MicroRNAs as Appropriate Discriminators in Non-Specific Alpha-Fetoprotein (AFP) Elevation in Testicular Germ Cell Tumor Patients
Abstract
1. Introduction
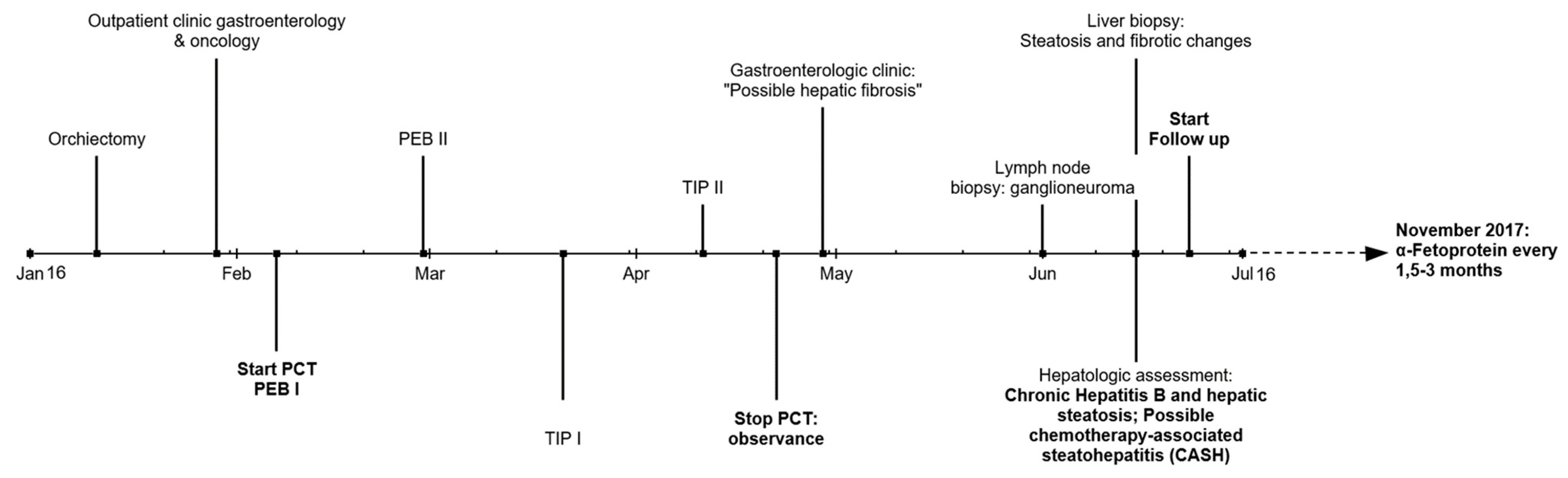
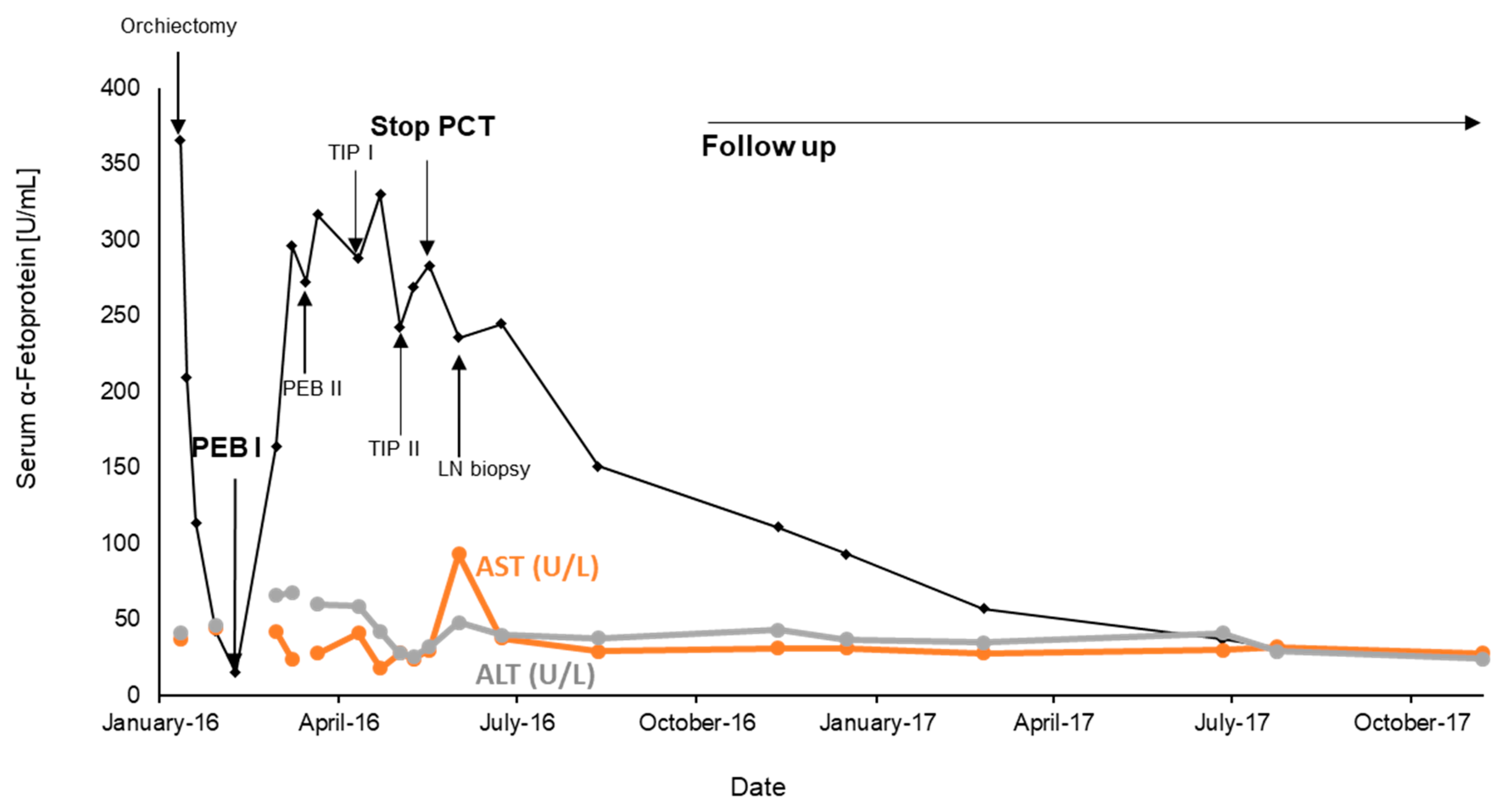
2. Case Report
3. Discussion
4. Materials and Methods
miRNA Isolation, cDNA Synthesis, and Quantitative RT PCR (qRT-PCR)
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| TGCT | Testicular germ cell tumors |
| IGCCCG | International Germ Cell Cancer Consensus Group |
| AFP | Alpha-fetoprotein |
| TGCT | Testicular germ cell tumors |
| LDH | Lactate dehydrogenase |
| PEB | Cisplatin, etoposide, and bleomycine |
| PCT | Polychemotherapy |
| TIP | Paclitaxel, ifosfamide, and cisplatin |
| HBs | Hepatitis B surface |
| CASH | Chemotherapy-associated steatohepatitis |
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA A Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Trabert, B.; Chen, J.; Devesa, S.S.; Bray, F.; McGlynn, K.A. International patterns and trends in testicular cancer incidence, overall and by histologic subtype, 1973–2007. Andrology 2015, 3, 4–12. [Google Scholar] [CrossRef]
- Boormans, J.L.; de Castro, J.M.; Marconi, L.; Yuan, Y.; Pes, M.P.L.; Bokemeyer, C.; Nicolai, N.; Algaba, F.; Oldenburg, J.; Albers, P. Testicular Tumor Size and Rete Testis Invasion as Prognostic Factors for the Risk of Relapse of Clinical Stage I Seminoma Testis Patients Under Surveillance: A Systematic Review by the Testicular Cancer Guidelines Panel. Eur. Urol. 2018, 73, 394–405. [Google Scholar] [CrossRef] [PubMed]
- Tandstad, T.; Kollmannsberger, C.K.; Roth, B.J.; Jeldres, C.; Gillessen, S.; Fizazi, K.; Daneshmand, S.; Lowrance, W.T.; Hanna, N.H.; Albany, C.; et al. Practice Makes Perfect: The Rest of the Story in Testicular Cancer as a Model Curable Neoplasm. J. Clin. Oncol. 2017, 35, 3525–3528. [Google Scholar] [CrossRef] [PubMed]
- Doherty, A.P.; Bower, M.; Christmas, T.J. The role of tumor markers in the diagnosis and treatment of testicular germ cell cancers. Br. J. Urol. 1997, 79, 247–252. [Google Scholar] [CrossRef]
- Milose, J.C.; Filson, C.P.; Weizer, A.Z.; Hafez, K.S.; Montgomery, J.S. Role of biochemical markers in testicular cancer: Diagnosis, staging, and surveillance. J. Urol. 2011, 4, 1–8. [Google Scholar]
- Gitlin, D.; Perricelli, A.; Gitlin, G.M. Synthesis of -fetoprotein by liver, yolk sac, and gastrointestinal tract of the human conceptus. Cancer Res. 1972, 32, 979–982. [Google Scholar]
- Germà, J.R.; Llanos, M.; Tabernero, J.M.; Mora, J. False elevations of alpha-fetoprotein associated with liver dysfunction in germ cell tumors. Cancer 1993, 72, 2491–2494. [Google Scholar] [CrossRef]
- Johnson, P. Role of alpha-fetoprotein in the diagnosis and management of hepatocellular carcinoma. J. Gastroenterol. Hepatol. 1999, 14, S32–S36. [Google Scholar] [CrossRef]
- Anfossi, S.; Fu, X.; Nagvekar, R.; Calin, G.A. MicroRNAs, Regulatory Messengers Inside and Outside Cancer Cells. Adv. Exp. Med. Biol. 2018, 1056, 87–108. [Google Scholar]
- Dragomir, M.P.; Knutsen, E.; Calin, G.A. SnapShot: Unconventional miRNA Functions. Cell 2018, 174, 1038–1038.e1. [Google Scholar] [CrossRef] [PubMed]
- Anheuser, P.; Radtke, A.; Wulfing, C.; Kranz, J.; Belge, G.; Dieckmann, K.P. Serum Levels of MicroRNA371a-3p: A Highly Sensitive Tool for Diagnosing and Staging Testicular Germ Cell Tumors: A Clinical Case Series. Urol. Int. 2017, 99, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Ling, H.; Krassnig, L.; Bullock, M.D.; Pichler, M. MicroRNAs in Testicular Cancer Diagnosis and Prognosis. Urol. Clin. North Am. 2016, 43, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Dieckmann, K.P.; Radtke, A.; Geczi, L.; Matthies, C.; Anheuser, P.; Eckardt, U.; Sommer, J.; Zengerling, F.; Trenti, E.; Pichler, R.; et al. Serum Levels of MicroRNA-371a-3p (M371 Test) as a New Biomarker of Testicular Germ Cell Tumors: Results of a Prospective Multicentric Study. J. Clin. Oncol. 2019, 37, 1412–1423. [Google Scholar] [CrossRef] [PubMed]
- Terbuch, A.; Adiprasito, J.B.; Stiegelbauer, V.; Seles, M.; Klec, C.; Pichler, G.P.; Resel, M.; Posch, F.; Lembeck, A.L.; Stöger, H.; et al. MiR-371a-3p Serum Levels Are Increased in Recurrence of Testicular Germ Cell Tumor Patients. Int. J. Mol. Sci. 2018, 19, 3130. [Google Scholar] [CrossRef]
- Belge, G.; Dieckmann, K.-P.; Spiekermann, M.; Balks, T.; Bullerdiek, J. Serum Levels of MicroRNAs miR-371-3: A Novel Class of Serum Biomarkers for Testicular Germ Cell Tumors? Eur. Urol. 2012, 61, 1068–1069. [Google Scholar] [CrossRef]
- Dieckmann, K.P.; Spiekermann, M.; Balks, T.; Flor, I.; Loning, T.; Bullerdiek, J.; Belge, G. MicroRNAs miR-371-3 in serum as diagnostic tools in the management of testicular germ cell tumors. Br. J. Cancer 2012, 107, 1754–1760. [Google Scholar] [CrossRef]
- Gillis, A.J.; Rijlaarsdam, M.A.; Eini, R.; Dorssers, L.C.; Biermann, K.; Murray, M.J.; Nicholson, J.C.; Coleman, N.; Dieckmann, K.-P.; Belge, G.; et al. Targeted serum miRNA (TSmiR) test for diagnosis and follow-up of (testicular) germ cell cancer patients: A proof of principle. Mol. Oncol. 2013, 7, 1083–1092. [Google Scholar] [CrossRef]
- Palmer, R.D.; Murray, M.J.; Saini, H.K.; Van Dongen, S.; Abreu-Goodger, C.; Muralidhar, B.; Pett, M.R.; Thornton, C.M.; Nicholson, J.C.; Enright, A.J.; et al. Malignant germ cell tumors display common microRNA profiles resulting in global changes in expression of messenger RNA targets. Cancer Res. 2010, 70, 2911–2923. [Google Scholar] [CrossRef]
- Myklebust, M.P.; Rosenlund, B.; Gjengstø, P.; Bercea, B.S.; Karlsdottir, Á.; Brydøy, M.; Dahl, O. Quantitative PCR Measurement of miR-371a-3p and miR-372-p Is Influenced by Hemolysis. Front. Genet. 2019, 10, 463. [Google Scholar] [CrossRef]
- Faraldi, M.; Gomarasca, M.; Sansoni, V.; Perego, S.; Banfi, G.; Lombardi, G. Normalization strategies differently affect circulating miRNA profile associated with the training status. Sci. Rep. 2019, 9, 1584. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lembeck, A.L.; Puchas, P.; Hutterer, G.; Barth, D.A.; Terbuch, A.; Bauernhofer, T.; Pichler, M. MicroRNAs as Appropriate Discriminators in Non-Specific Alpha-Fetoprotein (AFP) Elevation in Testicular Germ Cell Tumor Patients. Non-Coding RNA 2020, 6, 2. https://doi.org/10.3390/ncrna6010002
Lembeck AL, Puchas P, Hutterer G, Barth DA, Terbuch A, Bauernhofer T, Pichler M. MicroRNAs as Appropriate Discriminators in Non-Specific Alpha-Fetoprotein (AFP) Elevation in Testicular Germ Cell Tumor Patients. Non-Coding RNA. 2020; 6(1):2. https://doi.org/10.3390/ncrna6010002
Chicago/Turabian StyleLembeck, Anna L., Philip Puchas, Georg Hutterer, Dominik A. Barth, Angelika Terbuch, Thomas Bauernhofer, and Martin Pichler. 2020. "MicroRNAs as Appropriate Discriminators in Non-Specific Alpha-Fetoprotein (AFP) Elevation in Testicular Germ Cell Tumor Patients" Non-Coding RNA 6, no. 1: 2. https://doi.org/10.3390/ncrna6010002
APA StyleLembeck, A. L., Puchas, P., Hutterer, G., Barth, D. A., Terbuch, A., Bauernhofer, T., & Pichler, M. (2020). MicroRNAs as Appropriate Discriminators in Non-Specific Alpha-Fetoprotein (AFP) Elevation in Testicular Germ Cell Tumor Patients. Non-Coding RNA, 6(1), 2. https://doi.org/10.3390/ncrna6010002





