on 30 November 2025).
- Feinstein, A.R. Meta-analysis: Statistical alchemy for the 21st century. J. Clin. Epidemiol. 1995, 48, 71–79. [Google Scholar] [CrossRef]
- Collins, R.; Bowman, L.; Landray, M.; Peto, R. The magic of randomization versus the myth of real-world evidence. N. Engl. J. Med. 2020, 382, 674–678. [Google Scholar] [CrossRef]
- Nelson, L.D.; Simmons, J.; Simonsohn, U. Psychology’s renaissance. Ann. Rev. Psychol. 2018, 69, 511–534. [Google Scholar] [CrossRef]
- Greenland, S.; Senn, S.J.; Rothman, K.J.; Carlin, J.B.; Poole, C.; Goodman, S.N.; Altman, D.G. Statistical tests, P values, confidence intervals, and power: A guide to misinterpretations. Eur. J. Epidemiol. 2016, 31, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Wasserstein, R.; Schirm, A.; Lazar, N. Moving to a world beyond “p < 0.05”. Am. Stat. 2019, 73, 1–19. [Google Scholar] [CrossRef]
- Ioannidis, J. Why Most Published Research Findings Are False. PLoS Med. 2005, 2, e124. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, S.; de Gaetano, G.; Di Castelnuovo, A.; Djoussé, L.; Poli, A.; van Velden, D. Moderate alcohol consumption and lower total mortality risk: Justified doubts or established facts? Nutr. Metab. Cardiovasc. Dis. 2019, 29, 1003–1008. [Google Scholar] [CrossRef]
- Clay, J.M.; Stockwell, T.; Golder, S.; Lawrence, K.; McCambridge, J.; Vishnevsky, N.; Zuckermann, A.; Naimi, T. The International Scientific Forum on Alcohol Research (ISFAR) critiques of alcohol research: Promoting health benefits and downplaying harms. Addiction 2025, 120, 2319–2328. [Google Scholar] [CrossRef] [PubMed]
- Stockwell, T.; Zhao, J.; Clay, J.; Levesque, C.; Sanger, N.; Sherk, A.; Naimi, T. Why Do Only Some Cohort Studies Find Health Benefits From Low-Volume Alcohol Use? A Systematic Review and Meta-Analysis of Study Characteristics That May Bias Mortality Risk Estimates. J. Stud. Alcohol Drugs 2024, 85, 441–452. [Google Scholar] [CrossRef]
- Patel, A.; Figueredo, V. Alcohol and Cardiovascular Disease: Helpful or Hurtful. Rev. Cardiovasc. Med. 2023, 24, 121. [Google Scholar] [CrossRef] [PubMed]
- Kilian, C.; Buckley, C.; Lemp, J.; Kou, X.; Kerr, W.; Mulia, N.; Purshouse, R.; Rehm, J.; Probst, C. Targeting alcohol use in high-risk population groups: A US microsimulation study of beverage-specific pricing policies. Lancet Public Health 2025, 10, e815–e823. [Google Scholar] [CrossRef]
- Villacreces, S.; Blanco, C.; Caballero, I. Developments and characteristics of craft beer production processes. Food Biosci. 2022, 45, 101495. [Google Scholar] [CrossRef]
- Santos, O. Environmental health behavior as a unifying concept for public health and planetary health. In Environmental Health Behavior; Santos, O., Santos, R., Virgolino, A., Eds.; Academic Press: Cambridge, MA, USA, 2024; Chapter 5; pp. 45–60. [Google Scholar] [CrossRef]
- Ojeda-Linares, C.; Álvarez-Ríos, G.; Figueredo-Urbina, C.; Islas, L.; Lappe-Oliveras, P.; Nabhan, G.P.; Torres-García, I.; Vallejo, M.; Casas, A. Traditional Fermented Beverages of Mexico: A Biocultural Unseen Foodscape. Foods 2021, 10, 2390. [Google Scholar] [CrossRef]
- Griswold, M.; Gakidou, E. Alcohol and the global burden of disease—Authors’ reply. Lancet 2019, 393, 2391–2392. [Google Scholar] [CrossRef]
- Roerecke, M.; Rehm, J. On the evidence of a cardioprotective effect of alcohol consumption. Addiction 2013, 108, 429–431. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, J.P.A. Science Journalism and Advocacy: Disseminating, Facilitating, Defending, Shaping, Terrorizing, or Usurping Science? SSRN 2026. Available online: https://ssrn.com/abstract=6030516 (accessed on 10 March 2026). [CrossRef]
- Liew, Z.; Kioumourtzoglou, M.; Roberts, A.; O’Reilly, É.; Ascherio, A.; Weisskopf, M.G. Use of Negative Control Exposure Analysis to Evaluate Confounding: An Example of Acetaminophen Exposure and Attention-Deficit/Hyperactivity Disorder in Nurses’ Health Study II. Am. J. Epidemiol. 2019, 188, 768–775. [Google Scholar] [CrossRef]
- Ji, Y.; Azuine, R.; Zhang, Y.; Hou, W.; Hong, X.; Wang, G.; Riley, A.; Pearson, C.; Zuckerman, B.; Wang, X. Association of Cord Plasma Biomarkers of In Utero Acetaminophen Exposure With Risk of Attention-Deficit/Hyperactivity Disorder and Autism Spectrum Disorder in Childhood. JAMA Psychiatry 2020, 77, 180–189. [Google Scholar] [CrossRef]
- FDA. FDA Responds to Evidence of Possible Association Between Autism and Acetaminophen Use During Pregnancy. Available online: https://www.fda.gov/news-events/press-announcements/fda-responds-evidence-possible-association-between-autism-and-acetaminophen-use-during-pregnancy (accessed on 31 October 2025).
- Sheikh, J.; Allotey, J.; Sobhy, S.; Plana, M.; Martinez-Barros, H.; Naidu, H.; Junaid, F.; Sofat, R.; Mol, B.W.; Kenny, L.C.; et al. Maternal paracetamol (acetaminophen) use during pregnancy and risk of autism spectrum disorder and attention deficit/hyperactivity disorder in offspring: Umbrella review of systematic reviews. BMJ 2025, 391, e088141. [Google Scholar] [CrossRef] [PubMed]
- Nordhagen, E.; Flydal, E. WHO to build neglect of RF-EMF exposure hazards on flawed EHC reviews? Case study demonstrates how “no hazards” conclusion is drawn from data showing hazards. Rev. Environ. Health 2025, 40, 277–288. [Google Scholar] [CrossRef]
- Fausta, N.; Gianni, P.; Canali, R. Commentary: Association between wine consumption and cancer: A systematic review and meta-analysis. Front. Nutr. 2024, 11, 1335731. [Google Scholar] [CrossRef] [PubMed]
- Lucerón-Lucas-Torres, M.; Cavero-Redondo, I.; Martínez-Vizcaíno, V.; Bizzozero-Peroni, B.; Pascual-Morena, C.; Álvarez-Bueno, C. Association between wine consumption and cancer: A systematic review and meta-analysis. Front. Nutr. 2023, 10, 1197745. [Google Scholar] [CrossRef]
- Stockwell, T.; Priore, I.; Im, P. The U.S. National Academies of Science, Engineering, and Medicine Were Economical With the Truth About Alcohol and Health. J. Stud. Alcohol Drugs 2025, 86, 651–656. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Food and Nutrition Board; Committee on Review of Evidence on Alcohol and Health. Review of Evidence on Alcohol and Health; Stone, K.B., Calonge, B.N., Eds.; National Academies Press: Washington, DC, USA, 2025. [Google Scholar]
- Castriota, S.; Frumento, P.; Suppressa, F. How much is too much? A methodological investigation of the literature on alcohol consumption and health. J. Wine Econ. 2025, 20, 222–234. [Google Scholar] [CrossRef]
- GBD 2016 Alcohol Collaborators. Alcohol use and burden for 195 countries and territories, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2018, 392, 1015–1035. [Google Scholar] [CrossRef]
- Hendriks, H. Alcohol and Human Health: What Is the Evidence? Annu. Rev. Food Sci. Technol. 2020, 11, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Borenstein, M. How to understand and report heterogeneity in a meta-analysis: The difference between I-squared and prediction intervals. Integr. Med. Res. 2023, 12, 101014. [Google Scholar] [CrossRef]
- Bryazka, D.; Reitsma, M.B.; Griswold, M.G.; Abate, K.H.; Abbafati, C.; Abbasi-Kangevari, M.; Abbasi-Kangevari, Z.; Abdoli, A.; Abdollahi, M.; Abdullah, A.Y.M.; et al. Population-level risks of alcohol consumption by amount, geography, age, sex, and year: A systematic analysis for the Global Burden of Disease Study 2020. Lancet 2022, 400, 185–235. [Google Scholar] [CrossRef]
- Bagnardi, V.; Rota, M.; Botteri, E.; Tramacere, I.; Islami, F.; Fedirko, V.; Scotti, L.; Jenab, M.; Turati, F.; Pasquali, E.; et al. Alcohol consumption and site-specific cancer risk: A comprehensive dose–response meta-analysis. Br. J. Cancer 2015, 112, 580–593. [Google Scholar] [CrossRef]
- Kaltsas, A.; Chrisofos, M.; Symeonidis, E.N.; Zachariou, A.; Stavropoulos, M.; Kratiras, Z.; Giannakodimos, I.; Symeonidis, A.; Dimitriadis, F.; Sofikitis, N. To Drink or Not to Drink? Investigating Alcohol’s Impact on Prostate Cancer Risk. Cancers 2024, 16, 3453. [Google Scholar] [CrossRef]
- Shield, K.; Franklin, A.; Wettlaufer, A.; Sohi, I.; Bhulabhai, M.; Farkouh, E.K.; Radu, I.G.; Kassam, I.; Munnery, M.; Remtulla, R.; et al. National, regional, and global statistics on alcohol consumption and associated burden of disease 2000–20: A modelling study and comparative risk assessment. Lancet Public Health 2025, 10, e751–e761. [Google Scholar] [CrossRef]
- Sherk, A.; Churchill, S.; Cukier, S.; Grant, S.C.; Shield, K.; Stockwell, T. Distributions of alcohol use and alcohol-caused death and disability in Canada: Defining alcohol harm density functions and new perspectives on the prevention paradox. Addiction 2024, 119, 696–705. [Google Scholar] [CrossRef]
- Kilian, C.; Rehm, J.; Shield, K.; Manthey, J. Changes in Alcohol-Specific Mortality During the COVID-19 Pandemic in 14 European Countries. Sucht 2023, 69, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Hunt, D.; Rai, S. Testing threshold and hormesis in a random effects dose-response model applied to developmental toxicity data. Biom. J. 2005, 47, 319–328. [Google Scholar] [CrossRef]
- Manolis, T.; Manolis, A.A.; Manolis, A.S. Cardiovascular effects of alcohol: A double-edged sword/how to remain at the nadir point of the J-Curve? Alcohol 2019, 76, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, F.; Zhang, X.; Li, Z.; Li, H. Smoking increases risks of all-cause and breast cancer specific mortality in breast cancer individuals: A dose-response meta-analysis of prospective cohort studies involving 39725 breast cancer cases. Oncotarget 2016, 7, 83134. [Google Scholar] [CrossRef]
- Naimi, T.; Chikritzhs, T. Santa Claus, the Tooth Fairy, and purported lifetime nondrinkers: Ramifications for observational evidence about alcohol and health. Alcohol Clin. Exp. Res. 2025, 49, 92–94. [Google Scholar] [CrossRef]
- Rehm, J.; Greenfield, T.K.; Rogers, J.D. Average volume of alcohol consumption, patterns of drinking, and all-cause mortality: Results from the US national alcohol survey. Am. J. Epidemiol. 2001, 153, 64–71. [Google Scholar] [CrossRef]
- Makela, P.; Paljarvi, T.; Poikolainen, K. Heavy and nonheavy drinking occasions, all-cause and cardiovascular mortality and hospitalizations: A follow-up study in a population with a low consumption level. J. Stud. Alcohol 2005, 66, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, M.; Rehm, J.; Klipstein-Grobusch, K.; Boeing, H.; Schutze, M.; Drogan, D.; Overvad, K.; Tjonneland, A.; Halkjaer, J.; Fagherazzi, G.; et al. The association of pattern of lifetime alcohol use and cause of death in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Int. J. Epidemiol. 2013, 42, 1772–1790. [Google Scholar] [CrossRef]
- Zaridze, D.; Lewington, S.; Boroda, A.; Scelo, G.; Karpov, R.; Lazarev, A.; Konobeevskaya, I.; Igitov, V.; Terechova, T.; Boffetta, P.; et al. Alcohol and mortality in Russia: Prospective observational study of 151,000 adults. Lancet 2014, 383, 1465–1473. [Google Scholar] [CrossRef]
- Nakaya, N.; Kurashima, K.; Yamaguchi, J.; Ohkubo, T.; Nishino, Y.; Tsubono, Y.; Shibuya, D.; Fukudo, S.; Fukao, A.; Tsuji, I.; et al. Alcohol consumption and mortality in Japan: The Miyagi Cohort Study. J. Epidemiol. 2004, 14, S18–S25. [Google Scholar] [CrossRef][Green Version]
- Sempos, C.T.; Rehm, A.; Wu, T.J.; Crespo, C.J.; Trevisan, M. Average volume of alcohol consumption and all-cause mortality in African Americans: The NHEFS cohort. Alcohol. Clin. Exp. Res. 2003, 27, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.; Harrison, S.E.; Judd, N.; Bellis, M.A.; Hughes, K.; Jones, A. The impact of behavioural risk factors on communicable diseases: A systematic review of reviews. BMC Public Health 2021, 21, 2110. [Google Scholar] [CrossRef] [PubMed]
- Schutte, R.; Papageorgiou, M.; Najlah, M.; Huisman, H.; Ricci, C.; Zhang, J.; Milner, N.; Schutte, A. Drink types unmask the health risks associated with alcohol intake—Prospective evidence from the general population. Clin. Nutr. 2020, 39, 3168–3174. [Google Scholar] [CrossRef]
- Wall, T.L.; Ehlers, C.L. Genetic Influences Affecting Alcohol Use Among Asians. Alcohol Health Res. World 1995, 19, 184–189. [Google Scholar]
- Mensah, G.A. Cardiovascular Diseases in African Americans: Fostering Community Partnerships to Stem the Tide. Am. J. Kidney Dis. 2018, 72, S37–S42. [Google Scholar] [CrossRef]
- Song, J.W.; Chung, K.C. Observational studies: Cohort and case-control studies. Plast. Reconstr. Surg. 2010, 126, 2234–2242. [Google Scholar] [CrossRef]
- Biddinger, K.J.; Emdin, C.A.; Haas, M.E.; Wang, M.; Hindy, G.; Ellinor, P.T.; Kathiresan, S.; Khera, A.V.; Aragam, K.G. Association of habitual alcohol intake with risk of cardiovascular disease. JAMA Netw. Open 2022, 5, e223849. [Google Scholar] [CrossRef] [PubMed]
- Fourotan, F.; Guyatt, G.; Zuk, V.; Vandvik, P.; Alba, A.; Mustafa, R.; Vernooij, R.; Arevalo-Rodriguez, I.; Munn, Z.; Roshanov, P.; et al. GRADE Guidelines 28: Use of GRADE for the assessment of evidence about prognostic factors: Rating certainty in identification of groups of patients with different absolute risks. J. Clin. Epidemiol. 2020, 121, 62–70. [Google Scholar] [CrossRef]
- Higgins, J.; Thompson, S.; Deeks, J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Visontay, R.; Sunderland, M.; Slade, T.; Wilson, J.; Mewton, L. Are there non-linear relationships between alcohol consumption and long-term health?: A systematic review of observational studies employing approaches to improve causal inference. BMC Med. Res. Methodol. 2022, 22, 16. [Google Scholar] [CrossRef]
- Zheng, P.; Afshin, A.; Biryukov, S.; Bisignano, C.; Brauer, M.; Bryazka, D.; Burkart, K.; Cercy, K.M.; Cornaby, L.; Dai, X.; et al. The Burden of Proof studies: Assessing the evidence of risk. Nat. Med. 2022, 28, 2038–2044. [Google Scholar] [CrossRef] [PubMed]
- Carr, S.; Bryazka, D.; McLaughlin, S.A.; Zheng, P.; Bahadursingh, S.; Aravkin, A.Y.; Hay, S.I.; Lawlor, H.R.; Mullany, E.C.; Murray, C.J.L.; et al. A burden of proof study on alcohol consumption and ischemic heart disease. Nat. Commun. 2024, 15, 4082. [Google Scholar] [CrossRef]
- Rehm, J. Why the relationship between level of alcohol-use and all-cause mortality cannot be addressed with meta-analyses of cohort studies. Drug Alcohol Rev. 2019, 38, 3–4. [Google Scholar] [CrossRef]
- Gordon, K.S.; McGinnis, K.; Dao, C.; Rentsch, C.T.; Small, A.; Smith, R.V.; Kember, R.L.; Gelernter, J.; Kranzler, H.R.; Bryant, K.J.; et al. Differentiating types of self-reported alcohol abstinence. AIDS Behav. 2020, 24, 655–665. [Google Scholar] [CrossRef] [PubMed]
- Galán, I.; Fontán, J.; Ortiz, C.; López-Cuadrado, T.; Téllez-Plaza, M.; García-Esquinas, E. Volume of alcohol intake, heavy episodic drinking, and all-cause mortality in Spain: A longitudinal population-based study. Addict. Behav. 2024, 158, 108108. [Google Scholar] [CrossRef]
- Sarich, P.; Canfell, K.; Egger, S.; Banks, E.; Joshy, G.; Grogan, P.; Weber, M. Alcohol consumption, drinking patterns and cause-specific mortality in an Australian cohort of 181,607 participants aged 45 years and over. Public Health 2025, 239, 230–241. [Google Scholar] [CrossRef]
- Liu, X.; Ding, X.; Zhang, F.; Chen, L.; Luo, Q.; Xiao, M.; Liu, X.; Wu, Y.; Tang, W.; Qiu, J.; et al. Association between alcohol consumption and risk of stroke among adults: Results from a prospective cohort study in Chongqing, China. BMC Public Health 2023, 23, 1593. [Google Scholar] [CrossRef]
- Jani, B.; McQueenie, R.; Nicholl, B.; Field, R.; Hanlon, P.; Gallacher, K.; Mair, F.; Lewsey, J. Association between patterns of alcohol consumption (beverage type, frequency and consumption with food) and risk of adverse health outcomes: A prospective cohort study. BMC Med. 2021, 19, 8. [Google Scholar] [CrossRef]
- Wallach, J.; Serghiou, S.; Chu, L.; Egilman, A.; Vasiliou, V.; Ross, J.; Ioannidis, J. Evaluation of confounding in epidemiologic studies assessing alcohol consumption on the risk of ischemic heart disease. BMC Med. Res. Methodol. 2020, 20, 64. [Google Scholar] [CrossRef] [PubMed]
- Ortolá, R.; Sotos-Prieto, M.; García-Esquinas, E.; Galán, I.; Rodríguez-Artalejo, F. Alcohol Consumption Patterns and Mortality Among Older Adults With Health-Related or Socioeconomic Risk Factors. JAMA Netw. Open 2024, 7, e2424495. [Google Scholar] [CrossRef]
- Yun, J.; Han, K.; Ki, Y.; Hwang, D.; Kang, J.; Yang, H.; Park, K.; Kang, H.; Koo, B.; Kim, H.; et al. Impact of Alcohol Consumption on Cardiovascular Events in Patients Undergoing Percutaneous Coronary Intervention. J. Clin. Med. 2024, 13, 6542. [Google Scholar] [CrossRef] [PubMed]
- Matson, T.; Bobb, J.; Oliver, M.; Berger, D.; Jack, H.; Steel, T.; Bradley, K.; Hallgren, K. Alcohol consumption reported on routine healthcare screenings is associated with all-cause mortality in primary care patients: A retrospective cohort study. Alcohol Clin. Exp. Res. 2025, 49, 2875–2886. [Google Scholar] [CrossRef] [PubMed]
- Stattin, K.; Burger, B.; Eriksson, M.; Crockett, D.; Marks-Hultström, M.; Frithiof, R.; Kawati, R.; Lipcsey, M. The impact of lifestyle on infection risks and mortality: A UK biobank cohort study. Public Health 2025, 247, 5882. [Google Scholar] [CrossRef]
- Larsson, S.; Butterworth, A.; Burgess, S. Mendelian randomization for cardiovascular diseases: Principles and applications. Eur. Heart J. 2023, 44, 4913–4924. [Google Scholar] [CrossRef]
- Davey, S.; Hemani, G. Mendelian randomization: Genetic anchors for causal inference in epidemiological studies. Hum. Mol. Genet. 2014, 23, R89–R98. [Google Scholar] [CrossRef]
- van de Luitgaarden, I.; van Oort, S.; Bouman, E.; Schoonmade, L.J.; Schrieks, I.C.; Grobbee, D.E.; van der Schouw, Y.T.; Larsson, S.C.; Burgess, S.; van Ballegooijen, A.J.; et al. Alcohol consumption in relation to cardiovascular diseases and mortality: A systematic review of Mendelian randomization studies. Eur. J. Epidemiol. 2022, 37, 655–669. [Google Scholar] [CrossRef]
- Visontay, R.; Mewton, L.; Sunderland, M.; Chapman, C.; Slade, T. Is low-level alcohol consumption really health-protective? A critical review of approaches to promote causal inference and recent applications. Alcohol Clin. Exp. Res. 2024, 48, 771–780. [Google Scholar] [CrossRef]
- Kember, R.; Rentsch, C.; Lynch, J.; Vujkovic, M.; Voight, B.; Justice, A.; Assimes, T.; Kranzler, H. A Mendelian randomization study of alcohol use and cardiometabolic disease risk in a multi-ancestry population from the Million Veteran Program. Alcohol Clin. Exp. Res. 2024, 48, 2256–2268. [Google Scholar] [CrossRef]
- Larsson, S.C.; Carter, P.; Kar, S.; Vithayathil, M.; Mason, A.; Michaëlsson, K.; Burgess, S. Smoking, alcohol consumption, and cancer: A Mendelian randomisation study in UK Biobank and international genetic consortia participants. PLoS Med. 2020, 17, e1003178. [Google Scholar] [CrossRef]
- Larsson, S.C.; Mason, A.M.; Cronjé, H.T.; Bassett, E.; Horta, G.; Kar, S.; Burgess, S. Alcohol consumption and risk of cancer: A Mendelian randomization analysis of four biobanks and consortium data. BMC Med. 2025, 23, 676. [Google Scholar] [CrossRef] [PubMed]
- Miller, A. Still rethinking the J-shaped curve: A commentary on Kember et al., 2024. Alcohol Clin. Exp. Res. 2025, 49, 503–506. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Martínez-Urbistondo, D.; Barbería-Latasa, M.; Vázquez-Ruiz, Z.; Ruiz-Canela, M.; Bes-Rastrollo, M.; Martínez-González, M.Á. The Unfinished Debate on Wine and Other Alcoholic Beverages: Conflicting Evidence, Public Health Messages and the Missing Trial. Nutrients 2026, 18, 529. [Google Scholar] [CrossRef]
- Saunders, J.; Aasland, O.; Babor, T.; de la Fuente, J.; Grant, M. Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption–II. Addiction 1993, 88, 791–804. [Google Scholar] [CrossRef]
- Iliakis, P.; Stamou, E.; Vakka, A.; Ntalekou, K.; Kouremeti, M.; Ktenopoulos, N.; Karakasis, P.; Theofilis, P.; Pitsillidi, A.; Sakalidis, A.; et al. Alcohol Consumption and Acute Coronary Syndrome: Epidemiology, Pathophysiology, and Clinical Perspectives. J. Clin. Med. 2026, 15, 299. [Google Scholar] [CrossRef] [PubMed]
- Mayer, C.; Fontelo, P. Alcohol consumption and its correlation with medical conditions: A UK Biobank study. Front. Public Health 2024, 12, 1294492. [Google Scholar] [CrossRef]
- Timothy, C.; Durazzo, B.; Dieter, J.; Meyerhoff, J. Low level alcohol consumption is associated with lower regional brain volume and thickness and lower choline-containing compounds and myo-inositol levels in healthy adults. Alcohol 2025, 129, 157–165. [Google Scholar] [CrossRef]
- Chiva-Blanch, G.; Badimon, L. Benefits and Risks of Moderate Alcohol Consumption on Cardiovascular Disease: Current Findings and Controversies. Nutrients 2019, 12, 108. [Google Scholar] [CrossRef]
- Chu, L.; Ioannidis, J.; Egilman, A.; Vasiliou, V.; Ross, J.S.; Wallach, J.D. Vibration of effects in epidemiologic studies of alcohol consumption and breast cancer risk. Int. J. Epidemiol. 2020, 49, 608–618. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, J. Reforming Nutritional Epidemiologic Research—Reply. JAMA 2019, 321, 310. [Google Scholar] [CrossRef] [PubMed]
- Zavalis, E.; Pezzullo, A.; Ioannidis, J. Instability of Global Burden of Disease Estimates of Deaths and DALYs from Major Risk Factors. medRxiv 2025. [Google Scholar] [CrossRef]
- Ilak-Peršurić, A.S.; Težak-Damijanić, A.; Radeka, S. Relationship Between Health Benefit Perception Moderate Wine Consumption, Wine Label and Healthy Behaviour. Foods 2025, 14, 1937. [Google Scholar] [CrossRef]

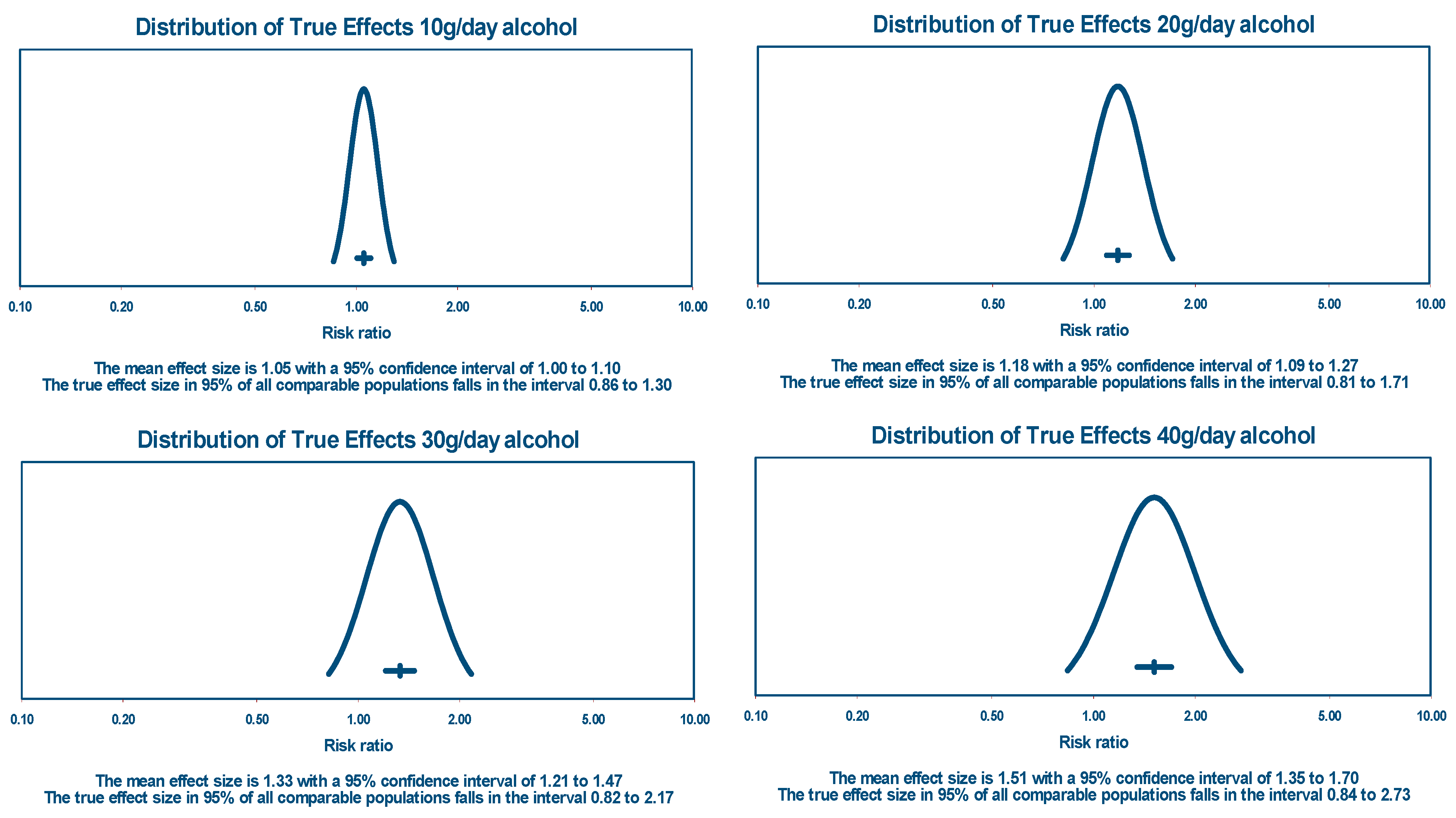
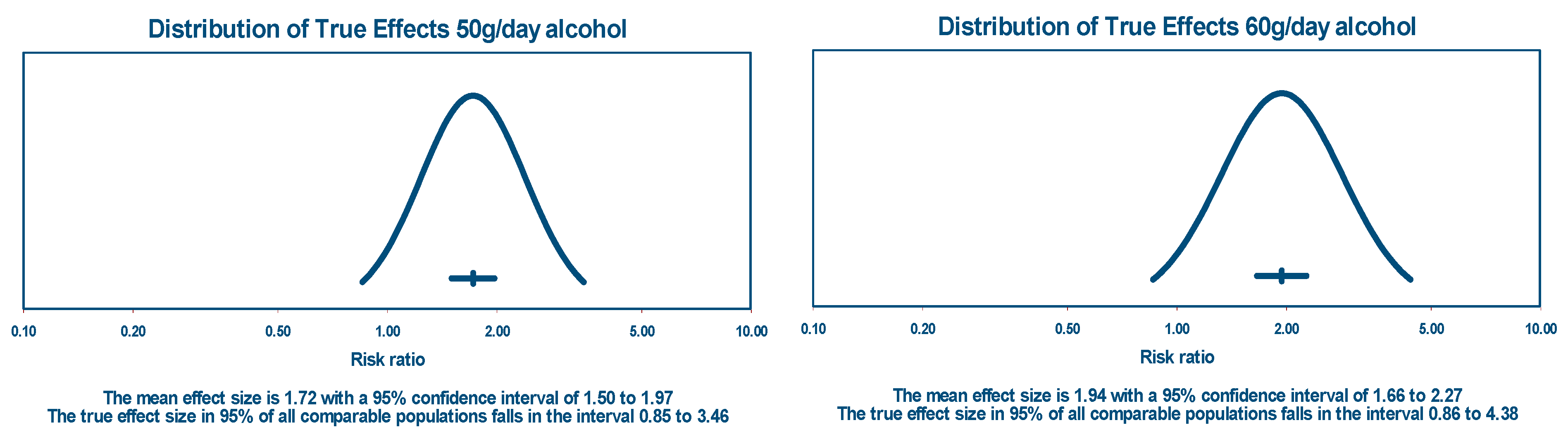

| TMREL | UI (95%) | NDE | UI (95%) | Main Causes | |
|---|---|---|---|---|---|
| Overall (GBD 2016 [55]) a | 0 | 0–0.800 | - | - | - |
| Overall (GBD 2020 [58]) a | 0.511 | 0.4–0.700 | 1.72 | 0.80–3.30 | - |
| 1.87 | 0.50–3.30 | - | |||
| Males aged 15–39 years | 0.136 | 0–0.400 | 0.249 | 0–1.00 | 66.3% all injuries b |
| Females aged 15–39 years | 0.273 | 0–0.500 | 0.546 | 0–1.30 | 47.9% all injuries |
| 40 years or older | 0.114 | 0–0.403 | 0.193 | 0–0.900 | - |
| 1.87 | 0.50–3.30 | 6.94 | 3.40–8.30 | - | |
| Males aged 40–64 years | 0.527 | 0.400–1.00 | 1.69 | 0.800–3.20 | 34.4% CVD c, 23% all injuries |
| Females aged 40–64 years | 0.562 | 0.400–0.800 | 1.82 | 1.00–3.10 | 30.85% CVD, 16.7% all injuries |
| Males 65 years or older | 0.636 | 0.500–1.00 | 3.19 | 1.50–5.20 | 57.3% CVD |
| Females 65 years or older | 0.656 | 0.50–1.00 | 3.51 | 1.70–5.50 | 56.6% CVD |
| Study Cohorts | Sociodemographic and Clinical Covariates | Reference Cohort | Main Conclusions | Reference |
|---|---|---|---|---|
| 22,091 individuals aged 30–79 from Chongqing, China | Age, sex, marital status, household income, education, smoking status, physical activity, red meat, fruits, vegetable intake, and spicy food intake, disease history (hypertension, diabetes) | Non-drinker (0 g/day) | Moderate drinking (13 to 36 g/day) and drinking 6–7 days per week were associated with a reduced risk of total stroke | [89] |
| 43,071 individuals from Spain, aged 15 years or older | Sex, age, education, marital status, size of the residence municipality, lifestyle behaviors, diet style, body mass index, health status, disease number | Infrequent occasional drinkers (consumption ≤ once/month) | Low-volume drinkers (<20 g/day) had a comparable mortality risk Never-drinkers, former drinkers, regular drinkers (>20 g/day), and those engaging in weekly heavy episodic drinking (HED), experienced higher mortality risk | [87] |
| 135,103 individuals of UK Biobank, with 60 years or older | Sex, age, race and ethnicity, education, drinking with meals, smoking, leisure-time, physical activity, time spent watching television, prevalent morbidities | Occasional drinkers (≤2.86 g/d) | No evidence of a beneficial association between low-risk (men: >2.86–20.00 g/d; women: >2.86–10.00 g/d) consumption and mortality Detrimental association of low-risk drinking in individuals with socioeconomic or health-related risk factors, especially for cancer deaths Preference for wine and drinking only during meals were associated with lower all-cause mortality | [92] |
| 77,409 individuals of the Republic of Korea during 4 years | Age, sex, social income, body mass index, regular exercise, smoking status, several blood analyses, hypertension, diabetes mellitus, dyslipidemia | Persistent non-drinkers | Both within-the-guideline (<8 g/day for women and <16 g/day for men) and above-the-guideline drinkers had a lower major adverse cardiovascular and cerebrovascular events (MACCE) risk than the non-drinkers Lowest risk with once-per-week and a mild amount per body weight (≤0.33 g/kg/week) | [93] |
| 531,851 health insured patients from Washington state (US) over 8 years | Age, sex, race and ethnicity, socioeconomic status, tobacco use, body mass index, substance use disorder and comorbidities | Low risk alcohol use | Patients with no use or very high-risk use had higher mortality Moderate-risk users had lower mortality Associations were stronger among young adults but not among sexes | [94] |
| 353,834 individuals of UK Biobank, 40–69 years, from 2006 to 2010 | Age, sex, education, household income, physical activity, smoking habits, comorbidities | Median of alcohol consumption (9.1 g/day) | Low and high consumption were associated with higher risk of contracting and dying of infectious diseases | [95] |
| 181,607 individuals of New South Wales, Australia, with 45 years or older (2005–2009), over a median of 11.4 years | Smoking, physical activity, eating habits, marital status, annual income, medical history | Low-volume drinkers (≥10 to ≤35 g/week) Pattern of drinking: cut-point of 1–3 vs. 4–7 days/week | J-shape for all alcohol-related cancer and diabetes U-shape for lower respiratory infection J-shaped for ischemic heart disease mortality: decreased risk from ≥40 to ≤200 g/week, and increased risk from ≥1170 g/week No significant effect for dementia and external causes (e.g., accidents, suicide, fall) All-cause mortality higher for >300 g/week | [88] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Malfeito-Ferreira, J.E.; Malfeito-Ferreira, M. Fermented Beverages, Ethanol and Health: A Critical Appraisal of Meta-Analytical Studies. Fermentation 2026, 12, 159. https://doi.org/10.3390/fermentation12030159
Malfeito-Ferreira JE, Malfeito-Ferreira M. Fermented Beverages, Ethanol and Health: A Critical Appraisal of Meta-Analytical Studies. Fermentation. 2026; 12(3):159. https://doi.org/10.3390/fermentation12030159
Chicago/Turabian StyleMalfeito-Ferreira, José Eduardo, and Manuel Malfeito-Ferreira. 2026. "Fermented Beverages, Ethanol and Health: A Critical Appraisal of Meta-Analytical Studies" Fermentation 12, no. 3: 159. https://doi.org/10.3390/fermentation12030159
APA StyleMalfeito-Ferreira, J. E., & Malfeito-Ferreira, M. (2026). Fermented Beverages, Ethanol and Health: A Critical Appraisal of Meta-Analytical Studies. Fermentation, 12(3), 159. https://doi.org/10.3390/fermentation12030159






