The Combined Influence of Dopant Species and Surface Termination on the Electronic Properties of Diamond Surfaces
Abstract
1. Introduction
2. Theoretical Methods
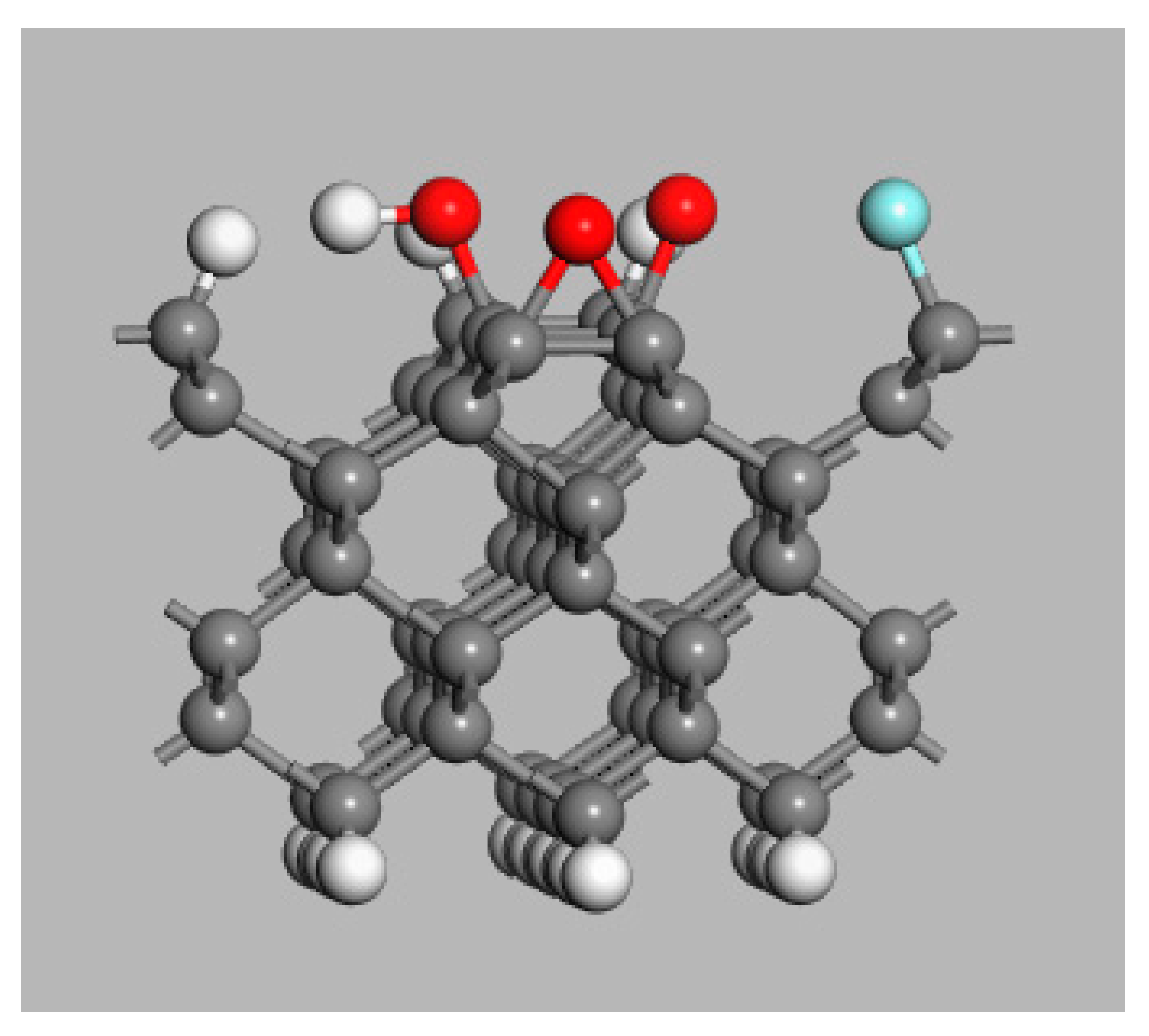
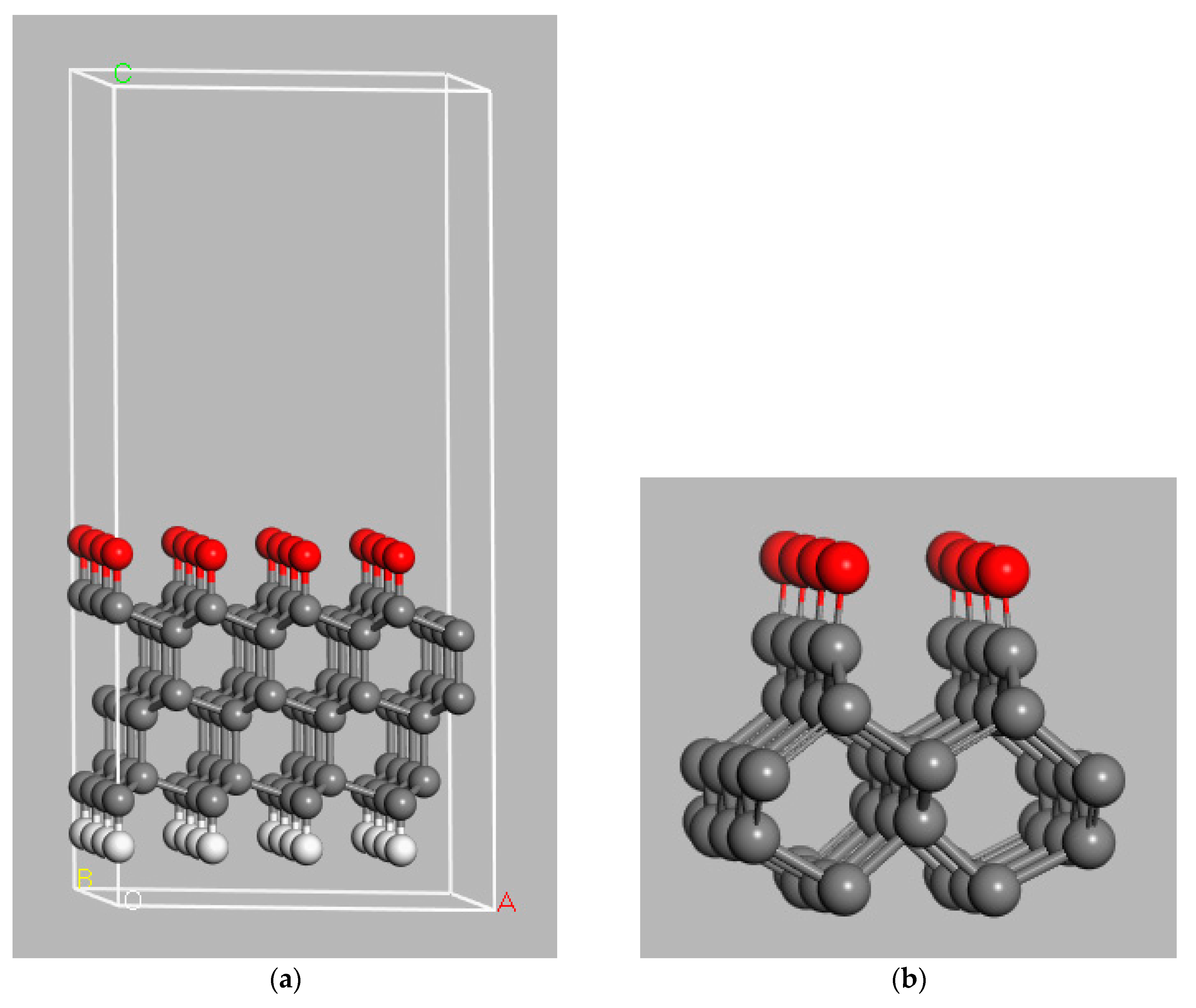
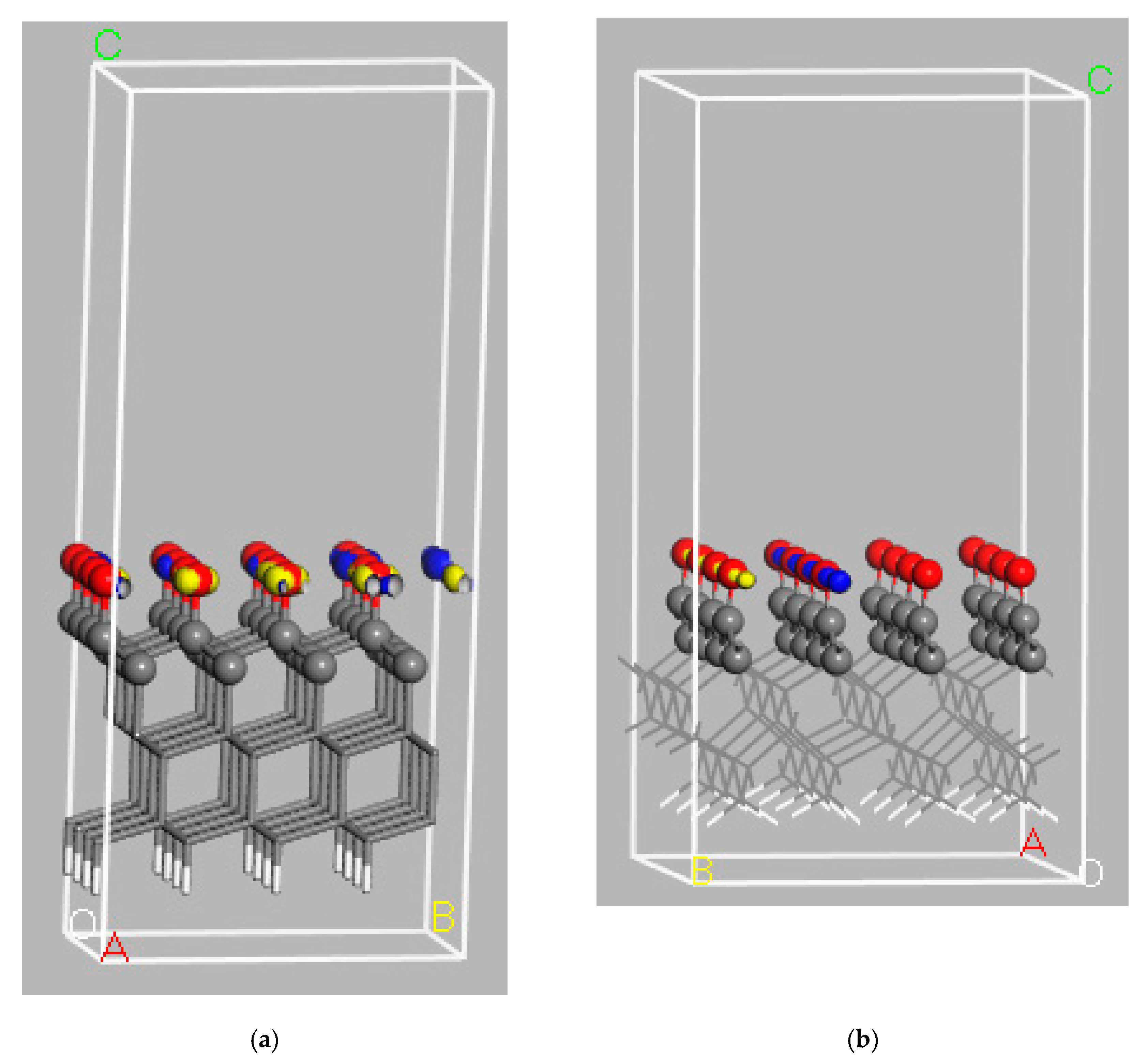
3. Model Systems
4. Diamond Surface Termination
5. Substitutional Doping of Diamond
6. Effect of Surface Terminating Species on the Surface Electronic Structure
6.1. General
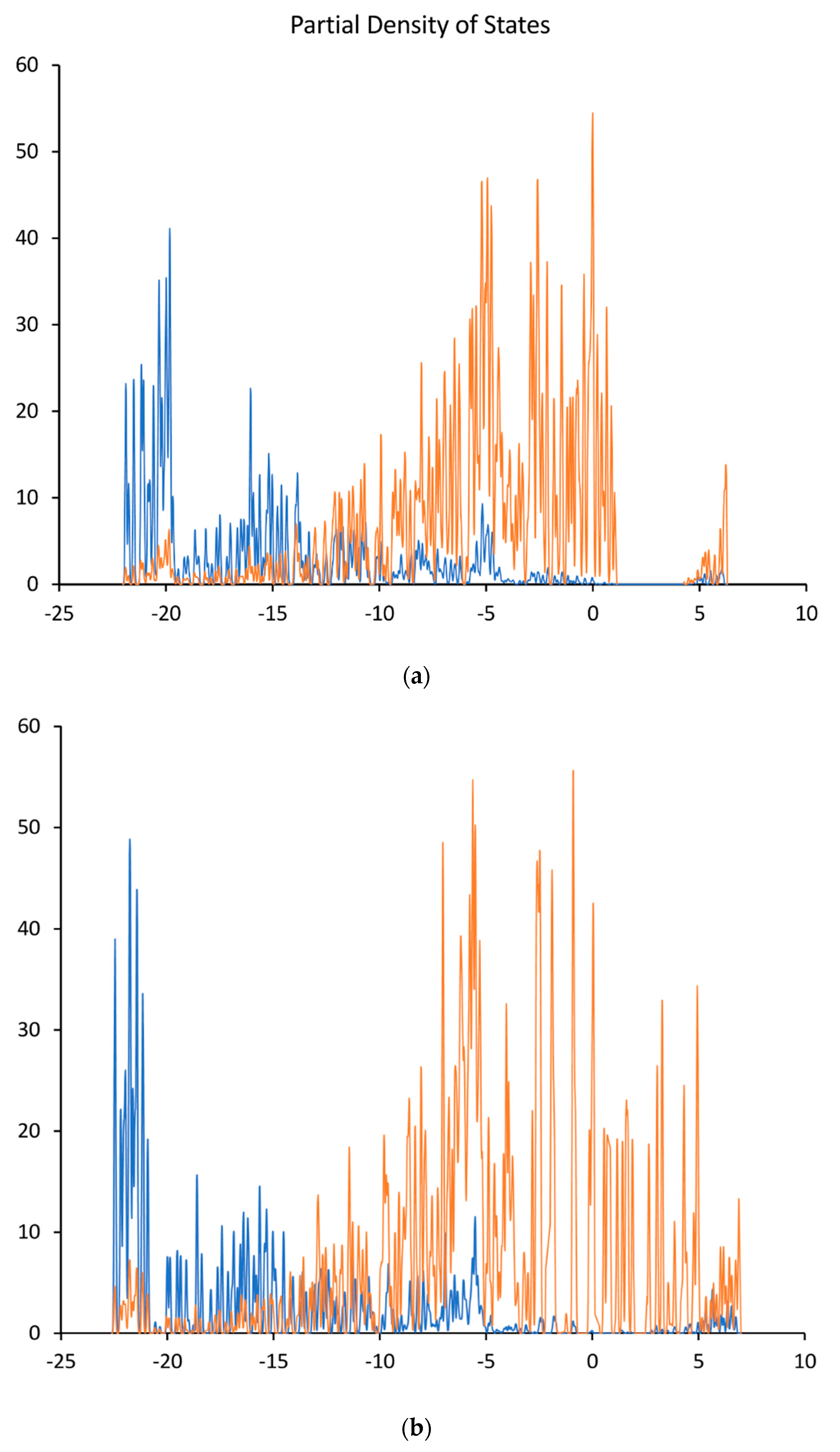
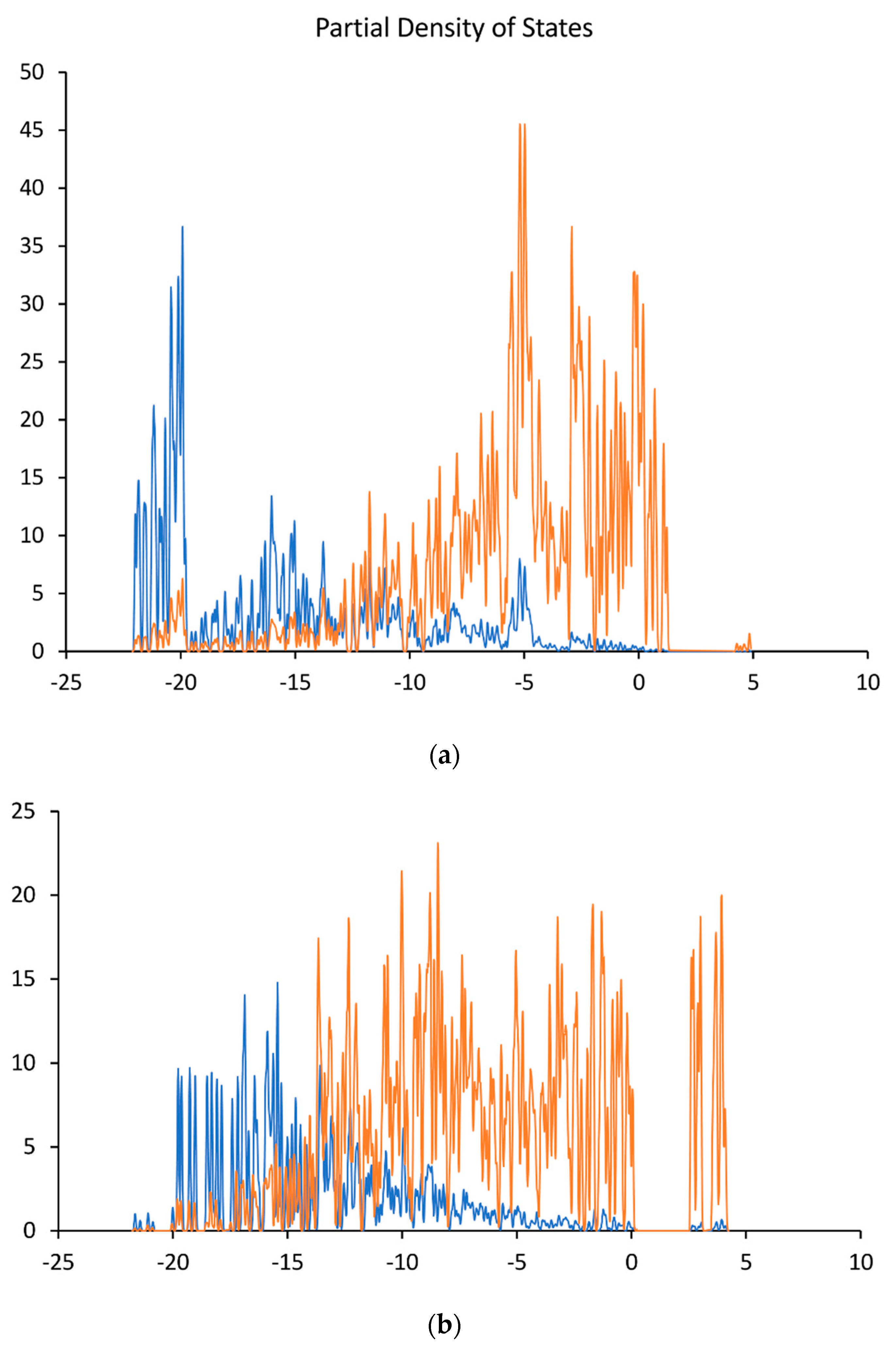
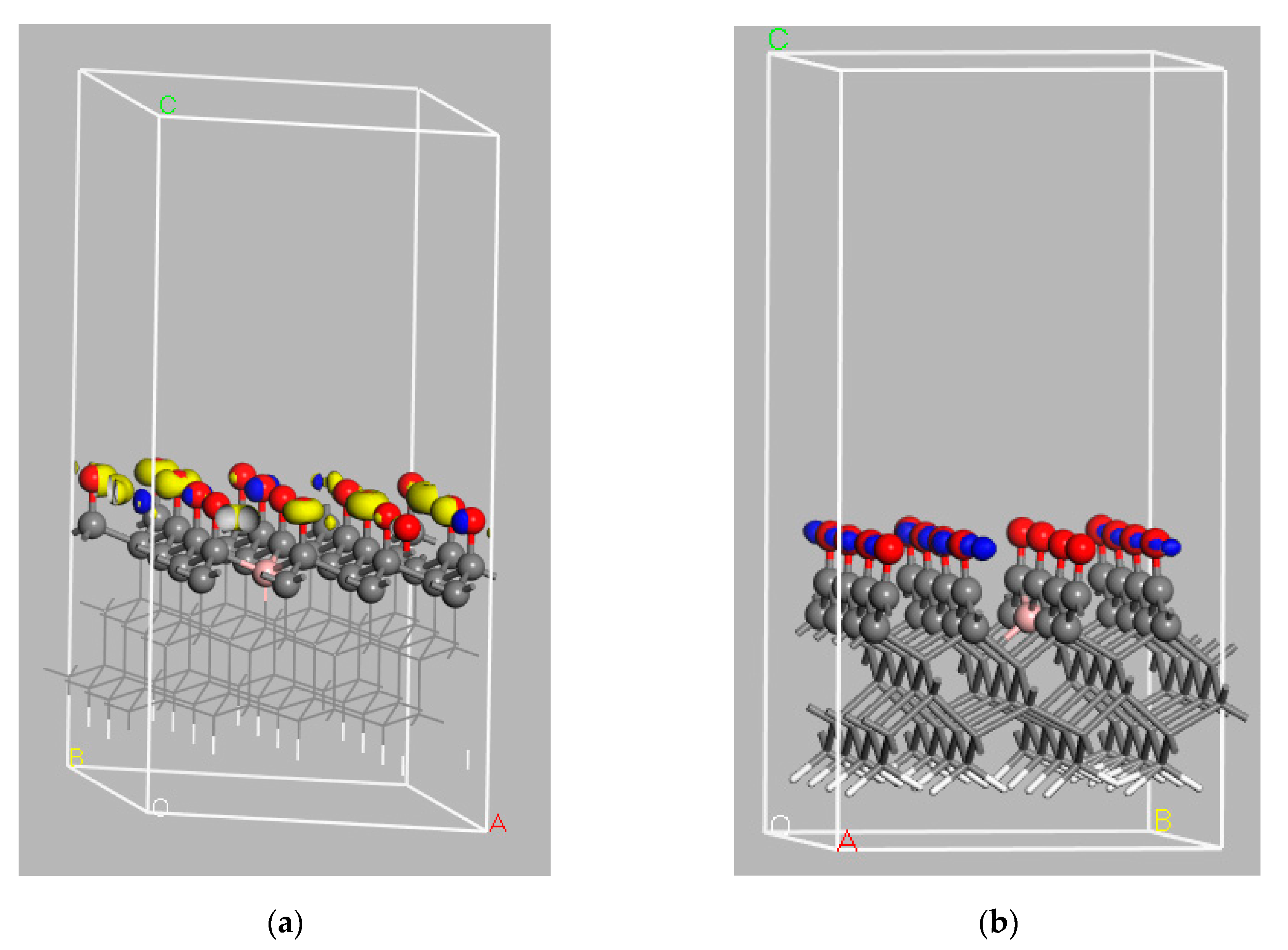
6.2. Analysis of Calculated Electronic Structures for Undoped Diamond Surfaces
7. The Combined Effect of p-Type Doping and Surface Termination on the Surface Electronic Structure
7.1. General
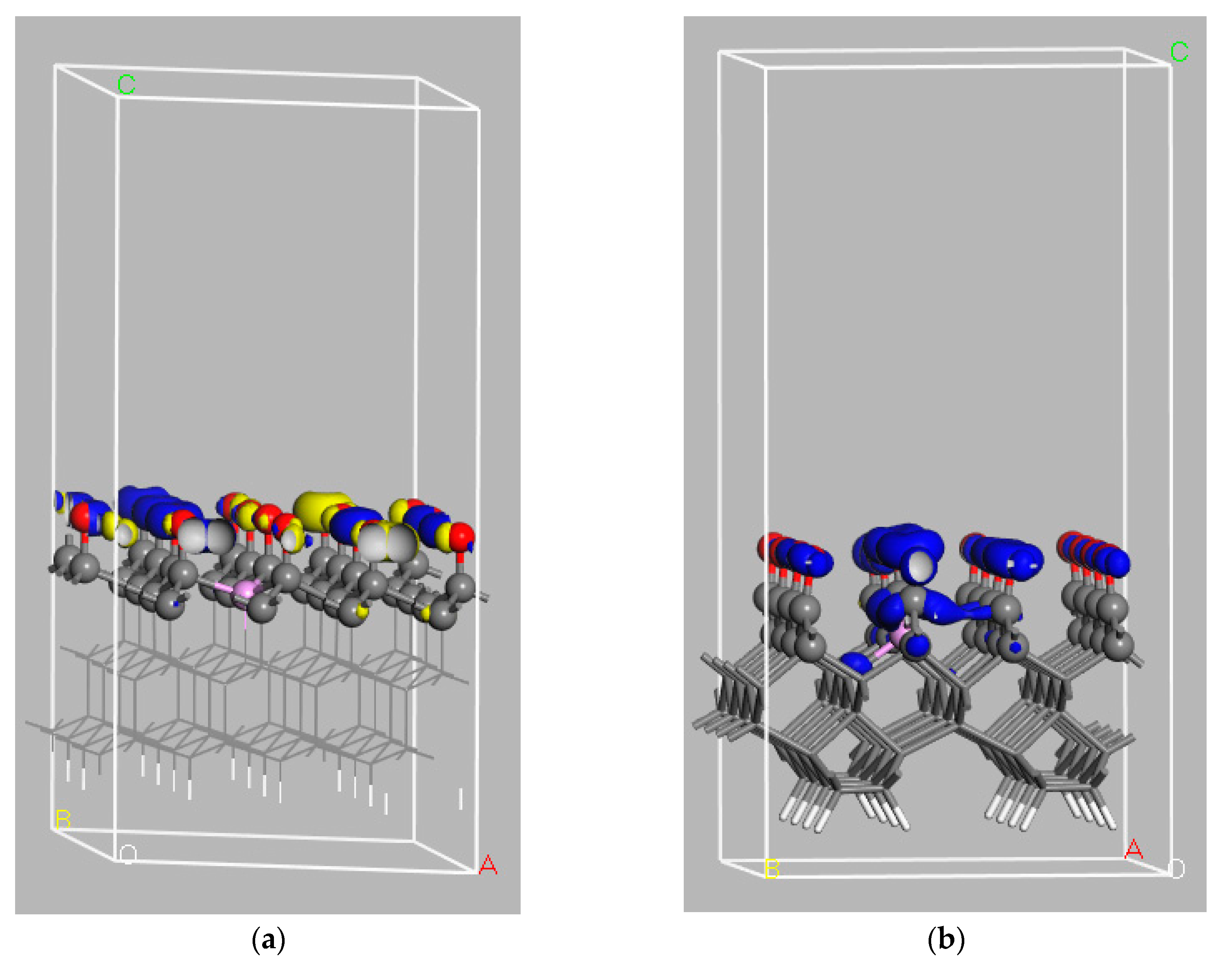
7.2. Analysis of Calculated Electronic Structures for B-Doped Diamond Surfaces
8. The Combined Effect of n-Type Doping and Surface Termination on the Surface Electronic Structure
8.1. General
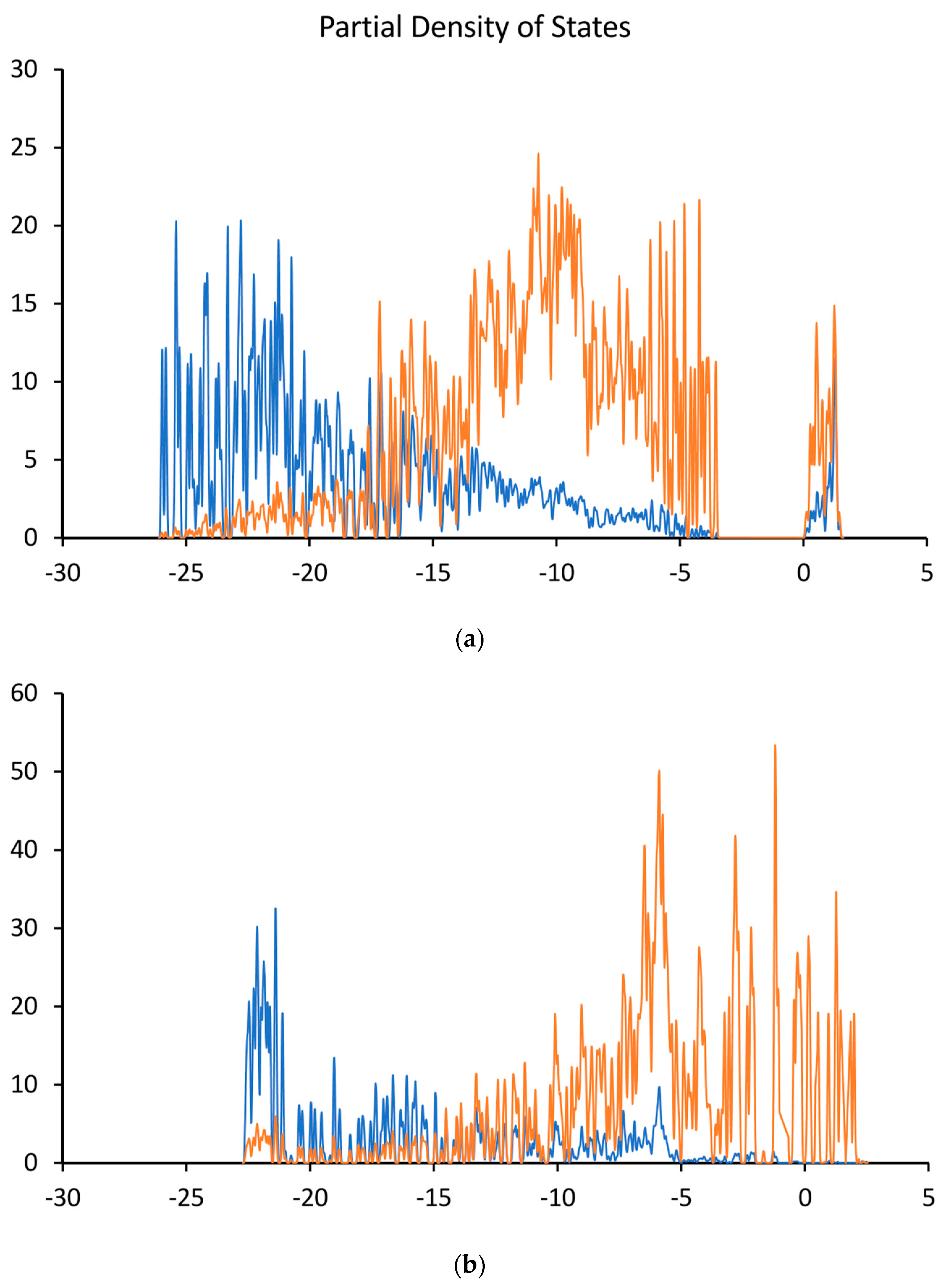
8.2. Analysis of Calculated Electronic Structures for n-Type Doped Diamond Surfaces
9. Summary and Conclusions
Supplementary Materials
Funding
Acknowledgments
Conflicts of Interest
References
- Shenai, K.; Scott, R.S.; Baliga, B.J. Optimum semiconductors for high-power electronics. Electron Devices IEEE Trans. 1989, 36, 1811–1823. [Google Scholar] [CrossRef]
- Ley, L.; Ristein, J.; Meier, F.; Riedel, M.; Strobel, P. Surface conductivity of the diamond: A novel transfer doping mechanism. Physica B 2006, 376–377, 262–267. [Google Scholar] [CrossRef]
- Uppireddi, K.; Westover, T.; Fisher, T.; Weiner, B.R.; Morell, G. Thermionic emission energy distribution from nanocrystalline diamond films for direct thermal-electrical energy conversion applications. J. Appl. Phys. 2009, 106, 043716. [Google Scholar] [CrossRef]
- Koeck, F.; Nemanich, R. Substrate-diamond interface consideration for enhanced thermionic electron emission from nitrogen doped diamond films. J. Appl. Phys. 2012, 112, 113717. [Google Scholar] [CrossRef]
- Neugebohrn, N.; Sun, T.; Koeck, F.; Hembree, G.G.; Nemanich, R.J.; Schmidt, T.; Falta, J. Spatial correlation of photo-induced and thermionic electron emission from low work function diamond films. Diam. Relat. Mater. 2013, 40, 12–16. [Google Scholar] [CrossRef]
- Suzuki, M.; Ono, T.; Sakuma, N.; Naoshi Sakai, T. Low-temperature thermionic emission from nitrogen-doped nanocrystalline diamond films on n-type Si grown by MPCVD. Diam. Relat. Mater. 2009, 18, 1274–1277. [Google Scholar] [CrossRef]
- Sowers, A.T.; Ward, B.L.; English, S.L.; Nemanich, R.J. Field emission properties of nitrogen-doped diamond films. J. Appl. Phys. 1999, 86, 3973–3982. [Google Scholar] [CrossRef]
- Koeck, F.A.M.; Nemanich, R. Emission characterization from nitrogen-doped diamond with respect to energy conversion. Diam. Relat. Mater. 2006, 15, 217–220. [Google Scholar] [CrossRef]
- Koeck, F.A.M.; Nemanich, R.J.; Balasubramaniam, Y.; Haenen, K.; Sharp, J. Enhanced thermionic energy conversion and thermionic emission from doped diamond films through methane exposure. Diam. Relat. Mater. 2011, 20, 1229–1233. [Google Scholar] [CrossRef]
- Sherehiy, A.; Dumpala, S.; Sunkara, M.K.; Jasinski, J.B.; Cohn, R.W.; Sumanasekera, G.U. Thermionic emission from phosphorous (P) doped diamond nanocrystals supported by conical carbon nanotubes and ultraviolet photoelectron spectroscopy study of P-doped diamond films. Diam. Relat. Mater. 2014, 50, 66–76. [Google Scholar] [CrossRef]
- Kono, S.; Takyo, G.; Amano, N.; Plusnin, N.I.; Mizuochi, K.; Aoyama, T.; Goto, T.; Abukawa, T.; Namba, A.; Tasumi, N.; et al. Mechanism of Field Emission from a Highly Phosphorous-Doped Chemical Vapor Deposition Diamond (111) Surface. Jpn. J. Appl. Phys. 2007, 46, L21–L24. [Google Scholar] [CrossRef]
- Yamada, T.; Masuzawa, T.; Mimura, H.; Okano, K. Electron emission from conduction band of heavily phosphorous doped diamond negative affinity surface. J. Phys. D Appl. Phys. 2016, 49, 045102. [Google Scholar] [CrossRef]
- Kato, H.; Takeuchi, D.; Ogura, M.; Yamada, T.; Kataoka, M.; Kimura, Y.; Sobue, S.; Nebel, C.E.; Yamasaki, S. Heavily phosphorus-doped nano-crystalline diamond electrode for thermionic emission application. Diam. Relat. Mater. 2016, 63, 165–168. [Google Scholar] [CrossRef]
- Haque, A.; Narayan, J. Electron field emission from Q-carbon. Diam. Relat. Mater. 2018, 86, 71–78. [Google Scholar] [CrossRef]
- Haque, A.; Narayan, J. Stability of electron field emission in Q-carbon. MRS Commun. 2018, 8, 1343–1351. [Google Scholar] [CrossRef]
- Narayan, J.; Bhaumik, A.; Gupta, S.; Haque, A.; Sachan, R. Progress in Q-carbon and related materials with extraordinary properties. Mater. Res. Lett. 2018, 6, 353–364. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Fischer, T.H.; Almlof, J. General methods for geometry and wave function optimization. J. Phys. Chem. 1992, 96, 9768. [Google Scholar] [CrossRef]
- Clark, S.J.; Segall, M.D.; Pickard, C.J.; Hasnip, P.J.; Probert, M.I.J.; Refson, K.; Payne, M.C. First principles methods using CASTEP. Z. für Krist. 2005, 220, 567–570. [Google Scholar] [CrossRef]
- Hohenberg, P.; Kohn, W. Inhomogeneous electron gas. Phys. Rev. B 1964, 136, 864–871. [Google Scholar] [CrossRef]
- Kohn, W.; Sham, L.J. Self-consistent equations including exchange and correlation effects. Phys. Rev. A 1965, 140, 1133–1138. [Google Scholar] [CrossRef]
- Vanderbilt, D. Soft self-consistent pseudopotentials in a generalized eigenvalue formalism. Phys. Rev. B 1990, 41, 7892–7895. [Google Scholar] [CrossRef] [PubMed]
- Monkhorst, H.J.; Pack, J.D. Special points for Brillouin-zone integrations. Phys. Rev. B 1976, 13, 5188–5192. [Google Scholar] [CrossRef]
- Parr, R.G.; Yang, W.T. Density functional-approach to the frontier-electron theory of chemical-reactivity. J. Am. Chem. Soc. 1984, 106, 4049–4050. [Google Scholar] [CrossRef]
- Yang, W.; Parr, R.G.; Pucci, R. Electron-density, Kohn-Sham Frontier Orbitals, and Fukui Functions. J. Chem. Phys. 1984, 81, 2862–2863. [Google Scholar] [CrossRef]
- Mulliken, R.S. Electronic population analysis on LCAO–MO molecular wave functions. I. J. Chem. Phys. 1955, 23, 1833–1840. [Google Scholar] [CrossRef]
- QuantumWise Atomistix ToolKit (ATK) with Virtual NanoLab, Version January 2018. Available online: https://www.synopsys.com/silicon/quantumatk.html (accessed on 1 January 2019).
- Ferreira, L.G.; Marques, M.; Teles, L.K. Approximation to density functional theory for the calculation of band gaps of semiconductors. Phys. Rev. B 2008, 78, 125116. [Google Scholar] [CrossRef]
- Delley, B. An all-electron numerical method for solving the local density functional for polyatomic molecules. J. Chem. Phys. 1990, 92, 508–517. [Google Scholar] [CrossRef]
- Delley, B. From molecules to solids with the DMol3 approach. J. Chem. Phys. 2000, 113, 7756–7764. [Google Scholar] [CrossRef]
- Tian, Y.; Larsson, K. Theoretical studies of size effects on surface chemical properties for nanoscale diamond particles. J. Phys. Chem. C 2014, 118, 26061. [Google Scholar]
- Larsson, K. Novel Aspects of Diamond. The Effect of Dopants on Diamond Surface Properties and Growth; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Zou, Y.; Larsson, F.; Larsson, K. Effect of CVD diamond growth by doping with nitrogen. Theor. Chem. Accounts 2014, 133, 1432. [Google Scholar]
- Zou, Y.; Larsson, K. Effect of boron doping on the CVD growth rate of diamond. J. Phys. Chem. C 2016, 120, 10658. [Google Scholar] [CrossRef]
- May, P.W.; Stone, J.C.; Ashfold, M.N.R.; Hallam, K.R.; Wang, W.N.; Fox, N.A. The effect of diamond surface termination species upon field emission properties. Diam. Relat. Mater. 1998, 7, 671–676. [Google Scholar] [CrossRef]
- Mackey, B.L.; Russell, J.N., Jr.; Crowell, J.E.; Butler, J.E. Effect of surface termination on the electrical conductivity and broad-band internal infrared reflectance of a diamond (110) surface. Phys. Rev. B 1995, 52, R17009–R17012. [Google Scholar] [CrossRef]
- Martin, H.B. Hydrogen and oxygen evolution on boron-doped diamond electrodes. J. Electrochem. Soc. 1996, 143, L133–L136. [Google Scholar] [CrossRef]
- Nebel, C.E.; Ertl, F.; Sauerer, C.; Stutzmann, M.; Graeff, C.F.O.; Bergonzo, P.; Williams, O.A.; Jackman, R.B. Low temperature properties of the p-type surface conductivity of diamond. Diam. Relat. Mater. 2002, 11, 351–354. [Google Scholar] [CrossRef]
- de Theije, F.K.; Roy, O.; van der Laag, N.J.; van Enckevort, W.J.P. Oxidative etching of diamond. Diam. Relat. Mater. 2000, 9, 929–934. [Google Scholar] [CrossRef]
- Wang, M.; Simon, N.; Decorse-Pascanut, C.; Bouttemy, M.; Etcheberry, A.; Li, M.; Boukherroub, R.; Szunerits, S. Comparison of the chemical composition of boron-doped diamond surfaces upon different oxidation processes. Electrochim. Acta 2009, 54, 5818–5824. [Google Scholar] [CrossRef]
- Ferro, S.; De Battisti, A. The 5-V window of polarizability of fluorinated diamond electrodes in aqueous solutions. Anal. Chem. 2003, 75, 7040–7042. [Google Scholar] [CrossRef]
- Kondo, T.; Ito, H.; Kusakabe, K.; Ohkawa, K.; Einaga, Y.; Fujishima, A.; Kawai, T. Plasma etching treatment for surface modification of boron-doped diamond electrodes. Electrochim. Acta. 2007, 52, 3841–3848. [Google Scholar] [CrossRef]
- Song, K.-S.; Degawa, M.; Nakamura, Y.; Kanazawa, H.; Umezawa, H.; Kawarada, H. Surface-modified diamond field-effect transistors for enzyme-immobilized biosensors. Jpn. J. Appl. Phys. 2004, 43, L814–L817. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Auciello, O.; Birrell, J.; Carlisle, J.A.; Curtiss, L.A.; Goyette, A.N.; Gruen, D.M.; Krauss, A.R.; Schlueter, J.; Sumant, A.; et al. Synthesis and characterization of highly-conducting nitrogen-doped ultrananocrystalline diamond films. Appl. Phys. Lett. 2001, 79, 1441–1443. [Google Scholar] [CrossRef]
- Rohrer, E.; Graeff, C.F.; Janssen, R.; Nebel, C.E.; Stutzmann, M.; Güttler, H.; Zachai, R. Nitrogen-related dopant and defect states in CVD diamond. Phys. Rev. B 1996, 54, 7874–7880. [Google Scholar] [CrossRef]
- Yu, B.D.; Miyamoto, Y.; Sugino, O. Efficient n-type doping of diamond using surfactant-mediated epitaxial growth. Appl. Phys. Lett. 2000, 76, 976–978. [Google Scholar] [CrossRef]
- Haase, A.; Peters, A.; Rosiwal, S. Growth and thermoelectric properties of nitrogen-doped diamond/graphite. Diam. Relat. Mater. 2016, 63, 222–226. [Google Scholar] [CrossRef]
- Haque, A.; Sumaiya, S. An overview on the formation and processing of nitrogen-vacancy photonic centers in diamond by ion implantation. J. Manuf. Mater. Proc. 2017, 1, 6. [Google Scholar] [CrossRef]
- Koizumi, S.; Kamo, M.; Sato, Y.; Ozaki, H.; Inuzuka, T. Growth and characterization of phosphorous doped {111} homoepitaxial diamond thin films. Appl. Phys. Lett. 1997, 71, 1065–1067. [Google Scholar] [CrossRef]
- Kato, H.; Yamasaki, S.; Okushi, H. Growth and characterization of phosphorus-doped diamond using organophosphorus gases. Phys. Stat. Sol. (a) 2005, 202, 2122–2128. [Google Scholar] [CrossRef]
- Frangieh, G.; Jomard, F.; Pinault, M.A. Influence of tertiarybutylphosphine (TBP) addition on the CVD growth of diamond. Phys. Stat. Sol. (a) 2009, 206, 1996–1999. [Google Scholar] [CrossRef]
- Haubner, R. Comparison of sulfur, boron, nitrogen and phosphorus additions during low-pressure diamond deposition. Diam. Relat. Mater. 2005, 14, 355–363. [Google Scholar] [CrossRef]
- Ohtani, R.; Yamamoto, T.; Janssens, S.D. Large improvement of phosphorus incorporation efficiency in n-type chemical vapor deposition of diamond. Appl. Phys. Lett. 2014, 105, 232106. [Google Scholar] [CrossRef]
- Matsumoto, T.; Kato, H.; Makino, T.; Ogura, M.; Takeuchi, D.; Okushi, H.; Yamasaki, S. Carrier transport in homoepitaxial diamond films with heavy phosphorus doping. Jpn. J. Appl. Phys. 2014, 53, 05FP05. [Google Scholar] [CrossRef]
- Grotjohn, T.A.; Tran, D.T.; Yaran, M.K.; Demlow, S.N.; Schuelke, T. Heavy phosphorus doping by epitaxial growth on the (111) diamond surface. Diam. Relat. Mater. 2014, 44, 129–133. [Google Scholar] [CrossRef]
- Hoshino, Y.; Kato, H.; Makino, T.; Ogura, M.; Iwasaki, T.; Hatano, M.; Yamasaki, S. Electrical properties of lateral p–n junction diodes fabricated by selective growth of n+ diamond. Phys. Stat. Sol. (a) 2012, 209, 1761–1764. [Google Scholar] [CrossRef]
- Makino, T.; Yoshino, K.; Sakai, N.; Uchida, K.; Koizumi, S.; Kato, H.; Takeuchi, D.; Ogura, M.; Oyama, K.; Matsumoto, T.; et al. Enhancement in emission efficiency of diamond deep-ultraviolet light emitting diode. Appl. Phys. Lett. 2011, 99, 061110. [Google Scholar] [CrossRef]
- Takeuchi, D.; Makino, T.; Kato, H.; Ogura, M.; Okushi, H.; Ohashi, H.; Yamasaki, S. High-voltage vacuum switch with a diamond p–i–n diode using negative electron affinity. Jpn. J. Appl. Phys. 2012, 51, 090113. [Google Scholar]
- Maier, F.; Ristein, J.; Ley, L. Electron affinity of plasma-hydrogenated and chemically oxidized diamond (100) surfaces. Phys. Rev. B 2001, 64, 165411. [Google Scholar] [CrossRef]
- Touhara, H.; Okino, F. Property control of carbon materials by fluorination. Carbon 2000, 38, 241–267. [Google Scholar] [CrossRef]
- Takeuchi, D.; Kato, H.; Ri, G.S.; Yamada, T.; Vinod, P.R.; Hwang, D.; Nebel, C.E.; Okushi, H.; Yamasaki, S. Direct observation of negative electron affinity in hydrogen-terminated diamond surfaces. Appl. Phys. Lett. 2005, 86, 152103. [Google Scholar] [CrossRef]
- McCreery, R.L. Advanced carbon electrode materials for molecular electrochemistry. Chem. Rev. 2008, 108, 2646–2687. [Google Scholar] [CrossRef]
- Pleskov, Y.V. Photoelectrochemical Properties of semiconductor diamond. J. Electroanalyt. Chem. 1987, 228, 19–27. [Google Scholar] [CrossRef]
- Swain, G.; Ramesham, R. The electrochemical activity of boron-doped polycrystalline diamond thin-film electrodes. Anal. Chem. 1993, 65, 345–351. [Google Scholar] [CrossRef]
- Williams, A.; Lightowl, E.; Collins, E. Impurity conduction in synthetic semiconducting diamond. J. Phys. Part C Solid State Phys. 1970, 3, 1727. [Google Scholar] [CrossRef]
- Chrenko, R. Boron, dominant acceptor in semiconducting diamond. Phys. Rev. B 1973, 7, 4560–4567. [Google Scholar] [CrossRef]
- Li, H.D. Investigation on crystalline structure, boron distribution, and residual stresses in freestanding boron-doped CVD diamond films. J. Cryst. Growth 2010, 312, 1986–1991. [Google Scholar] [CrossRef]
- Butler, J.E.; Vikharev, A.; Gorbachev, A.; Lobaev, M.; Muchnikov, A.; Radischev, D.; Isaev, V.; Chernov, V.; Bogdanov, S.; Drozdov, M.; et al. Nanometrical diamond delta doping with boron. Phys. Stat. Sol. 2017, 11, 1600329–1600333. [Google Scholar]
- Sakai, T.; Ono, T.; Sakuma, N.; Yoshida, H.; Suzuki, M.; Takeuchi, D.; Kono, S.; Yamasaki, S. Observation of negative electron affinity in low-voltage discharging boron-doped polycrystalline diamond. Jpn. J. Appl. Phys. 2014, 53, 05FP09. [Google Scholar] [CrossRef]
- Müller-Sebert, W.; Wörner, E.; Fuchs, F.; Wild, C.; Koidl, P. Nitrogen induced increase of growth rate in chemical vapor deposition of diamond. Appl. Phys. Lett. 1996, 68, 759–760. [Google Scholar] [CrossRef]
- Liu, T.; Raabe, D. Influence of nitrogen doping on growth rate and texture evolution of chemical vapor deposition diamond films. Appl. Phys. Lett. 2009, 94, 211191–211193. [Google Scholar] [CrossRef]
- Dunst, S.; Sternschulte, H.; Schreck, M. Growth rate enhancement by nitrogen in diamond chemical vapor deposition—A catalytic effect. Appl. Phys. Lett. 2009, 94, 224101–224103. [Google Scholar] [CrossRef]
- Bohr, S.; Haubner, R.; Lux, B. Influence of phosphorus addition on diamond CVD. Diam. Relat. Mater. 1995, 4, 133–144. [Google Scholar] [CrossRef]
- Kalish, R. The search for donors in diamond. Diam. Relat. Mater. 2001, 10, 1749–1755. [Google Scholar] [CrossRef]
- Kajihara, S.A.; Antonelli, A.; Bernholc, J.; Car, R. Nitrogen and potential n-type dopants in diamond. Phys. Rev. Lett. 1991, 66, 21101. [Google Scholar] [CrossRef]
- Jiang, N.; Hatta, A.; Ito, T. Nitrogen Doping Effects on Electrical Properties of Diamond Films. Jpn. J. Appl. Phys. 1998, 37, L1175–L1177. [Google Scholar] [CrossRef]
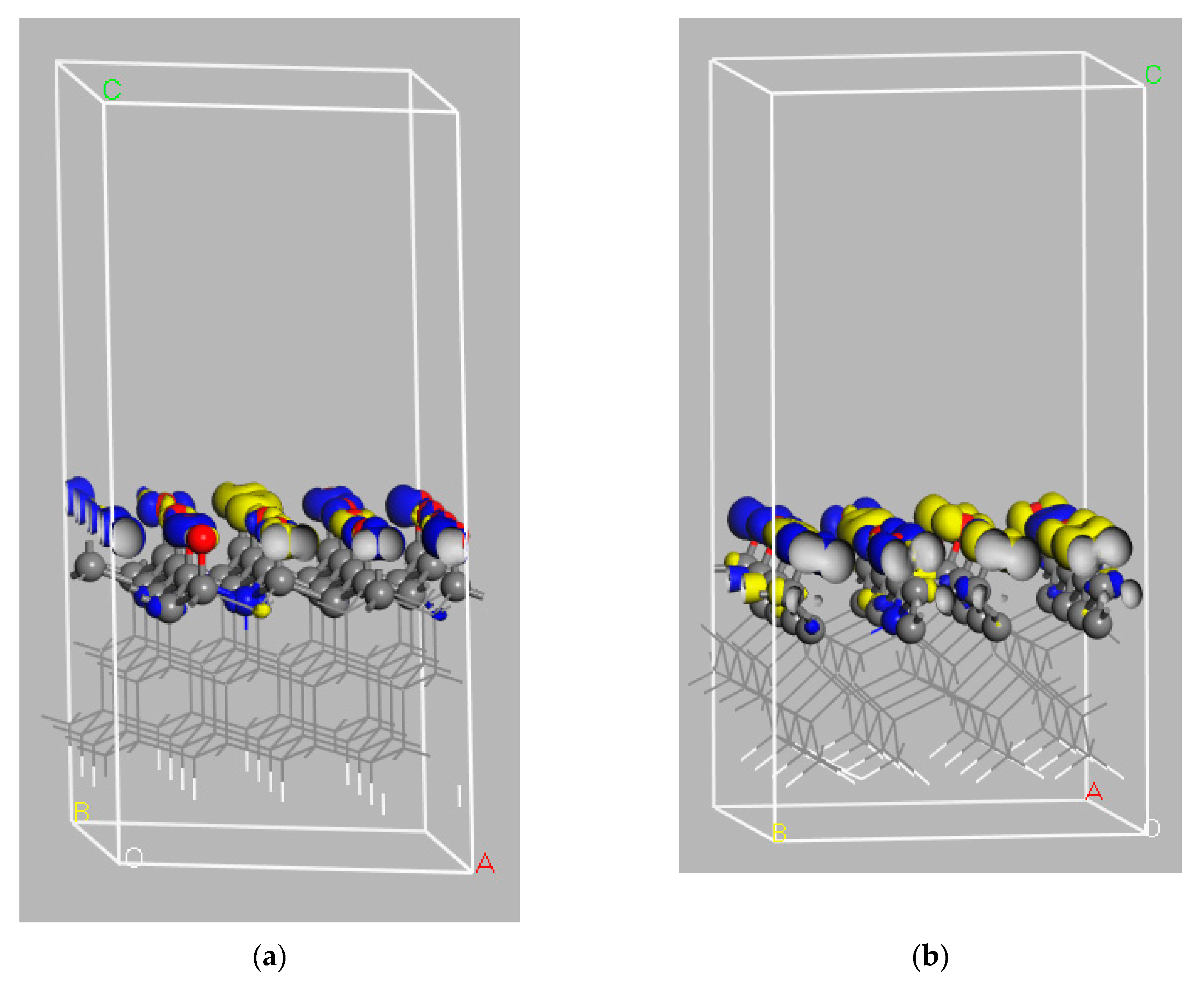
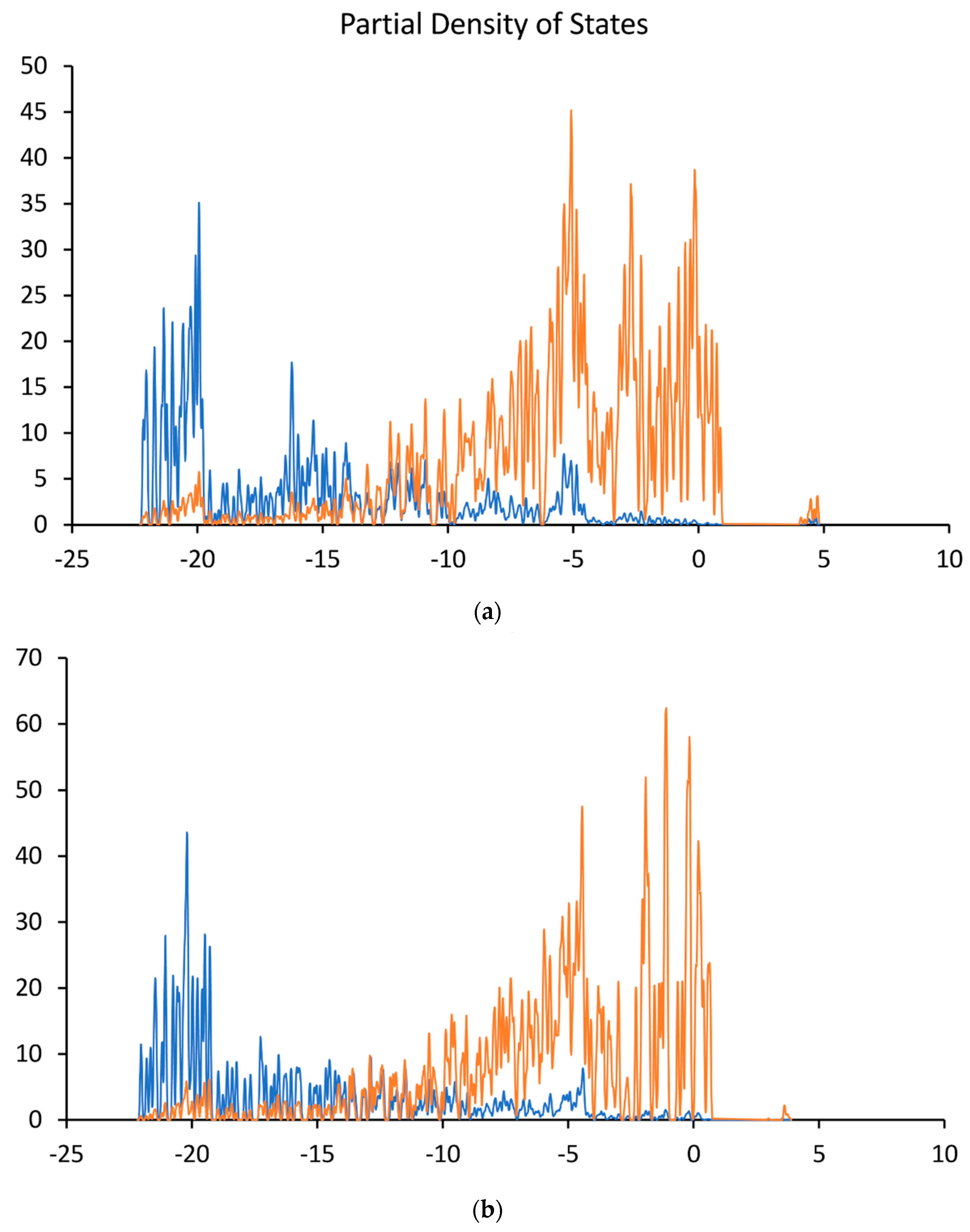
| (111) | Valence Band Maximum (VBM) | Conduction Band Minimum (CBM) | Bandgap (BG) | Electron Affinity (EA) | Work Function (Φ) |
|---|---|---|---|---|---|
| Non-doping | |||||
| H | −4.4 | +1.0 | 5.4 | −1.0 | 4.4 |
| F | −5.8 | −0.1 | 5.7 | +0.1 | 5.8 |
| OH | −4.3 | −0.3 | 4.6 | +0.3 | 4.3 |
| Oontop | −5.4 | −4.9 | 0.5 | +4.9 | 5.4 |
| NH2 | −2.8 | +0.7 | 3.4 | −0.7 | 2.8 |
| B-doping | |||||
| H | −3.2 | +0.3 | 3.5 | −0.3 | 3.2 |
| F | −5.3 | ~0 | 5.3 | ~0 | 5.3 |
| OH | −3.6 | 0.9 | 4.5 | −0.9 | 3.6 |
| Oontop | −5.4 | −5.0 | 0.4 | +5.0 | 5.4 |
| NH2 | −2.7 | −1.8 | 0.9 | +1.8 | 2.7 |
| N-doping | |||||
| H | −4.6 | −4.3 | 0.3 | +4.3 | 4.6 |
| F | −5.9 | −5.6 | 0.3 | +5.6 | 5.9 |
| OH | −4.6 | −4.2 | 0.5 | +4.2 | 4.6 |
| Oontop | −5.4 | −5.0 | 0.3 | +5.0 | 5.4 |
| NH2 | −3.1 | −2.7 | 0.4 | −2.7 | 3.1 |
| P-doping | |||||
| H | −4.9 | −4.2 | 0.6 | +4.2 | 4.9 |
| F | −6.1 | −5.1 | 1.0 | +5.1 | 6.1 |
| OH | −4.8 | −4.3 | 0.5 | +4.3 | 4.8 |
| Oontop | −5.4 | −5.0 | 0.4 | +5.0 | 5.4 |
| NH2 | −3.7 | −3.4 | 0.3 | +3.4 | 3.7 |
| (100) | Valence Band Maximum (VBM) | Conduction Band Minim (CBM) | Band Gap (BG) | Electron Affinity (EA) | Work Function (Φ) |
|---|---|---|---|---|---|
| Non-doping | |||||
| H | −3.9 | +0.9 | 4.8 | −0.9 | 3.9 |
| F | −4.8 | −1.8 | 3.0 | +1.8 | 4.8 |
| OH | −4.9 | +0.4 | 5.3 | −0.4 | 4.9 |
| Oontop | −5.0 | −4.2 | 0.8 | +4.2 | 5.0 |
| Obridge | −4.7 | −0.5 | 4.2 | +0.5 | 4.7 |
| NH2 | −3.7 | +0.8 | 4.5 | −0.8 | 3.7 |
| B-doping | |||||
| H | −3.4 | +0.6 | 4.0 | −0.6 | 3.4 |
| F | −5.0 | −0.7 | 4.3 | +0.7 | 5.0 |
| OH | −4.0 | +0.1 | 4.1 | −0.1 | 4.0 |
| Oontop | −5.0 | −4.7 | 0.3 | +4.7 | 5.0 |
| Obridge | −4.7 | −1.5 | 3.2 | +1.5 | 4.7 |
| NH2 | −3.2 | +0.8 | 4.0 | −0.8 | 3.2 |
| N-doping | |||||
| H | −3.7 | −3.5 | 0.2 | +3.5 | 3.7 |
| F | −5.3 | −5.0 | 0.2 | +5.0 | 5.3 |
| OH | −4.2 | −3.9 | 0.2 | +3.9 | 4.2 |
| Oontop | −5.3 | −5.0 | 0.3 | +5.0 | 5.1 |
| Obridge | −3.7 | −1.3 | 2.4 | +1.3 | 3.7 |
| NH2 | −3.8 | −3.7 | 0.1 | −3.7 | 3.8 |
| P-doping | |||||
| H | −4.0 | −2.9 | 1.1 | +2.9 | 4.0 |
| F | −5.5 | −4.3 | 1.2 | +4.3 | 5.5 |
| OH | −4.5 | −4.2 | 0.3 | +4.2 | 4.5 |
| Oontop | −5.3 | −5.2 | 0.1 | +5.2 | 5.3 |
| Obridge | −4.0 | −1.9 | 2.1 | +1.9 | 4.0 |
| NH2 | −4.1 | −4.0 | 0.9 | +4.0 | 4.1 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larsson, K. The Combined Influence of Dopant Species and Surface Termination on the Electronic Properties of Diamond Surfaces. C 2020, 6, 22. https://doi.org/10.3390/c6020022
Larsson K. The Combined Influence of Dopant Species and Surface Termination on the Electronic Properties of Diamond Surfaces. C. 2020; 6(2):22. https://doi.org/10.3390/c6020022
Chicago/Turabian StyleLarsson, Karin. 2020. "The Combined Influence of Dopant Species and Surface Termination on the Electronic Properties of Diamond Surfaces" C 6, no. 2: 22. https://doi.org/10.3390/c6020022
APA StyleLarsson, K. (2020). The Combined Influence of Dopant Species and Surface Termination on the Electronic Properties of Diamond Surfaces. C, 6(2), 22. https://doi.org/10.3390/c6020022






