Native Carbonic Anhydrase Activity Provides a Critical and Sufficient CO2 Concentrating Mechanism for Escherichia coli Succinate Fermentation
Abstract
1. Introduction
2. Materials and Methods
2.1. Media and Culture Conditions
2.2. Strain and Plasmid Construction
2.3. Cultivation on Agar Plates Under Controlled Atmospheric Conditions
2.4. Fermentation Product Analysis
3. Results and Discussion
3.1. E. coli’s Native CCM Is Critical for Efficient Succinate Fermentation
3.2. E. coli’s Native CCM Enables (An)Aerobic Growth in Low Extracellular Ci Environments
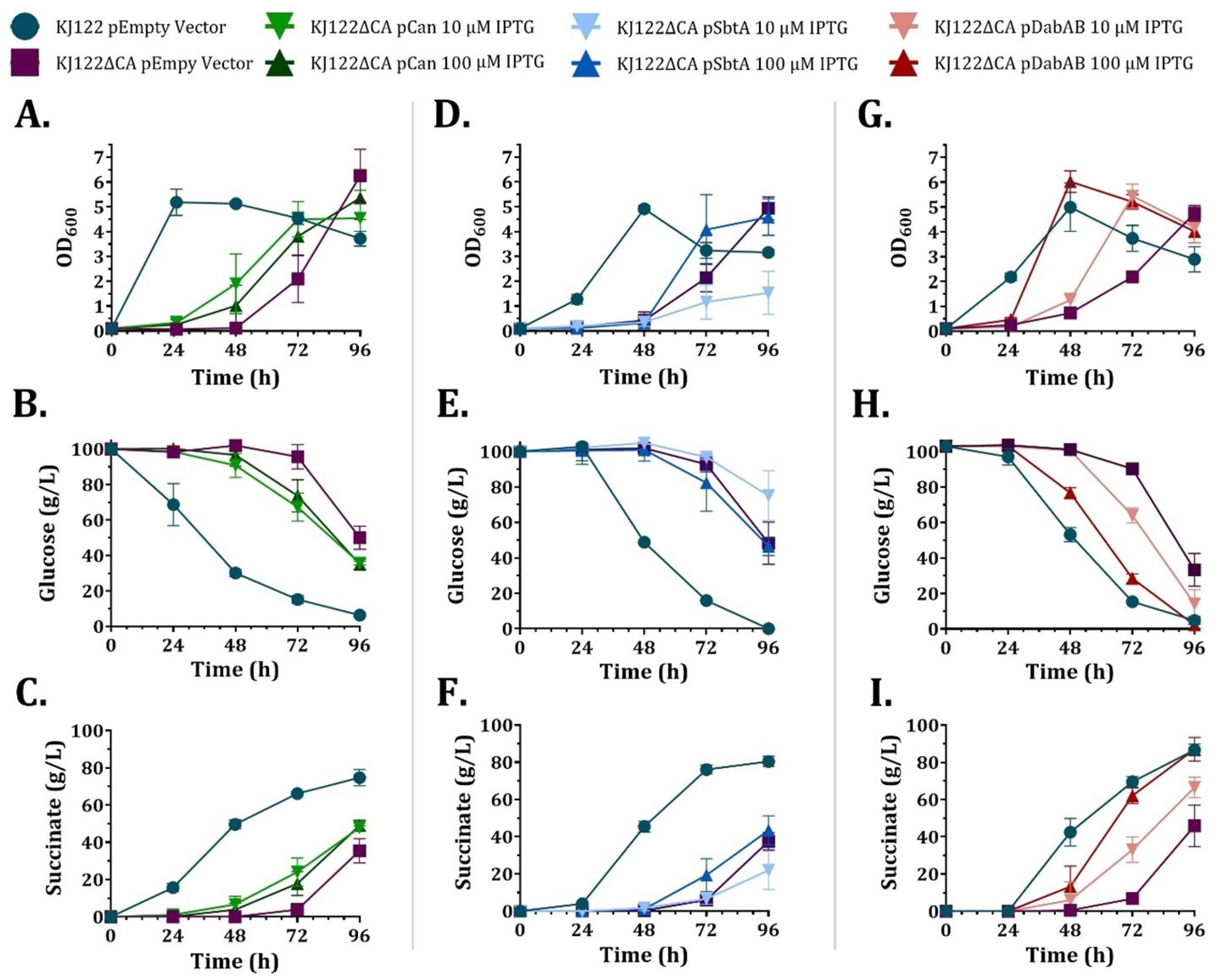
3.3. Heterologous CCM Components Restore Growth of KJ122ΔCA
3.4. Heterologous CCM Components Partially Restore Succinate Production in KJ122ΔCA
3.5. Functional Enhancement of E. coli’s Native CCM Does Not Improve Succinate Fermentation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Intergovernmental Panel on Climate Change (IPCC). Technical Summary. In Climate Change 2021—The Physical Science Basis; Cambridge University Press: Cambridge, UK, 2023; pp. 35–144. [Google Scholar] [CrossRef]
- Energy Institute. Statistical Review of World Energy; Energy Institute: London, UK, 2025; Available online: https://www.energyinst.org/statistical-review (accessed on 25 October 2025).
- Liu, H.; Jin, Y.; Zhang, R.; Ning, Y.; Yu, Y.; Xu, P.; Deng, L.; Wang, F. Recent advances and perspectives on production of value-added organic acids through metabolic engineering. Biotechnol. Adv. 2023, 62, 108076. [Google Scholar] [CrossRef]
- Ahn, J.H.; Seo, H.; Park, W.; Seok, J.; Lee, J.A.; Kim, W.J.; Kim, G.B.; Kim, K.-J.; Lee, S.Y. Enhanced succinic acid production by Mannheimia employing optimal malate dehydrogenase. Nat. Commun. 2020, 11, 1970. [Google Scholar] [CrossRef] [PubMed]
- Barletta, M.; Aversa, C.; Ayyoob, M.; Gisario, A.; Hamad, K.; Mehrpouya, M.; Vahabi, H. Poly(butylene succinate) (PBS): Materials, processing, and industrial applications. Prog. Polym. Sci. 2022, 132, 101579. [Google Scholar] [CrossRef]
- Silva, D.A.S.; Luque, R.; Delgado-Arcaño, Y.; Perpetuo, E.A.; Ruy, A.D.d.S.; de Souza, A.L.B.; Pontes, L.A. Prospecting technology and economic potential to produce bio-succinic acid: A review. Biotechnol. Rep. 2025, 48, e00920. [Google Scholar] [CrossRef]
- Pinazo, J.M.; Domine, M.E.; Parvulescu, V.; Petru, F. Sustainability metrics for succinic acid production: A comparison between biomass-based and petrochemical routes. Catal. Today 2015, 239, 17–24. [Google Scholar] [CrossRef]
- Ahn, J.H.; Jang, Y.S.; Lee, S.Y. Production of succinic acid by metabolically engineered microorganisms. Curr. Opin. Biotechnol. 2016, 42, 54–66. [Google Scholar] [CrossRef]
- Wu, N.; Wang, W.; Zhu, J. Progress on production of malic acid and succinic acid by industrially-important engineered microorganisms. J. Biotechnol. 2025, 400, 8–19. [Google Scholar] [CrossRef]
- Cok, B.; Tsiropoulos, I.; Roes, A.L.; Patel, M.K. Succinic acid production derived from carbohydrates: An energy and greenhouse gas assessment of a platform chemical toward a bio-based economy. Biofuels Bioprod. Biorefin. 2014, 8, 16–29. [Google Scholar] [CrossRef]
- Huang, K.; Peng, X.; Kong, L.; Wu, W.; Chen, Y.; Maravelias, C.T. Greenhouse Gas Emission Mitigation Potential of Chemicals Produced from Biomass. ACS Sustain. Chem. Eng. 2021, 9, 14480–14487. [Google Scholar] [CrossRef]
- Shen, N.; Wang, Q.; Qin, Y.; Zhu, J.; Zhu, Q.; Mi, H.; Wei, Y.; Huang, R. Optimization of succinic acid production from cane molasses by Actinobacillus succinogenes GXAS137 using response surface methodology (RSM). Food Sci. Biotechnol. 2014, 23, 1911–1919. [Google Scholar] [CrossRef]
- Liu, X.; Li, L.; Zhao, G.; Xiong, P. Optimization strategies for CO2 biological fixation. Biotechnol. Adv. 2024, 73, 108364. [Google Scholar] [CrossRef] [PubMed]
- Merlin, C.; Masters, M.; McAteer, S.; Coulson, A. Why is carbonic anhydrase essential to Escherichia coli? J. Bacteriol. 2003, 185, 6415–6424. [Google Scholar] [CrossRef]
- Slonczewski, J.L.; Rosen, B.P.; Alger, J.R.; Macnab, R.M. pH homeostasis in Escherichia coli: Measurement by 31P nuclear magnetic resonance of methylphosphonate and phosphate. Proc. Natl. Acad. Sci. USA 1981, 78, 6271–6275. [Google Scholar] [CrossRef] [PubMed]
- Tolleter, D.; Chochois, V.; Poiré, R.; Dean Price, G.; Badger, M.R. Measuring CO2 and HCO3− permeabilities of isolated chloroplasts using a MIMS-18O approach. J. Exp. Bot. 2017, 68, 3915–3924. [Google Scholar] [CrossRef]
- Patel, T.N.; Alissa Park, A.-H.; Banta, S. Periplasmic Expression of Carbonic Anhydrase in Escherichia coli: A New Biocatalyst for CO2 Hydration. Biotechnol. Bioeng. 2013, 110, 1865–1873. [Google Scholar] [CrossRef]
- Mitchell, M.J.; Jensen, O.E.; Cliffe, K.A.; Maroto-Valer, M.M. A model of carbon dioxide dissolution and mineral carbonation kinetics. Proc. R. Soc. A Math. Phys. Eng. Sci. 2010, 466, 1265–1290. [Google Scholar] [CrossRef]
- Guilloton, M.B.; Korte, J.J.; Lamblin, A.F.; Fuchs, J.A.; Anderson, P.M. Carbonic anhydrase in Escherichia coli. A product of the cyn operon. J. Biol. Chem. 1992, 267, 3731–3734. [Google Scholar] [CrossRef]
- Guilloton, M.B.; Lamblin, A.F.; Kozliak, E.I.; Gerami-Nejad, M.; Tu, C.; Silverman, D.; Anderson, P.M.; Fuchs, J.A. A physiological role for cyanate-induced carbonic anhydrase in Escherichia coli. J. Bacteriol. 1993, 175, 1443–1451. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Hou, W.-T.; Wang, L.; Li, B.; Chen, Y.; Chen, Y.; Jiang, Y.-L.; Zhou, C.-Z. Structures of cyanobacterial bicarbonate transporter SbtA and its complex with PII-like SbtB. Cell Discov. 2021, 7, 63. [Google Scholar] [CrossRef]
- Liu, L.-N. Advances in the bacterial organelles for CO2 fixation. Trends Microbiol. 2022, 30, 567–580. [Google Scholar] [CrossRef]
- Desmarais, J.J.; Flamholz, A.I.; Blikstad, C.; Dugan, E.J.; Laughlin, T.G.; Oltrogge, L.M.; Chen, A.W.; Wetmore, K.; Diamond, S.; Wang, J.Y.; et al. DABs are inorganic carbon pumps found throughout prokaryotic phyla. Nat. Microbiol. 2019, 4, 2204–2215. [Google Scholar] [CrossRef]
- Scott, K.M.; Leonard, J.M.; Boden, R.; Chaput, D.; Dennison, C.; Haller, E.; Harmer, T.L.; Anderson, A.; Arnold, T.; Budenstein, S.; et al. Diversity in CO2 Concentrating Mechanisms among Chemolithoautotrophs from the Genera Hydrogenovibrio, Thiomicrorhabdus, and Thiomicrospira, Ubiquitous in Sulfidic Habitats Worldwide. Appl. Environ. Microbiol. 2019, 85, e02096-18. [Google Scholar] [CrossRef]
- Fan, S.; Matsuo, M.; Huang, L.; Tribelli, P.M.; Götz, F. The MpsAB Bicarbonate Transporter Is Superior to Carbonic Anhydrase in Biofilm-Forming Bacteria with Limited CO2 Diffusion. Microbiol. Spectr. 2021, 9, e0030521. [Google Scholar] [CrossRef]
- Supuran, C.T. A simple yet multifaceted 90 years old, evergreen enzyme: Carbonic anhydrase, its inhibition and activation. Bioorganic Med. Chem. Lett. 2023, 93, 129411. [Google Scholar] [CrossRef] [PubMed]
- Jo, B.H.; Kim, I.G.; Seo, J.H.; Kang, D.G.; Cha, H.J. Engineered Escherichia coli with periplasmic carbonic anhydrase as a biocatalyst for CO2 sequestration. Appl. Environ. Microbiol. 2013, 79, 6697–6705. [Google Scholar] [CrossRef] [PubMed]
- Youse, M.S.; Holly, K.J.; Flaherty, D.P. Chapter Nine—Neisseria gonorrhoeae carbonic anhydrase inhibition. Enzymes 2024, 55, 243–281. [Google Scholar] [CrossRef]
- Jantama, K.; Zhang, X.; Moore, J.; Shanmugam, K.; Svoronos, S.; Ingram, L. Eliminating side products and increasing succinate yields in engineered strains of Escherichia coli C. Biotechnol. Bioeng. 2008, 101, 881–893. [Google Scholar] [CrossRef] [PubMed]
- Datsenko, K.A.; Wanner, B.L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA 2000, 97, 6640–6645. [Google Scholar] [CrossRef]
- Flores, A.D.; Ayla, E.Z.; Nielsen, D.R.; Wang, X. Engineering a Synthetic, Catabolically Orthogonal Coculture System for Enhanced Conversion of Lignocellulose-Derived Sugars to Ethanol. ACS Synth. Biol. 2019, 8, 1089–1099. [Google Scholar] [CrossRef]
- Gibson, D.G.; Young, L.; Chuang, R.-Y.; Venter, J.C.; Hutchison, C.A., III; Smith, H.O. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 2009, 6, 343–345. [Google Scholar] [CrossRef]
- Engler, C.; Kandzia, R.; Marillonnet, S. A One Pot, One Step, Precision Cloning Method with High Throughput Capability. PLoS ONE 2008, 3, e3647. [Google Scholar] [CrossRef]
- Flamholz, A.I.; Dugan, E.; Blikstad, C.; Gleizer, S.; Ben-Nissan, R.; Amram, S.; Antonovsky, N.; Ravishankar, S.; Noor, E.; Bar-Even, A.; et al. Functional reconstitution of a bacterial CO2 concentrating mechanism in Escherichia coli. eLife 2020, 9, e59882. [Google Scholar] [CrossRef]
- Flores, A.D.; Holland, S.C.; Mhatre, A.; Sarnaik, A.P.; Godar, A.; Onyeabor, M.; Varman, A.M.; Wang, X.; Nielsen, D.R. A coculture-coproduction system designed for enhanced carbon conservation through inter-strain CO2 recycling. Metab. Eng. 2021, 67, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Schmid, S.; Chaput, D.; Breitbart, M.; Hines, R.; Williams, S.; Gossett, H.K.; Parsi, S.D.; Peterson, R.; Whittaker, R.A.; Tarver, A.; et al. Dissolved Inorganic Carbon-Accumulating Complexes from Autotrophic Bacteria from Extreme Environments. J. Bacteriol. 2021, 203, e0037721. [Google Scholar] [CrossRef]
- Du, J.; Förster, B.; Rourke, L.; Howitt, S.M.; Price, G.D. Characterisation of cyanobacterial bicarbonate transporters in E. coli shows that SBTA homologs are functional in this heterologous expression system. PLoS ONE 2014, 9, e115905. [Google Scholar] [CrossRef]
- Park, S.; Lee, J.-U.; Cho, S.; Kim, H.; Bin Oh, H.; Pack, S.P.; Lee, J. Increased incorporation of gaseous CO2 into succinate by Escherichia coli overexpressing carbonic anhydrase and phosphoenolpyruvate carboxylase genes. J. Biotechnol. 2017, 241, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Kim, H.; Cho, S.; You, G.; Bin Oh, H.; Han, J.H.; Lee, J. Enhanced Incorporation of Gaseous CO2 to Succinate by a Recombinant Escherichia coli W3110. Biotechnol. Bioprocess Eng. 2019, 24, 103–108. [Google Scholar] [CrossRef]
- Zhu, L.W.; Zhang, L.; Wei, L.-N.; Li, H.-M.; Yuan, Z.-P.; Chen, T.; Tang, Y.-L.; Liang, X.-H.; Tang, Y.-J. Collaborative regulation of CO2 transport and fixation during succinate production in Escherichia coli. Sci. Rep. 2015, 5, 17321. [Google Scholar] [CrossRef]
- Xiao, M.; Zhu, X.; Bi, C.; Ma, Y.; Zhang, X. Improving Succinate Productivity by Engineering a Cyanobacterial CO2 Concentrating System (CCM) in Escherichia coli. Biotechnol. J. 2017, 12, 1700199. [Google Scholar] [CrossRef]





| Strain | Genotype/Features | Source |
| MG1655 | F-lambda-ilvG-rfb-50 rph-1 | Nielsen Lab |
| EDCM636 | MG1655 ΔlacZY, can< >FLK2 | CGSC |
| MG1655ΔCA | MG1655 Δcan cynT::FRT | This study |
| KJ122 | ATCC 8739, pck*, ptsI*, galR*, galS*, ΔldhA, ΔadhE, ΔackA, Δ(focA-pflB) ΔmgsA, ΔpoxB, ΔtdcDE, ΔcitF, ΔaspC, ΔsfcA; pck* stands for a mutated form of pck (G to A at position−64 relative to the ATG start codon), ptsI* stands for a mutated form of ptsI (single base deletion at position 1673 causing a frameshift mutation in the carboxyl-terminal region), galR* stands for an IS1 element inserted at position 261 in the galR ORF, galS* stands for an adenine insertion at position 231 in the galS ORF. | [29] |
| KJ122ΔCA | KJ122 Δcan cynT::FRT | This study |
| Plasmid | Genotype/Features | Source |
| pFE-dabAB2 | Source of dabAB | Addgene |
| pTrc99A | ColE1 ori, lacI, ampR, Ptrc | Nielsen Lab |
| pCan | pTrc99A-Trc-can | This study |
| pDabAB | pTrc99A-Trc-dabAB | This study |
| pCAN.g. | pTrc99A-Trc-CAN.g. | This study |
| pSbtA | pTrc99A-Trc-sbtA | This study |
| CCM Component Expressed | Glucose Consumed (g/L) | Succinate Produced (g/L) | Yield (gsuccinate/gglucose) | Succinate Production Rate (g/L-h) |
|---|---|---|---|---|
| Can | 99.6 ± 0.6 | 90.3 ± 0.7 | 0.91 ± 0.02 | 0.94 ± 0.02 |
| DabAB | 96.9 ± 0.7 | 83.5 ± 0.3 | 0.86 ± 0.00 | 0.87 ± 0.00 |
| SbtA | 97.2 ± 0.8 | 73.2 ± 3.8 | 0.75 ± 0.03 | 0.76 ± 0.04 |
| Empty Vector | 97.2 ± 2.9 | 84.4 ± 5.5 | 0.87 ± 0.06 | 0.87 ± 0.06 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Godar, A.G.; Cristobal, F.; Taquillo, L.; Wang, X.; Nielsen, D.R. Native Carbonic Anhydrase Activity Provides a Critical and Sufficient CO2 Concentrating Mechanism for Escherichia coli Succinate Fermentation. Fermentation 2026, 12, 101. https://doi.org/10.3390/fermentation12020101
Godar AG, Cristobal F, Taquillo L, Wang X, Nielsen DR. Native Carbonic Anhydrase Activity Provides a Critical and Sufficient CO2 Concentrating Mechanism for Escherichia coli Succinate Fermentation. Fermentation. 2026; 12(2):101. https://doi.org/10.3390/fermentation12020101
Chicago/Turabian StyleGodar, Amanda G., Francesca Cristobal, Luis Taquillo, Xuan Wang, and David R. Nielsen. 2026. "Native Carbonic Anhydrase Activity Provides a Critical and Sufficient CO2 Concentrating Mechanism for Escherichia coli Succinate Fermentation" Fermentation 12, no. 2: 101. https://doi.org/10.3390/fermentation12020101
APA StyleGodar, A. G., Cristobal, F., Taquillo, L., Wang, X., & Nielsen, D. R. (2026). Native Carbonic Anhydrase Activity Provides a Critical and Sufficient CO2 Concentrating Mechanism for Escherichia coli Succinate Fermentation. Fermentation, 12(2), 101. https://doi.org/10.3390/fermentation12020101






