Macadamia Nutshell Biochar for Nitrate Removal: Effect of Biochar Preparation and Process Parameters
Abstract
1. Introduction
2. Materials and Methods
2.1. Solution Preparation
2.2. MBC Preparation
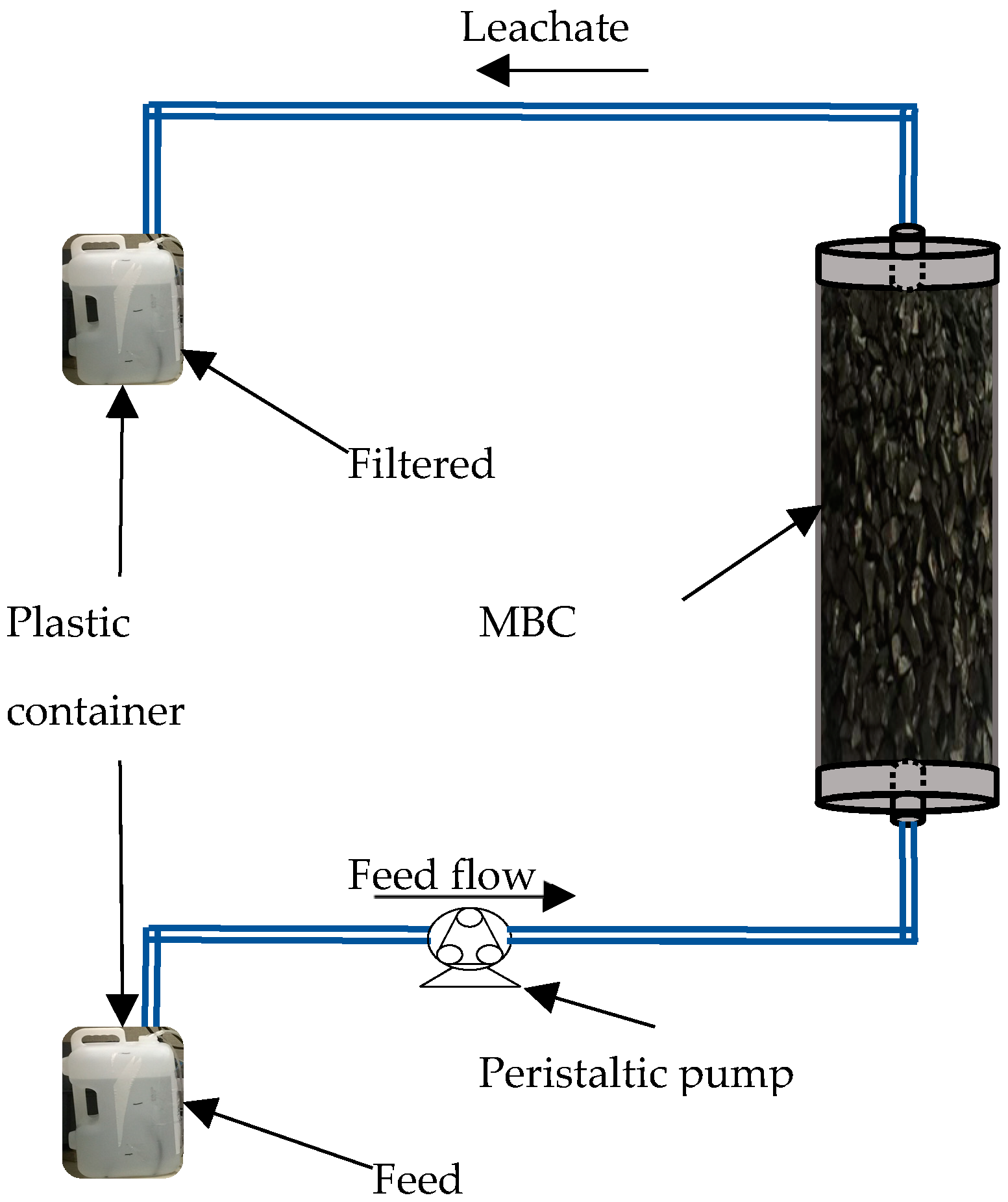
2.3. Column Preparation
2.4. Analytical Measurements
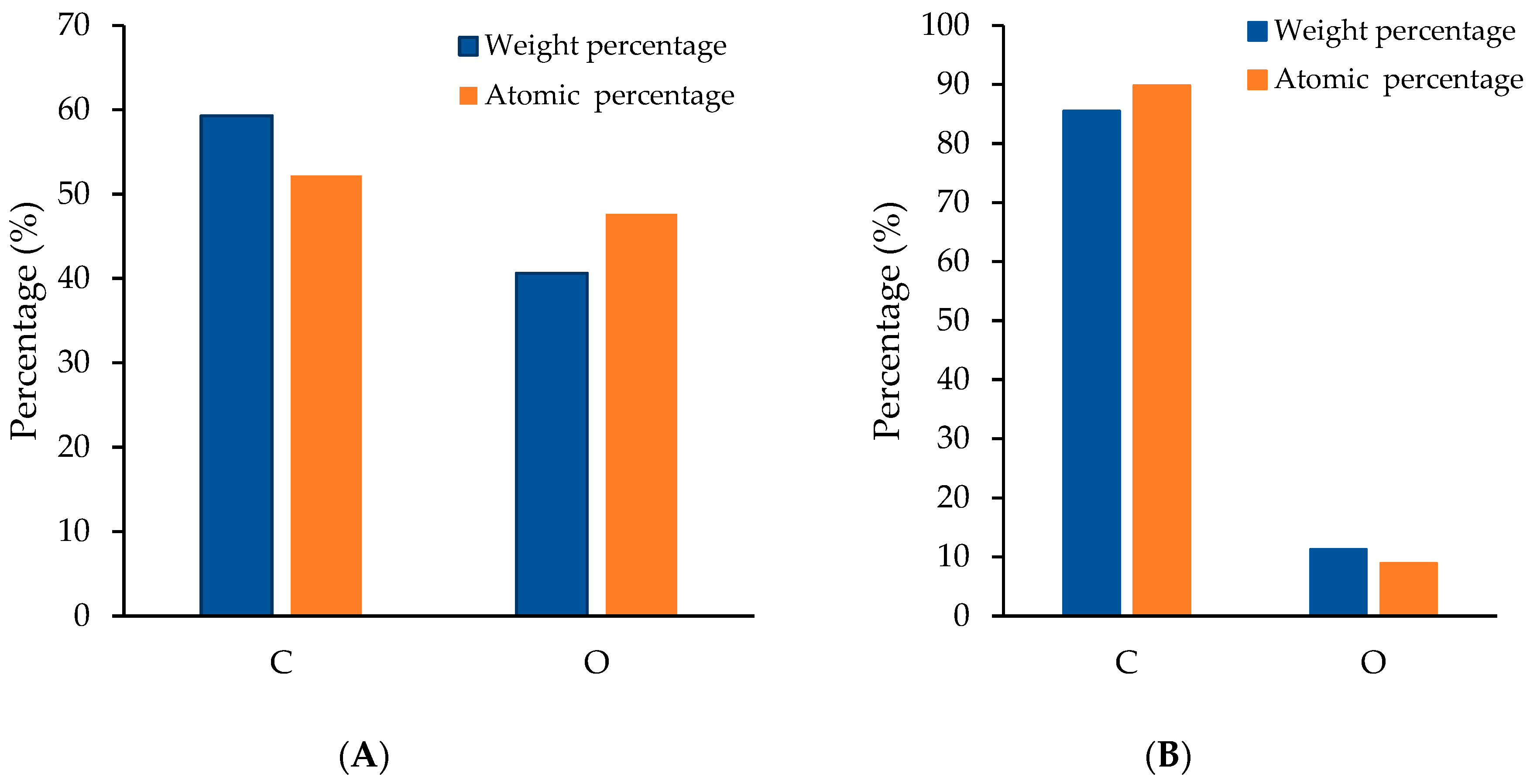
2.4.1. Morphology and Elemental Composition
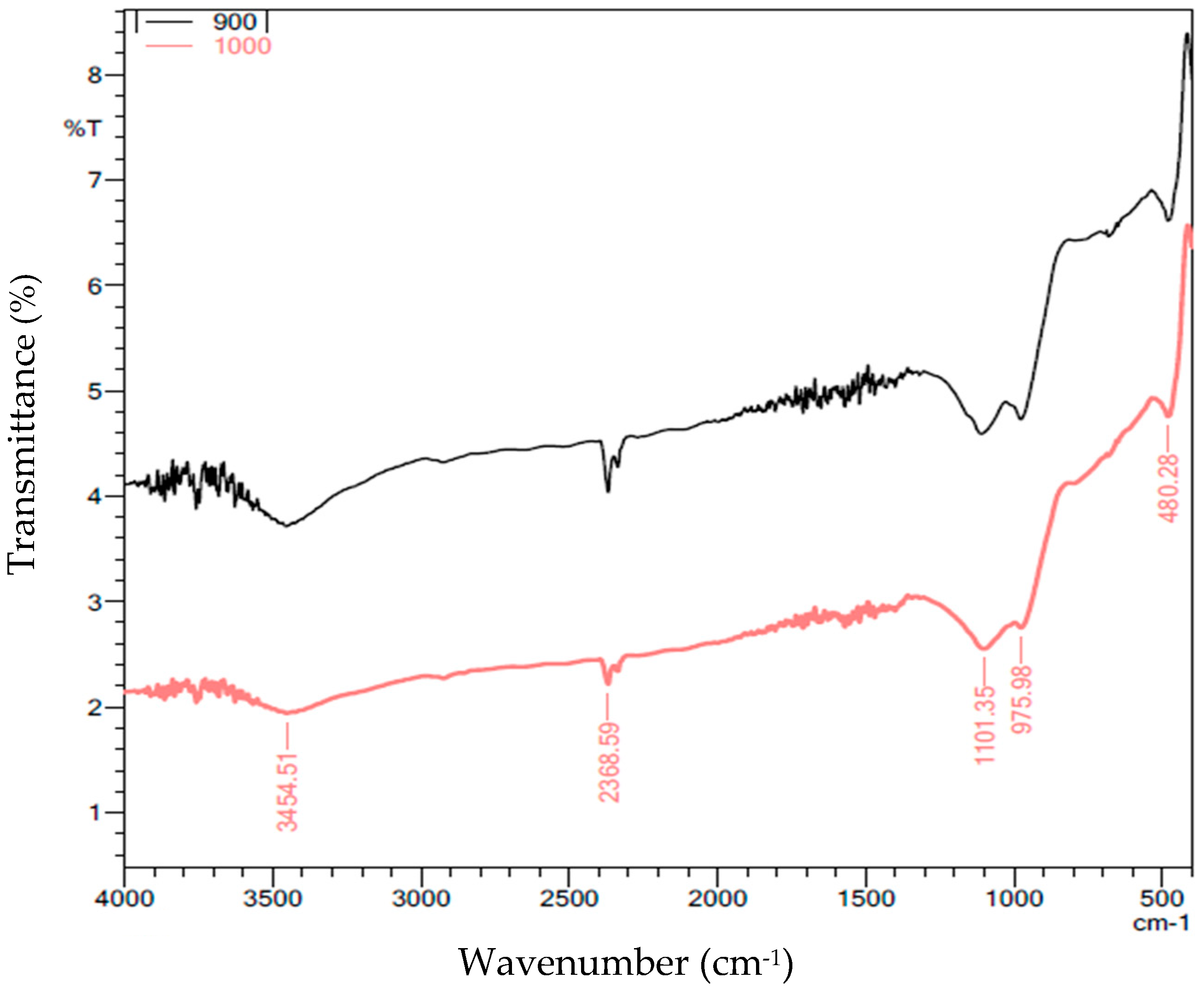
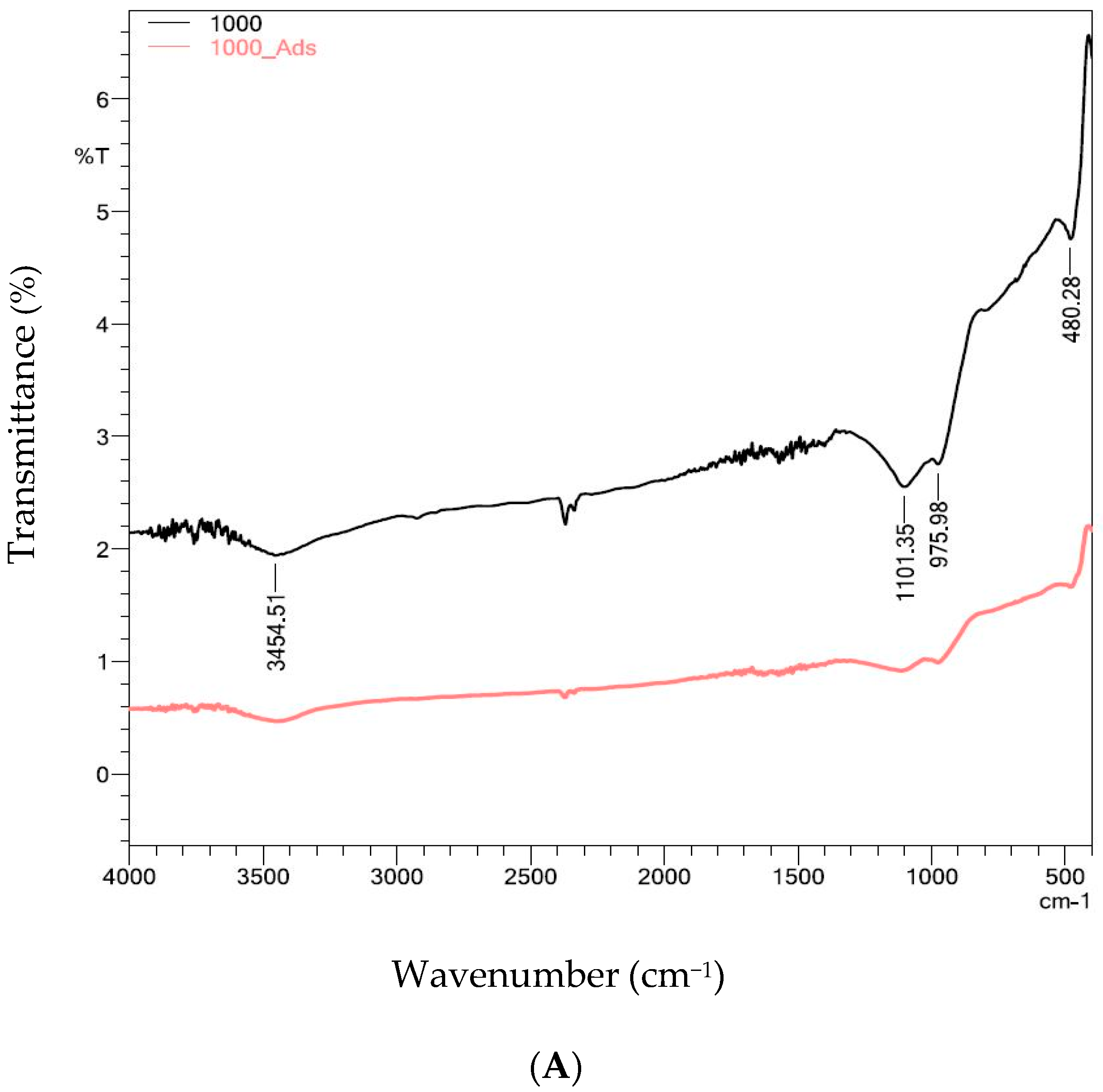
2.4.2. Functional Groups
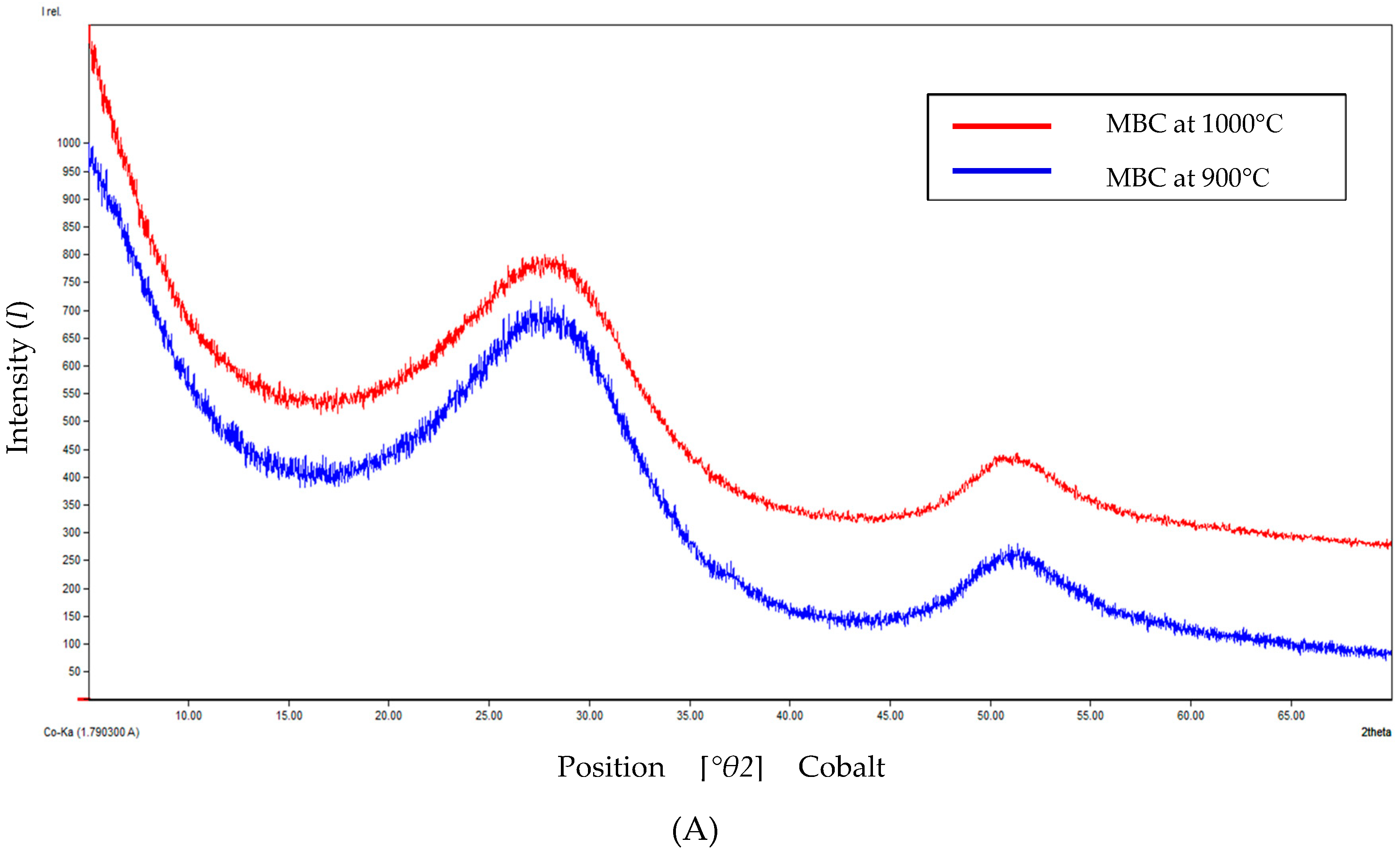
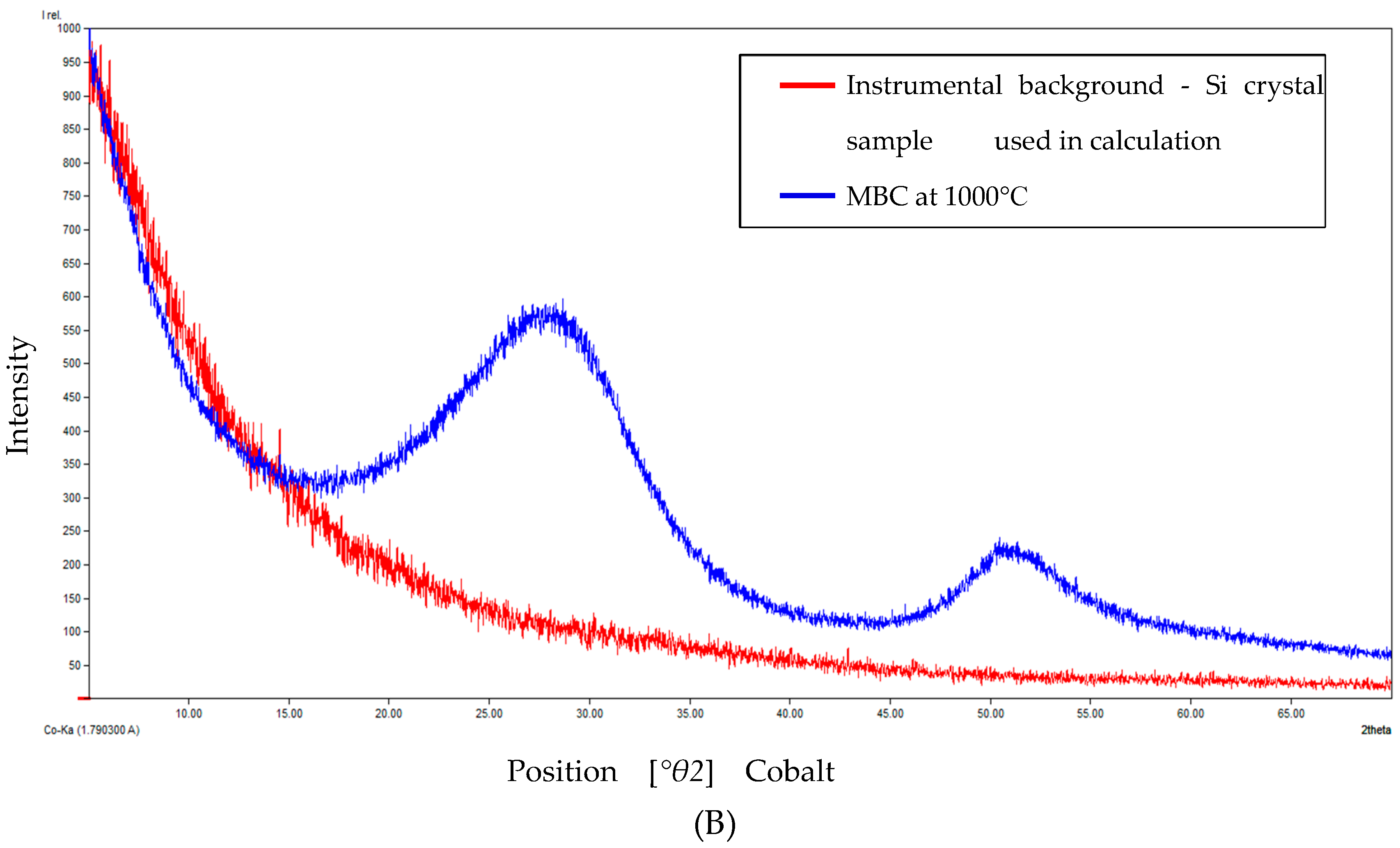
2.4.3. X-ray Diffraction Measurements
2.4.4. Elemental Composition
2.4.5. Nitrate Measurements
2.5. Kinetic Experiments
2.6. Column Experiments
2.7. Statistical Analyses
3. Results and Discussion
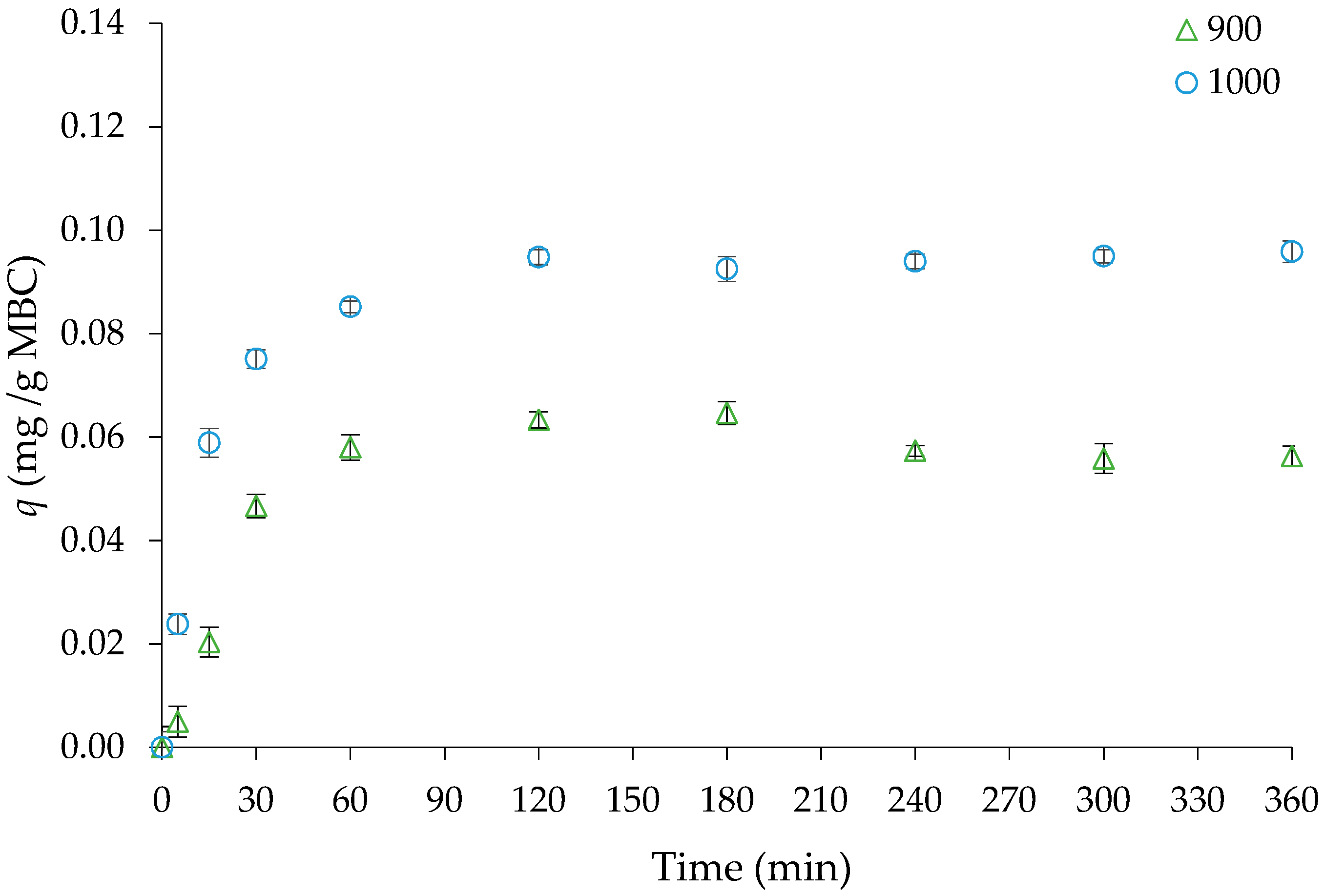
3.1. Kinetic Experiments—Effect of Pyrolysis Temperature
3.2. Column Experiments
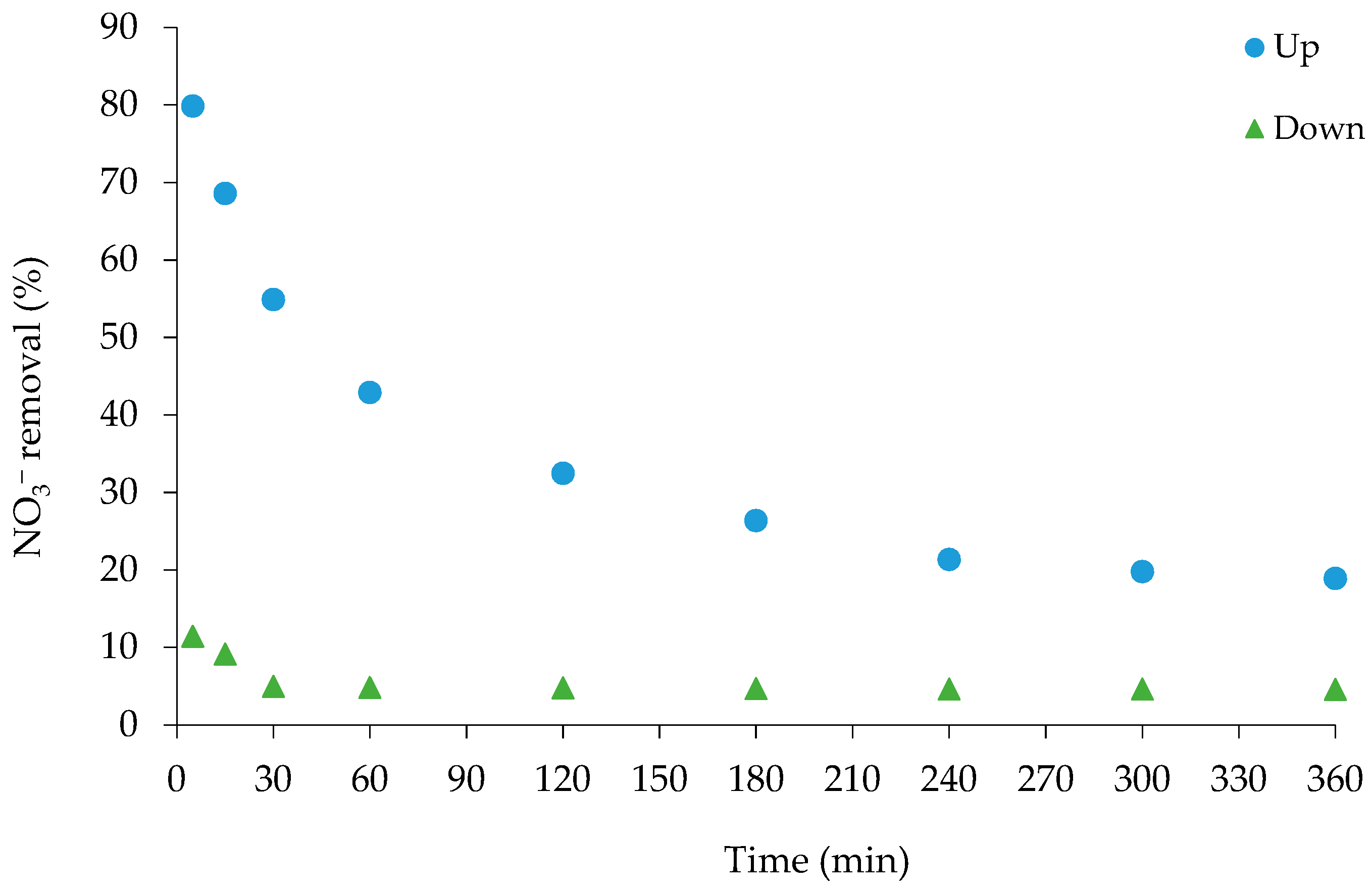
3.2.1. Preliminary Testing for Effect of Flow Direction on Nitrate Adsorption
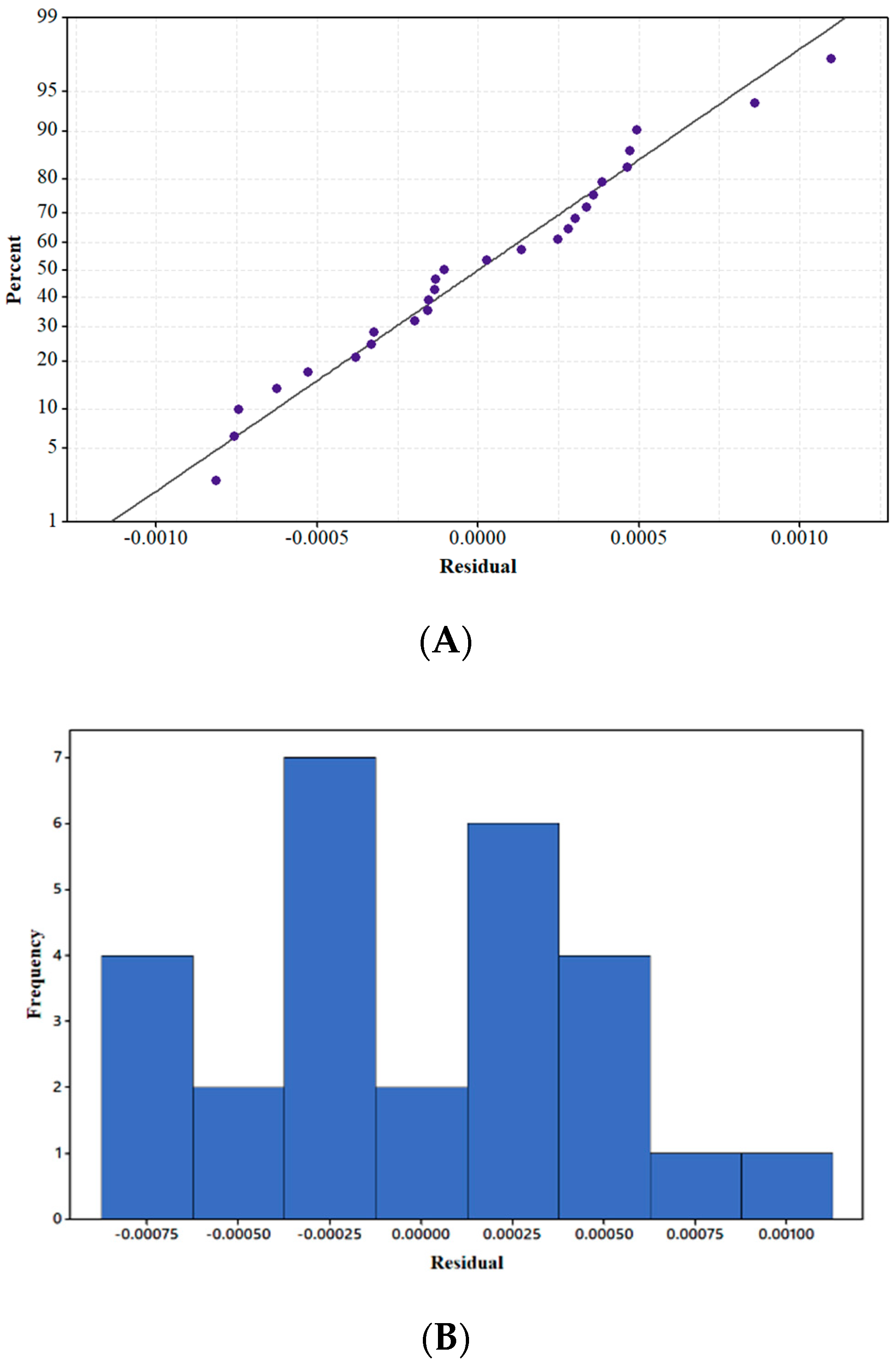
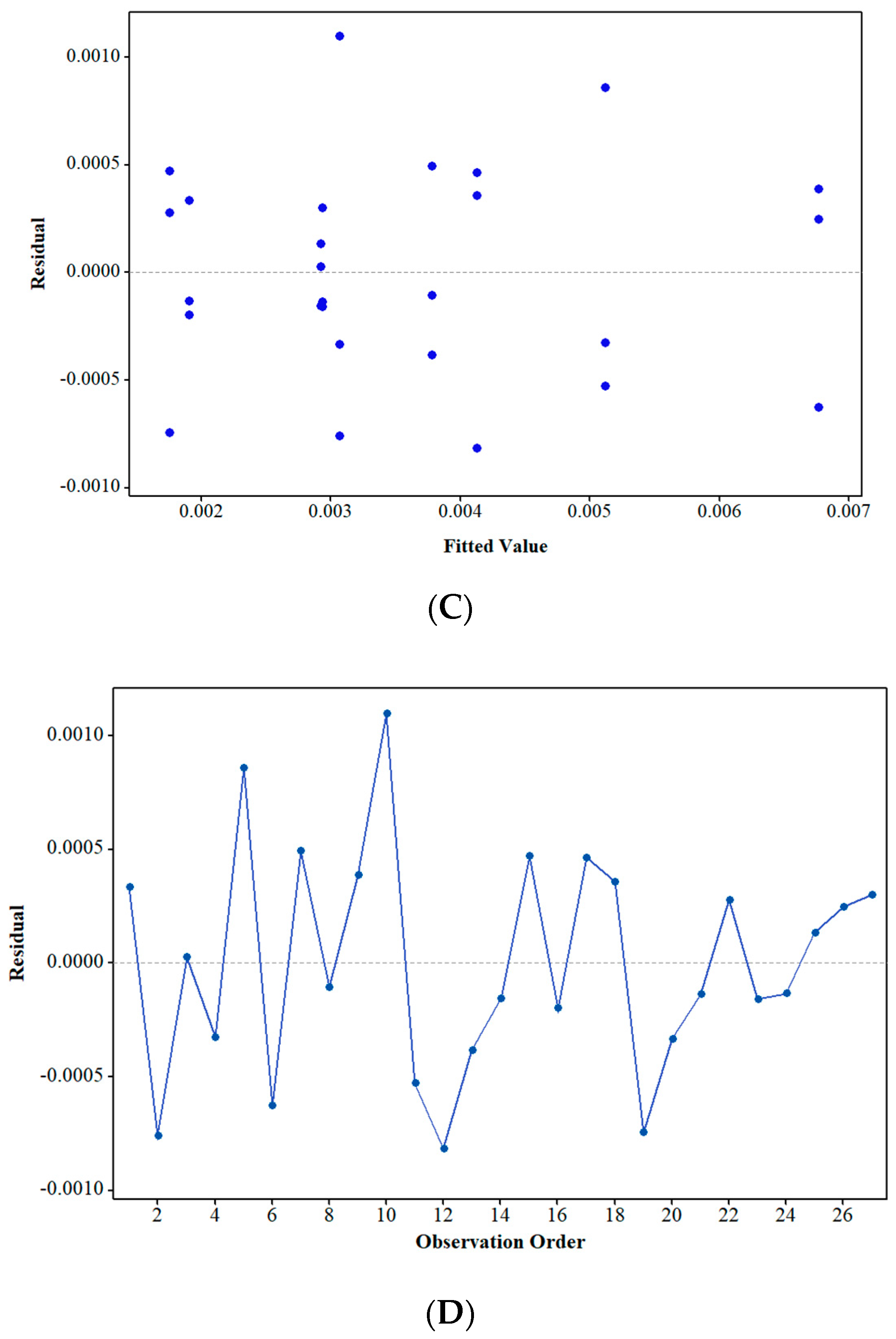
3.2.2. Normality and Residual Analyses
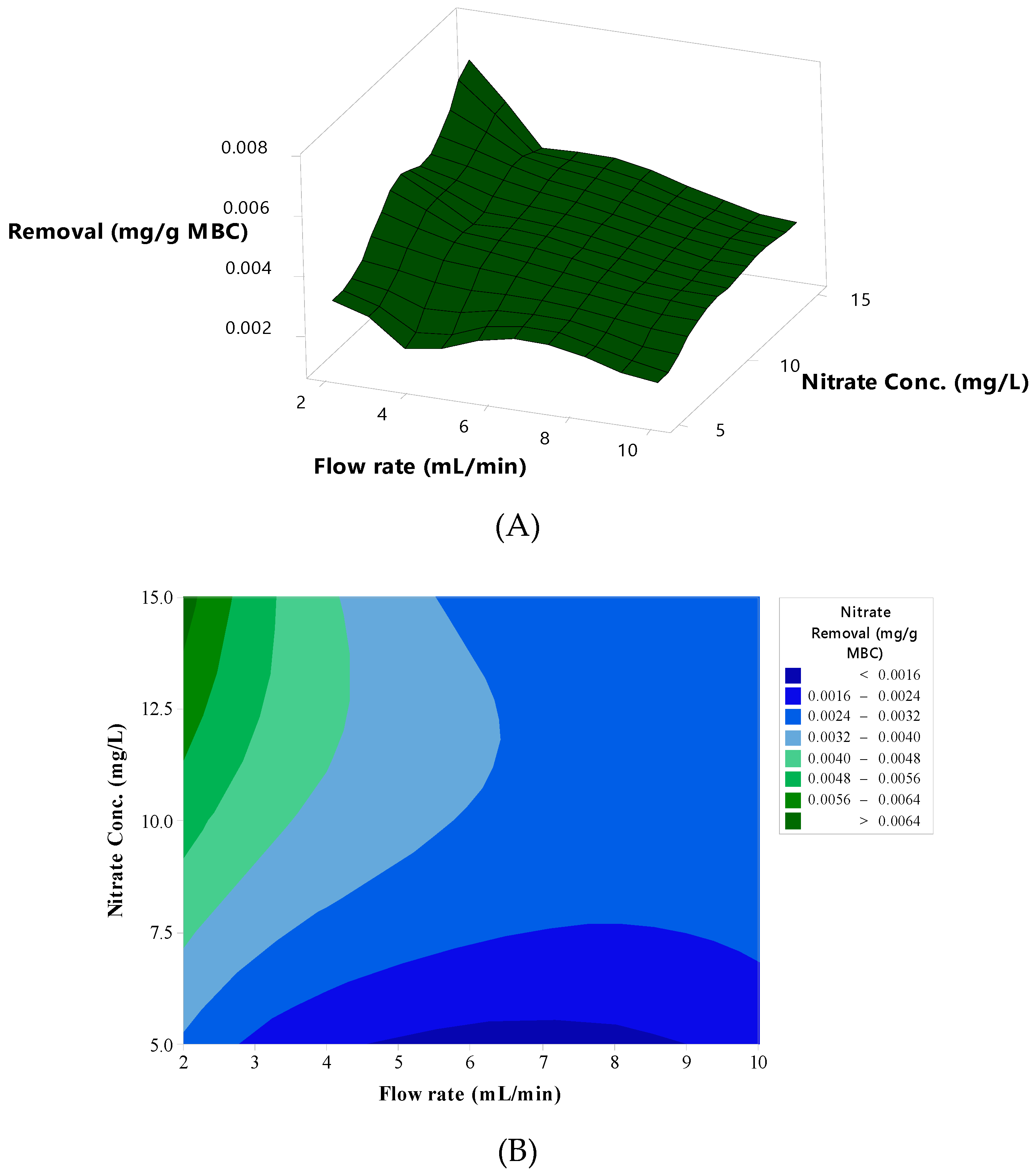
3.2.3. Surface and Contour Plots Analyses
3.2.4. Effective Operation Conditions for Column
4. Conclusions and Recommendations
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kleinman, P.J.A.; Sharpley, A.N.; Moyer, B.G.; Elwinger, G.F. Effect of Mineral and Manure Phosphorus Sources on Runoff Phosphorus. J. Environ. Qual. 2002, 31, 2026–2033. [Google Scholar] [CrossRef] [PubMed]
- Beman, J.M.; Arrigo, K.R.; Matson, P.A. Agricultural runoff fuels large phytoplankton blooms in vulnerable areas of the ocean. Nature 2005, 434, 211–214. [Google Scholar] [CrossRef] [PubMed]
- Hafshejani, L.D.; Hooshmand, A.; Naseri, A.A.; Mohammadi, A.S.; Abbasi, F.; Bhatnagar, A. Removal of nitrate from aqueous solution by modified sugarcane bagasse biochar. Ecol. Eng. 2016, 95, 101–111. [Google Scholar] [CrossRef]
- Chintala, R.; Mollinedo, J.; Schumacher, T.E.; Papiernik, S.K.; Malo, D.D.; Clay, D.E.; Kumar, S.; Gulbrandson, D.W. Nitrate sorption and desorption in biochars from fast pyrolysis. Microporous Mesoporous Mater. 2013, 179, 250–257. [Google Scholar] [CrossRef]
- Xu, X.; Gao, B.Y.; Yue, Q.Y.; Zhong, Q.Q. Preparation of agricultural by-product based anion exchanger and its utilization for nitrate and phosphate removal. Bioresour. Technol. 2010, 101, 8558–8564. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Gao, B.; Yao, Y.; Xue, Y.; Inyang, M. Synthesis of porous MgO-biochar nanocomposites for removal of phosphate and nitrate from aqueous solutions. Chem. Eng. J. 2012, 210, 26–32. [Google Scholar] [CrossRef]
- Water_Online. About Nutrient Removal. 2017. Available online: https://www.wateronline.com/solution/nutrient-removal (accessed on 15 July 2019).
- Good, A.G.; Beatty, P.H. Fertilizing Nature: A Tragedy of Excess in the Commons. PLoS Biol. 2011, 9, e1001124. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, A.; Kumar, E.; Sillanpää, M. Nitrate removal from water by nano-alumina: Characterization and sorption studies. Chem. Eng. J. 2010, 163, 317–323. [Google Scholar] [CrossRef]
- Olgun, A.; Atar, N.; Wang, S. Batch and column studies of phosphate and nitrate adsorption on waste solids containing boron impurity. Chem. Eng. J. 2013, 222, 108–119. [Google Scholar] [CrossRef]
- Plant Health Australia. Macadamias. 2012. Available online: http://www.planthealthaustralia.com.au/about-us/ (accessed on 29 June 2019).
- Fan, F.; Yang, Z.; Li, H.; Shi, Z.; Kan, H. Preparation and properties of hydrochars from macadamia nut shell via hydrothermal carbonization. R. Soc. Open Sci. 2018, 5, 181126. [Google Scholar] [CrossRef]
- Bada, S.; Falcon, R.; Falcon, L.; Makhula, M. Thermogravimetric investigation of macadamia nut shell, coal, and anthracite in different combustion atmospheres. J. South. Afr. Inst. Min. Met. 2015, 115, 741–746. [Google Scholar] [CrossRef]
- Kumar, U.; Maroufi, S.; Rajarao, R.; Mayyas, M.; Mansuri, I.; Joshi, R.K.; Sahajwalla, V. Cleaner production of iron by using waste macadamia biomass as a carbon resource. J. Clean. Prod. 2017, 158, 218–224. [Google Scholar] [CrossRef]
- Wrobel-Tobiszewska, A.; Boersma, M.; Sargison, J.; Adams, P.; Singh, B.; Franks, S.; Birch, C.J.; Close, D.C. Nutrient changes in potting mix and Eucalyptus nitens leaf tissue under macadamia biochar amendments. J. For. Res. 2018, 29, 383–393. [Google Scholar] [CrossRef]
- Spokas, K.A.; Baker, J.M.; Reicosky, D.C. Ethylene: Potential key for biochar amendment impacts. Plant Soil 2010, 333, 443–452. [Google Scholar] [CrossRef]
- Roser, M.B.; Feyereisen, G.W.; Spokas, K.A.; Mulla, D.J.; Strock, J.S.; Gutknecht, J. Carbon Dosing Increases Nitrate Removal Rates in Denitrifying Bioreactors at Low-Temperature High-Flow Conditions. J. Environ. Qual. 2018, 47, 856–864. [Google Scholar] [CrossRef] [PubMed]
- Roser, M. Using Unique Carbon Source Combinations to Increase Nitrate and Phosphate Removal in Bioreactors. Master’s Thesis, University of Minnesota, Minneapolis, MN, USA, 2016. [Google Scholar]
- Yang, J.; Li, H.; Zhang, D.; Wu, M.; Pan, B. Limited role of biochars in nitrogen fixation through nitrate adsorption. Sci. Total Environ. 2017, 592, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Beutel, M.W.; Newton, C.D.; Brouillard, E.S.; Watts, R.J. Nitrate removal in surface-flow constructed wetlands treating dilute agricultural runoff in the lower Yakima Basin, Washington. Ecol. Eng. 2009, 35, 1538–1546. [Google Scholar] [CrossRef]
- Lang, M.; Li, P.; Yan, X. Runoff concentration and load of nitrogen and phosphorus from a residential area in an intensive agricultural watershed. Sci. Total Environ. 2013, 458, 238–245. [Google Scholar] [CrossRef]
- Ghane, E.; Ranaivoson, A.Z.; Feyereisen, G.W.; Rosen, C.J.; Moncrief, J.F. Comparison of Contaminant Transport in Agricultural Drainage Water and Urban Stormwater Runoff. PLoS ONE 2016, 11, e0167834. [Google Scholar] [CrossRef]
- Department of Primary Industries. Farm Water Quality and Treatment. 2014. Available online: https://www.dpi.nsw.gov.au/__data/assets/pdf_file/0013/164101/Farm-water-quality-and-treatment.pdf (accessed on 1 June 2019).
- Bae, J.S.; Su, S. Macadamia nut shell-derived carbon composites for post combustion CO2 capture. Int. J. Greenh. Gas Control 2013, 19, 174–182. [Google Scholar] [CrossRef]
- Nick, T. Benefits of Biochar and its Applications for Runoff Water Management: Literature Review and Laboratory Report. Bachelor Thesis, Novia University of Applied Sciences, Vaasa, Finland, 2018. [Google Scholar]
- Shackley, S.; Sohi, S.; Brownsort, P.; Carter, S.; Cook, J.; Cunningham, C.; Gaunt, J.; Hammond, J.; Ibarrola, R.; Mašek, O. An Assessment of the Benefits and Issues Associated with the Application of Biochar to Soil. In A Report Commissioned by United Kingdom Department for Environment, Food and Rural Affairs, and Department of Energy and Climate Change; 2010; p. 132. [Google Scholar]
- Hagemann, N.; Spokas, K.; Schmidt, H.-P.; Kägi, R.; Böhler, M.A.; Bucheli, T.D. Activated Carbon, Biochar and Charcoal: Linkages and Synergies across Pyrogenic Carbon’s ABCs. Water 2018, 10, 182. [Google Scholar] [CrossRef]
- Shen, Y.S.; Wang, S.L.; Huang, S.T.; Tzou, Y.M.; Huang, J.H. Biosorption of Cr(VI) by coconut coir: Spectroscopic investigation on the reaction mechanism of Cr(VI) with lignocellulosic material. J. Hazard. Mater. 2010, 179, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Coates, J. Interpretation of infrared spectra, a practical approach. Encycl. Anal. Chem. 2000, 12, 10815–10837. [Google Scholar]
- Eaton, A.; Clesceri, E.W.; Rice, A.E; Greenberg, M.J.C.E.; Franson; APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater; APHA: Washington, DC, USA, 2005. [Google Scholar]
- Inyang, M.; Gao, B.; Wu, L.; Yao, Y.; Zhang, M.; Liu, L. Filtration of engineered nanoparticles in carbon-based fixed bed columns. Chem. Eng. J. 2013, 220, 221–227. [Google Scholar] [CrossRef]
- Zhao, H.; Xue, Y.; Long, L.; Hu, X. Adsorption of nitrate onto biochar derived from agricultural residuals. Water Sci. Technol. 2018, 77, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.; McKay, G. Pseudo-second order model for sorption processes. Process. Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Martins, A.C.; Pezoti, O.; Cazetta, A.L.; Bedin, K.C.; Yamazaki, D.A.; Bandoch, G.F.; Asefa, T.; Visentainer, J.V.; Almeida, V.C. Removal of tetracycline by NaOH-activated carbon produced from macadamia nut shells: Kinetic and equilibrium studies. Chem. Eng. J. 2015, 260, 291–299. [Google Scholar] [CrossRef]
- Mandal, S.; Thangarajan, R.; Bolan, N.S.; Sarkar, B.; Khan, N.; Ok, Y.S.; Naidu, R. Biochar-induced concomitant decrease in ammonia volatilization and increase in nitrogen use efficiency by wheat. Chemosphere 2016, 142, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Joseph, S.; Kammann, C.I.; Shepherd, J.G.; Conte, P.; Schmidt, H.-P.; Hagemann, N.; Rich, A.M.; Marjo, C.E.; Allen, J.; Munroe, P.; et al. Microstructural and associated chemical changes during the composting of a high temperature biochar: Mechanisms for nitrate, phosphate and other nutrient retention and release. Sci. Total Environ. 2018, 618, 1210–1223. [Google Scholar] [CrossRef]
- Sanford, J.; Larson, R.; Runge, T. Nitrate sorption to biochar following chemical oxidation. Sci. Total Environ. 2019, 669, 938–947. [Google Scholar] [CrossRef]
- Fidel, R.B.; Laird, D.A.; Spokas, K.A. Sorption of ammonium and nitrate to biochars is electrostatic and pH-dependent. Sci. Rep. 2018, 8, 17627. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, P.; Khoury, J.N. Reaction of phenol with nitrite ion: Pathways of formation of nitrophenols in environmental waters. Water Res. 2004, 38, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Cimo, G.; Haller, A.; Spokas, K.; Novak, J.; Ippolito, J.; Löhnertz, O.; Kammann, C. Mechanisms of Nitrate Capture in Biochar: Are they Related to Biochar Properties, Post-Treatment and Soil Environment? In Proceedings of the 19th EGU General Assembly Conference Abstracts, Vienna, Austria, 23–28 April 2017. [Google Scholar]
- Gai, X.; Wang, H.; Liu, J.; Zhai, L.; Liu, S.; Ren, T.; Liu, H. Effects of Feedstock and Pyrolysis Temperature on Biochar Adsorption of Ammonium and Nitrate. PLoS ONE 2014, 9, e113888. [Google Scholar] [CrossRef] [PubMed]
- Rafiq, M.K.; Bachmann, R.T.; Shang, Z.; Joseph, S.; Long, R. Influence of Pyrolysis Temperature on Physico-Chemical Properties of Corn Stover (Zea mays L.) Biochar and Feasibility for Carbon Capture and Energy Balance. PLoS ONE 2016, 11, e0156894. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.S.; Lee, S.M.; Lee, S.H.; Roh, J.S. X-ray diffraction analysis of the crystallinity of phenolic resin-derived carbon as a function of the heating rate during the carbonization process. Carbon Lett. 2018, 27, 108–111. [Google Scholar]
- Rajarao, R.; Mansuri, I.; Dhunna, R.; Khanna, R.; Sahajwalla, V. Study of structural evolution of chars during rapid pyrolysis of waste CDs at different temperatures. Fuel 2014, 134, 17–25. [Google Scholar] [CrossRef]
- Al-Juboori, R.A.; Yusaf, T.; Aravinthan, V.; Bowtell, L. Investigating natural organic carbon removal and structural alteration induced by pulsed ultrasound. Sci. Total Environ. 2016, 541, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- Ryan, B.F.; Joiner, B.L.; Cryer, J.D. MINITAB Handbook: Update for Release; Cengage Learning: Boston, MA, USA, 2012. [Google Scholar]
- Boubakri, A.; Helali, N.; Tlili, M.; Ben Amor, M. Fluoride removal from diluted solutions by Donnan dialysis using full factorial design. Korean J. Chem. Eng. 2014, 31, 461–466. [Google Scholar] [CrossRef]
- Mondal, M. Removal of Pb(II) ions from aqueous solution using activated tea waste: Adsorption on a fixed-bed column. J. Environ. Manag. 2009, 90, 3266–3271. [Google Scholar] [CrossRef]
- Xing, X.; Gao, B.Y.; Zhong, Q.Q.; Yue, Q.Y.; Li, Q. Sorption of nitrate onto amine-crosslinked wheat straw: Characteristics, column sorption and desorption properties. J. Hazard. Mater. 2011, 186, 206–211. [Google Scholar] [CrossRef]
- Karunarathne, H.; Amarasinghe, B. Fixed Bed Adsorption Column Studies for the Removal of Aqueous Phenol from Activated Carbon Prepared from Sugarcane Bagasse. Energy Procedia 2013, 34, 83–90. [Google Scholar] [CrossRef]

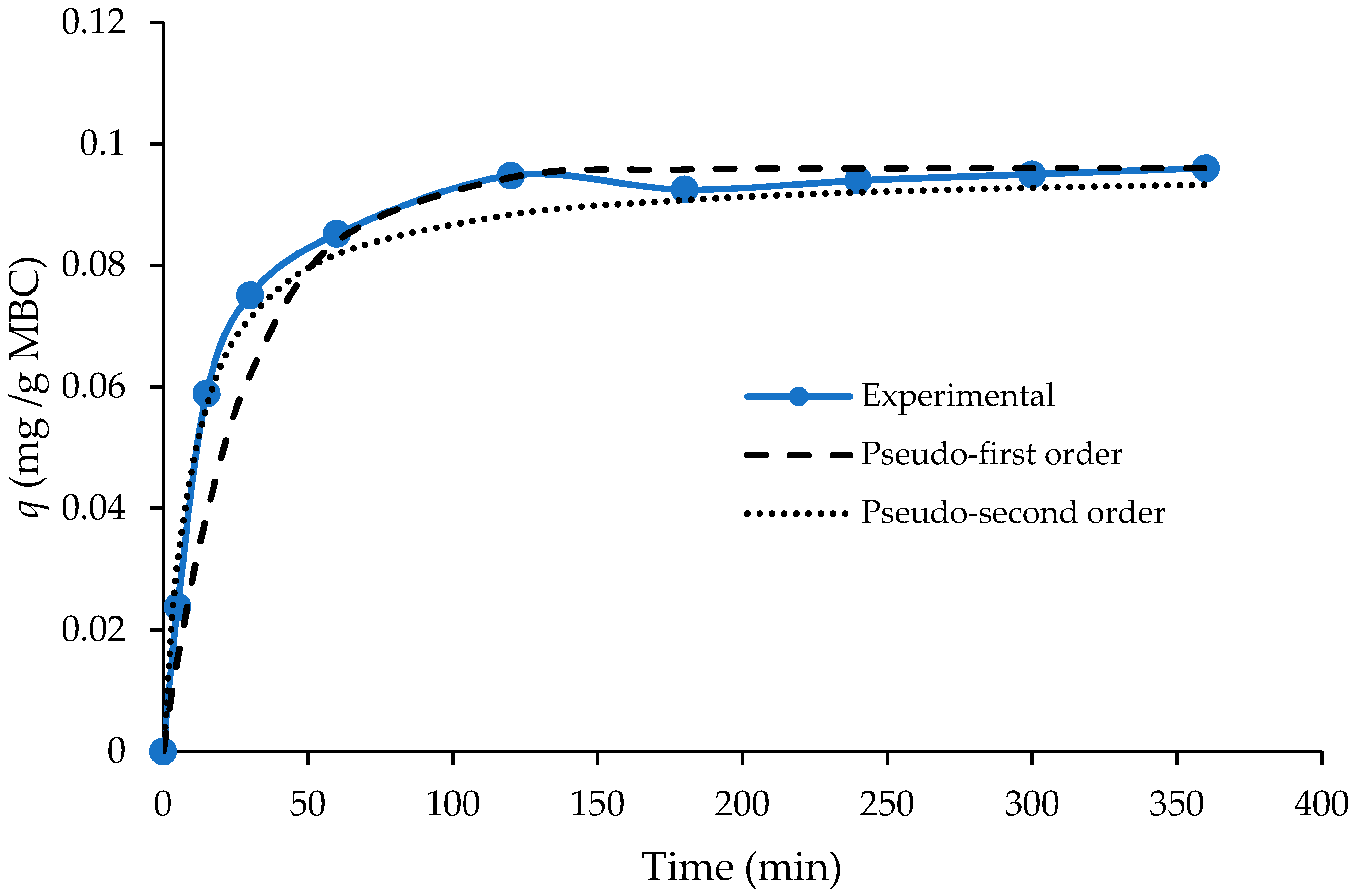

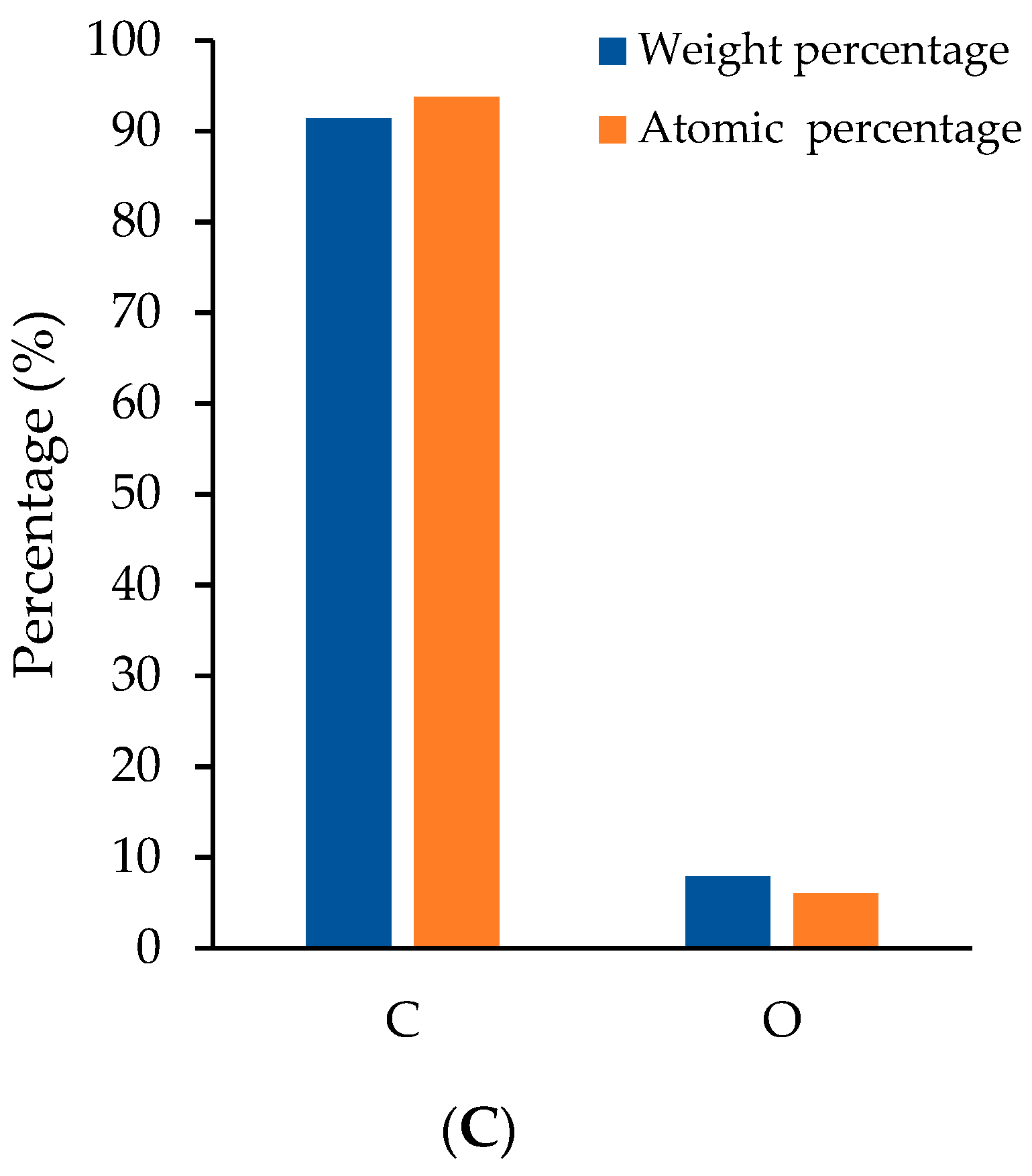

| Pseudo-First Order Model | Pseudo-Second Order Model | ||||
|---|---|---|---|---|---|
| qe (mg/g MBC) | K1 (min−1) | R2 | qe (mg/g MBC) | K2 (min−1) | R2 |
| 0.96 | 0.0328 | 0.92 | 0.93 | 1.004 | 0.98 |
| Temperature | Degree of Crystallinity (%) | Amorphous Content (%) |
|---|---|---|
| 900 °C | 18.07 | 81.93 |
| 1000 °C | 20.72 | 79.28 |
| No. | Flowrate (mL/min) | Nitrate Conc. (mg/L) | Nitrate Removal (%) | Removal NO3− mg/g MBC |
|---|---|---|---|---|
| 1 | 10 | 5 | 42.73 | 0.04 |
| 2 | 2 | 5 | 43.36 | 0.04 |
| 3 | 10 | 10 | 28.59 | 0.05 |
| 4 | 2 | 10 | 46.84 | 0.07 |
| 5 | 2 | 10 | 59.40 | 0.09 |
| 6 | 2 | 15 | 38.82 | 0.10 |
| 7 | 4 | 10 | 31.26 | 0.07 |
| 8 | 4 | 10 | 35.77 | 0.06 |
| 9 | 2 | 15 | 45.83 | 0.11 |
| 10 | 2 | 5 | 38.95 | 0.07 |
| 11 | 2 | 10 | 46.13 | 0.07 |
| 12 | 4 | 15 | 21.77 | 0.05 |
| 13 | 4 | 10 | 32.16 | 0.05 |
| 14 | 10 | 10 | 26.71 | 0.04 |
| 15 | 4 | 5 | 42.30 | 0.03 |
| 16 | 10 | 5 | 29.81 | 0.03 |
| 17 | 4 | 15 | 29.48 | 0.07 |
| 18 | 4 | 15 | 28.39 | 0.07 |
| 19 | 4 | 5 | 18.69 | 0.02 |
| 20 | 2 | 5 | 52.57 | 0.04 |
| 21 | 10 | 15 | 17.68 | 0.04 |
| 22 | 4 | 5 | 39.85 | 0.03 |
| 23 | 10 | 15 | 21.43 | 0.04 |
| 24 | 10 | 5 | 35.33 | 0.03 |
| 25 | 10 | 10 | 29.20 | 0.05 |
| 26 | 2 | 15 | 43.52 | 0.11 |
| 27 | 10 | 15 | 20.05 | 0.05 |
| Source | DF | Adj SS | Adj MS | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 8 | 0.000060 | 0.000008 | 21.79 | 0.000 |
| Linear | 4 | 0.000055 | 0.000014 | 39.43 | 0.000 |
| Flowrate (mL/min) | 2 | 0.000028 | 0.000014 | 40.12 | 0.000 |
| Nitrate Conc. (mg/L) | 2 | 0.000027 | 0.000013 | 38.74 | 0.000 |
| Flowrate (mL/min)*Nitrate Conc. (mg/L) | 4 | 0.000006 | 0.000001 | 4.16 | 0.015 |
| Error | 18 | 0.000006 | 0.000000 | ||
| Total | 26 | 0.000067 |
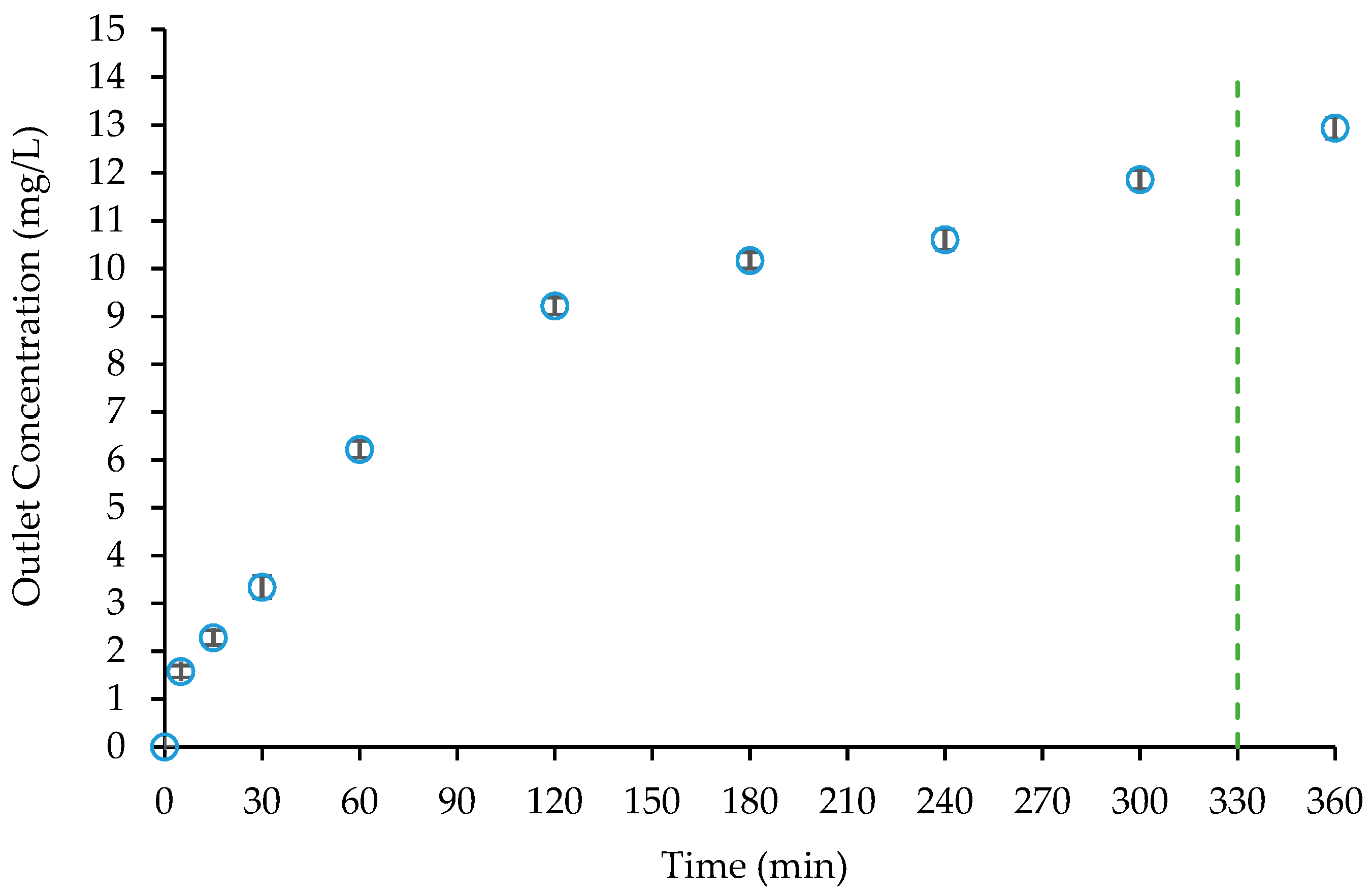
| Adsorbent Weight (g) | Ts (min) | Tb (min) at C/C0 = 0.20 | LUB (cm) | Amount Adsorbed at Tb | Amount Adsorbed at Full Bed Exhaustion | ||
|---|---|---|---|---|---|---|---|
| Total (mg) | mg/g | Total (mg) | mg/g | ||||
| 60 | 330 | 25 | 11.27 | 0.63 | 0.01 | 3.59 | 0.06 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bakly, S.; Al-Juboori, R.A.; Bowtell, L. Macadamia Nutshell Biochar for Nitrate Removal: Effect of Biochar Preparation and Process Parameters. C 2019, 5, 47. https://doi.org/10.3390/c5030047
Bakly S, Al-Juboori RA, Bowtell L. Macadamia Nutshell Biochar for Nitrate Removal: Effect of Biochar Preparation and Process Parameters. C. 2019; 5(3):47. https://doi.org/10.3390/c5030047
Chicago/Turabian StyleBakly, Salam, Raed A. Al-Juboori, and Les Bowtell. 2019. "Macadamia Nutshell Biochar for Nitrate Removal: Effect of Biochar Preparation and Process Parameters" C 5, no. 3: 47. https://doi.org/10.3390/c5030047
APA StyleBakly, S., Al-Juboori, R. A., & Bowtell, L. (2019). Macadamia Nutshell Biochar for Nitrate Removal: Effect of Biochar Preparation and Process Parameters. C, 5(3), 47. https://doi.org/10.3390/c5030047