Production and Characterization of Downgraded Maple Syrup-Based Synbiotic Containing Bacillus velezensis FZB42 for Animal Nutrition
Abstract
1. Introduction
2. Materials and Methods
2.1. Strain and Preliminary Assays
2.2. Culture Conditions for Media Optimization
2.3. B. velezensis Viability
2.4. Scaled-Up Validation Experiment
2.5. Spray Drying
2.6. B. velezensis Survival during In Vitro Digestion of the Spray-Dried Product
3. Results and Discussion
3.1. Preliminary Assays
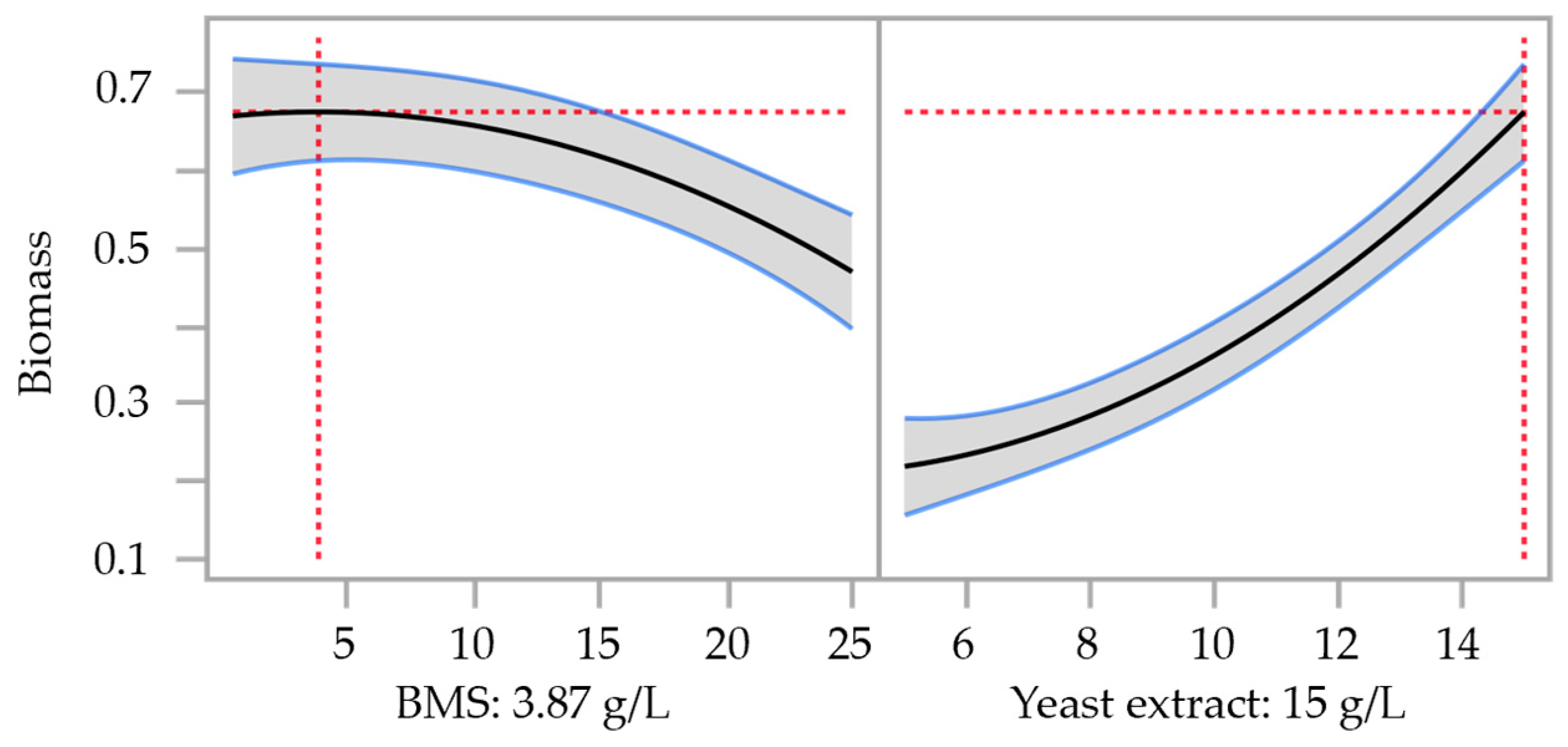
3.2. Optimization of Nutrient Medium by Central Composite Design (CCD)
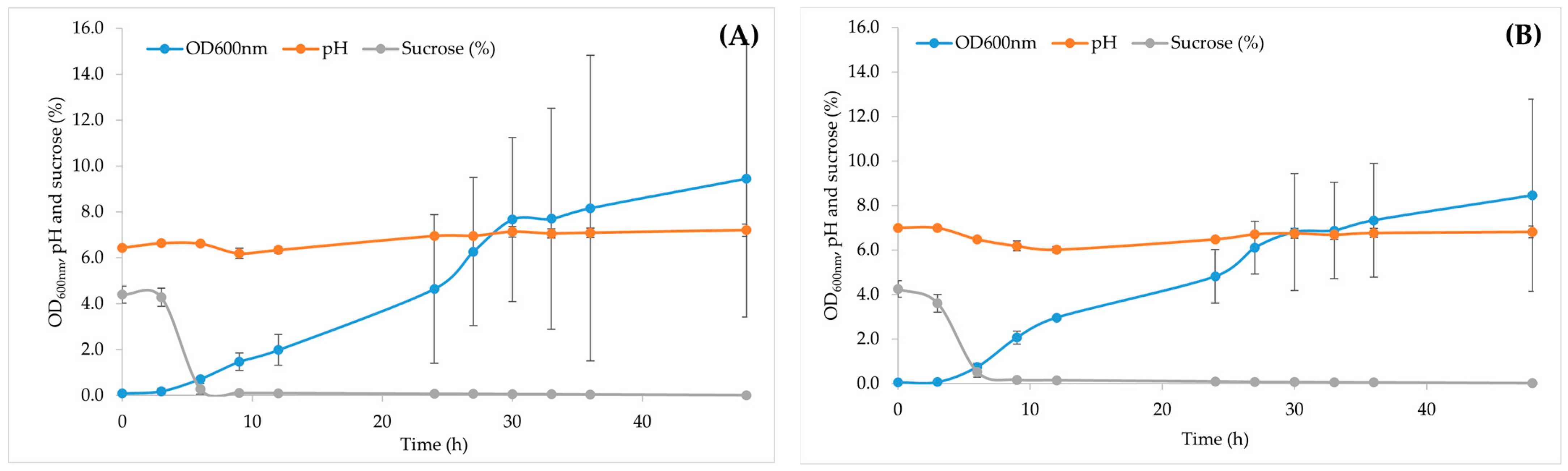
3.3. Large-Scale Production of Fermented Downgraded Maple Syrup
3.4. Spray Drying
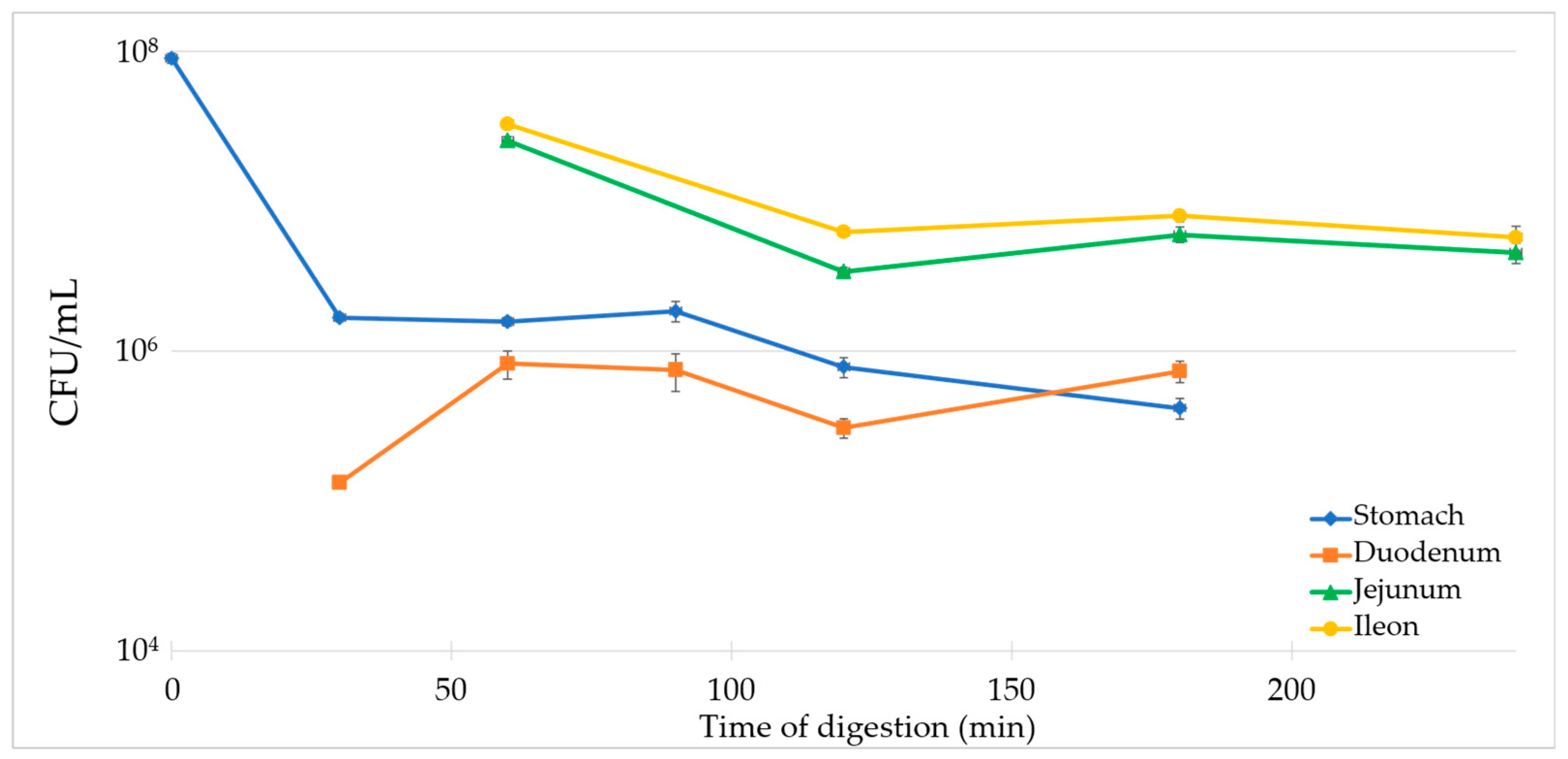
3.5. Survival Evaluation of the Dry Product with TIM-1
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cromwell, G.L. Why and how antibiotics are used in swine production. Anim. Biotechnol. 2002, 13, 7–27. [Google Scholar] [CrossRef] [PubMed]
- Marshall, B.M.; Levy, S.B. Food animals and antimicrobials: Impacts on human health. Clin. Microbiol. Rev. 2011, 24, 718–733. [Google Scholar] [CrossRef]
- World Health Organization. Antibiotic Resistance. Available online: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 9 April 2024).
- FAO/WHO. Probiotics in Food: Health and Nutritional Properties and Guidelines for Evaluation; FAO/WHO: Rome, Italy, 2001. [Google Scholar]
- Huyghebaert, G.; Ducatelle, R.; Van Immerseel, F. An update on alternatives to antimicrobial growth promoters for broilers. Vet. J. 2011, 187, 182–188. [Google Scholar] [CrossRef]
- Yang, Y.; Iji, P.A.; Choct, M. Dietary modulation of gut microflora in broiler chickens: A review of the role of six kinds of alternatives to in-feed antibiotics. World’s Poult. Sci. J. 2019, 65, 97–114. [Google Scholar] [CrossRef]
- Yoo, H.; Rheem, I.; Rheem, S.; Oh, S. Optimizing Medium Components for the Maximum Growth of Lactobacillus plantarum JNU 2116 Using Response Surface Methodology. Korean J. Food Sci. Anim. Resour. 2018, 38, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Piantini, E.; Rodríguez-Díez, E.; Chavarri, M.; López-de-Armentia, I.; Villaran, M.C.; Lombraña, J.I. Preparation of Hydrolyzed Sugarcane Molasses as a Low-Cost Medium for the Mass Production of Probiotic Lactobacillus paracasei ssp. paracasei F19. Separations 2023, 10, 33. [Google Scholar] [CrossRef]
- Maragkoudakis, P.; Vendramin, V.; Bovo, B.; Treu, L.; Corich, V.; Giacomini, A. Potential use of scotta, the by-product of the ricotta cheese manufacturing process, for the production of fermented drinks. J. Dairy Res. 2016, 83, 104–108. [Google Scholar] [CrossRef]
- Meral, H.; Savaş, İ.; Çiçek, Ş.K.; Demirdöven, A. Peach pomace: A potential probiotic carrier for fiber enrichment in milk. J. Food Meas. Charact. 2023, 18, 1933–1946. [Google Scholar] [CrossRef]
- Nagai, N.; Yamamoto, T.; Tanabe, W.; Ito, Y.; Kurabuchi, S.; Mitamura, K.; Taga, A. Changes in plasma glucose in Otsuka Long-Evans Tokushima Fatty rats after oral administration of maple syrup. J. Oleo Sci. 2015, 64, 331–335. [Google Scholar] [CrossRef]
- Legault, J.; Girard-Lalancette, K.; Grenon, C.; Dussault, C.; Pichette, A. Antioxidant activity, inhibition of nitric oxide overproduction, and in vitro antiproliferative effect of maple sap and syrup from Acer saccharum. J. Med. Food 2010, 13, 460–468. [Google Scholar] [CrossRef]
- Filteau, M.; Charron, G.; Landry, C.R. Identification of the fitness determinants of budding yeast on a natural substrate. ISME J. 2017, 11, 959–971. [Google Scholar] [CrossRef] [PubMed]
- Adams, G.; Bishop, C. Constitution of an arabinogalactan from maple sap. Can. J. Chem. 1960, 38, 2380–2386. [Google Scholar] [CrossRef]
- Sun, J.; Ma, H.; Seeram, N.P.; Rowley, D.C. Detection of Inulin, a Prebiotic Polysaccharide, in Maple Syrup. J. Agric. Food Chem. 2016, 64, 7142–7147. [Google Scholar] [CrossRef] [PubMed]
- Robinson, R.R.; Feirtag, J.; Slavin, J.L. Effects of dietary arabinogalactan on gastrointestinal and blood parameters in healthy human subjects. J. Am. Coll. Nutr. 2001, 20, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.-M.; Kim, A.-J.; Kim, Y.-O.; Hwang, J.-K. Immunomodulating activity of arabinogalactan and fucoidan in vitro. J. Med. Food 2005, 8, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Whistler, R.L.; Bushway, A.A.; Singh, P.P.; Nakahara, W.; Tokuzen, R. Noncytotoxic, antitumor polysaccharides. Adv. Carbohydr. Chem. Biochem. 1976, 32, 235–275. [Google Scholar] [PubMed]
- Ball, D.W. The Chemical Composition of Maple Syrup. J. Chem. Educ. 2007, 84, 1647. [Google Scholar] [CrossRef]
- Saraiva, A.; Carrascosa, C.; Ramos, F.; Raheem, D.; Lopes, M.; Raposo, A. Maple Syrup: Chemical Analysis and Nutritional Profile, Health Impacts, Safety and Quality Control, and Food Industry Applications. Int. J. Environ. Res. Public Health 2022, 19, 13684. [Google Scholar] [CrossRef]
- Decabooter, G.; Aspirault, C.; Filteau, M.; Fliss, I. The Physicochemical Characterization and In Vitro Digestibility of Maple Sugar Sand and Downgraded Maple Syrups. Foods 2023, 12, 3528. [Google Scholar] [CrossRef]
- Huang, S.; Vignolles, M.-L.; Chen, X.D.; Le Loir, Y.; Jan, G.; Schuck, P.; Jeantet, R. Spray drying of probiotics and other food-grade bacteria: A review. Trends Food Sci. Technol. 2017, 63, 1–17. [Google Scholar] [CrossRef]
- Barbosa, J.; Borges, S.; Amorim, M.; Pereira, M.J.; Oliveira, A.; Pintado, M.E.; Teixeira, P. Comparison of spray drying, freeze drying and convective hot air drying for the production of a probiotic orange powder. J. Funct. Foods 2015, 17, 340–351. [Google Scholar] [CrossRef]
- Liu, H.; Cui, S.W.; Chen, M.; Li, Y.; Liang, R.; Xu, F.; Zhong, F. Protective approaches and mechanisms of microencapsulation to the survival of probiotic bacteria during processing, storage and gastrointestinal digestion: A review. Crit. Rev. Food Sci. Nutr. 2019, 59, 2863–2878. [Google Scholar] [CrossRef] [PubMed]
- de Vos, P.; Faas, M.M.; Spasojevic, M.; Sikkema, J. Encapsulation for preservation of functionality and targeted delivery of bioactive food components. Int. Dairy J. 2010, 20, 292–302. [Google Scholar] [CrossRef]
- Jang, K.B.; Purvis, J.M.; Kim, S.W. Dose-response and functional role of whey permeate as a source of lactose and milk oligosaccharides on intestinal health and growth of nursery pigs. J. Anim. Sci. 2021, 99, skab008. [Google Scholar] [CrossRef] [PubMed]
- Mingmongkolchai, S.; Panbangred, W. Bacillus probiotics: An alternative to antibiotics for livestock production. J. Appl. Microbiol. 2018, 124, 1334–1346. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Garcia, C.; Bejar, V.; Martinez-Checa, F.; Llamas, I.; Quesada, E. Bacillus velezensis sp. nov., a surfactant-producing bacterium isolated from the river Velez in Malaga, southern Spain. Int. J. Syst. Evol. Microbiol. 2005, 55, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.T.; Lee, F.L.; Tai, C.J.; Kuo, H.P. Bacillus velezensis is a later heterotypic synonym of Bacillus amyloliquefaciens. Int. J. Syst. Evol. Microbiol. 2008, 58, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Rabbee, M.F.; Ali, M.S.; Choi, J.; Hwang, B.S.; Jeong, S.C.; Baek, K.-h. Bacillus velezensis: A valuable member of bioactive molecules within plant microbiomes. Molecules 2019, 24, 1046. [Google Scholar] [CrossRef]
- Dunlap, C.A.; Kim, S.J.; Kwon, S.W.; Rooney, A.P. Bacillus velezensis is not a later heterotypic synonym of Bacillus amyloliquefaciens; Bacillus methylotrophicus, Bacillus amyloliquefaciens subsp. plantarum and ‘Bacillus oryzicola’ are later heterotypic synonyms of Bacillus velezensis based on phylogenomics. Int. J. Syst. Evol. Microbiol. 2016, 66, 1212–1217. [Google Scholar] [CrossRef] [PubMed]
- Fan, B.; Wang, C.; Song, X.; Ding, X.; Wu, L.; Wu, H.; Gao, X.; Borriss, R. Bacillus velezensis FZB42 in 2018: The Gram-Positive Model Strain for Plant Growth Promotion and Biocontrol. Front. Microbiol. 2018, 9, 2491. [Google Scholar] [CrossRef]
- Chen, X.H.; Vater, J.; Piel, J.; Franke, P.; Scholz, R.; Schneider, K.; Koumoutsi, A.; Hitzeroth, G.; Grammel, N.; Strittmatter, A.W.; et al. Structural and functional characterization of three polyketide synthase gene clusters in Bacillus amyloliquefaciens FZB 42. J. Bacteriol. 2006, 188, 4024–4036. [Google Scholar] [CrossRef] [PubMed]
- Koumoutsi, A.; Chen, X.H.; Henne, A.; Liesegang, H.; Hitzeroth, G.; Franke, P.; Vater, J.; Borriss, R. Structural and functional characterization of gene clusters directing nonribosomal synthesis of bioactive cyclic lipopeptides in Bacillus amyloliquefaciens strain FZB42. J. Bacteriol. 2004, 186, 1084–1096. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.; Chen, X.-H.; Vater, J.; Franke, P.; Nicholson, G.; Borriss, R.; Süssmuth, R.D. Macrolactin is the polyketide biosynthesis product of the pks2 cluster of Bacillus amyloliquefaciens FZB42. J. Nat. Prod. 2007, 70, 1417–1423. [Google Scholar] [CrossRef]
- Chen, X.H.; Koumoutsi, A.; Scholz, R.; Eisenreich, A.; Schneider, K.; Heinemeyer, I.; Morgenstern, B.; Voss, B.; Hess, W.R.; Reva, O.; et al. Comparative analysis of the complete genome sequence of the plant growth-promoting bacterium Bacillus amyloliquefaciens FZB42. Nat. Biotechnol. 2007, 25, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Michiels, J.; Possemiers, S.; Degroote, J.; Ovyn, A.; De Smet, S.; Nakamura, N. Feeding Bacillus subtilis C-3102 to sows and suckling piglets and to weaned piglets improves parameters of gut health and feed: Gain ratio in weaners. J. Anim. Sci. 2016, 94, 135–137. [Google Scholar] [CrossRef]
- Thurlow, C.M.; Williams, M.A.; Carrias, A.; Ran, C.; Newman, M.; Tweedie, J.; Allison, E.; Jescovitch, L.N.; Wilson, A.E.; Terhune, J.S. Bacillus velezensis AP193 exerts probiotic effects in channel catfish (Ictalurus punctatus) and reduces aquaculture pond eutrophication. Aquaculture 2019, 503, 347–356. [Google Scholar] [CrossRef]
- Blanquet, S.; Zeijdner, E.; Beyssac, E.; Meunier, J.-P.; Denis, S.; Havenaar, R.; Alric, M. A dynamic artificial gastrointestinal system for studying the behavior of orally administered drug dosage forms under various physiological conditions. Pharm. Res. 2004, 21, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Kimoto-Nira, H.; Suzuki, C.; Sasaki, K.; Kobayashi, M.; Mizumachi, K. Survival of a Lactococcus lactis strain varies with its carbohydrate preference under in vitro conditions simulated gastrointestinal tract. Int. J. Food Microbiol. 2010, 143, 226–229. [Google Scholar] [CrossRef]
- Herigstad, B.; Hamilton, M.; Heersink, J. How to optimize the drop plate method for enumerating bacteria. J. Microbiol. Methods 2001, 44, 121–129. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists. AOAC Official Method 926.12 Moisture and Volatile Matter in Oils and Fats: Vacuum Oven Method. In Official Methods of Analysis of AOAC INTERNATIONAL; Latimer, G.W., Jr., Ed.; Oxford University Press: Oxford, UK, 2023. [Google Scholar]
- Minekus, M.; Marteau, P.; Havenaar, R.; in’t Veld, J.H.J.H. A Multicompartmental Dynamic Computer-controlled Model Simulating the Stomach and Small Intestine. Altern. Lab. Anim. 1995, 23, 197–209. [Google Scholar] [CrossRef]
- Meunier, J.P.; Manzanilla, E.G.; Anguita, M.; Denis, S.; Perez, J.F.; Gasa, J.; Cardot, J.M.; Garcia, F.; Moll, X.; Alric, M. Evaluation of a dynamic in vitro model to simulate the porcine ileal digestion of diets differing in carbohydrate composition. J. Anim. Sci. 2008, 86, 1156–1163. [Google Scholar] [CrossRef] [PubMed]
- de Medeiros Dantas, J.M.; Álvares Monge Neto, A.; Ghislain, T.; Lavoie, J.-M. Bioethanol Production as an Alternative End for Maple Syrups with Flavor Defects. Fermentation 2022, 8, 58. [Google Scholar] [CrossRef]
- Zeng, X.; Small, D.P.; Wan, W. Statistical optimization of culture conditions for bacterial cellulose production by Acetobacter xylinum BPR 2001 from maple syrup. Carbohydr. Polym. 2011, 85, 506–513. [Google Scholar] [CrossRef]
- Papizadeh, M.; Rohani, M.; Hosseini, S.N.; Shojaosadati, S.A.; Nahrevanian, H.; Talebi, M.; Pourshafie, M.R. Screening for efficient nitrogen sources for overproduction of the biomass of the functionally probiotic L. plantarum strain RPR42 in a cane molasses-based medium. AMB Express 2020, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Shih, I.-L.; Lin, C.-Y.; Wu, J.-Y.; Hsieh, C. Production of antifungal lipopeptide from Bacillus subtilis in submerged fermentation using shake flask and fermentor. Korean J. Chem. Eng. 2009, 26, 1652–1661. [Google Scholar] [CrossRef]
- Singh, S.; Sithole, B.; Lekha, P.; Permaul, K.; Govinden, R. Optimization of cultivation medium and cyclic fed-batch fermentation strategy for enhanced polyhydroxyalkanoate production by Bacillus thuringiensis using a glucose-rich hydrolyzate. Bioresour. Bioprocess. 2021, 8, 11. [Google Scholar] [CrossRef]
- Yánez-Mendizábal, V.; Viñas, I.; Usall, J.; Torres, R.; Solsona, C.; Teixidó, N. Production of the postharvest biocontrol agent Bacillus subtilis CPA-8 using low cost commercial products and by-products. Biol. Control 2012, 60, 280–289. [Google Scholar] [CrossRef]
- Azaroual, S.E.; El Mernissi, N.; Zeroual, Y.; Bouizgarne, B.; Meftah Kadmiri, I. Effect of Bacillus spp. strains on wheat nutrient assimilation and bioformulation by new spray drying approach using natural phosphate powder. Dry. Technol. 2021, 40, 2630–2644. [Google Scholar] [CrossRef]
- Luise, D.; Bertocchi, M.; Motta, V.; Salvarani, C.; Bosi, P.; Luppi, A.; Fanelli, F.; Mazzoni, M.; Archetti, I.; Maiorano, G.; et al. Bacillus sp. probiotic supplementation diminish the Escherichia coli F4ac infection in susceptible weaned pigs by influencing the intestinal immune response, intestinal microbiota and blood metabolomics. J. Anim. Sci. Biotechnol. 2019, 10, 74. [Google Scholar] [CrossRef]
- Jang, K.B.; Duarte, M.E.; Purvis, J.M.; Kim, S.W. Impacts of weaning age on dietary needs of whey permeate for pigs at 7 to 11 kg body weight. J. Anim. Sci. Biotechnol. 2021, 12, 111. [Google Scholar] [CrossRef]
- Mahan, D.C.; Fastinger, N.D.; Peters, J.C. Effects of diet complexity and dietary lactose levels during three starter phases on postweaning pig performance123. J. Anim. Sci. 2004, 82, 2790–2797. [Google Scholar] [CrossRef] [PubMed]
- Barajas-Álvarez, P.; González-Ávila, M.; Espinosa-Andrews, H. Recent Advances in Probiotic Encapsulation to Improve Viability under Storage and Gastrointestinal Conditions and Their Impact on Functional Food Formulation. Food Rev. Int. 2021, 39, 992–1013. [Google Scholar] [CrossRef]
- Maathuis, A.; Keller, D.; Farmer, S. Survival and metabolic activity of the GanedenBC30 strain of Bacillus coagulans in a dynamic in vitro model of the stomach and small intestine. Benef. Microbes 2010, 1, 31–36. [Google Scholar] [CrossRef]
- Keller, D.; Verbruggen, S.; Cash, H.; Farmer, S.; Venema, K. Spores of Bacillus coagulans GBI-30, 6086 show high germination, survival and enzyme activity in a dynamic, computer-controlled in vitro model of the gastrointestinal tract. Benef. Microbes 2019, 10, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, M.; Nakamura, Y.; Maathuis, A.; Venema, K.; Murota, I.; Yamamoto, N. Influence of Bacillus subtilis C-3102 on microbiota in a dynamic in vitro model of the gastrointestinal tract simulating human conditions. Benef. Microbes 2012, 3, 229–236. [Google Scholar] [CrossRef]
- Venema, K.; Verhoeven, J.; Beckman, C.; Keller, D. Survival of a probiotic-containing product using capsule-within-capsule technology in an in vitro model of the stomach and small intestine (TIM-1). Benef. Microbes 2020, 11, 403–409. [Google Scholar] [CrossRef]
| Standard Order | Downgraded Maple Syrup (g/L) | Yeast Extract (g/L) |
|---|---|---|
| 1 | 12.75 | 15 |
| 2 | 12.75 | 10 |
| 3 | 25 | 15 |
| 4 | 12.75 | 10 |
| 5 | 0.5 | 15 |
| 6 | 12.75 | 5 |
| 7 | 0.5 | 10 |
| 8 | 0.5 | 5 |
| 9 | 25 | 5 |
| 10 | 25 | 10 |
| 11 | 12.75 | 10 |
| 12 | 12.75 | 10 |
| Source | Nparm | DF | Sum of Squares | F Ratio | Prob > F |
|---|---|---|---|---|---|
| BMS (0.5, 25) | 1 | 1 | 0.02343750 | 10.4313 | 0.0179 |
| YE (5, 15) | 1 | 1 | 0.24200417 | 107.7090 | <0.0001 |
| BMS × BMS | 1 | 1 | 0.01269600 | 5.6506 | 0.0550 |
| BMS × YE | 1 | 1 | 0.00562500 | 2.5035 | 0.1647 |
| YE × YE | 1 | 1 | 0.01926667 | 8.5750 | 0.0263 |
| Step | CFU/mL or g |
|---|---|
| Erlenmeyer fermentation | 7.75 ± 0.35 × 108 |
| Bioreactor fermentation | 6.15 ± 0.46 × 109 |
| Spray drying | 2.00 ± 0.42 × 108 |
| TIM-1 a | 2.19 ± 1.09 × 109 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Decabooter, G.; Theiri, M.; Groleau, D.; Filteau, M.; Fliss, I. Production and Characterization of Downgraded Maple Syrup-Based Synbiotic Containing Bacillus velezensis FZB42 for Animal Nutrition. Fermentation 2024, 10, 221. https://doi.org/10.3390/fermentation10040221
Decabooter G, Theiri M, Groleau D, Filteau M, Fliss I. Production and Characterization of Downgraded Maple Syrup-Based Synbiotic Containing Bacillus velezensis FZB42 for Animal Nutrition. Fermentation. 2024; 10(4):221. https://doi.org/10.3390/fermentation10040221
Chicago/Turabian StyleDecabooter, Gautier, Mariem Theiri, Denis Groleau, Marie Filteau, and Ismail Fliss. 2024. "Production and Characterization of Downgraded Maple Syrup-Based Synbiotic Containing Bacillus velezensis FZB42 for Animal Nutrition" Fermentation 10, no. 4: 221. https://doi.org/10.3390/fermentation10040221
APA StyleDecabooter, G., Theiri, M., Groleau, D., Filteau, M., & Fliss, I. (2024). Production and Characterization of Downgraded Maple Syrup-Based Synbiotic Containing Bacillus velezensis FZB42 for Animal Nutrition. Fermentation, 10(4), 221. https://doi.org/10.3390/fermentation10040221







