Modulation of Alcohol Content in Wines Using Mixed Cultures
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeasts
2.2. Molecular Identification
2.3. Microvinifications
2.4. Descriptive Odor Analysis
2.5. Chemical Analysis
2.6. Mixed Cultures
2.7. Killer Activity
2.8. S02 and Ethanol Tolerance
2.9. Statistical and Data Analysis
3. Results
3.1. Screening of NS Yeast
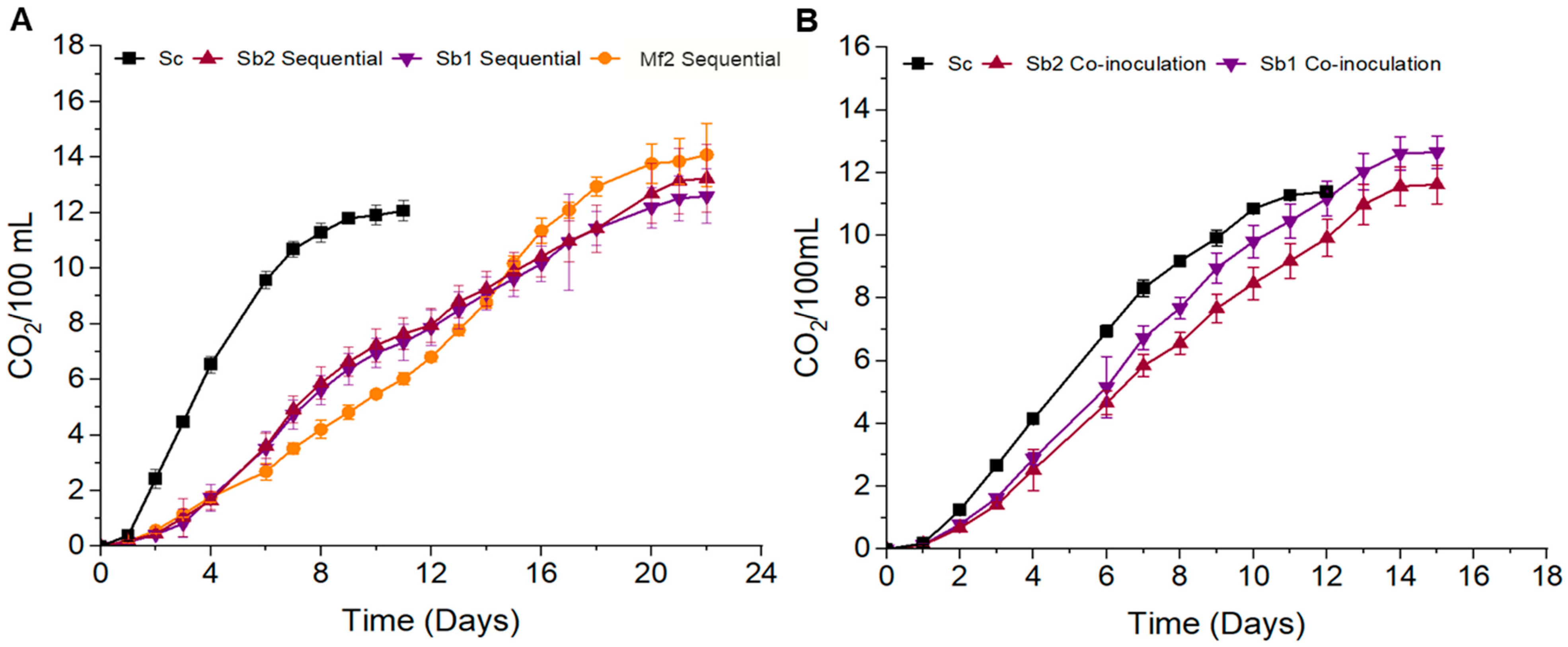
3.2. Mixed-Culture Strategy
3.3. Oenological Characterization of the Selected Strains
4. Discussion
4.1. Interpretation of the Results of Non-Saccharomyces Yeast Screening
4.2. Performance of Mixed-Culture Strategies
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Silva, P. Low-Alcohol and Nonalcoholic Wines: From Production to Cardiovascular Health, along with Their Economic Effects. Beverages 2024, 10, 49. [Google Scholar] [CrossRef]
- Global Status Report on Alcohol and Health and Treatment of Substance Use Disorders; World Health Organization: Geneva, Switzerland, 2024.
- Catarino, M.; Mendes, A. Dealcoholizing Wine by Membrane Separation Processes. Innov. Food Sci. Emerg. Technol. 2011, 12, 330–337. [Google Scholar] [CrossRef]
- Testa, B.; Coppola, F.; Succi, M.; Iorizzo, M. Biotechnological Strategies for Ethanol Reduction in Wine. Fermentation 2025, 11, 159. [Google Scholar] [CrossRef]
- An, J.; Zhang, Z.; Jin, A.; Tan, M.; Jiang, S.; Li, Y. A Critical Review of Alcohol Reduction Methods for Red Wines from the Perspective of Phenolic Compositions. Food Sci. Nutr. 2025, 13, e70401. [Google Scholar] [CrossRef]
- Listur, B.; Baldivia, G.; Coniberti, A.; Martín, V.; Boido, E.; Medina, K.; Carrau, F.; Dellacassa, E.; Fariña, L. Estrategias Combinadas Para La Obtención de Un Vino Tannat Con Un Contenido Moderado de Alcohol. BIO Web Conf. 2023, 68, 02045. [Google Scholar] [CrossRef]
- Carrau, F.; Boido, E.; Gaggero, C.; Medina, K.; Disegna, E.; Dellacassa, E. Vitis Vinifera Tannat, Chemical Characterization and Functional Properties. Ten Years of Research. In Multidisciplinary Approaches on Food Science and Nutrition for the 21st Century; Rosana, F., Ed.; Research Signpost/Transworld Research Network: Kerala, India, 2011; Volume 661, pp. 53–71. ISBN 9788178955049. [Google Scholar]
- González-Neves, G.; Charamelo, D.; Balado, J.; Barreiro, L.; Bochicchio, R.; Gatto, G.; Gil, G.; Tessore, A.; Carbonneau, A.; Moutounet, M. Phenolic Potential of Tannat, Cabernet-Sauvignon and Merlot Grapes and Their Correspondence with Wine Composition. Anal. Chim. Acta 2004, 513, 191–196. [Google Scholar] [CrossRef]
- Contreras, A.; Hidalgo, C.; Henschke, P.A.; Chambers, P.J.; Curtin, C.; Varela, C. Evaluation of Non-Saccharomyces Yeasts for the Reduction of Alcohol Content in Wine. Appl. Environ. Microbiol. 2014, 80, 1670–1678. [Google Scholar] [CrossRef] [PubMed]
- Rossouw, D.; Bauer, F.F. Exploring the Phenotypic Space of Non-Saccharomyces Wine Yeast Biodiversity. Food Microbiol. 2016, 55, 32–46. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Wei, Y.; Feng, W.; Zhang, H.; Jiang, J.; Qin, Y. Screening of Indigenous Hanseniaspora Strains from China for Ethanol Reduction in Wine. Foods 2025, 14, 1113. [Google Scholar] [CrossRef]
- Paramithiotis, S.; Patra, J.K.; Kotseridis, Y.; Dimopoulou, M. Utilization of Non-Saccharomyces to Address Contemporary Winemaking Challenges: Species Characteristics and Strain Diversity. Fermentation 2025, 11, 665. [Google Scholar] [CrossRef]
- Rolle, L.; Englezos, V.; Torchio, F.; Cravero, F.; Río Segade, S.; Rantsiou, K.; Giacosa, S.; Gambuti, A.; Gerbi, V.; Cocolin, L. Alcohol Reduction in Red Wines by Technological and Microbiological Approaches: A Comparative Study. Aust. J. Grape Wine Res. 2018, 24, 62–74. [Google Scholar] [CrossRef]
- Englezos, V.; Rantsiou, K.; Cravero, F.; Torchio, F.; Ortiz-Julien, A.; Gerbi, V.; Rolle, L.; Cocolin, L. Starmerella bacillaris and Saccharomyces cerevisiae Mixed Fermentations to Reduce Ethanol Content in Wine. Appl. Microbiol. Biotechnol. 2016, 100, 5515–5526. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Arneborg, N.; Toldam-Andersen, T.B.; Petersen, M.A.; Bredie, W.L. Effect of Sequential Fermentations and Grape Cultivars on Volatile Compounds and Sensory Profiles of Danish Wines. J. Sci. Food Agric. 2017, 97, 3594–3602. [Google Scholar] [CrossRef]
- Vicente, J.; Ruiz, J.; Belda, I.; Benito-Vázquez, I.; Marquina, D.; Calderón, F.; Santos, A.; Benito, S. The Genus Metschnikowia in Enology. Microorganisms 2020, 8, 1038. [Google Scholar] [CrossRef]
- Harlé, O.; Legrand, J.; Tesnière, C.; Pradal, M.; Mouret, J.-R.; Nidelet, T. Investigations of the Mechanisms of Interactions between Four Non-Conventional Species with Saccharomyces cerevisiae in Oenological Conditions. bioRxiv 2019. bioRxiv:363531. [Google Scholar] [CrossRef]
- Shi, W.K.; Wang, J.; Chen, F.S.; Zhang, X.Y. Effect of Issatchenkia Terricola and Pichia Kudriavzevii on Wine Flavor and Quality through Simultaneous and Sequential Co-Fermentation with Saccharomyces cerevisiae. LWT 2019, 116, 108477. [Google Scholar] [CrossRef]
- Pallmann, C.; Brown, J.; Olineka, T.; Cocolin, L.; Mills, D.; Bisson, L. Use of WL Medium to Profile Native Flora Fermentations. Am. J. Altern. Agric. 2001, 3, 198–203. [Google Scholar] [CrossRef]
- Godoy, A. Aislamiento e Identificación Molecular de Especies de Levaduras No-Saccharomyces Presentes En Uvas. Bachelor’s Thesis, Universidad de la República, Montevideo, Uruguay, 2013. [Google Scholar]
- Kurtzman, C.; Robnett, C. Identification of Clinically Important Ascomycetous Yeasts Based on Nucleotide Divergence in the 5′ End of the Large-Subunit (26S) Ribosomal DNA Gene. J. Clin. Microbiol. 1997, 35, 1216–1223. [Google Scholar] [CrossRef]
- Listur, M.B.; Martín, V.; Medina, K.; Carrau, F.; Boido, E.; Dellacassa, E.; Fariña, L. A Novel Approach for Enhancing the Terpenoid Content in Wine Using Starmerella bacillaris. Fermentation 2025, 11, 496. [Google Scholar] [CrossRef]
- ISO 3591:1977; Sensory analysis—Apparatus—Wine-tasting glass. International Organization for Standardization (ISO): Geneva, Switzerland, 1977.
- Comisión Nacional de Ética en Investigación. Decreto 158.019 Investigación en Seres Humanos; Ministerio de Salud Pública: Montevideo, Uruguay, 2019. [Google Scholar]
- Zhu, X.; Torija, M.J.; Mas, A.; Beltran, G.; Navarro, Y. Effect of a Multistarter Yeast Inoculum on Ethanol Reduction and Population Dynamics in Wine Fermentation. Foods 2021, 10, 623. [Google Scholar] [CrossRef]
- International Organisation of Vine and Wine. OIV-OENO 370-2012—Guidelines for the Characterization of Wine Yeasts of the Genus Saccharomyces Isolated from Vitivinicultural Environments; International Organisation of Vine and Wine: Paris, France, 2012. [Google Scholar]
- Medina, K.; Carrau, F.M.; Gioia, O.; Bracesco, N. Nitrogen Availability of Grape Juice Limits Killer Yeast Growth and Fermentation Activity during Mixed-Culture Fermentation with Sensitive Commercial Yeast Strains. Appl. Environ. Microbiol. 1997, 63, 2821–2825. [Google Scholar] [CrossRef]
- Mauriello, G.; Capece, A.; D’Auria, M.; Garde-Cerdán, T.; Romano, P. SPME-GC Method as a Tool to Differentiate VOC Profiles in Saccharomyces cerevisiae Wine Yeasts. Food Microbiol. 2009, 26, 246–252. [Google Scholar] [CrossRef]
- Englezos, V.; Rantsiou, K.; Torchio, F.; Rolle, L.; Gerbi, V.; Cocolin, L. Exploitation of the Non-Saccharomyces Yeast Starmerella bacillaris (Synonym Candida Zemplinina) in Wine Fermentation: Physiological and Molecular Characterizations. Int. J. Food Microbiol. 2015, 199, 33–40. [Google Scholar] [CrossRef]
- Ivit, N.N.; Longo, R.; Kemp, B. The Effect of Non-Saccharomyces and Saccharomyces Non-Cerevisiae Yeasts on Ethanol and Glycerol Levels in Wine. Fermentation 2020, 6, 77. [Google Scholar] [CrossRef]
- Contreras, A.; Hidalgo, C.; Schmidt, S.; Henschke, P.A.; Curtin, C.; Varela, C. The Application of Non-Saccharomyces Yeast in Fermentations with Limited Aeration as a Strategy for the Production of Wine with Reduced Alcohol Content. Int. J. Food Microbiol. 2015, 205, 7–15. [Google Scholar] [CrossRef]
- Raymond Eder, M.L.; Rosa, A.L. Genetic, Physiological, and Industrial Aspects of the Fructophilic Non-Saccharomyces Yeast Species, Starmerella bacillaris. Fermentation 2021, 7, 87. [Google Scholar] [CrossRef]
- Quirós, M.; Rojas, V.; Gonzalez, R.; Morales, P. Selection of Non-Saccharomyces Yeast Strains for Reducing Alcohol Levels in Wine by Sugar Respiration. Int. J. Food Microbiol. 2014, 181, 85–91. [Google Scholar] [CrossRef]
- Zhu, X.; Navarro, Y.; Mas, A.; Torija, M.J.; Beltran, G. A Rapid Method for Selecting Non-Saccharomyces Strains with a Low Ethanol Yield. Microorganisms 2020, 8, 658. [Google Scholar] [CrossRef] [PubMed]
- Swiegers, J.H.; Pretorius, I.S. Modulation of Volatile Sulfur Compounds by Wine Yeast. Appl. Microbiol. Biotechnol. 2007, 74, 954–960. [Google Scholar] [CrossRef] [PubMed]
- Kinzurik, M.I.; Herbst-Johnstone, M.; Gardner, R.C.; Fedrizzi, B. Hydrogen Sulfide Production during Yeast Fermentation Causes the Accumulation of Ethanethiol, S-Ethyl Thioacetate and Diethyl Disulfide. Food Chem. 2016, 209, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Ailer, Š.; Jakabová, S.; Benešová, L.; Ivanova-Petropulos, V. Wine Faults: State of Knowledge in Reductive Aromas, Oxidation and Atypical Aging, Prevention, and Correction Methods. Molecules 2022, 27, 3535. [Google Scholar] [CrossRef] [PubMed]
- Ciani, M.; Comitini, F.; Mannazzu, I.; Domizio, P. Controlled Mixed Culture Fermentation: A New Perspective on the Use of Non-Saccharomyces Yeasts in Winemaking. FEMS Yeast Res. 2010, 10, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Lleixà, J.; Manzano, M.; Mas, A.; del C. Portillo, M. Saccharomyces and Non-Saccharomyces Competition during Microvinification under Different Sugar and Nitrogen Conditions. Front. Microbiol. 2016, 7, 1959. [Google Scholar] [CrossRef] [PubMed]
- Gobbi, M.; De Vero, L.; Solieri, L.; Comitini, F.; Oro, L.; Giudici, P.; Ciani, M. Fermentative Aptitude of Non-saccharomyces Wine Yeast for Reduction in the Ethanol Content in Wine. Eur. Food Res. Technol. 2014, 239, 41–48. [Google Scholar] [CrossRef]
- Canonico, L.; Comitini, F.; Ciani, M. Metschnikowia Pulcherrima Selected Strain for Ethanol Reduction in Wine: Influence of Cell Immobilization and Aeration Condition. Foods 2019, 8, 378. [Google Scholar] [CrossRef]
- Hranilovic, A.; Gambetta, J.M.; Jeffery, D.W.; Grbin, P.R.; Jiranek, V. Lower-Alcohol Wines Produced by Metschnikowia Pulcherrima and Saccharomyces cerevisiae Co-Fermentations: The Effect of Sequential Inoculation Timing. Int. J. Food Microbiol. 2020, 329, 108651. [Google Scholar] [CrossRef]
- Guindal, A.M.; Morales, P.; Tronchoni, J.; Gonzalez, R. Reduction of Ethanol Content in Wine with an Improved Combination of Yeast Strains and Process Conditions. Food Microbiol. 2023, 115, 104344. [Google Scholar] [CrossRef]
- Kurtzman, C.P.; Droby, S. Metschnikowia Fructicola, a New Ascosporic Yeast with Potential for Biocontrol of Postharvest Fruit Rots. Syst. Appl. Microbiol. 2001, 24, 395–399. [Google Scholar] [CrossRef]
- Liu, J.; Wisniewski, M.; Droby, S.; Tian, S.; Hershkovitz, V.; Tworkoski, T. Effect of Heat Shock Treatment on Stress Tolerance and Biocontrol Efficacy of Metschnikowia Fructicola. FEMS Microbiol. Ecol. 2011, 76, 145–155. [Google Scholar] [CrossRef]
- Martín, V.; Boido, E.; Listur, B.; Carrau, F.; Medina, K. Exploring the Bioprotective Potential of Native Non-Saccharomyces Yeasts in Winemaking: Preliminary Results. In Proceedings of the Centenary of the OIV 45th World Congress of Vine and Wine, Dijon, France, 14–18 October 2024. [Google Scholar] [CrossRef]
- Boscaino, F.; Ionata, E.; La Cara, F.; Guerriero, S.; Marcolongo, L.; Sorrentino, A. Impact of Saccharomyces cerevisiae and Metschnikowia Fructicola Autochthonous Mixed Starter on Aglianico Wine Volatile Compounds. J. Food Sci. Technol. 2019, 56, 4982–4991. [Google Scholar] [CrossRef] [PubMed]
- García-Luque, E.; González, R.; Cao, R.; Soto, E.; Blanco, P. Sequential Fermentation with Non-Saccharomyces Yeasts Improves the Chemical and Sensory Characteristics of Albariño and Lado Wines. Fermentation 2025, 11, 73. [Google Scholar] [CrossRef]
- Mestre Furlani, M.V.; Maturano, Y.P.; Combina, M.; Mercado, L.A.; Toro, M.E.; Vazquez, F. Selection of Non-Saccharomyces Yeasts to Be Used in Grape Musts with High Alcoholic Potential: A Strategy to Obtain Wines with Reduced Ethanol Content. FEMS Yeast Res. 2017, 17, 10. [Google Scholar] [CrossRef]
- Scanes, K.T.; Hohmann, S.; Priori, B.A. Glycerol Production by the Yeast Saccharomyces cerevisiae and Its Relevance to Wine: A Review. S. Afr. J. Enol. Vitic. 2017, 19, 17–24. [Google Scholar] [CrossRef]
- Nadai, C.; da Silva Duarte, V.; Sica, J.; Vincenzi, S.; Carlot, M.; Giacomini, A.; Corich, V. Starmerella bacillaris Released in Vineyards at Different Concentrations Influences Wine Glycerol Content Depending on the Vinification Protocols. Foods 2023, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Englezos, V.; Rantsiou, K.; Cravero, F.; Torchio, F.; Giacosa, S.; Ortiz-Julien, A.; Gerbi, V.; Rolle, L.; Cocolin, L. Volatile Profiles and Chromatic Characteristics of Red Wines Produced with Starmerella bacillaris and Saccharomyces cerevisiae. Food Res. Int. 2018, 109, 298–309. [Google Scholar] [CrossRef]
- Bagheri, B.; Zambelli, P.; Vigentini, I.; Bauer, F.F.; Setati, M.E. Investigating the Effect of Selected Non-Saccharomyces Species on Wine Ecosystem Function and Major Volatiles. Front. Bioeng. Biotechnol. 2018, 6, 422413. [Google Scholar] [CrossRef]
- Tronchoni, J.; Gamero, A.; Arroyo-López, F.N.; Barrio, E.; Querol, A. Differences in the Glucose and Fructose Consumption Profiles in Diverse Saccharomyces Wine Species and Their Hybrids during Grape Juice Fermentation. Int. J. Food Microbiol. 2009, 134, 237–243. [Google Scholar] [CrossRef]
- Sipiczki, M. Candida Zemplinina Sp. Nov., an Osmotolerant and Psychrotolerant Yeast That Ferments Sweet Botrytized Wines. Int. J. Syst. Evol. Microbiol. 2003, 53, 2079–2083. [Google Scholar] [CrossRef]
- Horváth, B.O.; Sárdy, D.N.; Kellner, N.; Magyar, I. Effects of High Sugar Content on Fermentation Dynamics and Some Metabolites of Wine-Related Yeast Species Saccharomyces cerevisiae, S. Uvarum and Starmerella bacillaris. Food Technol. Biotechnol. 2020, 58, 76. [Google Scholar] [CrossRef]
- Rantsiou, K.; Dolci, P.; Giacosa, S.; Torchio, F.; Tofalo, R.; Torriani, S.; Suzzi, G.; Rolle, L.; Cocolina, L. Candida Zemplinina Can Reduce Acetic Acid Produced by Saccharomyces cerevisiae in Sweet Wine Fermentations. Appl. Environ. Microbiol. 2012, 78, 1987–1994. [Google Scholar] [CrossRef]
- Padilla, B.; Gil, J.V.; Manzanares, P. Past and Future of Non-Saccharomyces Yeasts: From Spoilage Microorganisms to Biotechnological Tools for Improving Wine Aroma Complexity. Front. Microbiol. 2016, 7, 411. [Google Scholar] [CrossRef] [PubMed]
- Jolly, N.P.; Varela, C.; Pretorius, I.S. Not Your Ordinary Yeast: Non-Saccharomyces Yeasts in Wine Production Uncovered. FEMS Yeast Res. 2013, 14, 215–237. [Google Scholar] [CrossRef]
- Ciani, M.; Morales, P.; Comitini, F.; Tronchoni, J.; Canonico, L.; Curiel, J.A.; Oro, L.; Rodrigues, A.J.; Gonzalez, R. Non-Conventional Yeast Species for Lowering Ethanol Content of Wines. Front. Microbiol. 2016, 7, 642. [Google Scholar] [CrossRef]
- Puyo, M.; Simonin, S.; Bach, B.; Klein, G.; Alexandre, H.; Tourdot-Maréchal, R. Bio-Protection in Oenology by Metschnikowia Pulcherrima: From Field Results to Scientific Inquiry. Front. Microbiol. 2023, 14, 1252973. [Google Scholar] [CrossRef]
- Medina, K.; Boido, E.; Fariña, L.; Gioia, O.; Gomez, M.E.; Barquet, M.; Gaggero, C.; Dellacassa, E.; Carrau, F. Increased Flavour Diversity of Chardonnay Wines by Spontaneous Fermentation and Co-Fermentation with Hanseniaspora Vineae. Food Chem. 2013, 141, 2513–2521. [Google Scholar] [CrossRef]
- Vejarano, R.; Gil-Calderón, A. Commercially Available Non-Saccharomyces Yeasts for Winemaking: Current Market, Advantages over Saccharomyces, Biocompatibility, and Safety. Fermentation 2021, 7, 171. [Google Scholar] [CrossRef]
- Ciani, M.; Comitini, F. Yeast Interactions in Multi-Starter Wine Fermentation. Curr. Opin. Food Sci. 2015, 1, 1–6. [Google Scholar] [CrossRef]
- Englezos, V.; Rantsiou, K.; Giacosa, S.; Río Segade, S.; Rolle, L.; Cocolin, L. Cell-to-Cell Contact Mechanism Modulates Starmerella bacillaris Death in Mixed Culture Fermentations with Saccharomyces cerevisiae. Int. J. Food Microbiol. 2019, 289, 106–114. [Google Scholar] [CrossRef] [PubMed]
- El Dana, F.; Hayar, S.; Alexandre, H. Killer Yeast in Winemaking: A Comprehensive Review. Food Biosci. 2025, 73, 107631. [Google Scholar] [CrossRef]
- Capece, A.; Pietrafesa, A.; Pietrafesa, R.; Garrigós, V.; Tedesco, F.; Romano, P.; Matallana, E.; Siesto, G.; Aranda, A. Impact of Starmerella bacillaris and Zygosaccharomyces Bailii on Ethanol Reduction and Saccharomyces cerevisiae Metabolism during Mixed Wine Fermentations. Food Res. Int. 2022, 159, 111649. [Google Scholar] [CrossRef] [PubMed]
- Varela, C.; Dry, P.R.; Kutyna, D.R.; Francis, I.L.; Henschke, P.A.; Curtin, C.D.; Chambers, P.J. Strategies for Reducing Alcohol Concentration in Wine. Aust. J. Grape Wine Res. 2015, 21, 670–679. [Google Scholar] [CrossRef]
- Akhtar, W.; Ceci, A.T.; Longo, E.; Marconi, M.A.; Lonardi, F.; Boselli, E. Dealcoholized Wine: Techniques, Sensory Impacts, Stability, and Perspectives of a Growing Industry. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70171. [Google Scholar] [CrossRef]
- Longo, R.; Blackman, J.W.; Torley, P.J.; Rogiers, S.Y.; Schmidtke, L.M. Changes in Volatile Composition and Sensory Attributes of Wines during Alcohol Content Reduction. J. Sci. Food Agric. 2017, 97, 8–16. [Google Scholar] [CrossRef]
- Coppola, F.; Testa, B.; Succi, M.; Paventi, G.; Di Martino, C.; Iorizzo, M. Viticultural and Pre-Fermentation Strategies to Reduce Alcohol Levels in Wines. Foods 2025, 14, 2647. [Google Scholar] [CrossRef]
- Ozturk, B.; Anli, E. Different Techniques for Reducing Alcohol Levels in Wine: A Review. BIO Web Conf. 2014, 3, 02012. [Google Scholar] [CrossRef]


| Abbreviation | Species | Isolation Code |
|---|---|---|
| Sb1 | Starmerella bacillaris | M10_02G |
| Sb2 | Starmerella bacillaris | C10_46F |
| Sb3 | Starmerella bacillaris | CF10_31F |
| Sb4 | Starmerella bacillaris | T10_25F |
| Sb5 | Starmerella bacillaris | T10_20F |
| Sb6 | Starmerella bacillaris | A10_74F |
| Mf1 | Metschnikowia fructicola | T11_193F |
| Mf2 | Metschnikowia fructicola | A10_77F |
| Mf3 | Metschnikowia fructicola | T11_191F |
| Mf4 | Metschnikowia fructicola | M12_01G |
| Mf5 | Metschnikowia fructicola | M12_02G |
| Mf6 | Metschnikowia fructicola | M12_03G |
| Mp1 | Metschnikowia pulcherrima | M11_56F |
| Mp2 | Metschnikowia pulcherrima | T11_233G |
| Mp3 | Metschnikowia pulcherrima | T11_235G |
| Mp4 | Metschnikowia pulcherrima | M00_19G |
| Hu1 | Hanseniaspora uvarum | M18_161F |
| Hu2 | Hanseniaspora uvarum | C12_203G |
| Hu3 | Hanseniaspora uvarum | T18_147G |
| Hu4 | Hanseniaspora uvarum | M12_04G |
| Hu5 | Hanseniaspora uvarum | M12_05G |
| Hu6 | Hanseniaspora uvarum | M18_158G |
| Hu7 | Hanseniaspora uvarum | C12_208G |
| Hu8 | Hanseniaspora uvarum | T12_152F |
| Ho1 | Hanseniaspora opuntiae | T12_153F |
| Ho2 | Hanseniaspora opuntiae | M18_13G |
| It | Issatchenkia terricola | M16_21G |
| Strategy | Strain Combination | Initial Inoculum (Cells/mL) | Supplementation (Time and Type) |
|---|---|---|---|
| Sequential | Sb1 + Sc | NS: 1 × 106 | At 48 h: 50 mg N/L DAP + 50 mg N/L AAs + DAP; 2.5 mg/L yeast extract; 0.3 mg/L thiamine |
| Sc: 1 × 106 (48 h) | |||
| Sequential | Sb2 + Sc | NS: 1 × 106 | At 48 h: 50 mg N/L DAP + 50 mg N/L AAs + DAP; 2.5 mg/L yeast extract; 0.3 mg/L thiamine |
| Sc: 1 × 106 (48 h) | |||
| Sequential | Mf2 + Sc | NS: 1 × 106 | At 48 h: 50 mg N/L DAP + 50 mg N/L AAs + DAP; 2.5 mg/L yeast extract; 0.3 mg/L thiamine |
| Sc: 1 × 106 (48 h) | |||
| Control | Sc (monoculture) | 1 × 106 | At 72 h: 50 mg N/L DAP; 2.5 mg/L yeast extract; 0.3 mg/L thiamine |
| Co-inoculation | Sb1 + Sc | NS: 1 × 106 | At 72 h: 50 mg N/L DAP; 2.5 mg/L yeast extract; 0.3 mg/L thiamine |
| Sc: 1 × 105 | |||
| Co-inoculation | Sb2 + Sc | NS: 1 × 106 | At 72 h: 50 mg N/L DAP; 2.5 mg/L yeast extract; 0.3 mg/L thiamine |
| Sc: 1 × 105 |
| Strategy | Mixed Culture Strain | Glucose (g/L) | Fructose (g/L) | Glycerol (g/L) | Ethanol Reduction (95% CI vs. Control) |
|---|---|---|---|---|---|
| Sequential | Sb1 + Sc | 6.1 ± 0.5 X | ND | 8.2 ± 0.3 X | 0.3–4.0 * |
| Sb2 + Sc | 13.9 ± 2.9 X | ND | 8.6 ± 0.3 X | – | |
| Mf2 + Sc | ND | 1.9 ± 0.1 X | 5.7 ± 0.1 X | 0.7–4.0 * | |
| Co-inoculation | Sb1 + Sc | 1.3 ± 0.1 | ND | 5.2 ± 0.2 X | 0.2–0.7 * |
| Sb2 + Sc | 11.7 ± 0.7 X | ND | 5.6 ± 0.1 X | 0.7–1.0 * |
| Strain | % Growth in Ethanol (% v/v) | % Growth in SO2 (mg/L) | K2 Sensitivity | Killer Phenotype | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | |||||||||||||||
| 3 | 6 | 10 | 12 | 14 | 25 | 40 | 75 | 100 | 25 | 40 | 75 | 100 | 150 | |||
| Sb1 | 100 | 76 | 29 | 13 | 10 | 90 | 59 | 19 | NR | 99 | 94 | 77 | 21 | NR | N | N |
| Sb2 | 90 | 67 | 18 | 12 | 10 | 89 | 57 | 22 | NR | 97 | 91 | 50 | 30 | NR | N | N |
| Mf2 | 57 | 18 | 10 | NR | NR | 85 | 69 | 51 | 10 | 97 | 88 | 85 | 71 | 17 | N | N |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Listur, M.B.; Martín, V.; Fariña, L.; Boido, E.; Dellacassa, E.; Carrau, F.; Medina, K. Modulation of Alcohol Content in Wines Using Mixed Cultures. Fermentation 2026, 12, 57. https://doi.org/10.3390/fermentation12010057
Listur MB, Martín V, Fariña L, Boido E, Dellacassa E, Carrau F, Medina K. Modulation of Alcohol Content in Wines Using Mixed Cultures. Fermentation. 2026; 12(1):57. https://doi.org/10.3390/fermentation12010057
Chicago/Turabian StyleListur, María Belén, Valentina Martín, Laura Fariña, Eduardo Boido, Eduardo Dellacassa, Francisco Carrau, and Karina Medina. 2026. "Modulation of Alcohol Content in Wines Using Mixed Cultures" Fermentation 12, no. 1: 57. https://doi.org/10.3390/fermentation12010057
APA StyleListur, M. B., Martín, V., Fariña, L., Boido, E., Dellacassa, E., Carrau, F., & Medina, K. (2026). Modulation of Alcohol Content in Wines Using Mixed Cultures. Fermentation, 12(1), 57. https://doi.org/10.3390/fermentation12010057









