4.1. Implications from the Comprehensive Evaluation of On-Farm Silage Quality
In this study, GRA was applied to comprehensively evaluate 25 on-farm Juncao silage samples. This method allows for a more holistic and accurate objective assessment [
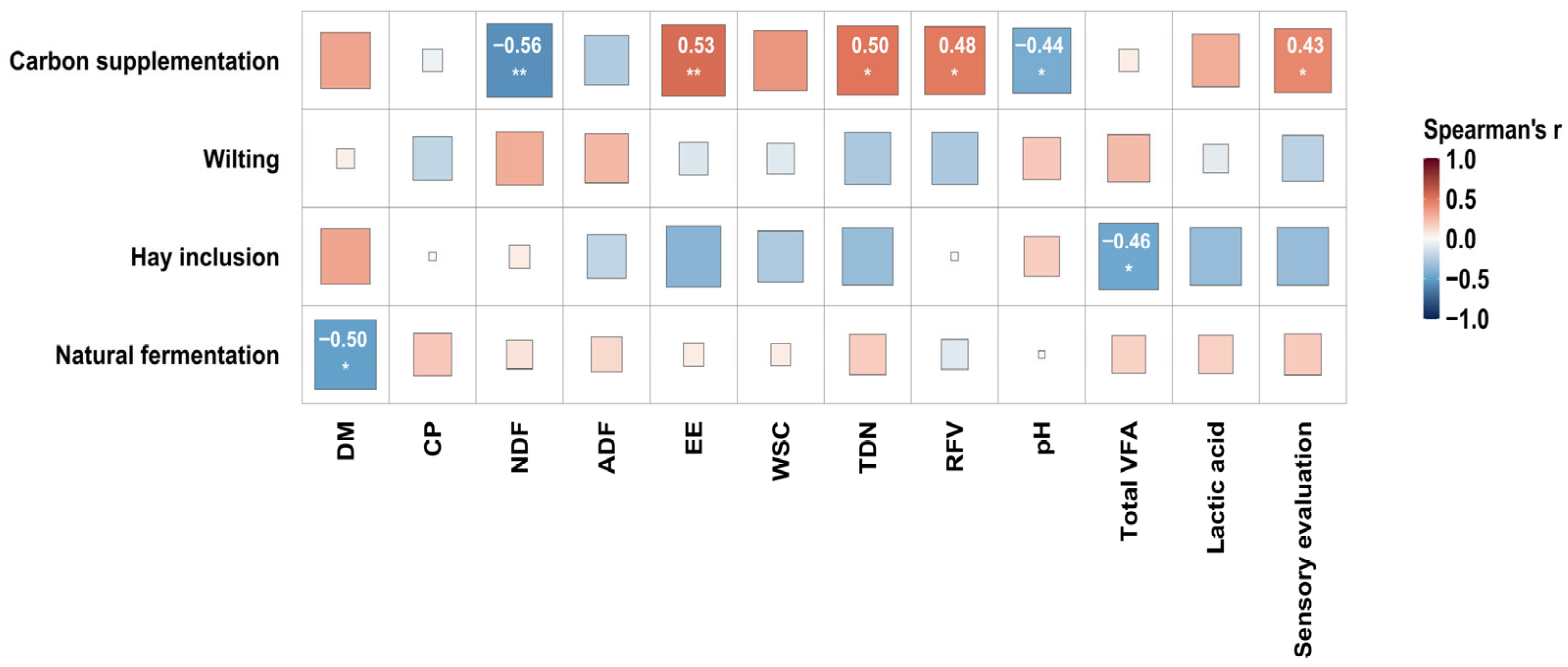
22]. The comprehensive analysis revealed that the top-ranked treatments were J2, J3, J6, X6, J5, J4, J1, X7, C4, and S1. These treatments all had sensory evaluation scores above 15, TDN contents greater than 52.8%, pH values below 4.2, total VFA contents above 7.14%, and DM contents ranging from 18.4% to 29.0%. Furthermore, carbon supplementation during ensiling, through materials such as cornmeal or rice bran, increased the DM content and enhanced the nutritional quality of the silage. For example, compared with the untreated group (J1), the addition of 4% cornmeal (J2) increased DM content by 25.37%, TDN content by 10.16%, reduced NDF content by 27.20% and ADF content by 26.98%, lowered pH by 12.32%, and raised lactic acid content by 9.14%. These findings are consistent with those reported by Wu et al. [
23], who also observed that adding 3–9% cornmeal significantly improved fermentation characteristics and nutritional compositions in silage.
Additionally, correlation analysis was conducted on these 25 samples, which further identified that natural fermentation was significantly negatively correlated with DM content, while hay inclusion showed a significant negative correlation with total VFA. This could be attributed to the fact that lower DM content often led to contamination in silage, which subsequently reduced the concentration of total VFA [
24]; or to the fact that hay inclusion increased the DM content during ensiling, but the resulting poor compaction might have resulted in inferior fermentation characteristics [
25]. Also, undesirable microorganisms attached to the hay surface might have also contributed, the underlying mechanisms of which require further investigation. Furthermore, carbon supplementation showed a significant positive correlation with sensory quality and nutritional compositions. Current research generally maintains that incorporating carbon supplementation during the ensiling process can accelerate fermentation and enhance silage quality [
23,
26]. These additives not only provide sufficient substrates for the rapid accumulation of lactic acid and a sharp decline in pH but also promote the synthesis of microbial proteins, thereby improving the overall nutritional profile [
27]. These findings are consistent with our results and demonstrate that carbon supplementation effectively improves Juncao silage quality.
4.2. Balancing Nutritional Compositions and Fermentation Characteristics at Optimal Cutting Height
Ensiling is one of the effective methods for improving nutritional quality and extending storage life. Generally, ensiling requires controlling moisture content between 60% and 70% and having a certain buffering capacity. However, for Juncao, its relatively high contents of WSC and CP make it a suitable substrate for anaerobic fermentation [
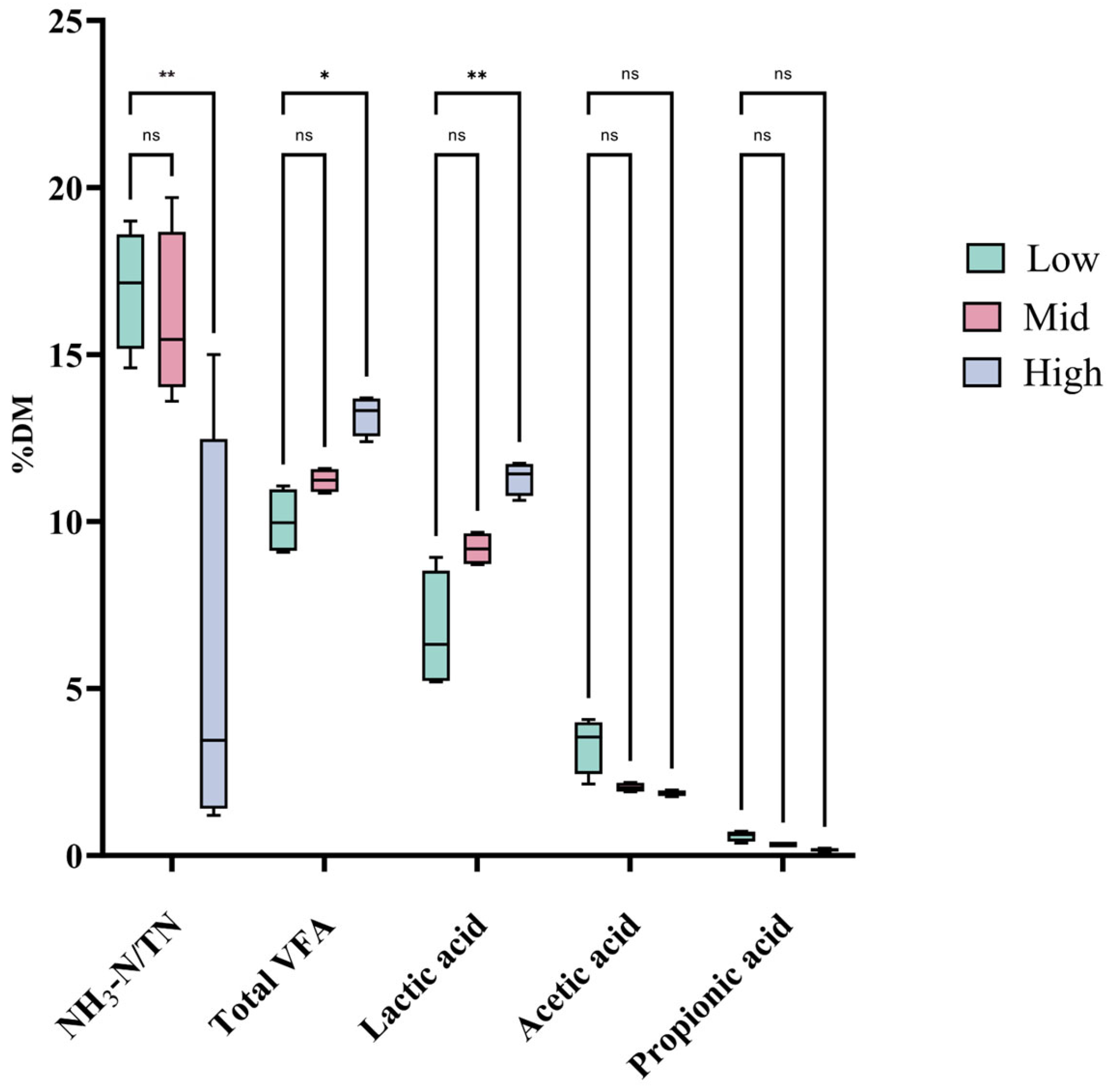
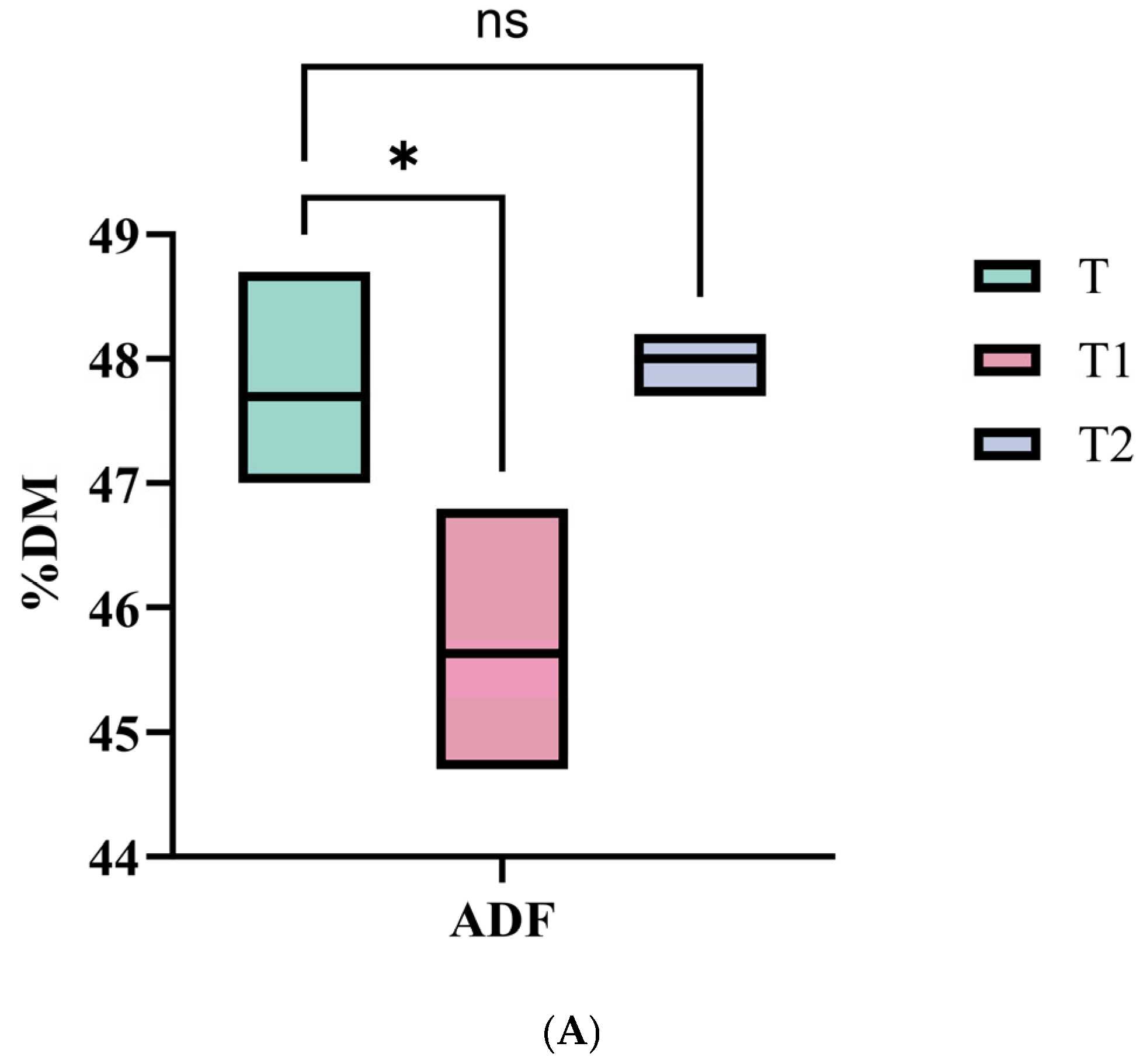
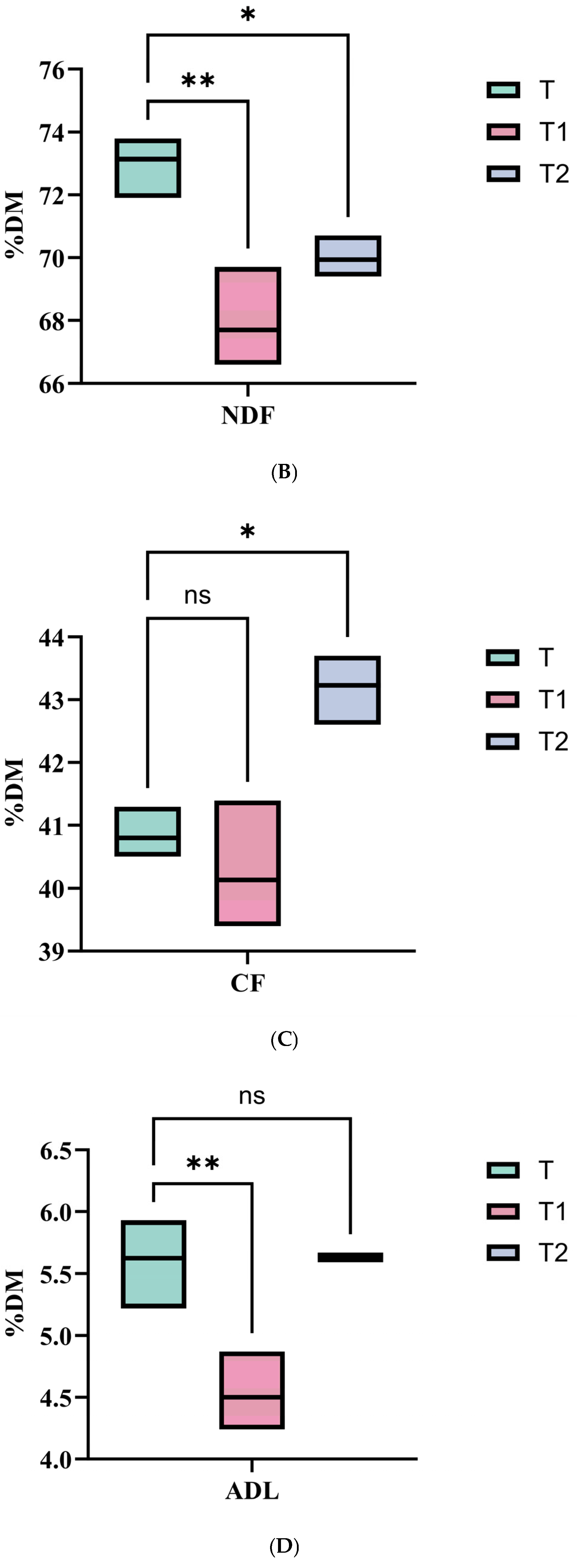
28]. In this study, the nutritional compositions of Juncao silage showed that DM, ADF, ADL, and CF increased with cutting height, while CP, EE, TDN, and RFV decreased. Regarding fermentation characteristics, total VFA and lactic acid contents increased with cutting height, whereas the NH
3-N/TN, propionic acid content, and pH decreased. These trends were corroborated by our correlation analysis, showing significant positive correlations of cutting height with DM, ADF, ADL, CF, total VFA, and lactic acid, and significant negative correlations with CP, EE, TDN, RFV, pH, NH
3-N/TN, acetic acid, and propionic acid. These results were consistent with the findings of Zhao et al. [
29]. Furthermore, the directional trends observed in our study for CP, ADF, RFV, and fermentation characteristics were in agreement with those reported by Li et al. [
30] in a separate study.
The pH serves as a primary visual and biochemical indicator for evaluating silage fermentation quality. In this study, the pH of Juncao silage decreased significantly as the cutting height increased, reaching 4.01 and 3.33 in the 150–200 cm and 200–250 cm groups, respectively. These results indicate that cutting Juncao at a height between 150 cm and 250 cm resulted in a pH decrease to a favorable range (approximately 4.0 or below). This change was primarily driven by lactic acid. Current research generally identifies lactic acid as the predominant factor influencing silage pH [
31], which was consistent with the lactic acid results observed in this study. This indicated that higher levels of lactic acid were produced during the ensiling of Juncao when cut above 150 cm. The resulting low-pH environment helped stabilize fermentation by inhibiting acid-intolerant microorganisms [
32]. Regarding the effect of cut height on pH, previous studies on buffel grass silage reported that higher cut heights resulted in significantly higher pH values compared to lower heights; however, similar to our findings, pH showed a linear decrease as height increased [
33]. This phenomenon could be attributed to the linear increase in DM content with cut height. Although the DM content in this study was relatively low, the reduction in CP and WSC after ensiling provided energy for Juncao fermentation, which aligned with the research of Zhao et al. [
28,
29]. Furthermore, Juncao contains a certain amount of starch. A higher carbohydrate content typically increases microbial abundance in silage, allowing LAB to utilize WSC to produce substantial amounts of lactic acid [
34], thereby promoting a rapid decline in pH [
35].
NH
3-N/TN is considered the most reliable variable for detecting the fermentation quality of silage. Our results demonstrated that the NH
3-N/TN concentration in Juncao silage from the 200–250 cm group was extremely significantly lower than in the other two groups. A lower proportion of NH
3-N indicated reduced protein degradation, which enhanced the apparent digestibility and overall quality of the silage [
36]. Some researchers have pointed out that the NH
3-N/TN in Juncao silage produced at a later growth stage was significantly reduced by 40.12%, and therefore recommended harvesting Juncao at a later growth stage for anaerobic fermentation to produce silage, in order to optimize fermentation quality [
29].
Silage fermentation is generally divided into six distinct stages: aerobic phase, acetic acid fermentation, initiation of lactic acid fermentation, completion of lactic acid fermentation, storage, and feeding [
37]. This general finding diverged from the pattern of higher acetic acid concentrations in low-DM silage [
38], where acetic acid was reported as a crucial precursor for milk fat synthesis in ruminants [
39]. However, our findings diverged from this pattern. In this study, although DM content increased with cutting height, correlation analysis indicated a significant negative correlation between acetic acid content and cutting height. This discrepancy may be attributed to the specific types of LAB present. Mainstream theory classifies LAB into homofermentative and heterofermentative strains [
40]. Certain heterofermentative LAB were reported to degrade lactic acid into acetic acid and propionic acid [
41]. In our results, the trends for propionic and acetic acids were similar, while the lactic acid trend was the opposite. This suggests that during Juncao ensiling, the present LAB did not degrade lactic acid into other organic acids but instead facilitated lactic acid accumulation, further depressing the pH.
In conclusion, while elevated cutting heights led to a decline in nutritional components such as CP, TDN, and RFV along with an increase in fiber fractions including ADF, ADL, and CF, they consistently promoted enhanced fermentation, as evidenced by desirable pH, high lactic acid, and low NH3-N/TN. Balancing these effects, a cutting height of 200–250 cm is proposed to maximize the overall fermentation quality of Juncao.
4.3. Efficacy and Mechanisms of Natural Fermentation vs. Microbial Inoculant
For Juncao with high moisture content, strategies such as adding materials to increase DM [
25], wilting to reduce moisture [
42], applying microbial inoculant [
43], and mixing microorganisms with fermentation substrates [
23] can promote lactic acid production during ensiling, inhibit harmful microorganisms, and enhance the nutritional value and fermentation quality of the silage.
While adding microbial inoculants is generally expected to increase protein content and minimize nutrient loss [
44], the CP content in the microbial inoculant group of this study was significantly lower than that in the natural fermentation group, contradicting this expectation. This finding aligns with the conclusion of Chen et al. [
45], who reported that adding
Lactiplantibacillus plantarum and
Bacillus licheniformis to
Pennisetum sinese silage significantly reduced NH
3-N, NDF, and ADF contents and pH compared to natural fermentation, but also resulted in decreased CP content. The reduced CP content observed in both studies might be attributed to the abundance of epiphytic (endogenous) bacteria naturally present on the forage, which may exhibit an antagonistic effect when exogenous LAB strains are supplemented.
Regarding fiber degradation, natural fermentation significantly reduced the contents of NDF, ADF, and ADL, whereas the addition of microbial inoculants showed no significant impact. This finding contrasts with several mainstream studies in which additives are typically employed to degrade cellulose. For instance, Dong et al. [
46] found that adding
Lactobacillus plantarum and cellulase significantly reduced fiber content in
Pennisetum Purpureum silage. Similarly, Silva et al. [
47] reported a linear decrease in NDF with increasing cellulase levels, achieving the lowest ADF at a 4.5% inclusion rate. The inverse results observed in this study may be explained by the higher lactic acid levels in the natural fermentation group; the sustained low pH likely facilitated the acid hydrolysis of plant cell walls, leading to the reduction in NDF and ADF values. However, other studies have also indicated that additives such as
Lacticaseibacillus rhamnosus and
Lentilactobacillus buchneri had no significant effect on NDF and ADF contents [
23].
Regarding fermentation quality, in this study, the pH of naturally fermented Juncao silage was significantly lower than that of the groups with microbial inoculants added, while the contents of total VFA and lactic acid were significantly higher. In contrast, some studies suggest that adding microbial inoculants can improve fermentation quality, increase the abundance of dominant bacterial communities, elevate lactic acid content, and lower pH [
48]. However, Tian et al. [
49] reported that among additives such as malic acid, glucose, cellulase, or
Bacillus subtilis applied to hybrid
Pennisetum silage, only malic acid significantly reduced pH, while the other methods showed no significant effect. Zhu et al. [
50] found that adding
Lactobacillus plantarum or
Bacillus licheniformis during ensiling did not significantly lower pH, and an increase in lactic acid content was only observed with
L. plantarum, and only when added at 1 × 10
7 CFU/g. Wu et al. [
23] concluded that adding microbial inoculants along with an appropriate amount of cornmeal could improve fermentation quality. However, when the cornmeal addition reached 6%, the effect of the microbial inoculants diminished, showing no clear impact on pH, whereas the role of cornmeal increased, and the lactic acid content peaked when no inoculant was added but with 6% cornmeal.
Therefore, we speculate that in this study, adding microbial inoculants such as
Lactococcus lactis and
Lactobacillus buchneri to Juncao silage did not significantly improve its post-ensiling nutritional compositions or fermentation characteristics. This may be related to the types and dosage of microorganisms added, as well as the type and amount of fermentation substrates. Additionally, the DM content of Juncao in this study was only 15.27%, which is below 20%. The lack of significant improvement in Juncao silage quality upon adding microbial inoculant may be due to the fact that the key factors determining successful ensiling are WSC and DM contents, rather than the addition of microbial inoculant [
51].
Although this study comprehensively evaluated on-farm silage quality, determined the optimal cutting height, and compared natural fermentation with microbial inoculant, certain limitations remain. The selection of microbial inoculant was not exhaustive; instead, a mainstream microbial inoculant was utilized. Given that Juncao is a tropical plant rich in endogenous microbiota, future research should prioritize comprehensive microbial profiling and strain identification to develop specialized inoculant tailored to this species. Nevertheless, this study demonstrates that compared to conventional microbial inoculant additives, natural fermentation of Juncao can achieve excellent results. This finding not only suggests a potential reduction in input and labor costs but also provides a theoretical foundation and new perspectives for subsequent research on high-quality Juncao silage production.