Theobroma bicolor (Pataxte) Fermentation: A Novel Source of Promising Probiotic Lactic Acid Bacteria
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Biological Material
2.3. Methods
2.3.1. Fermentation
2.3.2. Physicochemical Properties of Fermentation
pH
Titratable Acidity
2.3.3. Isolation of Lactic Acid Bacteria
2.3.4. Morphological Examination
Gram Staining
2.3.5. Biochemical Identification
Catalase Test
2.3.6. Antimicrobial Activity
Kirby-Bauer Test
Minimum Inhibitory Concentration
2.3.7. Identification of Isolated Bacteria by MALDI-TOF MS
2.3.8. Molecular Identification of the Selected LAB Strain and Phylogenetic Analysis
2.3.9. Bigel Preparation
2.3.10. Encapsulation of Bacterial Strains
2.3.11. In Vitro Gastrointestinal Simulation
2.3.12. Statistical Analysis
3. Results and Discussion
3.1. Endogenous Fermentation
3.2. Isolation of Lactic Acid Bacteria
3.3. Antagonistic Activity
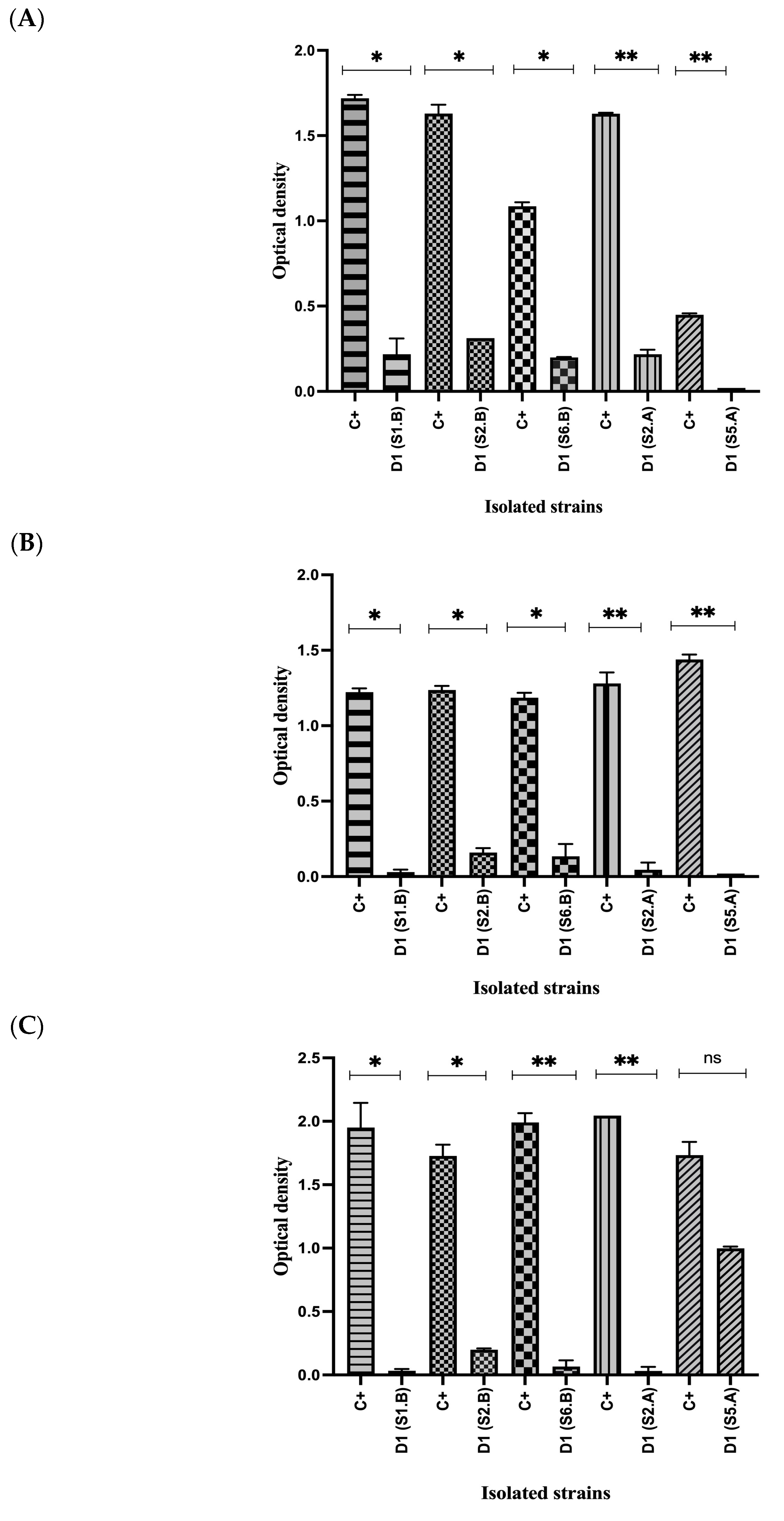
3.4. In Vitro Gastrointestinal Digestion
3.5. Identification of Isolated Bacteria by MALDI-TOF MS
3.6. Molecular Identification of the Potential Probiotic Isolated
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Díaz, R.O.; Hernández, M.S. Theobromas de la Amazonia Colombiana: Una alternativa saludable. Inf. Tecnol. 2020, 31, 3–10. [Google Scholar] [CrossRef]
- Gálvez-Marroquín, L.A.; Reyes-Reyes, A.L.; Avendaño-Arrazate, C.H.; Hernández-Goméz, E. Pataxte (Theobroma bicolor Humb. Bonpl.): Especie Subutilizada en México. Agro Product. 2016, 9, 41–47. [Google Scholar]
- Tinajero-Carrizales, C.; González-Pérez, A.L.; Rodríguez-Castillejos, G.C.; Castañón-Nájera, G.; Ruíz Salazar, R. Comparación próximal en Cacao (Theobroma cacao) y Pataxte (T. bicolor) de Tabasco y Chiapas, México. Polibotánica 2021, 52, 135–149. [Google Scholar] [CrossRef]
- Tigrero, B.; Sanclemente, B. Sustitución de Theobroma cacao Por Theobroma bicolor y su Aplicación en Repostería. Master’s Thesis, University of Guayaquil, Guayaquil, Ecuador, 2018. [Google Scholar]
- Ponce-Sánchez, J.; Zurita-Benavides, M.G.; Peñuela, M.C. Reproductive ecology of white cacao (Theobroma bicolor Humb. Bonpl.) in Ecuador, western Amazonia: Floral visitors and the impact of fungus and mistletoe on fruit production. Braz. J. Bot. 2021, 44, 479–489. [Google Scholar] [CrossRef]
- Benlloch-Tinoco, M.; Ramírez, J.M.N.; García, P.; Gentile, P.; Girón-Hernández, J. Theobroma genus: Exploring the therapeutic potential of T. grandiflorum and T. bicolor in biomedicine. Food Biosci. 2024, 61, 104755. [Google Scholar] [CrossRef]
- Vera-Chang, J.F.; Vásquez-Cortez, L.H.; Zapata-Quevedo, K.L.; Rodríguez-Cevallos, S.L. Caracterización morfológica, fisicoquímica y microbiológica del cacao Macambo (Theobroma bicolor Humb. Bonpl.) en Ecuador. Rev. Agrotecnológica Amaz. 2024, 4, e657. [Google Scholar] [CrossRef]
- Yang, F.; Chen, C.; Ni, D.; Yang, Y.; Tian, J.; Li, Y.; Chen, S.; Ye, X.; Wang, L. Effects of Fermentation on Bioactivity and the Composition of Polyphenols Contained in Polyphenol-Rich Foods: A Review. Foods 2023, 12, 3315. [Google Scholar] [CrossRef]
- Montaño, A.C.; Pérez, D.M.; Montalvo, I.G.; Medina, M.A.S.; Bautista, E.H. Evaluación del Proceso Fermentativo Tradicional del Theobroma bicolor en Teotitlán del Valle, Oaxaca, México. Cienc. Lat. Rev. Cient. Multidiscip. 2025, 9, 7899–7909. [Google Scholar] [CrossRef]
- De Vuyst, L.; Weckx, S. The cocoa bean fermentation process: From ecosystem analysis to starter culture development. J. Appl. Microbiol. 2016, 121, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Guzmán-Alvarez, R.; Márquez-Ramos, J.G. Fermentation of Cocoa Beans. In Fermentation—Processes, Benefits and Risks; IntechOpen: London, UK, 2021; Volume 1. [Google Scholar] [CrossRef]
- Batista, N.N.; Ramos, C.L.; Dias, D.R.; Pinheiro, A.C.M.; Schwan, R.F. The impact of yeast starter cultures on the microbial communities and volatile compounds in cocoa fermentation and the resulting sensory attributes of chocolate. J. Food Sci. Technol. 2015, 53, 1101–1110. [Google Scholar] [CrossRef] [PubMed]
- Schwan, R.F.; Wheals, A.E. The Microbiology of Cocoa Fermentation and its Role in Chocolate Quality. Crit. Rev. Food Sci. Nutr. 2004, 44, 205–221. [Google Scholar] [CrossRef]
- Echegaray, N.; Yilmaz, B.; Sharma, H.; Kumar, M.; Pateiro, M.; Ozogul, F.; Lorenzo, J.M. A novel approach to Lactiplantibacillus plantarum: From probiotic properties to the omics insights. Microbiol. Res. 2023, 268, 127289. [Google Scholar] [CrossRef]
- Agyirifo, D.S.; Wamalwa, M.; Otwe, E.P.; Galyuon, I.; Runo, S.; Takrama, J.; Ngeranwa, J. Metagenomics analysis of cocoa bean fermentation microbiome identifying species diversity and putative functional capabilities. Heliyon 2019, 5, e02170. [Google Scholar] [CrossRef] [PubMed]
- Lima, L.J.R.; Almeida, M.H.; Nout, M.J.R.; Zwietering, M.H. Theobroma cacao L., “The Food of the Gods”: Quality Determinants of Commercial Cocoa Beans, with Particular Reference to the Impact of Fermentation. Crit. Rev. Food Sci. Nutr. 2011, 51, 731–761. [Google Scholar] [CrossRef]
- Ouattara, H.G.; Ouattara, H.; Yao, W.; Williams, N.; Reverchon, S.; Niamke, S.; Elias, R.; Edwards, D. Lactic Acid Bacteria (LAB) strains are actively involved in the occurrence of aroma compounds during cocoa fermentation. Microbiol. Nat. 2019, 1, 63–72. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, J.; Lv, M.; Shao, Z.; Hungwe, M.; Wang, J.; Bai, X.; Xie, J.; Wang, Y.; Geng, W. Metabolism Characteristics of Lactic Acid Bacteria and the Expanding Applications in Food Industry. Front. Bioeng. Biotechnol. 2021, 9, 612285. [Google Scholar] [CrossRef]
- Garrote, A.; Bonet, R. Probióticos. Farm. Prof. 2017, 31, 13–16. Available online: https://dialnet.unirioja.es/servlet/articulo?codigo=7718744 (accessed on 7 March 2024).
- Tormo Carnicé, R. Probióticos. Concepto y mecanismos de acción. An. Pediatría 2006, 4, 30–41. Available online: https://www.analesdepediatria.org/es-probioticos-concepto-mecanismos-accion-articulo-13092364 (accessed on 7 March 2024).
- Tripathi, M.K.; Giri, S.K. Probiotic functional foods: Survival of probiotics during processing and storage. J. Funct. Foods 2014, 9, 225–241. [Google Scholar] [CrossRef]
- Cook, M.T.; Tzortzis, G.; Charalampopoulos, D.; Khutoryanskiy, V.V. Microencapsulation of probiotics for gastrointestinal delivery. J. Control. Release 2012, 162, 56–67. [Google Scholar] [CrossRef]
- Hashemi, B.; Assadpour, E.; Jafari, S.M. Bigels as novel carriers of bioactive compounds: Applications and research trends. Food Hydrocoll. 2024, 147, 109427. [Google Scholar] [CrossRef]
- Bollom, M.A. Development and Characterization of a Novel, Edible Bigel System with the Potential to Protect Probiotics During In Vitro Digestion. Master’s Thesis, Iowa State University, Ames, Iowa, 2020; pp. 1–105. [Google Scholar]
- Singh, V.K.; Qureshi, D.; Nayak, S.K.; Pal, K. Bigels. In Polymeric Gels; Woodhead Publishing: Sawston, UK, 2018; pp. 265–282. [Google Scholar] [CrossRef]
- Li, P.; Chen, F.; Yuan, S.; Dai, W.; Yin, L.; Dai, Q.; Li, Z.; Liu, H.; Guo, Q.; Zhu, Q. Development of novel Lactobacillus plantarum-encapsulated Bigel based on soy lecithin-beeswax oleogel and flaxseed gum hydrogel for enhanced survival during storage and gastrointestinal digestion. Food Hydrocoll. 2025, 163, 111052. [Google Scholar] [CrossRef]
- Guerrero, P.D.; Cardoso-Ugarte, G.A.; Pérez-Armendáriz, B. Producción de bebida fermentada simbiótica de pataxte (Theobroma bicolor) adicionada con bacterias ácido lácticas. Rev. Cienc. Tecnol. Soc. 2024, 4, 1–11. [Google Scholar] [CrossRef]
- Pérez-Pérez, M.A.; Sánchez-Chino, X.M.; García-Bautista, M.; González-Díaz, A.Á.; Ruíz-Montoya, L.; Castellanos-Morales, G. Phenotypic and genetic variation in pataxte (Theobroma bicolor Humb. Bonpl.) from cacao growing regions from southeastern Mexico. Bot. Sci. 2025, 103, 858–875. [Google Scholar] [CrossRef]
- Velásquez-Reyes, D.; Gschaedler, A.; Kirchmayr, M.; Avendaño-Arrazate, C.; Rodríguez-Campos, J.; Calva-Estrada, S.; Lugo-Cervantes, E. Cocoa bean turning as a method for redirecting the aroma compound profile in artisanal cocoa fermentation. Heliyon 2021, 7, e07694. [Google Scholar] [CrossRef] [PubMed]
- AOAC International. Official Methods of Analysis of AOAC International, 17th ed.; Horwitz, W., Ed.; AOAC International: Rockville, MD, USA, 2000. [Google Scholar]
- De Man, J.C.; Rogosa, M.; Sharpe, M.E. A medium for the cultivation of lactobacilli. J. Appl. Bacteriol. 1960, 23, 130–135. [Google Scholar] [CrossRef]
- Holzapfel, W.H.; Wood, B.J. (Eds.) Lactic Acid Bacteria; Wiley: Hoboken, NJ, USA, 2014. [Google Scholar] [CrossRef]
- Tripathi, N.; Sapra, A. Gram Staining; National Library of Medicine; StatPearls Publishing: Orlando, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK562156/ (accessed on 7 March 2024).
- Halima, K.; Anokhe, A.; Kalia, V. Catalase Test: A Biochemical Protocol for Bacterial Identification. AgriCos 2022, 3, 53–55. [Google Scholar]
- Herrera, M.L. Pruebas de sensibilidad antimicrobiana: Métodología de laboratorio. Rev. Médica Hosp. Nac. Niños Dr. Carlos Sáenz Herrera 1999, 34, 33–41. Available online: https://www.scielo.sa.cr/scielo.php?script=sci_arttext&pid=S1017-85461999000100010 (accessed on 7 March 2024).
- Singhal, N.; Kumar, M.; Kanaujia, P.K.; Virdi, J.S. MALDI-TOF Mass spectrometry: An Emerging Technology for Microbial Identification and Diagnosis. Front. Microbiol. 2015, 6, 791. [Google Scholar] [CrossRef]
- Alizadeh, M.; Yousefi, L.; Pakdel, F.; Ghotaslou, R.; Rezaee, M.A.; Khodadadi, E.; Oskouei, M.A.; Soroush Barhaghi, M.H.; Kafil, H.S. MALDI-TOF Mass Spectroscopy Applications in Clinical Microbiology. Adv. Pharmacol. Pharm. Sci. 2021, 2021, 9928238. [Google Scholar] [CrossRef] [PubMed]
- Woese, C.R.; Kandler, O.; Wheelis, M.L. Towards a natural system of organisms: Proposal for the domains Archaea, Bacteria, and Eucarya. Proc. Natl. Acad. Sci. USA 1990, 87, 4576–4579. [Google Scholar] [CrossRef]
- Weisburg, W.G.; Barns, S.M.; Pelletier, D.A.; Lane, D.J. 16S ribosomal DNA amplification for phylogenetic study. J. Bacteriol. 1991, 173, 697–703. [Google Scholar] [CrossRef]
- Clarridge, J.E. Impact of 16S rRNA Gene Sequence Analysis for Identification of Bacteria on Clinical Microbiology and Infectious Diseases. Clin. Microbiol. Rev. 2004, 17, 840–862. [Google Scholar] [CrossRef] [PubMed]
- Sanger, F.; Nicklen, S.; Coulson, A.R. DNA sequencing with chain-terminating inhibitors. Proc. Nat. Acad. Sci. USA 1977, 74, 5463–5467. [Google Scholar] [CrossRef]
- Martínez-Ortiz, V.M.; Trujillo-López, M.A.; El-Kassis, E.G.; Bautista-Rodríguez, E.; Kirchmayr, M.R.; Hernández-Carranza, P.; Pérez-Armendáriz, B. Bacillus mojavensis isolated from aguamiel and its potential as a probiotic bacterium. Electron. J. Biotechnol. 2024, 67, 42–49. [Google Scholar] [CrossRef]
- Ho, V.T.T.; Fleet, G.H.; Zhao, J. Unravelling the contribution of lactic acid bacteria and acetic acid bacteria to cocoa fermentation using inoculated organisms. Int. J. Food Microbiol. 2018, 279, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Cortez, D.; Quispe-Sanchez, L.; Mestanza, M.; Oliva-Cruz, M.; Yoplac, I.; Torres, C.; Chavez, S.G. Changes in bioactive compounds during fermentation of cocoa (Theobroma cacao) harvested in Amazonas-Peru. Curr. Res. Food Sci. 2023, 6, 100494. [Google Scholar] [CrossRef]
- Castro-Alayo, E.M.; Idrogo-Vásquez, G.; Siche, R.; Cardenas-Toro, F.P. Formation of aromatic compounds precursors during fermentation of Criollo and Forastero cocoa. Heliyon 2019, 5, e01157. [Google Scholar] [CrossRef] [PubMed]
- Visintin, S.; Alessandria, V.; Valente, A.; Dolci, P.; Cocolin, L. Molecular identification and physiological characterization of yeasts, lactic acid bacteria and acetic acid bacteria isolated from heap and box cocoa bean fermentations in West Africa. Int. J. Food Microbiol. 2016, 216, 69–78. [Google Scholar] [CrossRef]
- ben Omar, N.; Ampe, F. Microbial Community Dynamics during Production of the Mexican Fermented Maize Dough Pozol. Appl. Environ. Microbiol. 2000, 66, 3664–3673. [Google Scholar] [CrossRef]
- Lamei, S.; Hu, Y.O.O.; Olofsson, T.C.; Andersson, A.F.; Forsgren, E.; Vásquez, A. Improvement of identification methods for honeybee specific Lactic Acid Bacteria; future approaches. PLoS ONE 2017, 12, e0174614. [Google Scholar] [CrossRef]
- Abdullah, D.; Poddar, S.; Rai, R.P.; Purwati, E.; Dewi, N.P.; Pratama, Y.E. Molecular Identification of Lactic Acid Bacteria an Approach to Sustainable Food Security. J. Public Health Res. 2022, 10, jphr.2021.2508. [Google Scholar] [CrossRef]
- Von Wright, A.; Axelsson, L. Lactic Acid Bacteria: Microbiological and Functional Aspects; CRC Press: Boca Raton, FL, USA, 2011; p. 798. [Google Scholar] [CrossRef]
- Sharma, A.; Lee, H.-J. Antimicrobial Activity of Probiotic Bacteria Isolated from Plants: A Review. Foods 2025, 14, 495. [Google Scholar] [CrossRef]
- Lee, J.; Jo, J.; Wan, J.; Seo, H.; Han, S.-W.; Shin, Y.-J.; Kim, D.-H. In Vitro Evaluation of Probiotic Properties and Anti-Pathogenic Effects of Lactobacillus and Bifidobacterium Strains as Potential Probiotics. Foods 2024, 13, 2301. [Google Scholar] [CrossRef] [PubMed]
- Simões, L.; Fernandes, N.; de Souza, A.; dos Santos, L.; Magnani, M.; Abrunhosa, L.; Teixeira, J.; Schwan, R.F.; Dias, D.R. Probiotic and Antifungal Attributes of Lactic Acid Bacteria Isolates from Naturally Fermented Brazilian Table Olives. Fermentation 2022, 8, 277. [Google Scholar] [CrossRef]
- Kelidkazeran, E.; Bouri Yildiz, M.; Sahin, F. In Vitro Assessment of Biological and Functional Properties of Potential Probiotic Strains Isolated from Commercial and Dairy Sources. Microorganisms 2025, 13, 970. [Google Scholar] [CrossRef]
- Abdel-Daim, A.; Hassouna, N.; Hafez, M.; Ashor, M.S.A.; Aboulwafa, M.M. Antagonistic Activity of Lactobacillus Isolates against Salmonella typhi In Vitro. BioMed Res. Int. 2013, 2013, 680605. [Google Scholar] [CrossRef]
- Piedra, V.; Usaga, J.; Redondo-Solano, M.; Uribe-Lorío, L.; Valenzuela-Martínez, C.; Barboza, N. Inhibiting potential of selected lactic acid bacteria isolated from Costa Rican agro-industrial waste against Salmonella sp. in yogurt. Ital. J. Food Saf. 2024, 14, 12494. [Google Scholar] [CrossRef]
- Yilmaz, B.; Bangar, S.P.; Echegaray, N.; Suri, S.; Tomasevic, I.; Manuel Lorenzo, J.; Melekoglu, E.; Rocha, J.M.; Ozogul, F. The Impacts of Lactiplantibacillus plantarum on the Functional Properties of Fermented Foods: A Review of Current Knowledge. Microorganisms 2022, 10, 826. [Google Scholar] [CrossRef]
- Seddik, H.A.; Bendali, F.; Gancel, F.; Fliss, I.; Spano, G.; Drider, D. Lactobacillus plantarum and Its Probiotic and Food Potentialities. Probiotics Antimicrob. Proteins 2017, 9, 111–122. [Google Scholar] [CrossRef]
- Kim, S.W.; Kang, S.I.; Shin, D.H.; Oh, S.Y.; Lee, C.W.; Yang, Y.; Son, Y.K.; Yang, H.-S.; Lee, B.-H.; An, H.-J.; et al. Potential of Cell-Free Supernatant from Lactobacillus plantarum NIBR97, Including Novel Bacteriocins, as a Natural Alternative to Chemical Disinfectants. Pharmaceuticals 2020, 13, 266. [Google Scholar] [CrossRef]
- Nandha, M.C.; Shukla, R.M. Exploration of probiotic attributes in lactic acid bacteria isolated from fermented Theobroma cacao L. fruit using in vitro techniques. Front. Microbiol. 2023, 14, 1274636. [Google Scholar] [CrossRef] [PubMed]
- Escobar-Ramírez, M.C.; Jaimez-Ordaz, J.; Escorza-Iglesias, V.A.; Rodríguez-Serrano, G.M.; Contreras-López, E.; Ramírez-Godínez, J.; Castañeda-Ovando, A.; Morales-Estrada, A.I.; Felix-Reyes, N.; González-Olivares, L.G. Lactobacillus pentosus ABHEAU-05: An in vitro digestion resistant lactic acid bacterium isolated from a traditional fermented Mexican beverage. Rev. Argent. De Microbiol. 2020, 52, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, X.; Clark, S.; Acevedo, N. Bigels—Oleocolloid matrices—As probiotic protective systems in yogurt. J. Food Sci. 2021, 86, 4892–4900. [Google Scholar] [CrossRef]
- De Koster, C.G.; Brul, S. MALDI-TOF MS identification and tracking of food spoilers and food-borne pathogens. Curr. Opin. Food Sci. 2016, 10, 76–84. [Google Scholar] [CrossRef]
- Fahrurrozi; Rahayu, E.P.; Nugroho, I.B.; Lisdiyanti, P. Lactic acid bacteria (LAB) isolated from fermented cocoa beans prevent the growth of model food-contaminating bacteria. AIP Conf. Proc. 2019, 2099, 020005. [Google Scholar] [CrossRef]


| Macroscopic Characteristics | Microscopic Characteristics | ||||||
|---|---|---|---|---|---|---|---|
| Strain | Color | Shape | Size | Elevation | Shape | Gram Stain | Catalase |
| S1.B | White | Circular | Small | Convex | Bacillus | (+/− ) | (+) |
| S2.A | White | Circular | Big | Convex | Bacillus | (+/−) | (+) |
| S2.B | Transparent | Circular | Small | Convex | Bacillus | (+/−) | (+) |
| S4.A | Transparent | Circular | Small | Convex | Bacillus | (+/−) | (+) |
| S5.A | White | Irregular | Big | Convex | Bacillus | (+/−) | (+) |
| S6.B | White | Irregular | Big | High | Bacillus | (+/−) | (+) |
| S7.A | White | Circular | Big | High | Bacillus | (+/−) | (+) |
| S8.A | White | Circular | Big | High | Bacillus | (+/−) | (+) |
| S9.B | White | Circular | Big | Convex | Bacillus | (+/−) | (+) |
| S10.A | White | Circular | Big | High | Bacillus | (+/−) | (+) |
| S11.B | White | Irregular | Big | High | Bacillus | (+/−) | (+) |
| S12.A | White | Circular | Small | High | Bacillus | (+/−) | (+) |
| S13.A | White | Irregular | Big | High | Bacillus | (+/−) | (+) |
| Macroscopic Characteristics | Microscopic Characteristics | ||||||
|---|---|---|---|---|---|---|---|
| Strain | Color | Shape | Size | Elevation | Shape | Gram Stain | Catalase |
| S1.B | White/Transparent | Circular | Small | Convex | Bacillus | (+) | (−) |
| S2.A | White | Circular | Small | High | Bacillus | (+) | (−) |
| S2.B | White | Circular | Small | High | Bacillus | (+) | (−) |
| S5.A | White | Circular | Big | Convex | Bacillus | (+) | (−) |
| S6.B | White/Transparent | Circular | Small | High | Cocci | (+) | (−) |
| S1.B (mm) | S2.B (mm) | S6.B (mm) | S2.A (mm) | S5.A (mm) | |
|---|---|---|---|---|---|
| S. Enteritidis | 12.00 ± 2.0 M | 7.67 ± 2.1 W | 7.00 ± 1.0 W | 10.57 ± 2.0 M | 7.33 ± 2.3 W |
| E. coli | 8.03 ± 2.3 W | 6.67 ± 1.1 W | 11.00 ± 3.6 M | 7.33 ± 2.3 W | 8.67 ± 2.3 W |
| S. aureus | 7.33 ± 2.31 W | 7.67 ± 2.65 W | 8.00 ± 1.73 W | 6.00 ± 0.0 W | 8.33 ± 2.5 W |
| S1.B (% v/v) | S2.B (% v/v) | S6.B (% v/v) | S2.A (% v/v) | S5.A (% v/v) | |
|---|---|---|---|---|---|
| E.coli | 50.85 | 46.47 | 48.20 | 44.17 | 7.41 |
| S. aureus | 38.13 | 49.62 | 45.54 | 38.80 | 99.42 |
| S. Enteritidis | 38.24 | 42.53 | 42.52 | 41.15 | 32.56 |
| Oral Phase | Gastric Phase | Intestinal Phase | ||||
|---|---|---|---|---|---|---|
| Strain | N/B (Log CFU/mL) | B (Log CFU/mL) | N/B (Log CFU/mL) | B (Log CFU/mL) | N/B (Log CFU/mL) | B (Log CFU/mL) |
| S2.A | TNTC | TNTC | 0.34 ± 0.49 bA | 3.40 ± 0.06 aA | 0.34 ± 0.49 bA | 3.04 ± 0.04 aA |
| S1.B | TNTC | TNTC | 3.64 ± 0.49 bB | 5.18 ± 0.01 aB | 3.61 ± 0.00 bB | 5.08 ± 0.10 aB |
| S5.A | 5.63 ± 0.13 | TNTC | 0.00 ± 0.00 bA | 4.37 ± 0.00 aC | 0.00 ± 0.00 bA | 4.34 ± 0.11 aC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Rosas-Ordaz, M.F.; Pérez-Armendáriz, B.; Meza-Jiménez, M.d.L.; Contreras-Mioni, L.; Cardoso-Ugarte, G.A. Theobroma bicolor (Pataxte) Fermentation: A Novel Source of Promising Probiotic Lactic Acid Bacteria. Fermentation 2026, 12, 41. https://doi.org/10.3390/fermentation12010041
Rosas-Ordaz MF, Pérez-Armendáriz B, Meza-Jiménez MdL, Contreras-Mioni L, Cardoso-Ugarte GA. Theobroma bicolor (Pataxte) Fermentation: A Novel Source of Promising Probiotic Lactic Acid Bacteria. Fermentation. 2026; 12(1):41. https://doi.org/10.3390/fermentation12010041
Chicago/Turabian StyleRosas-Ordaz, María Fernanda, Beatriz Pérez-Armendáriz, María de Lourdes Meza-Jiménez, Laura Contreras-Mioni, and Gabriel Abraham Cardoso-Ugarte. 2026. "Theobroma bicolor (Pataxte) Fermentation: A Novel Source of Promising Probiotic Lactic Acid Bacteria" Fermentation 12, no. 1: 41. https://doi.org/10.3390/fermentation12010041
APA StyleRosas-Ordaz, M. F., Pérez-Armendáriz, B., Meza-Jiménez, M. d. L., Contreras-Mioni, L., & Cardoso-Ugarte, G. A. (2026). Theobroma bicolor (Pataxte) Fermentation: A Novel Source of Promising Probiotic Lactic Acid Bacteria. Fermentation, 12(1), 41. https://doi.org/10.3390/fermentation12010041







