Controlling the Incorporation of Phosphorus Functionalities on Carbon Nanofibers: Effects on the Catalytic Performance of Fructose Dehydration
Abstract
:1. Introduction
2. Results
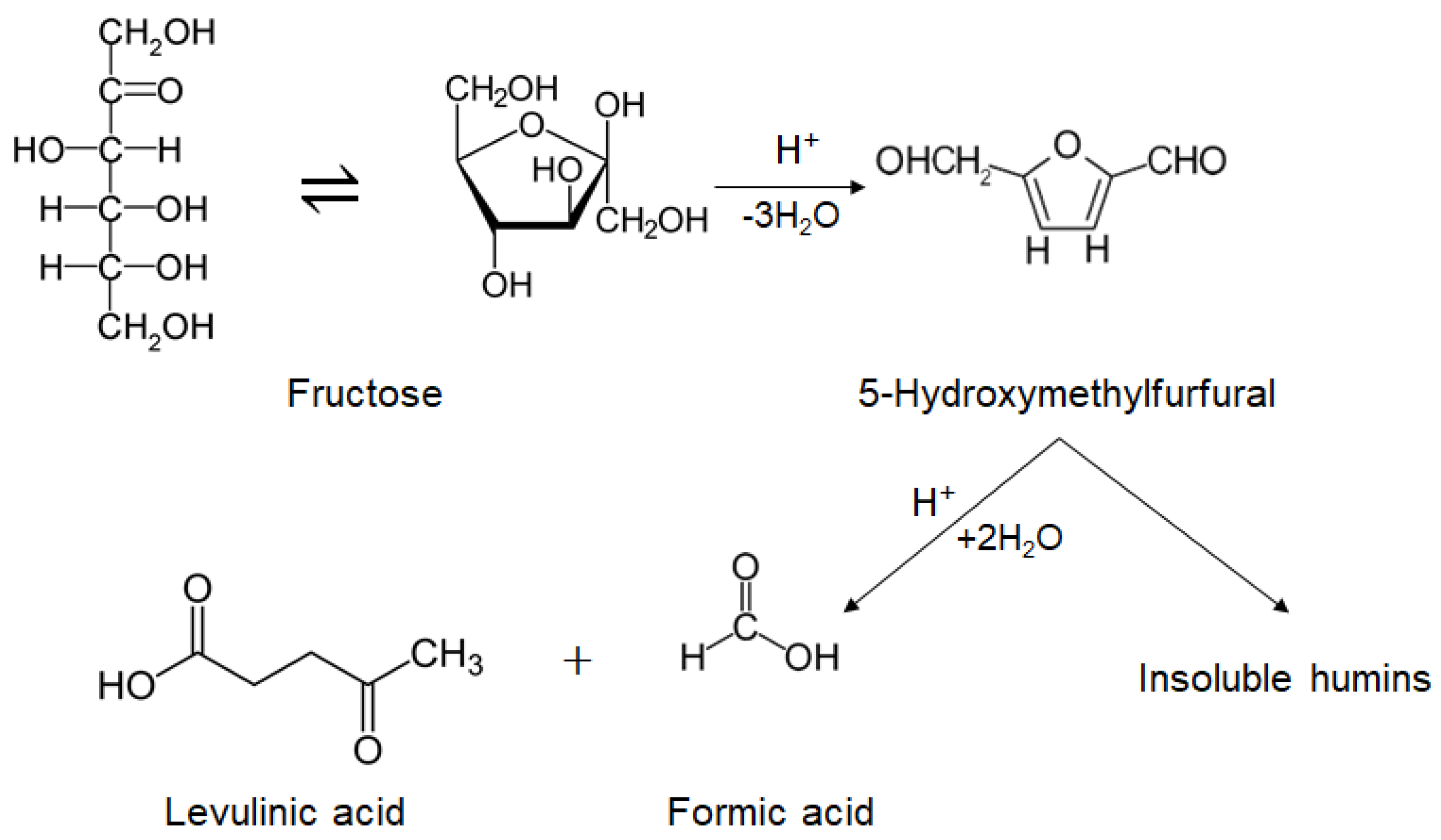
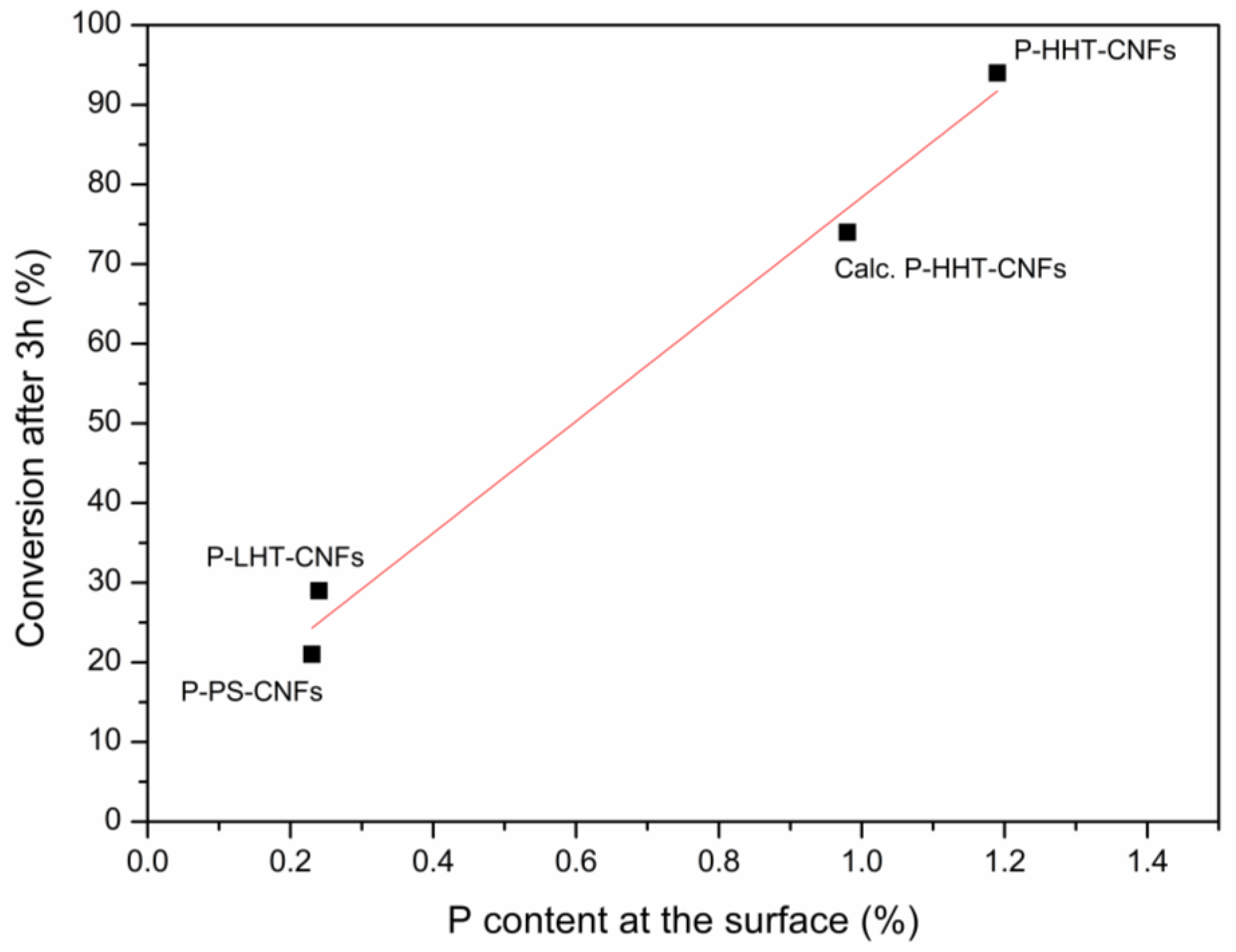
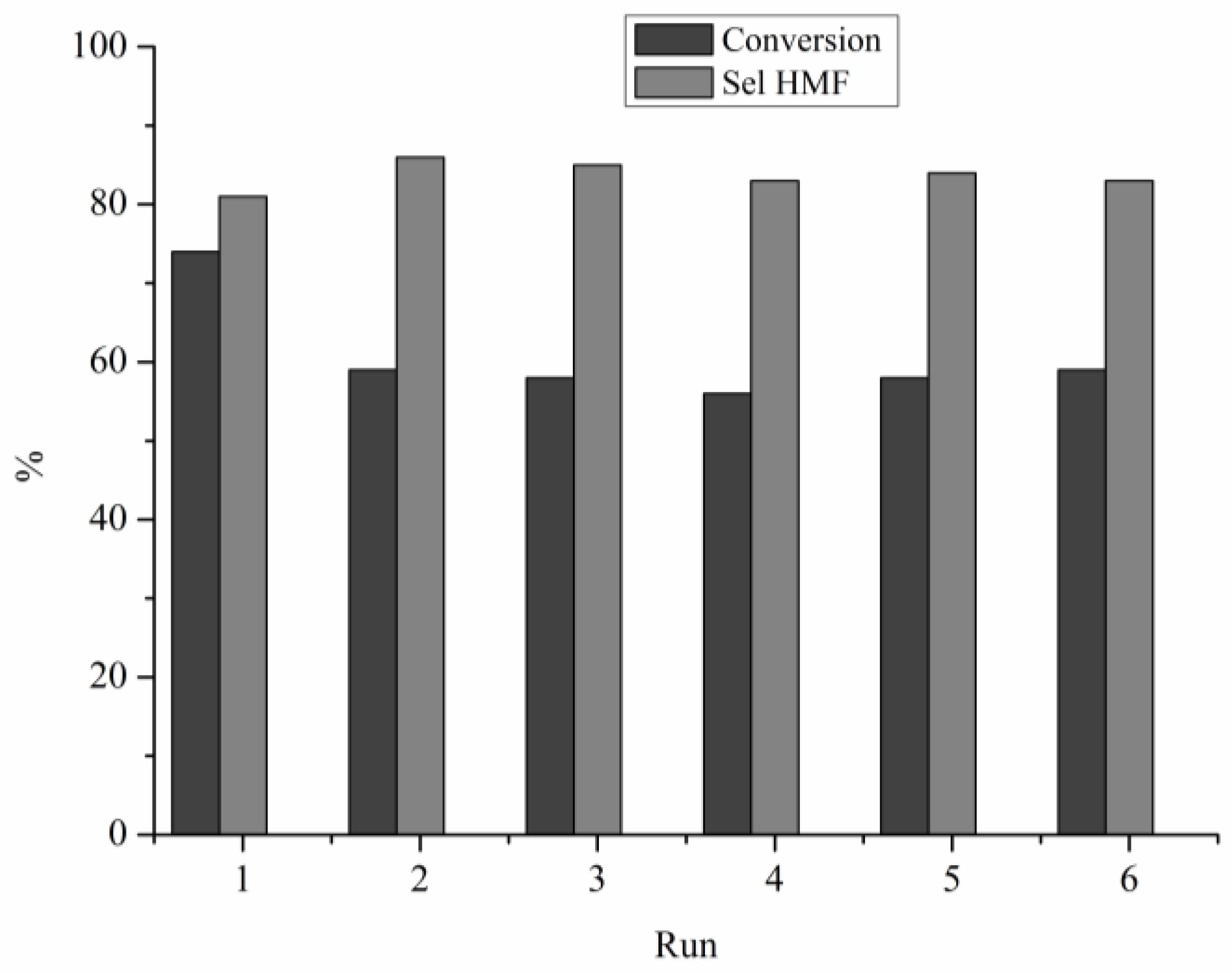
2.1. Catalytic Fructose Dehydration
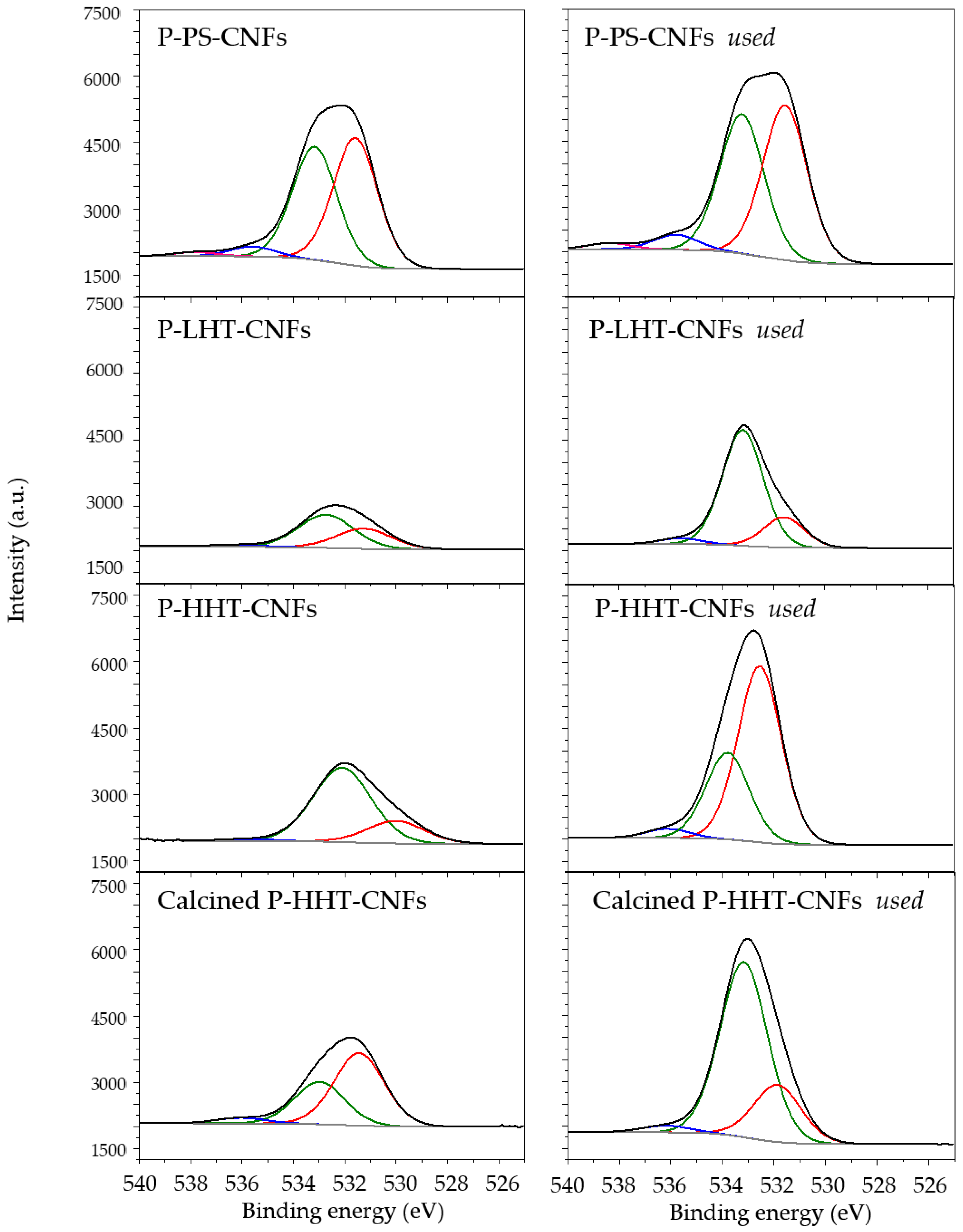
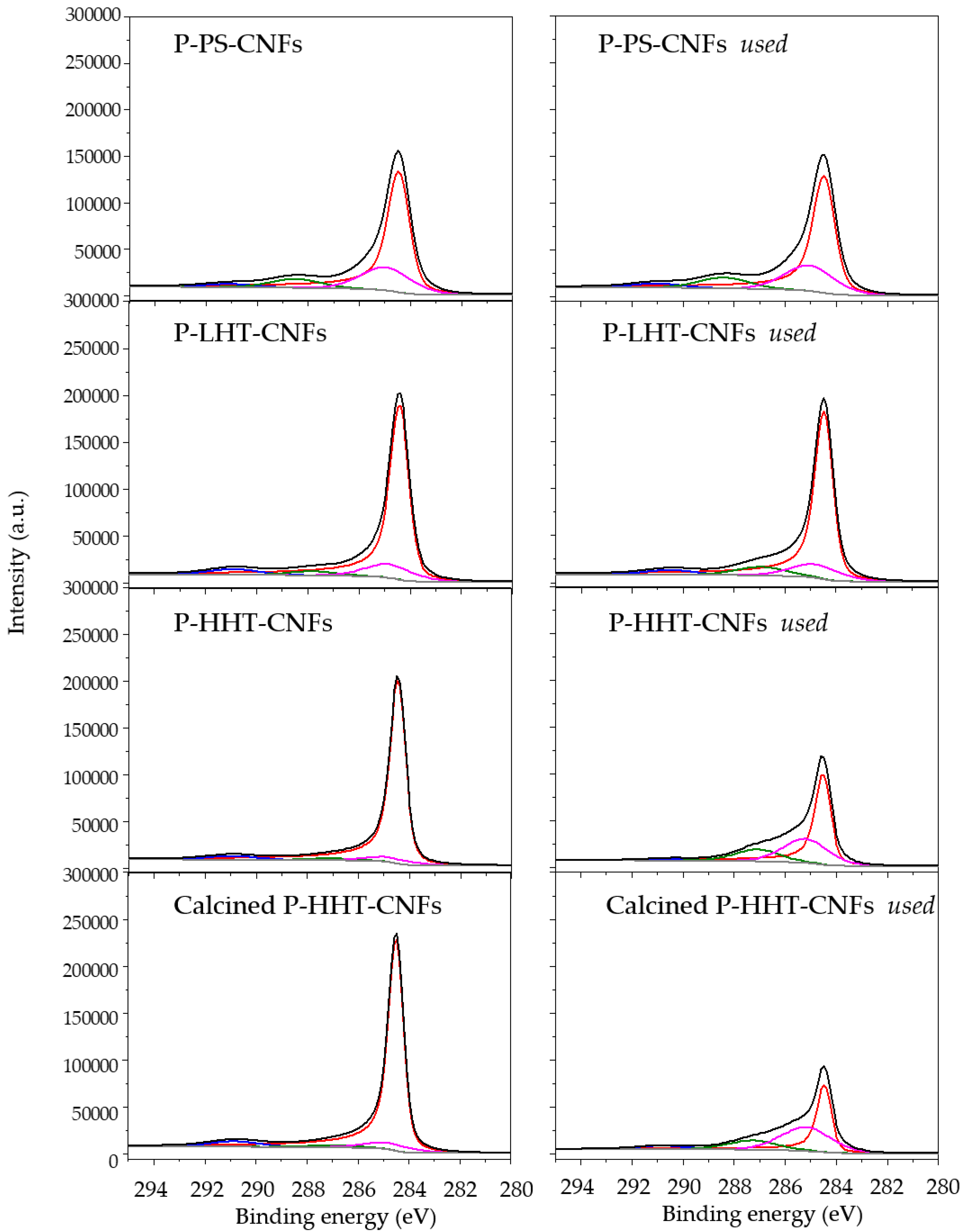
2.2. Characterization of P-Functionalized Carbon Nanofibers (CNFs)
3. Discussion
4. Materials and Methods
4.1. Material Functionalization
4.2. Catalytic Tests
4.3. Catalyst Characterization
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van Santen, R.A. Modern Heterogeneous Catalysis: An Introduction, 1st ed.; Wiley-VCH: Weinheim, Germany, 2017; pp. 1–166. ISBN 978-3-527-33961-7. [Google Scholar]
- Rodríguez-Reinoso, F. The role of carbon materials in heterogeneous catalysis. Carbon 1998, 36, 159–175. [Google Scholar] [CrossRef]
- Serp, P.; Figueiredo, J.L. Carbon Materials for Catalysis, 1st ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2009; pp. 1–218. ISBN 9780470178850. [Google Scholar]
- Liu, X.; Dai, L. Carbon-based metal-free catalysts. Nat. Rev. Mater. 2016, 1, 16064. [Google Scholar] [CrossRef]
- Lam, E.; Luong, J.H.T. Carbon materials as catalyst supports and catalysts in the transformation of biomass to fuels and chemicals. ACS Catal. 2014, 4, 3393–3410. [Google Scholar] [CrossRef]
- Su, D.S.; Zhang, J.; Frank, B.; Thomas, A.; Wang, X.; Paraknowitsch, J.; Schlögl, R. Metal-Free heterogeneous catalysis for sustainable chemistry. ChemSusChem 2010, 3, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Pierson, H.O. Handbook of Carbon, Graphite, Diamonds and Fullerenes: Processing, Properties and Applications, 1st ed.; Noyes Publications: New York, NY, USA, 1993; pp. 70–86. ISBN 0815513399. [Google Scholar]
- Bitter, J.H. Nanostructured carbons in catalysis a Janus material—Industrial applicability and fundamental insights. J. Mater. Chem. 2010, 20, 7312–7321. [Google Scholar] [CrossRef]
- Serp, P.; Machado, B. Nanostructured Carbon Materials for Catalysis, 1st ed.; Royal Society of Chemistry: Cambridge, UK, 2015; pp. 1–267. [Google Scholar]
- Yu, D.; Nagelli, E.; Du, F.; Dai, L. Metal-Free carbon nanomaterials become more active than metal catalysts and last longer. J. Phys. Chem. Lett. 2010, 1, 2165–2173. [Google Scholar] [CrossRef]
- Pan, X.; Bao, X. The effects of confinement inside carbon nanotubes on catalysis. Acc. Chem. Res. 2011, 44, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Fan, Z.; Pan, X.; Bao, X. Effect of confinement in carbon nanotubes on the activity of Fischer—Tropsch iron catalyst. J. Am. Chem. Soc. 2008, 130, 9414–9419. [Google Scholar] [CrossRef] [PubMed]
- Villa, A.; Wang, D.; Chan-Thaw, C.E.; Campisi, S.; Veith, G.M.; Prati, L. The confinement effect on the activity of Au NPs in polyol oxidation. Catal. Sci. Technol. 2016, 6, 598–601. [Google Scholar] [CrossRef]
- Guo, S.; Pan, X.; Gao, H.; Yang, Z.; Zhao, J.; Bao, X. Probing the electronic effect of carbon nanotubes in catalysis: NH3 synthesis with Ru nanoparticles. Chem. Eur. J. 2010, 16, 5379–5384. [Google Scholar] [CrossRef] [PubMed]
- Toebes, M.L.; Zhang, Y.; Hájek, J.; Nijhuis, T.A.; Bitter, J.H.; Van Dillen, A.J.; Murzin, D.Y.; de Jong, K.P. Support effects in hydrogenation of cinnamaldehyde over carbon nanofiber-supported platinum catalysts: Characterization and catalysis. J. Catal. 2004, 226, 215–225. [Google Scholar] [CrossRef]
- Zhu, J.; Holmen, A.; Chen, D. Carbon nanomaterials in catalysis: Proton affinity, chemical and electronic properties, and their catalytic consequences. ChemCatChem 2013, 5, 378–401. [Google Scholar] [CrossRef]
- Zhang, J.; Su, D.S.; Blume, R.; Schlögl, R.; Wang, R.; Yang, X.; Gajović, A. Surface Chemistry and Catalytic Reactivity of a Nanodiamond in the Steam-Free Dehydrogenation of Ethylbenzene. Angew. Chem. Int. Ed. 2010, 49, 8640–8644. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Li, B.; Su, D. The Unexpected Reactivity of the Carbon Sites on the Nanostructured Carbon Catalysts towards the C–H Bond Activation from the Analysis of the Aromaticity. Chem. Asian J. 2016, 11, 1668–1671. [Google Scholar] [CrossRef] [PubMed]
- Axet, M.R.; Dechy-Cabaret, O.; Durand, J.; Gouygou, M.; Serp, P. Coordination chemistry on carbon surfaces. Coord. Chem. Rev. 2016, 308, 236–345. [Google Scholar] [CrossRef]
- Gao, J.; Ji, Y.; Li, Y.; Zhong, J.; Sun, X. The morphological effect on electronic structure and electrical transport properties of one-dimensional carbon nanostructures. RSC Adv. 2017, 7, 21079–21084. [Google Scholar] [CrossRef]
- Li, J.; Vergne, M.J.; Mowles, E.D.; Zhong, W.H.; Hercules, D.M.; Lukehart, C.M. Surface functionalization and characterization of graphitic carbon nanofibers (GCNFs). Carbon 2005, 43, 2883–2893. [Google Scholar] [CrossRef]
- Balasubramanian, K.; Burghard, M. Chemically functionalized carbon nanotubes. Small 2005, 1, 180–192. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Maiti, U.N.; Lee, J.M.; Lim, J.; Han, T.H.; Kim, S.O. Nitrogen-doped carbon nanotubes and graphene composite structures for energy and catalytic applications. Chem. Commun. 2014, 50, 6818–6830. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Yang, L.; Huang, Z.; Luo, J.; Li, M.; Wang, D.; Chen, Y. Enhancing Electrocatalytic Oxygen Reduction on Nitrogen-Doped Graphene by Active Sites Implantation. Sci. Rep. 2013, 3, 3306. [Google Scholar] [CrossRef] [PubMed]
- Fraga, M.A.; Jordao, E.; Mendes, M.J.; Freitas, M.M.A.; Faria, J.L.; Figueiredo, J.L. Properties of carbon-supported platinum catalysts: Role of carbon surface sites. J. Catal. 2002, 209, 355–364. [Google Scholar] [CrossRef]
- Hara, M.; Yoshida, T.; Takagaki, A.; Takata, T.; Kondo, J.N.; Hayashi, S.; Domen, K. A carbon material as a strong protonic acid. Angew. Chem. Int. Ed. 2004, 43, 2955–2958. [Google Scholar] [CrossRef] [PubMed]
- Villa, A.; Schiavoni, M.; Prati, L. Material science for the support design: A powerful challenge for catalysis. Catal. Sci. Technol. 2012, 2, 673–682. [Google Scholar] [CrossRef]
- Arrigo, R.; Hävecker, M.; Wrabetz, S.; Blume, R.; Lerch, M.; McGregor, J.; Parrott, E.P.J.; Zeitler, J.A.; Gladden, L.F.; Knop-Gericke, A.; et al. Tuning the acid/base properties of nanocarbons by functionalization via amination. J. Am. Chem. Soc. 2010, 132, 9616–9630. [Google Scholar] [CrossRef] [PubMed]
- Campisi, S.; Marzorati, S.; Spontoni, P.; Chan-Thaw, C.E.; Longhi, M.; Villa, A.; Prati, L. Tailored N-Containing Carbons as Catalyst Supports in Alcohol Oxidation. Materials 2016, 9, 114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Dai, L. Heteroatom-doped graphitic carbon catalysts for efficient electrocatalysis of oxygen reduction reaction. ACS Catal. 2015, 5, 7244–7253. [Google Scholar] [CrossRef]
- Villa, A.; Schiavoni, M.; Chan-Thaw, C.E.; Fulvio, P.F.; Mayes, R.T.; Dai, S.; More, K.L.; Veith, G.M.; Prati, L. Acid-functionalized mesoporous carbon: An efficient support for ruthenium-catalyzed γ-valerolactone production. ChemSusChem 2015, 8, 2520–2528. [Google Scholar] [CrossRef] [PubMed]
- Prati, L.; Chan-Thaw, C.E.; Campisi, S.; Villa, A. N-Modified Carbon-Based Materials: Nanoscience for Catalysis. Chem. Rec. 2016, 16, 2187–2197. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.K.; Thakur, M.K. Chemical Functionalization of Carbon Nanomaterials: Chemistry and Applications; CRC Press: Boca Raton, FL, USA, 2015; p. 237. ISBN 1482253968. [Google Scholar]
- Carniti, P.; Gervasini, A.; Biella, S.; Auroux, A. Niobic acid and niobium phosphate as highly acidic viable catalysts in aqueous medium: Fructose dehydration reaction. Catal. Today 2006, 118, 373–378. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J.J.; Ren, J.W.; Liu, X.H.; Li, X.C.; Xia, Y.J.; Lu, G.Z.; Wang, Y.Q. Mesoporous niobium phosphate: An excellent solid acid for the dehydration of fructose to 5-hydroxymethylfurfural in water. Catal. Sci. Technol. 2012, 2, 2485–2491. [Google Scholar] [CrossRef]
- Carlini, C.; Patrono, P.; Galletti, A.M.R.; Sbrana, G. Heterogeneous catalysts based on vanadyl phosphate for fructose dehydration to 5-hydroxymethyl-2-furaldehyde. Appl. Catal. A 2004, 275, 111–118. [Google Scholar] [CrossRef]
- Villa, A.; Schiavoni, M.; Fulvio, P.F.; Mahurin, S.M.; Dai, S.; Mayes, R.T.; Veith, G.M.; Prati, L. Phosphorylated mesoporous carbon as effective catalyst for the selective fructose dehydration to HMF. J. Energy Chem. 2013, 22, 305–311. [Google Scholar] [CrossRef]
- Aronne, A.; Di Serio, M.; Vitiello, R.; Clayden, N.J.; Minieri, L.; Imparato, C.; Piccolo, A.; Pernice, P.; Carniti, P.; Gervasini, A. An environmentally friendly Nb–P–Si solid catalyst for acid demanding reactions. J. Phys. Chem. C 2017, 121, 17378–17389. [Google Scholar] [CrossRef]
- Tessonnier, J.-P.; Rosenthal, D.; Hansen, T.W.; Hess, C.; Schuster, M.E.; Blume, R.; Girgsdies, F.; Pfaender, N.; Timpe, O.; Su, D.S.; et al. Analysis of the structure and chemical properties of some commercial carbon nanostructures. Carbon 2009, 47, 1779–1798. [Google Scholar] [CrossRef]
- Carniti, P.; Gervasini, A.; Marzo, M. Absence of expected side-reactions in the dehydration reaction of fructose to HMF in water over niobic acid catalyst. Catal. Commun. 2011, 12, 1122–1126. [Google Scholar] [CrossRef]
- Moreau, C.; Durand, R.; Razigade, S.; Duhamet, J.; Faugeras, P.; Rivalier, P.; Ros, P.; Avignon, G. Dehydration of fructose to 5-hydroxymethylfurfural over H-mordenites. Appl. Catal. A Gen. 1996, 145, 211–224. [Google Scholar] [CrossRef]
- Van Dam, H.E.; Kieboom, A.P.G.; van Bekkum, H. The Conversion of Fructose and Glucose in Acidic Media: Formation of Hydroxymethylfurfural. Starch Stärke 1986, 38, 95–101. [Google Scholar] [CrossRef]
- Zhao, Q.; Wang, L.; Zhao, S.; Wang, X.; Wang, S. High selective production of 5-hydroymethylfurfural from fructose by a solid heteropolyacid catalyst. Fuel 2011, 90, 2289–2293. [Google Scholar] [CrossRef]
- Wang, L.; Dong, X.; Jiang, H.; Li, G.; Zhang, M. Phosphorylated ordered mesoporous carbon as a novel solid acid catalyst for the esterification of oleic acid. Catal. Commun. 2014, 56, 164–167. [Google Scholar] [CrossRef]
- Bedia, J.; Rosas, J.M.; Márquez, J.; Rodríguez-Mirasol, J.; Cordero, T. Preparation and characterization of carbon based acid catalysts for the dehydration of 2-propanol. Carbon 2009, 47, 286–294. [Google Scholar] [CrossRef]
- Kundu, S.; Wang, Y.; Xia, W.; Muhler, M. Thermal Stability and Reducibility of Oxygen-Containing Functional Groups on Multiwalled Carbon Nanotube Surfaces: A Quantitative High-Resolution XPS and TPD/TPR Study. J. Phys. Chem. C 2008, 112, 16869–16878. [Google Scholar] [CrossRef]
- Walczyk, M.; Świąkatkowski, A.; Pakuła, M.; Biniak, S. Electrochemical studies of the interaction between a modified activated carbon surface and heavy metal ions. J. Appl. Electrochem. 2005, 35, 123–130. [Google Scholar] [CrossRef]
- Tuinstra, F.; Koenig, J.L. Raman Spectrum of Graphite. J. Chem. Phys. 1970, 53, 1126–1130. [Google Scholar] [CrossRef]
- Lim, S.; Yoon, S.; Mochida, I. Surface Modification of Carbon Nanofiber with High Degree of Graphitization. J. Phys. Chem. 2004, 1533–1536. [Google Scholar] [CrossRef]
- Szekeres, G.P.; Nemeth, K.; Kinka, A.; Magyar, M.; Reti, B.; Varga, E.; Szegletes, Z.; Erdohelyi, A.; Nagy, L.; Hernadi, K. Segmental nitrogen doping and carboxyl functionalization of multi-walled carbon nanotubes. Phys. Status Solidi Basic Res. 2015, 252, 2472–2478. [Google Scholar] [CrossRef]
- Tikhomirov, A.S.; Sorokina, N.E.; Avdeev, V.V. Surface modification of carbon fibers with nitric acid solutions. Inorg. Mater. 2011, 47, 684–688. [Google Scholar] [CrossRef]
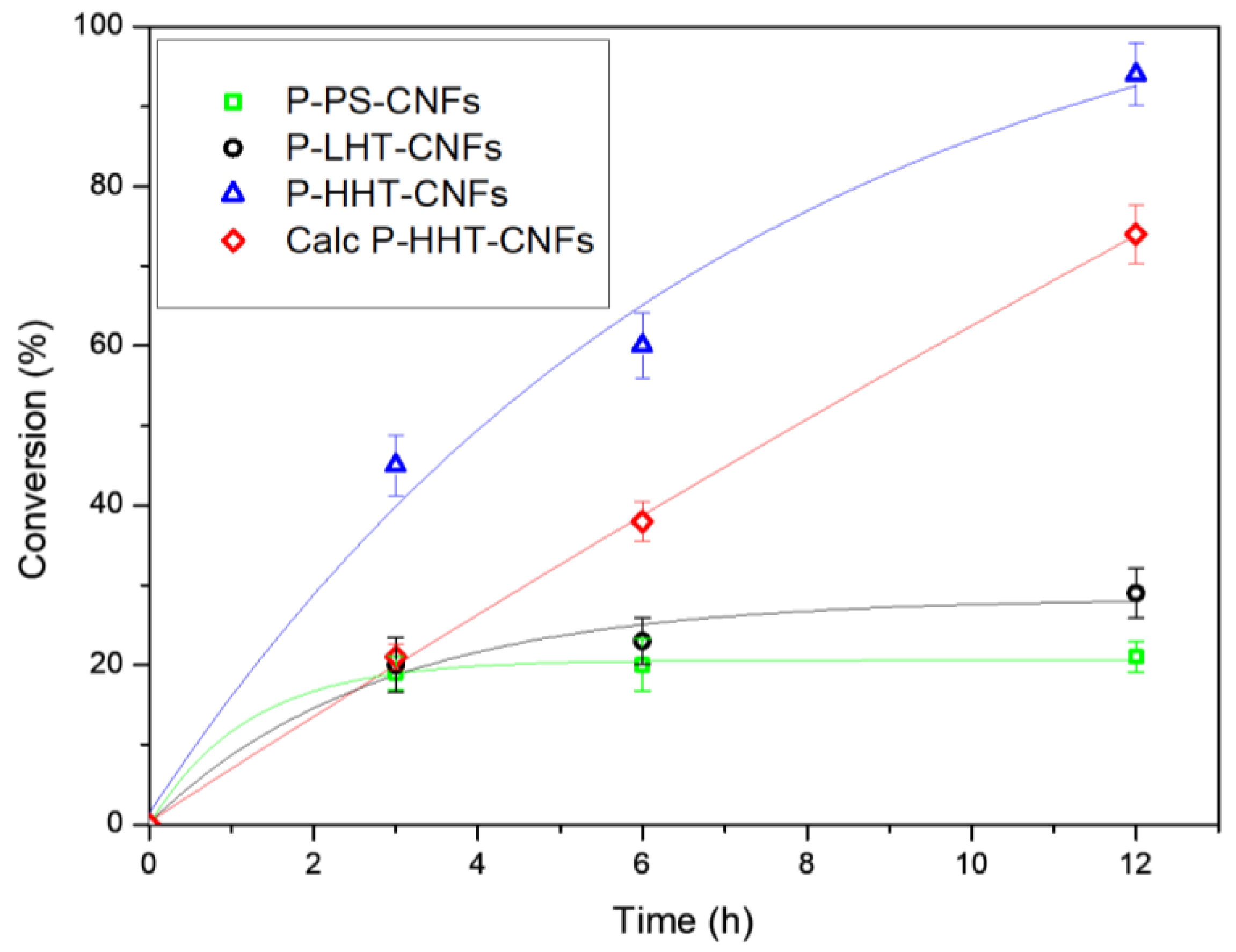



| Catalyst | Time of Reaction (h) | Conversion (%) | Selectivity | ||
|---|---|---|---|---|---|
| HMF | Levulinic Acid | Formic Acid | |||
| P-PS-CNFs | 3 | 17 | 54 | 2 | 7 |
| 6 | 18 | 50 | 5 | 9 | |
| 12 | 19 | 48 | 7 | 11 | |
| P-LHT-CNFs | 3 | 20 | 53 | 1 | 9 |
| 6 | 23 | 45 | 1 | 5 | |
| 12 | 29 | 49 | 2 | 3 | |
| P-HHT-CNFs | 3 | 45 | 50 | 2 | 2 |
| 6 | 60 | 49 | 6 | 4 | |
| 12 | 94 | 46 | 10 | 7 | |
| Calc P-HHT-CNFs | 3 | 21 | 87 | 2 | 2 |
| 6 | 38 | 86 | 3 | 5 | |
| 12 | 74 | 81 | 6 | 8 | |
| H3PO4 0.1M | 3 | 88 | 52 | 7 | 8 |
| Sample | Specific Surface Area (m2 g−1) |
|---|---|
| PS-CNFs | 43 ± 2.3 |
| P-PS-CNFs | 45 ± 1.8 |
| LHT-CNFs | 38 ± 3.3 |
| P-LHT-CNFs | 39 ± 4.2 |
| HHT-CNFs | 37 ± 0.9 |
| P-HHT-CNFs | 36 ± 2.1 |
| Sample | Atomic Ratio % C:O:P |
|---|---|
| P-PS-CNFs | 87.12:12.65:0.23 |
| P-PS-CNFs-used | 85.08:14.85:0.07 |
| P-LHT-CNFs | 96.06:3.70:0.24 |
| P-LHT-CNFs-used | 92.74:7.25:0.01 |
| P-HHT-CNFs | 90.89:7.92:1.19 |
| P-HHT-CNFs-used | 81.84:18.11:0.05 |
| calc P-HHT-CNFs | 91.65:7.37:0.98 |
| calc P-HHT-CNFs-used | 80.32:19.56:0.12 |
| Sample | O1S | P1S | C1s | ||||||
|---|---|---|---|---|---|---|---|---|---|
| C=O, P=O, P–O | C–O, C–O–C, P–O–C | H2O | C–O–PO3 C–P | sp2 | sp3 C–P | C=O | C=C | ||
| P-PS-CNFs | BE | 531.6 | 533.3 | 535.6 | 133.7 | 284.5 | 285.0 | 288.5 | 291.1 |
| Atom % | (50.1) | (44.5) | (5.4) | (100) | (77.4) | (14.4) | (5.7) | (2.5) | |
| P-PS-CNFs-used | BE | 531.6 | 533.3 | 535.9 | 133.7 | 284.5 | 285.1 | 288.4 | 291.1 |
| Atom % | (49.1) | (44.2) | (6.7) | (100) | (73.9) | (15.9) | (7.1) | (3.1) | |
| P-LHT-CNFs | BE | 531.3 | 532.8 | 536.2 | 133.8 | 284.4 | 284.9 | 287.9 | 290.1 |
| Atom % | (35.6) | (58.2) | (6.2) | (100) | (86.8) | (7.4) | (2.6) | (3.2) | |
| P-LHT-CNFs-used | BE | 531.6 | 533.2 | 535.6 | 133.7 | 284.5 | 285.0 | 287.6 | 290.5 |
| Atom % | (20.0) | (76.2) | (3.8) | (100) | (85.5) | (7.3) | (4.8) | (2.4) | |
| P-HHT-CNFs | BE | 532.5 | 533.8 | 536.1 | 133.4 | 284.5 | 285.1 | 287.7 | 291.0 |
| Atom % | (31.9) | (64.7) | (3.3) | (100) | (94.5) | (2.4) | (1.1) | (2.0) | |
| P-HHT-CNFs-used | BE | 532.5 | 533.8 | 536.0 | 133.4 | 284.5 | 285.2 | 287.5 | 290.9 |
| Atom % | (70.3) | (26.8) | (2.9) | (100) | (70.2) | (19.0) | (9.5) | (1.3) | |
| calc P-HHT-CNFs | BE | 531.4 | 533.0 | 536.0 | 134.2 | 284.5 | 285.0 | 287.1 | 290.9 |
| Atom % | (61.3) | (33.2) | (5.5) | (100) | (93.6) | (2.9) | (1.1) | (2.4) | |
| calc P-HHT-CNFs-used | BE | 531.9 | 533.2 | 536.2 | 134.2 | 284.5 | 285.2 | 287.3 | 290.9 |
| Atom % | (23.4) | (73.0) | (3.6) | (100) | (64.6) | (22.9) | (9.0) | (3.5) | |
| Sample Name | Graphitization Degree (ID/IG) 1 | ||
|---|---|---|---|
| Pristine | P-Functionalised | After Reaction | |
| PS-CNFs | 0.75 | 0.58 | 1.16 |
| LHT CNFs | 0.71 | 1.01 | 1.11 |
| HHT CNFs | 0.11 | 0.09 | 0.34 |
| Calcined Funct-HHT CNFs | 0.11 | 0.36 | 0.38 |
| Sample | Acid Site (mmol g−1) |
|---|---|
| P-PS-CNFs | 0.116 ± 0.093 |
| P-LHT-CNFs | 0.343 ± 0.032 |
| P-HHT-CNFs | 1.257 ± 0.009 |
| Calc. P-HHT-CNFs | 0.797 ± 0.015 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campisi, S.; Sanchez Trujillo, F.J.; Motta, D.; Davies, T.E.; Dimitratos, N.; Villa, A. Controlling the Incorporation of Phosphorus Functionalities on Carbon Nanofibers: Effects on the Catalytic Performance of Fructose Dehydration. C 2018, 4, 9. https://doi.org/10.3390/c4010009
Campisi S, Sanchez Trujillo FJ, Motta D, Davies TE, Dimitratos N, Villa A. Controlling the Incorporation of Phosphorus Functionalities on Carbon Nanofibers: Effects on the Catalytic Performance of Fructose Dehydration. C. 2018; 4(1):9. https://doi.org/10.3390/c4010009
Chicago/Turabian StyleCampisi, Sebastiano, Felipe J Sanchez Trujillo, Davide Motta, Thomas E. Davies, Nikolaos Dimitratos, and Alberto Villa. 2018. "Controlling the Incorporation of Phosphorus Functionalities on Carbon Nanofibers: Effects on the Catalytic Performance of Fructose Dehydration" C 4, no. 1: 9. https://doi.org/10.3390/c4010009
APA StyleCampisi, S., Sanchez Trujillo, F. J., Motta, D., Davies, T. E., Dimitratos, N., & Villa, A. (2018). Controlling the Incorporation of Phosphorus Functionalities on Carbon Nanofibers: Effects on the Catalytic Performance of Fructose Dehydration. C, 4(1), 9. https://doi.org/10.3390/c4010009









