Enhanced Squalene Production by Thraustochytrium sp. RT2316-16 by Polyphenols from Barley Bagasse
Abstract
1. Introduction
2. Materials and Methods
2.1. Culture Experiments
2.2. Effect of Terbinafine and Polyphenols from Barley Bagasse on Squalene Production by Thraustochytrium sp. RT2316-16
2.3. RNA Extraction
2.4. Conversion of RNA to cDNA
2.5. Conventional PCR
2.6. qRT-PCR
2.7. Concentration of Biomass
2.8. Concentration of Glucose
2.9. Extraction of Total Lipids and Determination of the Fatty Acid Profile
2.10. Squalene Content in the Biomass
2.11. Analysis of the Residual Concentration of Amino Acids
2.12. Extraction and Quantification of Polyphenols from Barley Bagasse
2.13. Statistical Analysis
2.14. Kinetic Parameters
3. Results
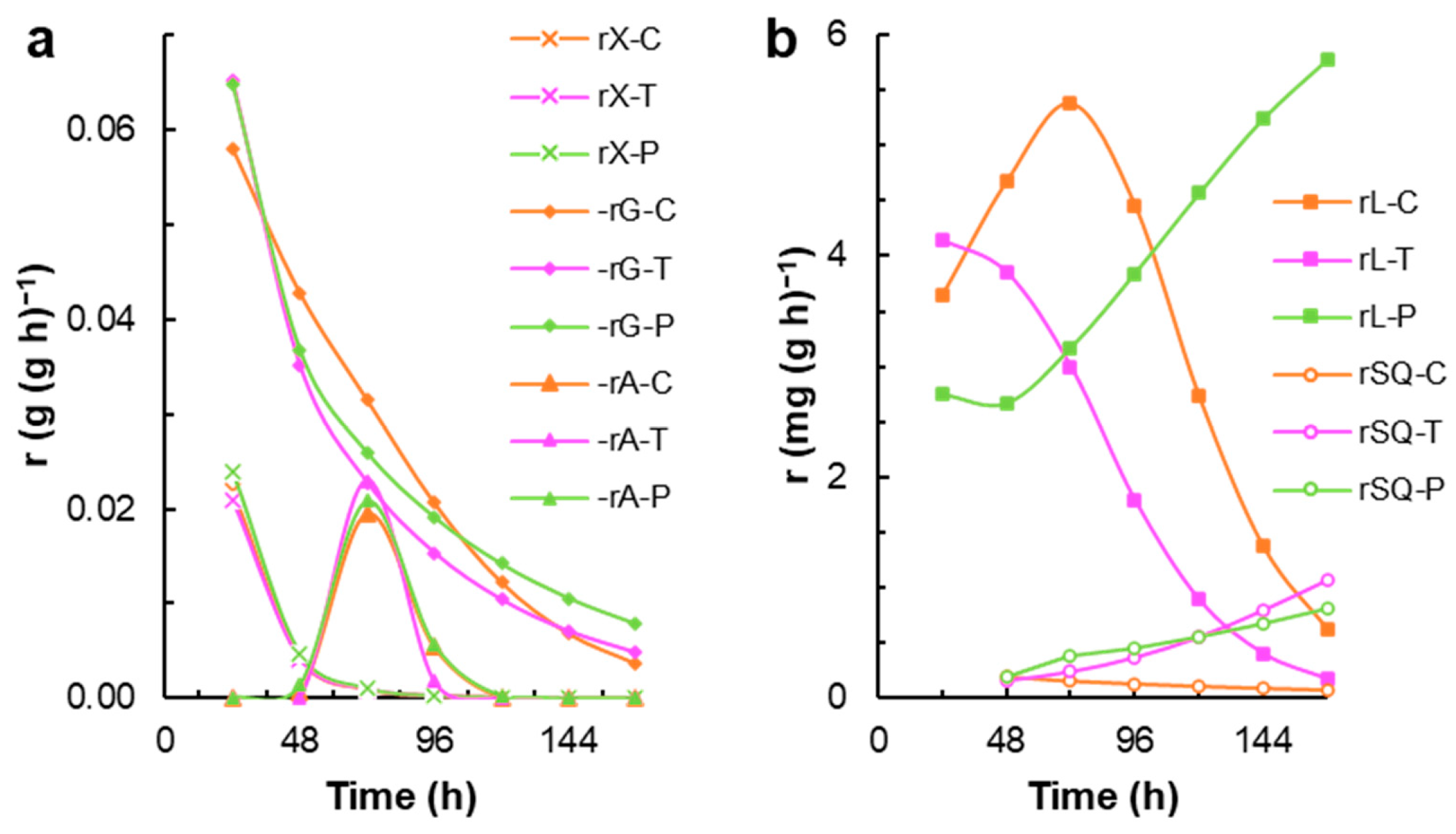
3.1. Effect of Terbinafine and Polyphenols from Barley Bagasse on the Biomass Growth and Squalene Content in Thraustochytrium sp. RT2316-16
3.2. Effect of Terbinafine and the Polyphenols from Barley Bagasse on the Fatty Acid Composition of the Total Lipids in Thraustochytrium sp. RT2316-16
3.3. Relative Expression of Genes in the Mevalonate Pathway in Thraustochytrium sp. RT2316-16
4. Discussion
4.1. Terbinafine and Polyphenols from Barley Bagasse Enhancers of the Squalene Content in Thraustochytrium sp. RT2316-16
4.2. Fatty Acid Profile of the Lipids Produced by Thraustochytrium sp. RT2316-16
4.3. Relative Expression of Some Genes in the Mevalonate and Sterol/Cholesterol Pathway in Thraustochytrium sp. RT2316-16
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Huang, Z.R.; Lin, Y.K.; Fang, J.Y. Biological and pharmacological activities of squalene and related compounds: Potential uses in cosmetic dermatology. Molecules 2009, 14, 540–554. [Google Scholar] [CrossRef]
- Kim, S.K.; Karadeniz, F. Biological importance and applications of squalene and squalane. Adv. Food Nutr. Res. 2012, 65, 223–233. [Google Scholar] [CrossRef]
- Zhu, J.; Wang, Y.; Zhu, K.; Zhang, C. Advances in understanding the role of squalene epoxidase in cancer prognosis and resistance. Mol. Biol. Rep. 2025, 52, 162. [Google Scholar] [CrossRef]
- Ahmed, B.S.; Mhammad, H. Squalene Exploring Its Vital Roles in Vaccine Production, Skincare, Cholesterol Metabolism, Anti-Cancer Strategies, Cardiovascular Health, and Antioxidant Potency. Bull. Pharm. Sci. Assiut Univ. 2024, 47, 437–448. [Google Scholar] [CrossRef]
- Ibrahim, N.I.; Mohamed, M.I. Interdependence of anti-inflammatory and antioxidant properties of squalene–implication for cardiovascular health. Life 2021, 11, 103. [Google Scholar] [CrossRef]
- Grand View Research, Inc. Squalene Market Size and Share|Industry Report, 2030. Grand View Research. Available online: https://www.grandviewresearch.com/industry-analysis/squalene-market (accessed on 8 January 2026).
- Deprez, P.P.; Volkman, J.K.; Davenport, S.R.B. Squalene Content and Neutral Lipid Composition of Livers from Deep-sea Sharks Caught in Tasmanian Waters. Aust. J. Mar. Freshwater Res. 1990, 41, 375–387. [Google Scholar] [CrossRef]
- Hernández, M.L.; Muñoz-Ocaña, C.; Posada, P.; Sicardo, M.D.; Hornero-Méndez, D.; Gómez-Coca, R.B.; Belaj, A.; Moreda, W.; Martínez-Rivas, J.M. Functional Characterization of Four Olive Squalene Synthases with Respect to the Squalene Content of the Virgin Olive Oil. J. Agric. Food Chem. 2023, 71, 15701–15712. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Sreerama, Y.N.; Dharmaraj, U. Effect of processing on squalene content of grain amaranth fractions. J. Cereal Sci. 2021, 100, 103218. [Google Scholar] [CrossRef]
- Chabrol, R. The Hideous Price of Beauty. An Investigation into the Market of Deep-Sea Shark Liver Oil. Bloom Association. 2012. Available online: https://www.google.com/url?sa=t&source=web&rct=j&opi=89978449&url=https://www.bloomassociation.org/en/wp-content/uploads/2013/10/ENG_Squalene_4-pager.pdf&ved=2ahUKEwichorB-5iSAxX2mSYFHXp3GgYQFnoECCQQAQ&usg=AOvVaw1WtxmXQy2dRtbJGq9Q3iVW (accessed on 18 January 2026).
- DataVagyanik. Squalane (Hydrogenated Squalene) Market Size, Production, Price, Market Share, Import vs Export, and Top Latest Trends, Till 2035. DataVagyanik. 2025. Available online: https://datavagyanik.com/reports/squalane-hydrogenated-squalene-market-size-production-sales-average-product-price-market-share-import-vs-export (accessed on 8 January 2026).
- PW Consulting Chemical & Energy Research Center. Squalene Market. 2024. Available online: https://pmarketresearch.com/chemi/squalene-market (accessed on 8 January 2026).
- Olzmann, J.A.; Carvalho, P. Dynamics and functions of lipid droplets. Nat. Rev. Mol. 2019, 20, 137–155. [Google Scholar] [CrossRef] [PubMed]
- Csáky, Z.; Garaiová, M.; Kodedová, M.; Valachovič, M.; Sychrová, H.; Hapala, I. Squalene lipotoxicity in a lipid droplet-less yeast mutant is linked to plasma membrane dysfunction. Yeast 2020, 37, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.-J.; Solbiati, J.O.; Ramamoorthy, G.; Hillerich, B.S.; Seidel, R.D.; Cronan, J.E.; Almo, S.C.; Poulter, C.D. Biosynthesis of squalene from farnesyl diphosphate in bacteria: Three steps catalyzed by three enzymes. ACS Cent. Sci. 2015, 1, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, K.; Saito, N.; Shibuya, S.; Kobayashi, W.; Amano, R.; Hirai, T.; Sasaki, S.; Nakano, C.; Hoshino, T. Biochemical characterization of the water-soluble squalene synthase from Methylococcus capsulatus and the functional analyses of its two DXXD (E) D motifs and the highly conserved aromatic amino acid residues. FEBS J. 2014, 281, 5479–5497. [Google Scholar] [CrossRef]
- Lee, S.; Poulter, C.D. Cloning, solubilization, and characterization of squalene synthase from Thermosynechococcus elongates BP-1. J. Bacteriol. 2008, 190, 3808–3816. [Google Scholar] [CrossRef]
- Padyana, A.K.; Gross, S.; Jin, L.; Cianchetta, G.; Narayanaswamy, R.; Wang, F.; Wang, R.; Fang, C.; Lv, X.; Biller, S.A.; et al. Structure and inhibition mechanism of the catalytic domain of human squalene epoxidase. Nat. Commun. 2019, 10, 97. [Google Scholar] [CrossRef]
- Grundy, S.M. HMG-CoA reductase inhibitors for treatment of hypercholesterolemia. N. Engl. J. Med. 1988, 319, 24–33. [Google Scholar] [CrossRef]
- Wang, L.; Sha, Y.; Wu, D.; Wei, Q.; Chen, D.; Yang, S.; Jia, F.; Yuan, Q.; Han, X.; Wang, J. Surfactant induces ROS-mediated cell membrane permeabilization for the enhancement of mannatide production. Process. Biochem. 2020, 91, 172–180. [Google Scholar] [CrossRef]
- Wolkers, W.; Oldenhof, H.; Tang, F.; Han, J.; Bigalk, J.; Sieme, H. Factors affecting the membrane permeability barrier function of cells during preservation technologies. Langmuir 2018, 35, 7520–7528. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Fan, Z.; Deng, C.X. Effects of shear stress cultivation on cell membrane disruption and intracellular calcium concentration in sonoporation of endothelial cells. J. Biomech. 2011, 44, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Silver, S.; Wendt, L. Mechanism of action of phenethyl alcohol: Breakdown of the cellular permeability barrier. J. Bacteriol. 1967, 93, 560–566. [Google Scholar] [CrossRef]
- Nowosielski, M.; Hoffmann, M.; Wyrwicz, L.S.; Stepniak, P.; Plewczynski, D.M.; Lazniewski, M.; Ginalski, K.; Rychlewski, L. Detailed Mechanism of Squalene Epoxidase Inhibition by Terbinafine. J. Chem. Inf. Model. 2011, 51, 455–462. [Google Scholar] [CrossRef]
- Fan, K.W.; Aki, T.; Chen, F.; Jiang, Y. Enhanced production of squalene in the thraustochytrid Aurantiochytrium mangrovei by medium optimization and treatment with terbinafine. World J. Microbiol. Biotechnol. 2010, 26, 1303–1309. [Google Scholar] [CrossRef] [PubMed]
- Quilodrán, B.; Cortinez, G.; Bravo, A.; Silva, D. Characterization and comparison of lipid and PUFA production by native thraustochytrid strains using complex carbon sources. Heliyon 2020, 6, e05404. [Google Scholar] [CrossRef]
- Jakobsen, N.A.; Aasen, I.M.; Josefsen, K.D.; Strøm, A.R. Accumulation of docosahexaenoic acid-rich lipid in thraustochytrid Aurantiochytrium sp. strain T66: Effects of N and P starvation and O2 limitation. Appl. Microbiol. Biotechnol. 2008, 80, 297–306. [Google Scholar] [CrossRef]
- Leyton, A.; Flores, L.; Shene, C.; Chisti, Y.; Larama, G.; Asenjo, J.A.; Armenta, R.E. Antarctic thraustochytrids as sources of carotenoids and high-value fatty acids. Mar. Drugs 2021, 19, 386. [Google Scholar] [CrossRef]
- Flores, L.; Shene, C.; Asenjo, J.A.; Chisti, Y. Coenzyme Q in Thraustochytrium sp. RT2316-16: Effect of the Medium Composition. Mar. Drugs 2023, 21, 586. [Google Scholar] [CrossRef]
- Paredes, P.; Flores, L.; Bustamante, M.; Chisti, Y.; Asenjo, J.A.; Shene, C. Production of squalene and fatty acids by Thraustochytrium sp. RT2316-16: Effects of dissolved oxygen and the medium composition. Bioresour. Bioprocess. 2025, 12, 98. [Google Scholar] [CrossRef]
- Bi, Y.; Guo, P.; Liu, L.; Chen, L.; Zhang, W. Elucidation of sterol biosynthesis pathway and its co-regulation with fatty acid biosynthesis in the oleaginous marine protist Schizochytrium sp. Front. Bioeng. Biotechnol. 2023, 11, 1188461. [Google Scholar] [CrossRef] [PubMed]
- Shene, C.; Leyton, A.; Rubilar, M.; Pinelo, M.; Acevedo, F.; Morales, E. Production of lipids and docosahexaenoic acid by a native Thraustochytrium strain. Eur. J. Lipid Sci. Technol. 2013, 115, 890–900. [Google Scholar] [CrossRef]
- Li, S.; Hu, Z.; Yang, X.; Li, Y. Effect of nitrogen sources on Omega-3 polyunsaturated fatty acid biosynthesis and gene expression in Thraustochytriidae sp. Mar. Drugs 2020, 18, 612. [Google Scholar] [CrossRef]
- Ma, Z.; Tan, Y.; Cui, G.; Feng, Y.; Cui, Q.; Song, X. Transcriptome and gene expression analysis of DHA producer Aurantiochytrium under low temperature conditions. Sci. Rep. 2015, 5, 14446. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Leyton, A.; Shene, C.; Chisti, Y.; Asenjo, J.A. Production of carotenoids and phospholipids by Thraustochytrium sp. in batch and repeated-batch culture. Mar. Drugs 2022, 20, 416. [Google Scholar] [CrossRef]
- Budge, S.M.; Barry, C. Determination of squalene in edible oils by transmethylation and GC analysis. MethodsX 2019, 6, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, P.M.; Petersen, D.; Dambmann, C. Improved method for determining food protein degree of hydrolysis. J. Food Sci. 2001, 66, 642–646. [Google Scholar] [CrossRef]
- Meneses, N.G.; Martins, S.; Teixeira, J.A.; Mussatto, S.I. Influence of extraction solvents on the recovery of antioxidant phenolic compounds from brewer’s spent grains. Sep. Purif. Technol. 2013, 108, 152–158. [Google Scholar] [CrossRef]
- Han, J.Y.; Seo, S.H.; Song, J.M.; Lee, H.; Choi, E.S. High-level recombinant production of squalene using selected Saccharomyces cerevisiae strains. J. Ind. Microbiol. Biotechnol. 2018, 45, 239–251. [Google Scholar] [CrossRef]
- Tang, W.Y.; Wang, D.P.; Tian, Y.; Fan, X.; Wang, C.; Lu, X.Y.; Li, P.W.; Ji, X.J.; Liu, H.H. Metabolic engineering of Yarrowia lipolytica for improving squalene production. Bioresour. Technol. 2021, 323, 124652. [Google Scholar] [CrossRef]
- Lam, P.-L.; Wong, M.-M.; Hung, L.-K.; Yung, L.-H.; Tang, J.C.-O.; Lam, K.-H.; Chung, P.-Y.; Wong, W.-Y.; Ho, Y.-W.; Wong, R.S.-M.; et al. Miconazole and terbinafine induced reactive oxygen species accumulation and topical toxicity in human keratinocytes. Drug Chem. Toxicol. 2020, 45, 834–838. [Google Scholar] [CrossRef]
- Shekhova, E.; Kniemeyer, O.; Brakhage, A.A. Induction of mitochondrial reactive oxygen species production by itraconazole, terbinafine, and amphotericin B as a mode of action against Aspergillus fumigatus. Antimicrob. Agents Chemother. 2017, 61, 10–1128. [Google Scholar] [CrossRef]
- Nohl, H.; Gille, L.; Schonheit, K.; Liu, Y. Conditions allowing redoxcycling ubisemiquinone in mitochondria to establish a direct redox couple with molecular oxygen. Free Radic. Biol. Med. 1996, 20, 207–213. [Google Scholar] [CrossRef]
- Turrens, J.F.; Alexandre, A.; Lehninger, A.L. Ubisemiquinone is the electron donor for superoxide formation by complex III of heart mitochondria. Arch. Biochem. Biophys. 1985, 237, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Sagatova, A.A. Strategies to Better Target Fungal Squalene Monooxygenase. J. Fungi 2021, 7, 49. [Google Scholar] [CrossRef]
- Zhang, D.; Jian, Y.P.; Zhang, Y.N.; Li, Y.; Gu, L.T.; Sun, H.H.; Liu, M.D.; Zhou, H.L.; Wang, Y.S.; Xu, Z.X. Short-chain fatty acids in diseases. Cell Commun. Signal. 2023, 21, 212. [Google Scholar] [CrossRef] [PubMed]
- Gangopadhyay, N.; Rai, D.K.; Brunton, N.P.; Gallagher, E.; Hossain, M.B. Antioxidant-guided isolation and mass spectrometric identification of the major polyphenols in barley (Hordeum vulgare) grain. Food Chem. 2016, 210, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Kamel, E.M.; Allam, A.A.; Rudayni, H.A.; Alkhedhairi, S.; Alkhayl, F.F.A.; Alwaili, M.A.; Lamsabhi, A.M. Mechanistic Insights into Polyphenols-mediated Squalene Epoxidase Inhibition: Computational Models and Experimental Validation for Targeting Cholesterol Biosynthesis. Cell Biochem. Biophys. 2025, 83, 4531–4550. [Google Scholar] [CrossRef]
- Abe, I.; Seki, T.; Umehara, K.; Miyase, T.; Noguchi, H.; Sakakibara, J.; Ono, T. Green tea polyphenols: Novel and potent inhibitors of squalene epoxidase. Biochem. Biophys. Res. Commun. 2000, 268, 767–771. [Google Scholar] [CrossRef]
- Muhammad Abdul Kadar, N.N.; Ahmad, F.; Teoh, S.L.; Yahaya, M.F. Caffeic acid on metabolic syndrome: A review. Molecules 2021, 26, 5490. [Google Scholar] [CrossRef]
- Yoon, S.A.; Kang, S.I.; Shin, H.S.; Kang, S.W.; Kim, J.H.; Ko, H.C.; Kim, S.J. p-Coumaric acid modulates glucose and lipid metabolism via AMP-activated protein kinase in L6 skeletal muscle cells. Biochem. Biophys. Res. Commun. 2013, 432, 553–557. [Google Scholar] [CrossRef]
- Mgbechidinma, C.L.; Zheng, G.; Baguya, E.B.; Zhou, H.; Okon, S.U.; Zhang, C. Fatty acid composition and nutritional analysis of waste crude fish oil obtained by optimized milder extraction methods. Environ. Eng. Res. 2023, 28, 220034. [Google Scholar] [CrossRef]
- Abidi, I.; Mansouri, S.; Radhouane, L.; Riadh, K.; El Felah, M.; Bouzid, S. Phenolic, Flavonoid and Tannin Contents of Tunisian Barley Landraces. Int. J. Agric. Innov. Res. 2015, 3, 1417–1423. [Google Scholar]
- Zou, L.G.; Zheng, D.L.; Yao, Y.T.; Wen, F.F.; Li, D.W.; Yang, Y.F.; Yang, W.D.; Balamurugan, S.; Kwok, H.F.; Li, H.Y. Polyphenols modulate microalgae metabolism with a particular increment in lipid accumulation. Fuel 2023, 352, 129085. [Google Scholar] [CrossRef]
- Liu, S.; Zhu, Y.; Liu, N.; Fan, D.; Wang, M.; Zhao, Y. Antioxidative properties and chemical changes of quercetin in fish oil: Quercetin reacts with free fatty acids to form its ester derivatives. J. Agric. Food Chem. 2021, 69, 1057–1067. [Google Scholar] [CrossRef]
- Feng, J.; Cai, H.; Wang, H.; Li, C.; Liu, S. Improved oxidative stability of fish oil emulsion by grafted ovalbumin-catechin conjugates. Food Chem. 2018, 241, 60–69. [Google Scholar] [CrossRef]
- Qiu, X.; Hong, H.; MacKenzie, S.L. Identification of a Delta 4 fatty acid desaturase from Thraustochytrium sp. involved in the biosynthesis of docosahexanoic acid by heterologous expression in Saccharomyces cerevisiae and Brassica juncea. J. Biol. Chem. 2001, 276, 31561–31566. [Google Scholar] [CrossRef] [PubMed]
- Sugerman, D.T.; Livingston, E.H.; Lynm, C. Statins. J. Am. Med. Assoc. 2013, 309, 1419. [Google Scholar] [CrossRef]
- Gómez, M.; Baeza, M.; Cifuentes, V.; Alcaíno, J. The SREBP (Sterol Regulatory Element-Binding Protein) pathway: A regulatory bridge between carotenogenesis and sterol biosynthesis in the carotenogenic yeast Xanthophyllomyces dendrorhous. Biol. Res. 2021, 54, 34. [Google Scholar] [CrossRef]
- Pollier, J.; Vancaester, E.; Kuzhiumparambil, U.; Vickers, C.E.; Vandepoele, K.; Goossens, A.; Fabris, M. A widespread alternative squalene epoxidase participates in eukaryote steroid biosynthesis. Nat. Microbiol. 2019, 4, 226–233. [Google Scholar] [CrossRef] [PubMed]






| Gene | Abbreviation | Sequence | Tm (°C) | Reference |
|---|---|---|---|---|
| Mitochondrial folate transporter | MFT_F | ACACTACCGCAGCCTATCAC | 56.8 | [33] |
| MFT_R | ATCCATCTGTCAAGCCATCC | 54.7 | ||
| Ribosome-binding ATPase | RBA_F | CCTTGGGCATGTCTACTTCT | 54.3 | [33] |
| RBA_R | GTCTGAAACGAGCGAACACC | 56.3 | ||
| 3-Hydroxy-3-methylglutaryl-CoA synthase | HMGS_F | AACTCGTGCTACCTTCGC | 55.0 | [31] |
| HMGS_R | AAGTCCTCACCGTAAGCC | 54.3 | ||
| 3-Hydroxy-3-methylglutaryl-CoA reductase | HMGR_F | GACGCTACTGCTACCTGC | 55.5 | [31] |
| HMGR_R | CTGGGCTTCTTGTCGGTA | 54.3 | ||
| Alternative squalene epoxidase | AltSQE_F | GGACGTCAAGCACCATCTCA | 57.2 | [31] |
| AltSQE_R | TGCGGCCTTCAACTACAACT | 57.0 | ||
| Ribosomal RNA 18s | 18s_F | TGCCGACTTGCGATTGTTG | 56.4 | [34] |
| 18s_R | TTCAGCCTTGCGACCATACT | 56.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Paredes, P.; Iturra, J.; Shene, C. Enhanced Squalene Production by Thraustochytrium sp. RT2316-16 by Polyphenols from Barley Bagasse. Fermentation 2026, 12, 63. https://doi.org/10.3390/fermentation12010063
Paredes P, Iturra J, Shene C. Enhanced Squalene Production by Thraustochytrium sp. RT2316-16 by Polyphenols from Barley Bagasse. Fermentation. 2026; 12(1):63. https://doi.org/10.3390/fermentation12010063
Chicago/Turabian StyleParedes, Paris, Javiera Iturra, and Carolina Shene. 2026. "Enhanced Squalene Production by Thraustochytrium sp. RT2316-16 by Polyphenols from Barley Bagasse" Fermentation 12, no. 1: 63. https://doi.org/10.3390/fermentation12010063
APA StyleParedes, P., Iturra, J., & Shene, C. (2026). Enhanced Squalene Production by Thraustochytrium sp. RT2316-16 by Polyphenols from Barley Bagasse. Fermentation, 12(1), 63. https://doi.org/10.3390/fermentation12010063






