1. Introduction
Many biological cell types rely upon tail-like appendages known as flagella to propel them towards optimal environments or environments where the cell serves a specific function. These flagella must typically move through highly viscous fluids, perhaps in the presence of other objects and cells within the fluid. Collective motion of flagella has long been observed in cells. For example, in bacteria with multiple flagella, flagella can rotate in a helical fashion and form a bundle [
1,
2]. In mammalian sperm, cells have a single flagellum but flagellar motion may be impacted by neighboring sperm or other structures in the environment.
Sperm motility is a major indicating factor for fertility potential and thus sexual reproduction [
3]. Sperm must maneuver through a variety of environments in order to reach the egg and this process involves a complex choreography of biochemical reactions that mediate changes in motility patterns. These motility patterns are undulatory and often characterized by a nearly planar, sinusoidal beat form, though helical or “Figure 8” beat patterns have also been observed [
4,
5,
6,
7]. A planar waveform would result in sperm primarily traveling in a planar trajectory, but high resolution 3D trajectories of sperm have shown more intriguing patterns of helical or chiral ribbon trajectories over time [
8,
9,
10,
11].
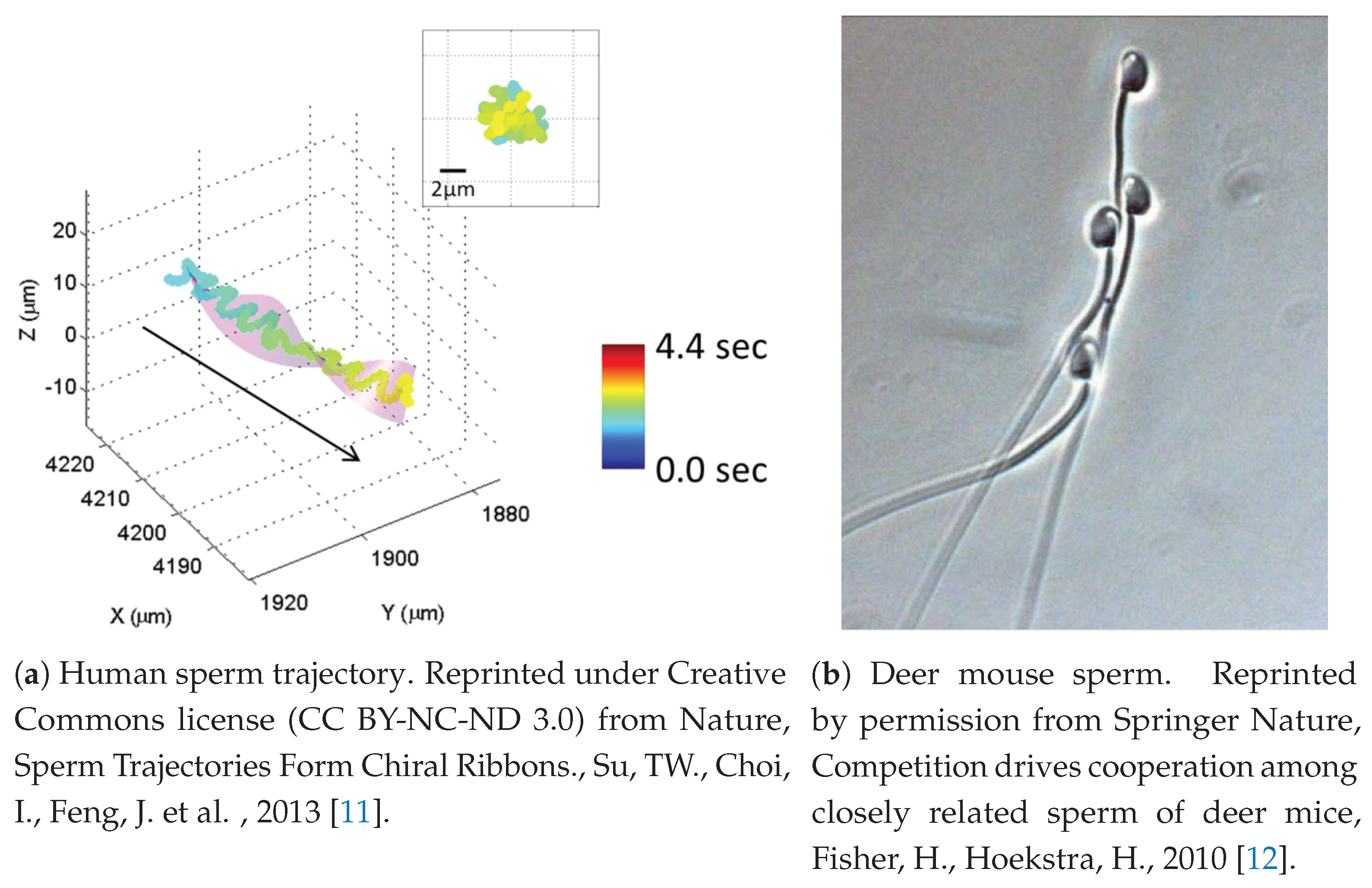
Figure 1a shows one example of a chiral ribbon trajectory observed in human sperm.
Species as diverse as deer mouse, cattle, fly, echidna and opossum have sperm that can collectively swim together [
12,
13,
14,
15,
16,
17,
18,
19].
Figure 1b shows an example of such behavior in the deer mouse, where apical hooks on sperm heads enable sperm to attach to a neighbor’s flagellum. This is sometimes referred to as a sperm train. Swimming with nearby neighbors makes for a highly complex fluid–structure environment in which there is feedback between the fluid and neighboring sperm that will effect velocities and trajectories significantly, and therefore has the potential to impact reproductive success. Moreover, complexity can arise from surface interactions between sperm and the oviductal epithelium, egg or other structures immersed in the fluid. For instance, it is known that sperm will repeatedly bind and unbind to the oviductal epithelium [
20,
21] and this behavior has an impact on sperm trajectories over time.
Undulating flagella and sperm motility have been the inspiration for mathematical modeling efforts for decades as an ideal system for studying motion in viscous fluids. Models for planar motion of flagella near surfaces includes investigations near a no-slip planar surface [
22,
23,
24,
25,
26] and a rigid sphere [
27]. In general, models have shown that individual sperm tend to be attracted to planar surfaces and stick to them unless the sperm change their waveform (i.e., increase amplitude, asymmetry, or wavenumber) and/or the sperm is kept a small distance away from the planar surface. Near a rigid sphere, sperm may more easily escape the spherical surface though this is dependent on geometry [
27].
Mathematical models for flagellar swimming in groups are an active area of research. Some studies have considered 2D fluids, which demonstrated a tendency of flagella to attract and that this attraction is energetically favorable [
23,
28,
29,
30]. In 3D fluids, models have shown both attraction and repulsion of neighboring flagellar pairs, but the results are dependent on configurations of the flagella, the fluid domain, timescales of simulation, and the flagellar model. In [
29], coplanar swimmers in 3D fluids had increased efficiency. The work of [
31] showed gains in velocity for shorter timescale simulations of both coplanar and parallel plane geometries, though results depended on phase. In [
32], it was proposed that coplanar attraction was an unstable state, as parallel and nearly coplanar swimmers tended to swim apart, in contrast to attraction seen for perfectly coplanar swimmers. Aggregation of sperm was shown to be a commonly observed behavior in a 2D-periodic thin film in [
33]. Interestingly, this aggregation was suppressed with the introduction of stochasticity in the beat frequency. A coarse-grained approach based on experimentally-derived beat patterns showed that sperm clustering is inhibited by low viscosity media and small changes in individual flagellar motion substantially affect clustering dynamics and collective motion [
34]. In [
35], the authors found evidence for stable pairwise swimming configurations and increases in velocities that may indicate an advantage to cooperative behavior. Moreover, transient attraction of pairs of swimmers and increases in velocities were also demonstrated for coplanar flagella in [
36].
Missing in these prior results are broader considerations of three-dimensional geometries of groups of flagellar swimmers with fine-grained models of flagellar motility on long timescales. This is primarily due to the high computational costs of these investigations but also due to restrictions in some modeling assumptions and frameworks. The purpose of this work is to address this gap and provide insight into how swimming in groups and near surfaces affects not only trajectories but also velocities, power expenditures, and efficiency.
The most realistic modeling framework for sperm motility would include a viscoelastic, non-Newtonian fluid model, detailed flagellar waveforms that can be modulated by interactions with other objects, the entire morphology of the sperm head, fully three-dimensional domains and geometries, external fluid flow, and biochemically-mediated interactions between sperm and their environment. At present, putting all of these components together is computationally prohibitive. Thus, prior work has all taken aim at specific pieces of the puzzle. As our interest lies in collective flagellar motion, our priorities are to consider a detailed model of the flagellum that allows for emergent waveforms as in [
32,
37] instead of prescribed waveforms as in [
38], simulate many flagella as done in [
33] but with a broader range of geometrical configurations as in [
31,
32,
35], with and without a surface present similar to [
25,
39] over longer time scales.
Recent work has simulated flagella in pairs for up to 10–20 beats [
35] but we will go beyond this here to address any lingering questions about long-range stability of flagellar interactions like those posed in [
30]. Simulations involving all of these components are computational expensive due to the finer scale details we aim to resolve but they are plausible, as long as we relax some of other ideal features of a model. In our case, we will focus on purely viscous, Newtonian fluid flow instead of non-Newtonian fluids like those modeled in [
30,
40]. We will also not consider sperm heads as done in [
26], external fluid flow such as [
41], or more detailed biochemical interactions like those described in [
37,
42]. These choices not only keep our simulations tractable, but also allow for direct comparison with prior, related work including [
32,
43].
As a first approximation, we will be using a planar flagellar motility model that is capable of bending in three dimensions when a flagellum is pushed out of its native plane. This model was developed in [
32] and is based on prior work developed in [
37,
44]. Here, we extend this work to consider groups of more than two flagella and add a new component to the model to allow neighboring flagella to attach along each other’s length to mimic apical hooks. This model is related to the work in [
33], but we do not consider a thin-film (a nearly 2D setting) nor do we include periodicity in our fluid environment. Therefore, the environments we are modeling are less spatially constrained and less dense than in [
33], but provide the ability to analyze a broad range of 3D configurations and yet still focus on individual interactions between swimmers. In comparison to [
34], our flagellar motility model is not coarse grained, allows for changes in the beat pattern when flagella encounter other objects within the fluid, and allows for a more detailed analysis of individual flagellar behaviors.
In this paper, we will consider groups of flagella up to 9, in a variety of configurations, both with and without surfaces present. We employ the method of regularized Stokeslets [
45] due to the need for computational solutions to solve the governing equations of viscous fluid environments. In this modeling framework, planar surface dynamics have been studied but only in more restrictive geometries for individual swimmers in [
25]. Therefore, this present work is a new exploration of surface dynamics for groups of flagellar swimmers, with no spatial restrictions for flagellar configurations. In this way, we will be able to explore the importance of swimming in groups in viscous environments and interpret the behavior of populations of sperm observed in 3D experimental imaging in [
8,
9,
10,
11] as well as the observed cooperative behavior we see in species discussed in [
12,
13,
14,
15,
16,
17].
2. Materials and Methods
The modeling framework we use is a simple extension of the work in [
32,
46]. We detail the components of the model below.
2.1. Fluid Model
Flagellar motility in realistic biological settings is characterized by highly viscoelastic, non-Newtonian fluid environments. In laboratory settings, fluid environments can be varied between viscous and viscoelastic regimes, both with low Reynolds numbers. For average mammalian sperm dimensions, the Reynolds number is on the order of
(see
Table 1). Recent work has highlighted that assuming a zero Reynolds number may not always be appropriate for modeling the motion of cilia and flagella [
47], but here we will assume a Newtonian fluid with zero Reynolds number for simplicity and to facilitate comparisons with prior work involving similar models. Thus, we model the fluid environment in 3D using the incompressible Stokes equations below:
In these equations, is the dynamic viscosity, is the fluid velocity, p is pressure, is the force density and and t are the spatial and temporal coordinates, respectively.
The flagella in our simulations exert forces along their length and therefore, we use the following formulation for
:
The integral term represents all forces that are exerted by the
flagellum.
L is the flagellum length,
s is the arc length along the flagellum, and
is the spatial position along the length of the
flagellum. The function
is an approximation to the Dirac delta function, often referred to as a blob function or regularized delta function. There are many choices for blob functions, but here we will employ one that has been used in previous studies related to this work [
32,
43]:
In simulations with a surface included, we require additional terms to enforce a no-slip boundary condition at the surface. We use the method of images to accomplish this, as derived in [
46].
2.2. Flagellum Model
The flagella in our model will all be identical, with the exception of their spatial positions and beat planes. Each flagellum is considered a three-dimensional curve in space with forces exerted along the curve. In order to conceptualize these forces, we define a local frame of reference for each flagellum m as follows: translate the flagellum position so its center of mass is at the origin, and rotate the flagellum so its beat plane is . We will refer to flagellar coordinates in the local frame of reference as .
The forces that govern the motion of the flagella include tensile forces that keep the curve connected and roughly inextensible, bending forces that actuate the motion of the curve, and planar restriction forces that keep the curve in a nearly planar configuration. Each of these forces is derived from the following continuous energy formulations:
Equation (
4) is the continuous version of a tensile energy of an ideal spring model. Equation (
5) is a bending energy that actuates the flagellum to move towards the planar preferred curvature given by
, which is chosen to mimic sinusoidal flagellar undulation by setting
The parameter
is used instead of
s here, because while the flagellum can be in a three-dimensional configuration, the beating behavior is planar and therefore
is the projection of the true arc length
s onto the flagellar beat plane. Similarly,
ℓ represents the full arc length of the flagellum after projection onto the beat plane. Equation (
6) is the planar restriction energy, which is an energy that measures local deviations out of the beat plane. The vector
is the normal vector to the beat plane in the local frame of reference. All
values are stiffness parameters. In particular,
is chosen to be relatively stiff to enforce inextensibility of the flagellum. For further details, see [
44] for the derivation of Equations (
4) and (
5) and [
32] for further explanation of Equation (
6).
In the local frame of reference, forces exerted along the flagellum length are then given by:
where
. The forces
are then found by rotating and translating
back into the original frame of reference for
. Such forces move the flagellum towards a minimal energy configuration.
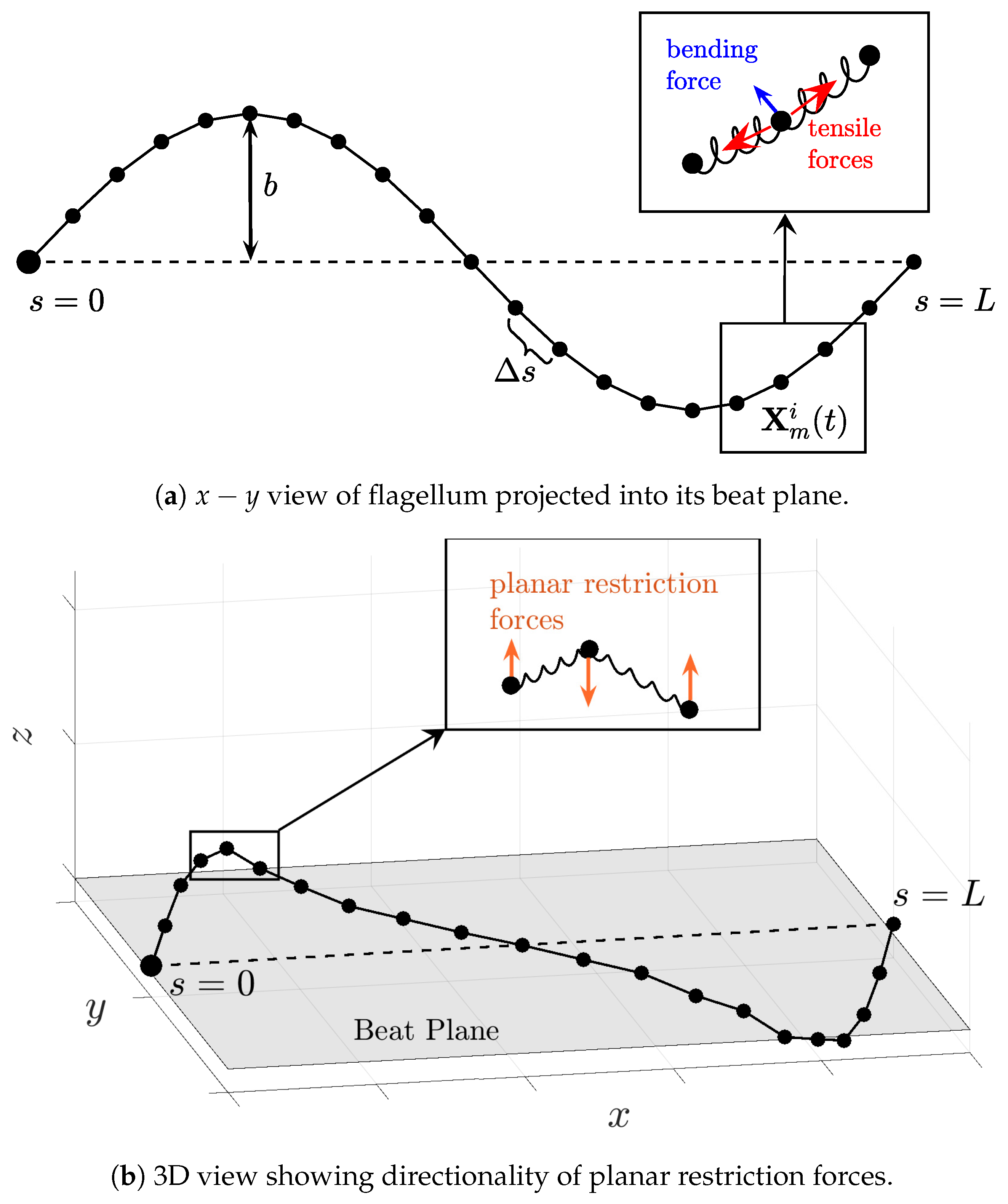
Because we will be simulating flagellar motion computationally, we discretize the spatial coordinates
and the forces
by evenly distributing points along the flagellum as depicted in
Figure 2. Thus, we will be modeling the motion of each point
(the
point along the
flagellum) that arises from discretized forces denoted by
, which are derived from discretizing the energies in Equations (
4)–(
6) along each flagellum.
Figure 2 is a schematic representing the forces derived from Equations (
4)–(
8) and what they mean in the 2D beat plane, as well as the forces out of the beat plane, resulting from Equation (
6) specifically.
As done in [
32], we add an additional repulsive force to each
to prevent flagella from unrealistically passing through each other. We denote this repulsion force by
and define it as:
where
is given by
and
is a small threshold distance in which the repulsion force is added. This effectively keeps individual points (denoted by
i and
j) along neighboring flagella (denoted by
m and
n, respectively) from overlapping.
This model for flagellar motion allows for waveforms to emerge and be modulated by their external environment. Such modulations would occur when flagella are close enough to surfaces and other flagella. This flagellar model is computationally expensive when compared to coarse-grained flagellar models (such as [
34]) or prescribed motion models such as [
31,
39], but necessary so we can pursue a more detailed analysis of flagellar cooperativity and surface interactions.
2.3. Coupling the Flagella and Fluid Environment
Forces that the flagella exert in our fluid will move the fluid in accordance with the incompressible Stokes Equation (
1). If each force were modeled as a point force, the solution to the fluid velocity field could be found using a summation of Stokeslets (the fundamental solution to the incompressible Stokes Equation (
1)), due to the linearity of these equations. As we aim to simulate flagellar motion computationally, we use the blob function given in Equation (
3) and therefore solutions will be a summation of regularized Stokelets, which are solutions to the incompressible Stokes Equations derived from the blob function [
45]. In our case, the fluid velocity in free space is:
where
and
is a small parameter. This is a summation of regularized Stokeslets over all
m flagella and all
i points along each flagellum.
2.4. Model for Apical Hooks
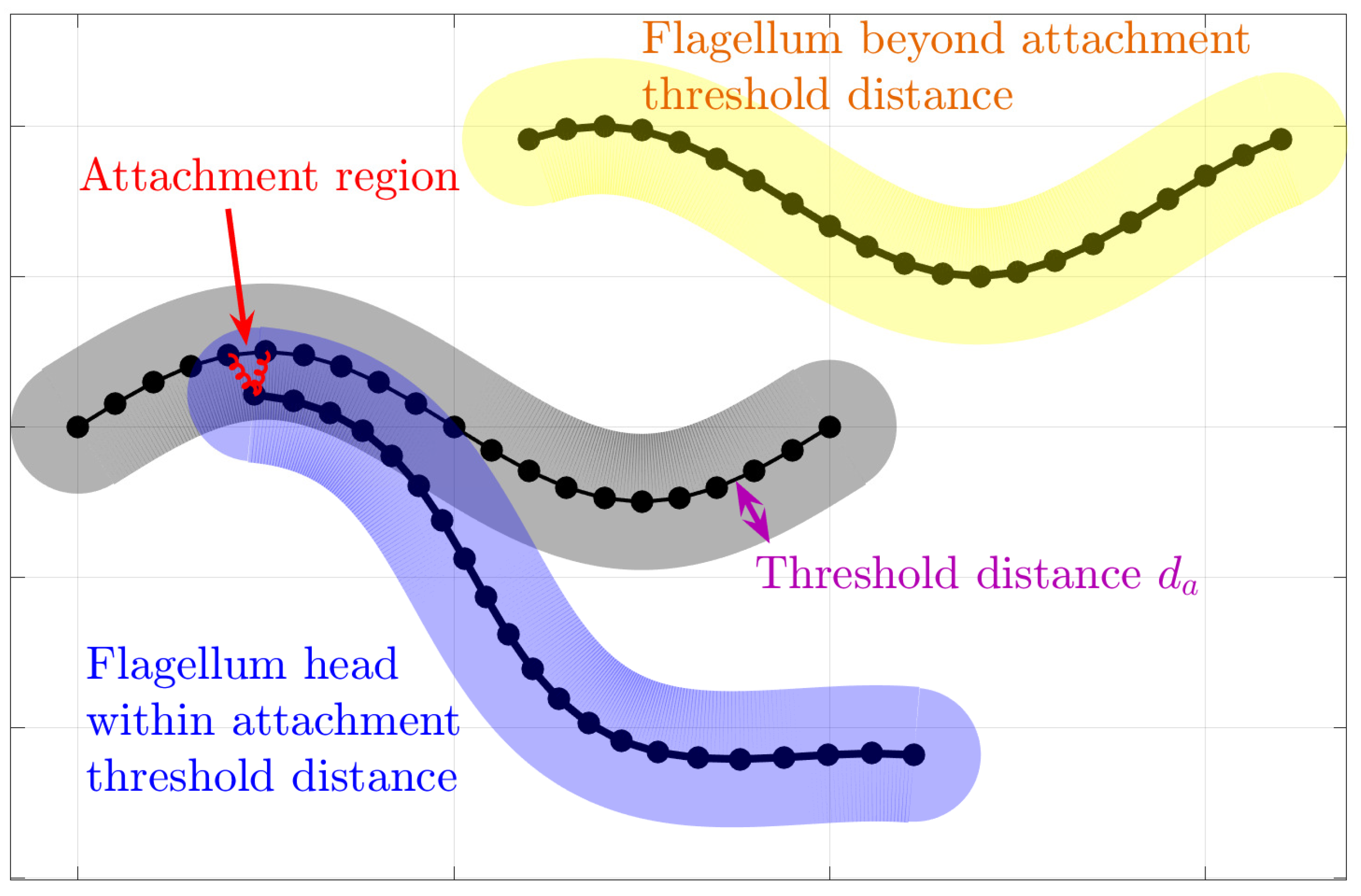
In an attempt to understand the collective motion of sperm that have been observed to attach to each other, we choose to focus on the sperm train behavior shown in
Figure 1b, which has been observed in deer mice as [
12]. These so-called trains arise, at least in part, due to an apical hook on the sperm head that allows a sperm to attach to the flagellum of another sperm. Instead of modeling the morphology of the hook that can attach to neighboring flagella, we simply add forces at the head (initial point) of flagellum
m and along the neighboring flagellum
n if the head of flagellum
m gets within a threshold distance,
, of the neighboring flagellum
n.
The forces added are a simple spring force model that effectively holds the head of the flagellum close to the neighboring flagellum points, with a fixed resting length
a. The mathematical formulation to model for this attachment behavior can be expressed as:
Note that this force is applied at the head (first point) of the flagellum
m but also in equal magnitude but opposite direction at nearby points
j along the neighboring flagellum
n, so both flagella will be impacted by the attachment.
Figure 3 shows a schematic of the attachment model.
This is not a permanent bond between the flagella, but rather forces that persist as long as the head of one flagellum remains close enough to another flagellum. This attachment does not prevent rotation of the flagella, or translational movement of the flagella along each other. These details are important as a hook around a flagellum might allow for some motion up and down the flagellum, and would not necessarily prevent rotation around the hook attachment region.
We note that the model described in Equation (
11) is similar to models used in other investigations involving cell adhesion. For instance, a similar spring-like adhesion force to model receptor-ligand binding between particles and walls in the context of blood flow has been used for many years [
48,
49,
50,
51]. Our model differs in four main ways from such work. First, these works are considering whole cells with distributions of many nano-structures (receptors) on their surface that can bind to ligands on epithelial surfaces. Our model has no surface of a cell, but rather a single point at the head of the flagellum that can bind. Second, bond formation and dissolution is different: for our model, the apical hook force is applied only when flagellar heads are within a fixed distance of another flagellum, whereas there is a probabilistic approach for forming and breaking bonds in [
48,
49,
50,
51]. Third, we are modeling a much larger structure than receptors and ligands. An apical hook is a macrostructure of the sperm head and would not necessarily experience the same types of probabilistic effects of forming and breaking bonds that we see in receptor-ligand dynamics. Lastly, our apical hook model enables binding between one flagellum and another flagellum, which are both freely moving curves in space, rather than a cell body and a surface.
2.5. Computational Method
Our computational method is as follows:
2.6. Parameters and Initial Configurations of Flagella
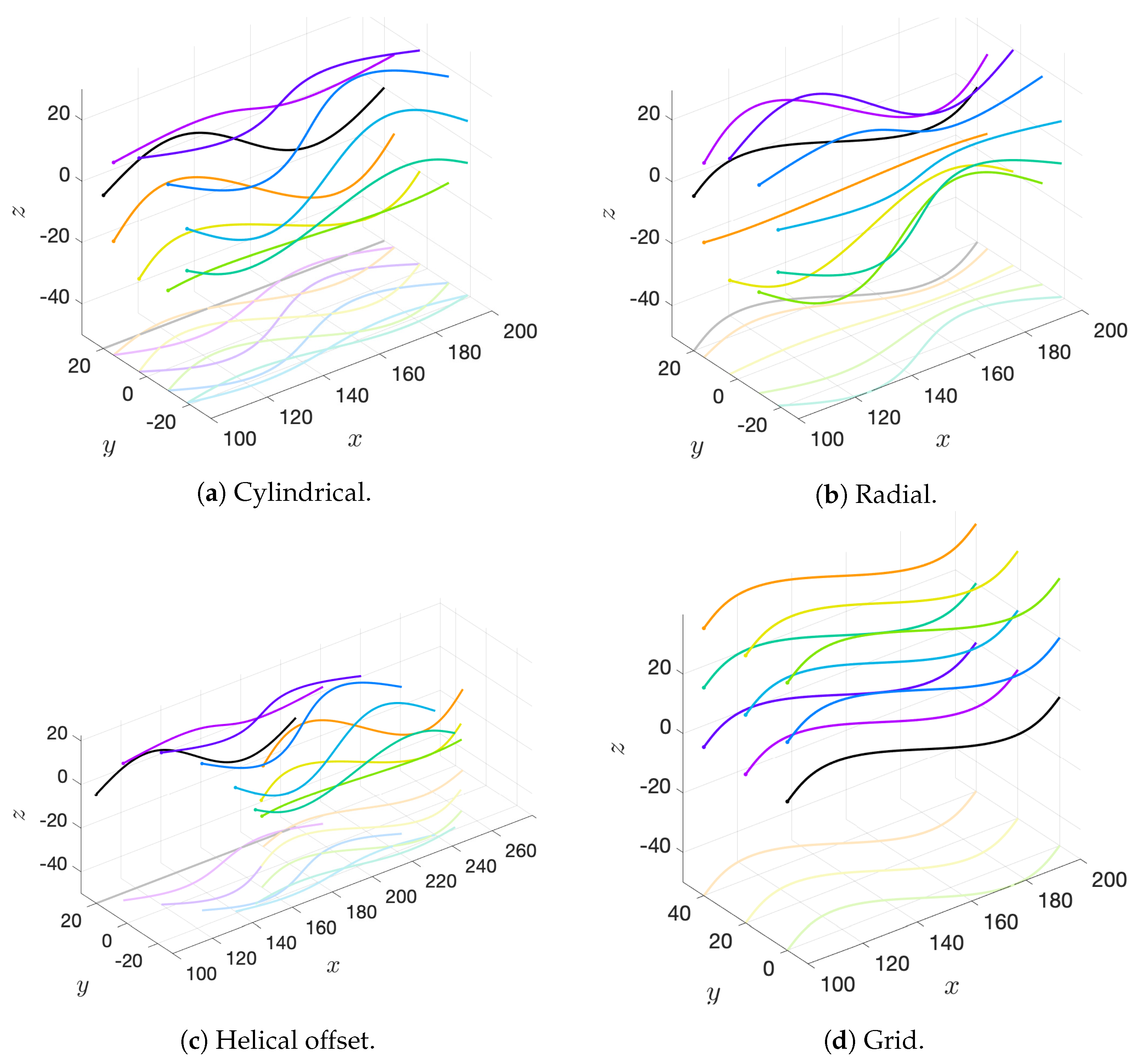
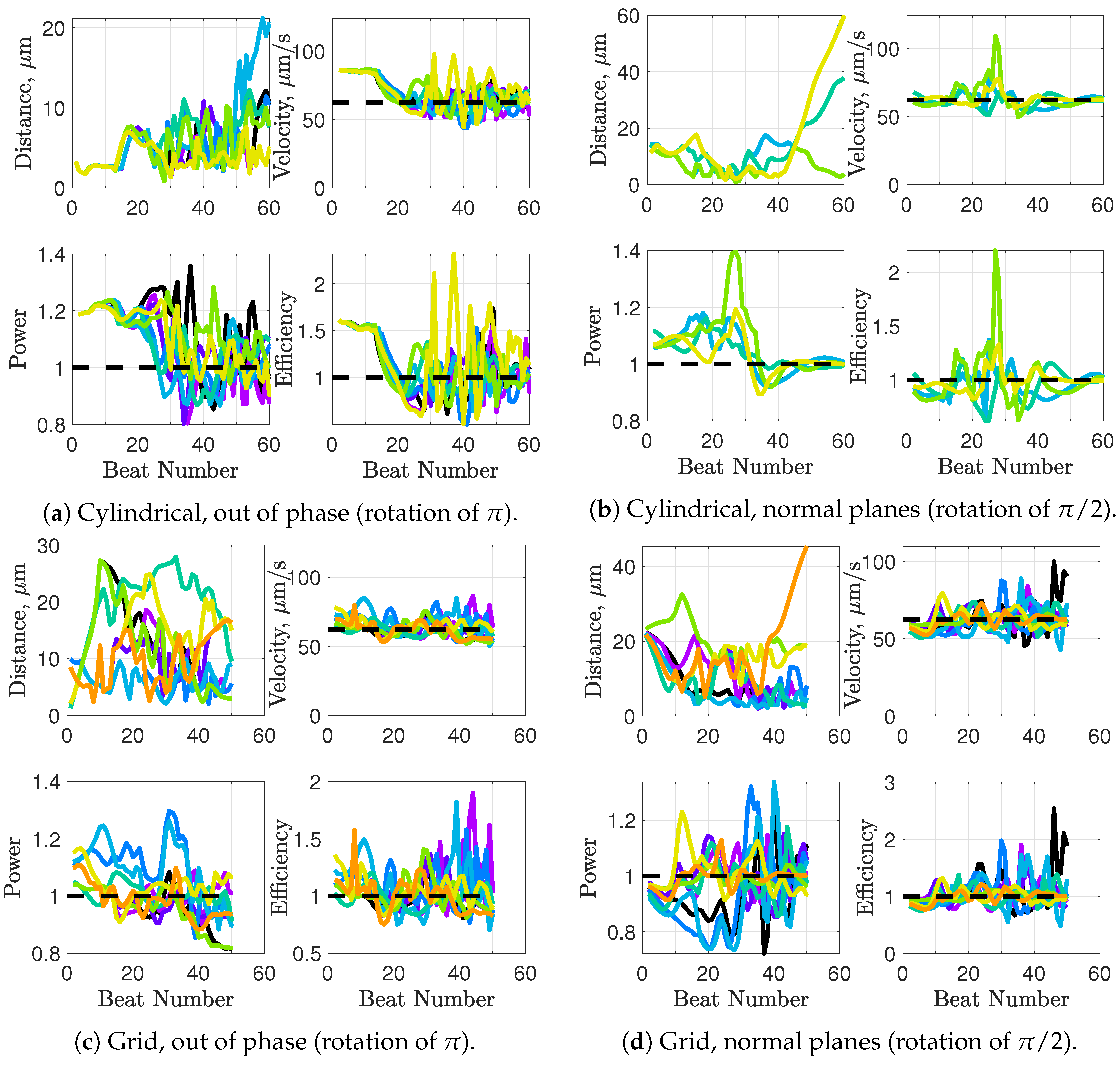
We will consider a variety of initial configurations of flagella, which are based on geometries that allow for an exploration of symmetry and phase. While there are infinitely many configurations that we could consider, we restrict ourselves to the following scenarios in order to exploit symmetry and compare and contrast effectively:
Cylindrical: flagellar planes tangent to a cylinder;
Radial: flagellar planes normal to a cylinder;
Helical offset: cylindrical pattern of neighboring flagella set back along a cylindrical surface to form a helical shape; and
Grid: all flagella are aligned with bodies in grid patterns.
These basic initial configurations are shown in
Figure 4 for groups of 9 swimmers.
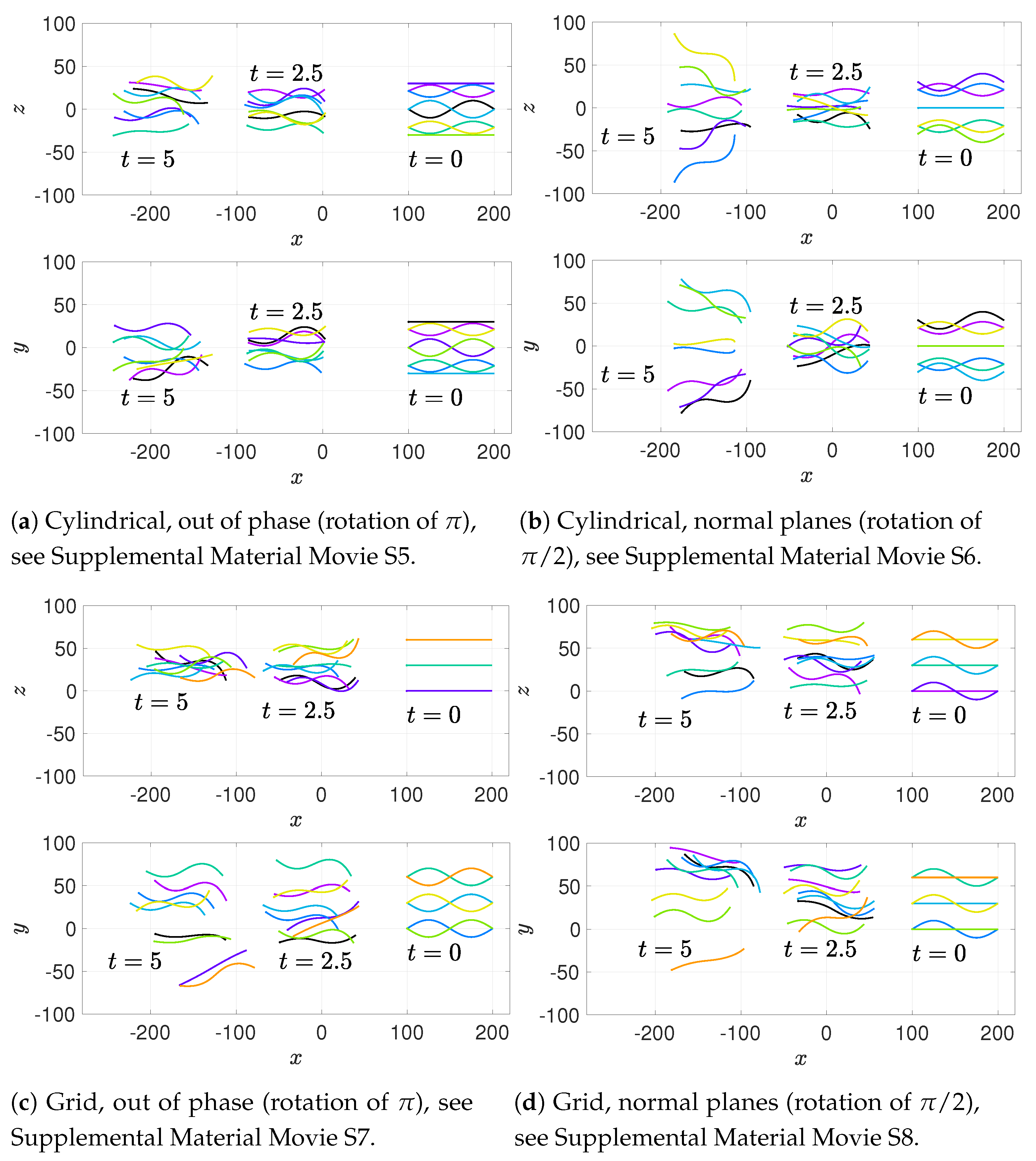
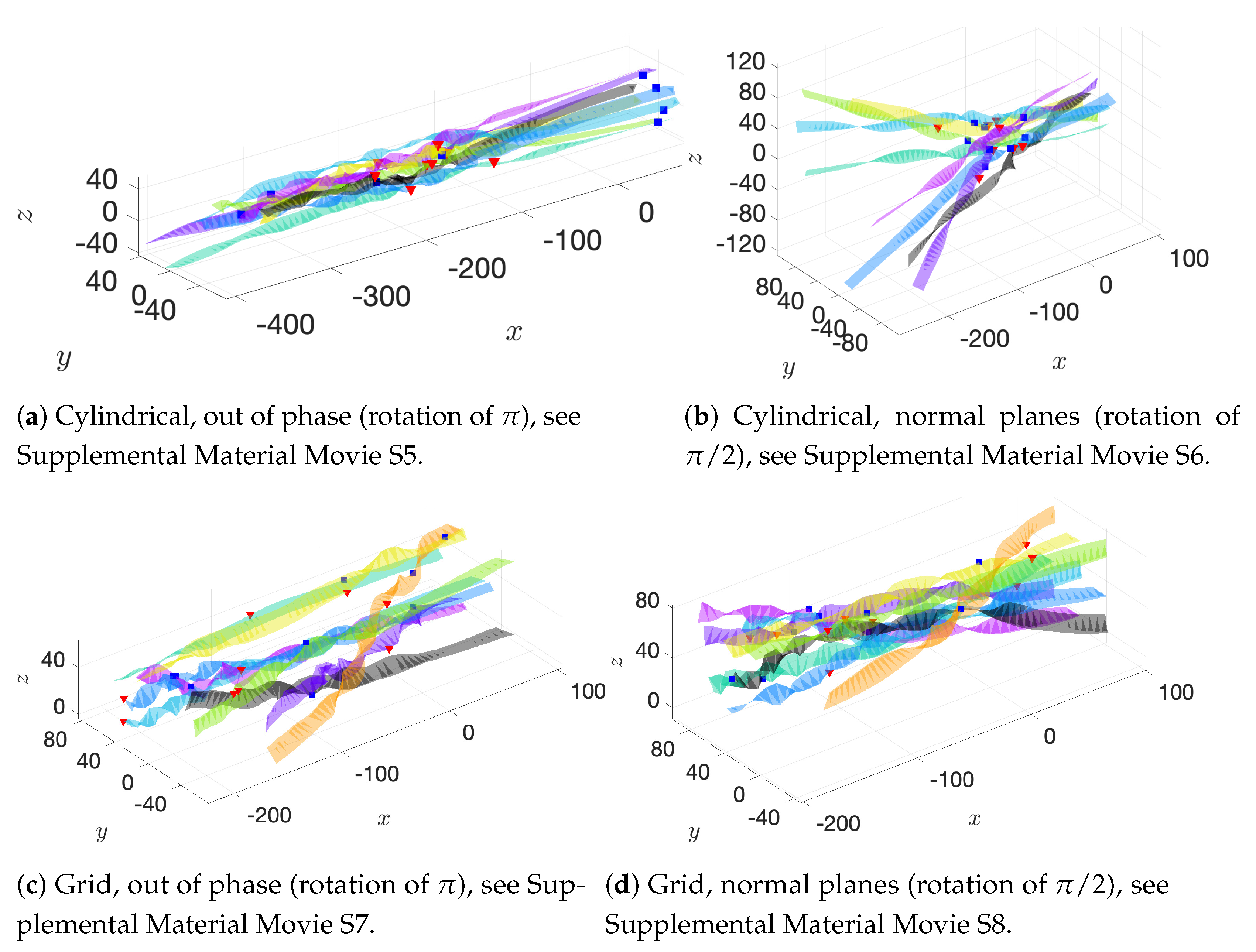
In addition to these basic initial configurations, we also wish to consider phase or alignment differences between flagella. To do so, we rotate nearest neighboring flagella along their centerline axis by one of two angles:
: we will refer to this as a “normal plane” configuration. Neighboring flagella have beat planes that are normal or nearly normal to each other. Motion of neighboring flagella are neither in phase (aligned) nor in opposition.
: we will refer to this rotation as an “out of phase” configuration. Neighboring flagella will be completely out of phase or nearly out of phase, but with similar beat planes. Similar here means identical, parallel, or nearly parallel beat planes. Motions of neighboring flagella are generally expected to push and pull in opposition to each other.
While we are not changing the phase of flagella explicitly, the rotations do enable some exploration of the role of phase. See
Figure 5 for a graphical representation of these rotations.
Figure 6a–c show depictions of how we combine the alignment schemes of
Figure 5 with the basic initial configurations introduced in
Figure 4.
Figure 6d–f show the initial configurations we will use when considering a planar surface (or wall) nearby. Lastly, all parameter values are listed in
Table 1.
Table 1.
Parameter values. Sperm parameters were set to that of a typical mammalian sperm with active motility. Wherever possible, biologically-measured values were used. In absence of biologically-measured values, values from prior modeling work were used, if they exist.
Table 1.
Parameter values. Sperm parameters were set to that of a typical mammalian sperm with active motility. Wherever possible, biologically-measured values were used. In absence of biologically-measured values, values from prior modeling work were used, if they exist.
| Parameter | Parameter | Parameter | Remark/Reference(s) |
|---|
| Regularization parameter | 1.3 m | [45] |
| | Diameter of sperm flagellum | 0.5 m | [52] |
| Viscosity (water) | 10 kg m s | |
| Spatial (arc length) discretization | 1 m | Set to be [25,32] |
| L | Flagellum length | 100 m | [53] |
| b | Amplitude | 10 m | [54,55] |
| Wavenumber | | [5] |
| Frequency | 20 (10 Hz) | [54,55] |
| Tensile stiffness | 2 pN m | [25,32] |
| Planar restriction stiffness | 1 pN m | [32] |
| Planar bending stiffness | 10 pN m | [37] |
| Repulsion stiffness | 5 aN m | [32] |
| Attachment stiffness | 20 fN m | Set to be , this work |
| Repulsion length | 3 m | Set to be [32] |
| Attraction threshold | 10 m | This work |
| a | Attachment length | 6 m | Close to sperm head dimensions [53] |
| V | Average path velocity | – m/s | [56] |
| Density (water) | kg m | |
| Reynolds number | | |
4. Discussion
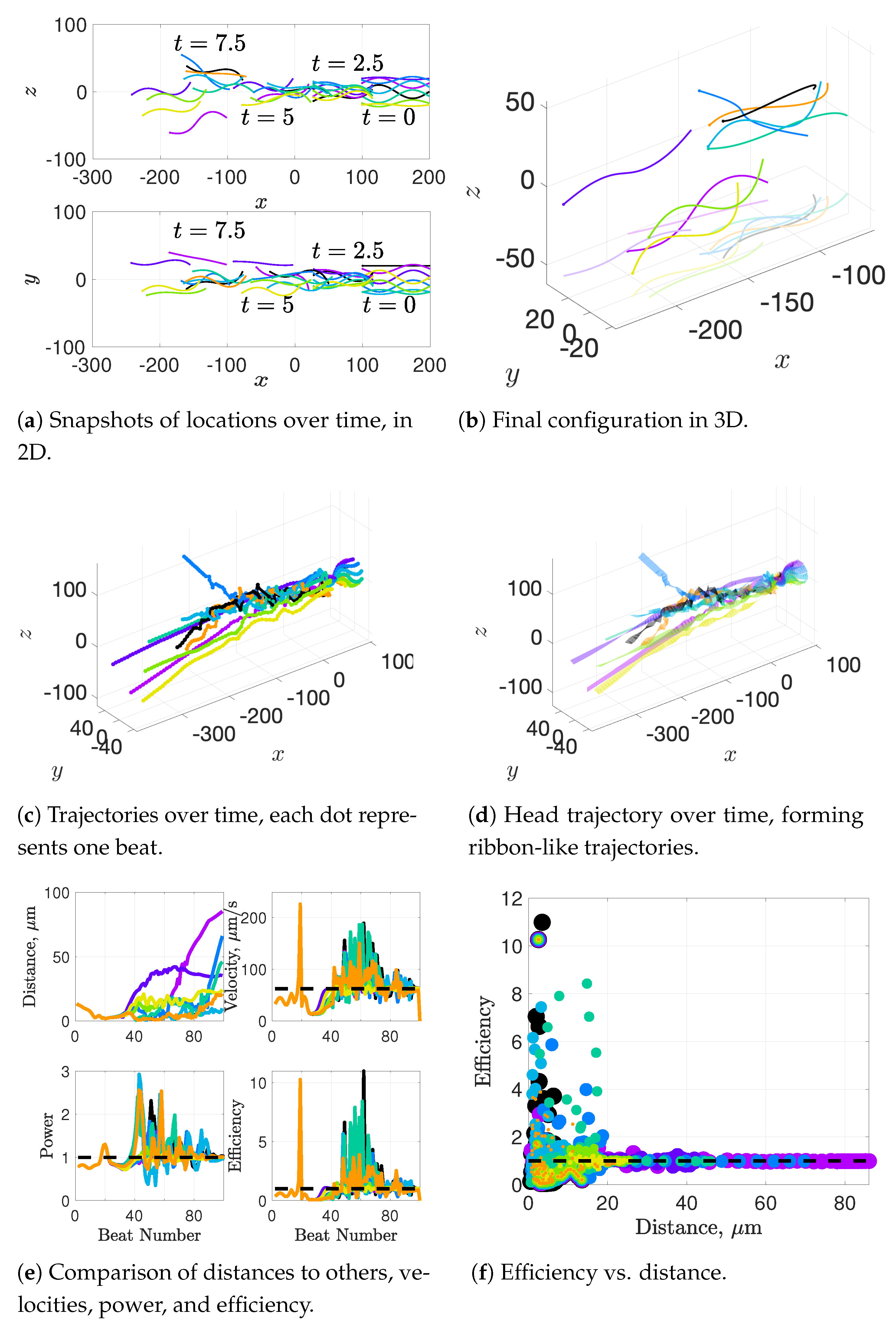
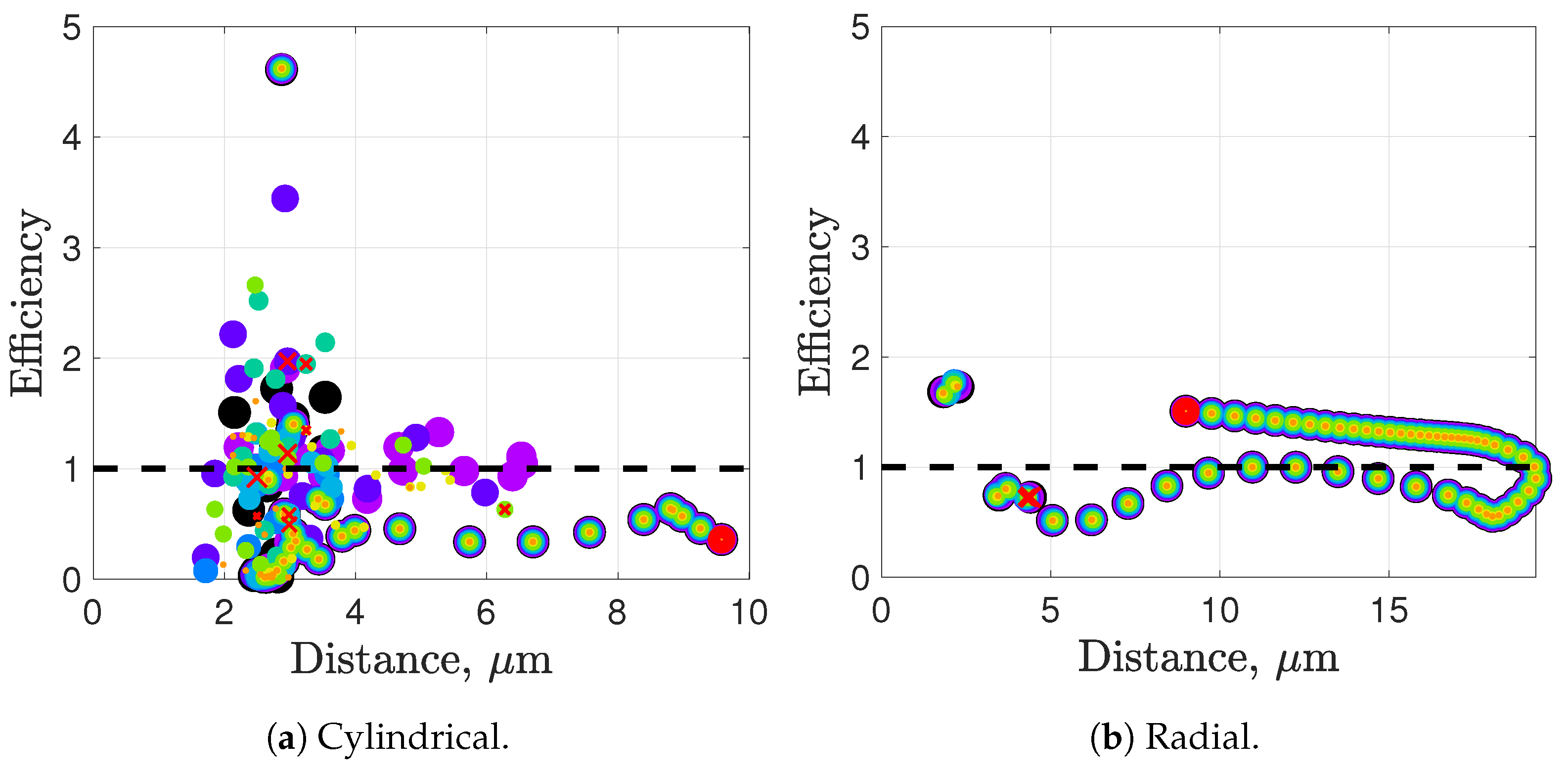
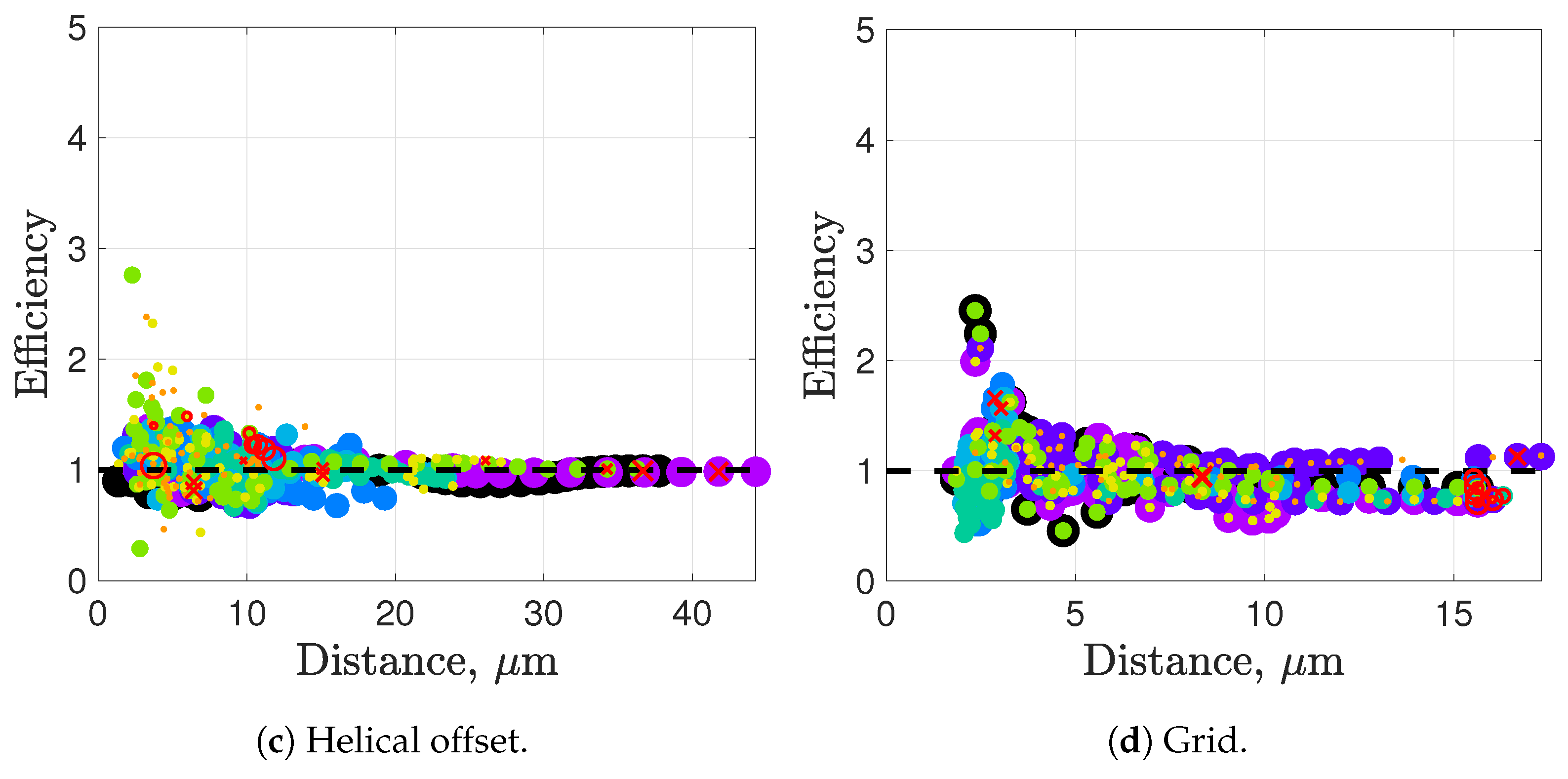
Synchronization and attraction of undulating, planar flagella have been studied for quite some time using both mathematical models and biological experiments. In particular, one theory has been that flagella tend to attract due to energetically favorable conditions, from the perspective of optimizing velocity or efficiency. Our general observations of flagella swimming in groups can be summarized as follows:
Flagella with similar beat planes tend to attract initially. If their beat planes are identical, attraction is a stable long-term behavior. In all other geometries, attraction is transient.
If beat planes are nearly identical, velocities and efficiencies decrease as flagella attract.
Flagella in non-identical beat planes tend to swim apart in the long term.
Flagella can rotate to have beat planes that are closer to nearby neighbors and this is often accompanied by a transient attraction phase, with decreases in velocities and efficiencies.
Flagella do not tend to swim in higher efficiency configurations unless there is another mechanism for keeping flagella near one another, such as apical hook binding.
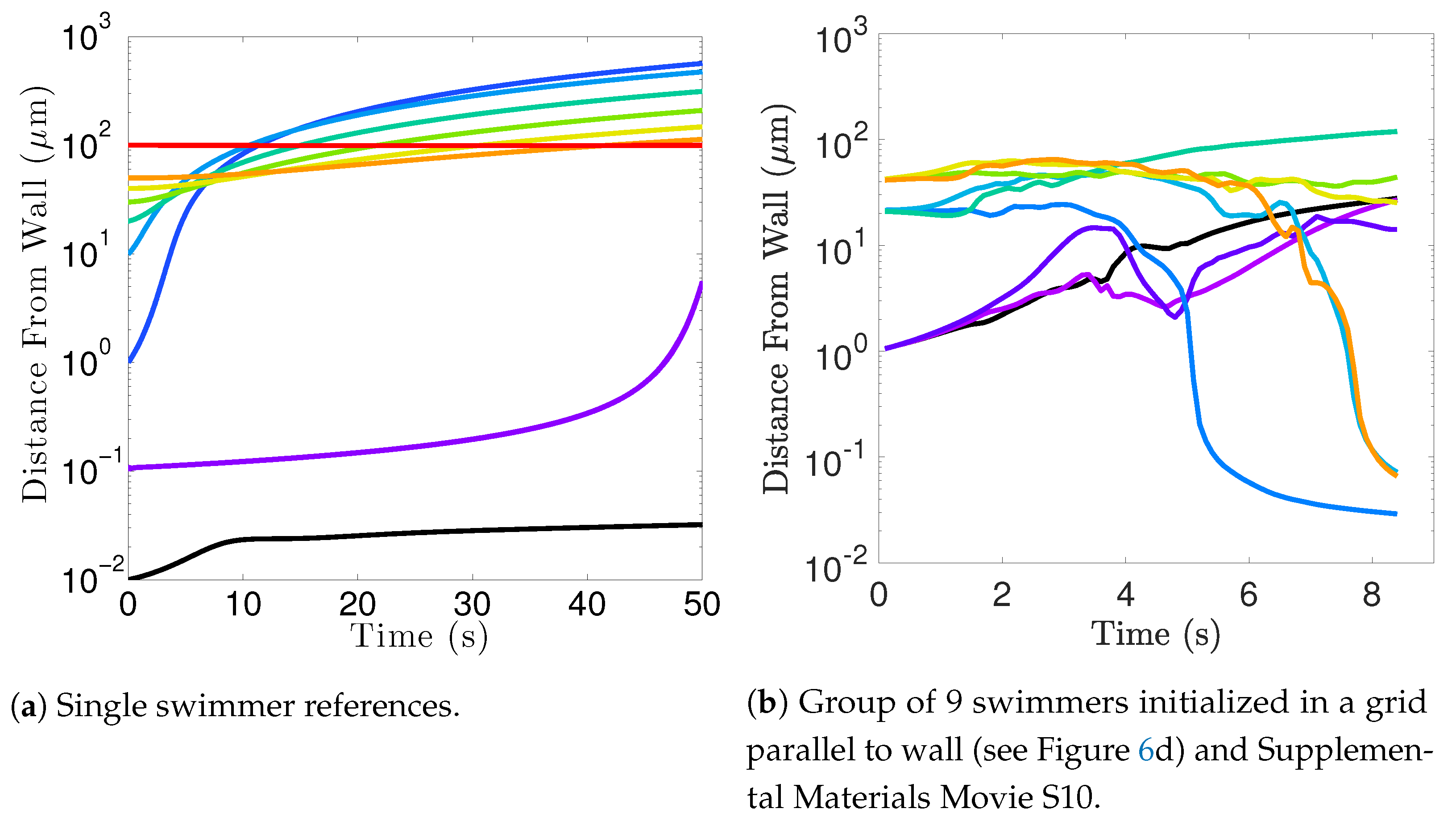
Near surfaces, having more than one flagella present may promote aggregation unless sperm are able to bind to one another and swim as a collective with higher velocities and powers.
These results are consistent with many prior results for coplanar flagellar geometries. For instance, in [
28], a 2D model showed attraction amongst many flagella. A comparison of pairs of coplanar flagella in 2D and 3D fluids was done in [
29], in which attraction and synchronization was observed in both fluids, and efficiency as well as velocity were shown to increase in some parameter regimes. In [
30], pairwise attraction of flagella was demonstrated in a 2D fluid with a resistance term meant to mimic swimming through a complex fluid containing other cells and polymers, though the question of long-term stability of this attraction was posed due to final configurations of the simulations. The work in [
33] also included a 2D framework with large populations of flagella (10–1000) and clustering phenomena were demonstrated, though the clustering was sensitive stochasticity in beat frequencies.
Transient attraction dynamics (followed by divergence in longer timescales) have more recently been observed in a model proposed in [
36] which considered two coplanar flagella–a scenario in which our model has been shown to have
stable attraction dynamics in prior studies [
32]. The flagella in [
36] are based on a geometric clutch model instead of the preferred curvature model we present here. This difference in modeling framework might explain the discrepancies between our coplanar results and that of [
36]. On the other hand, the authors in [
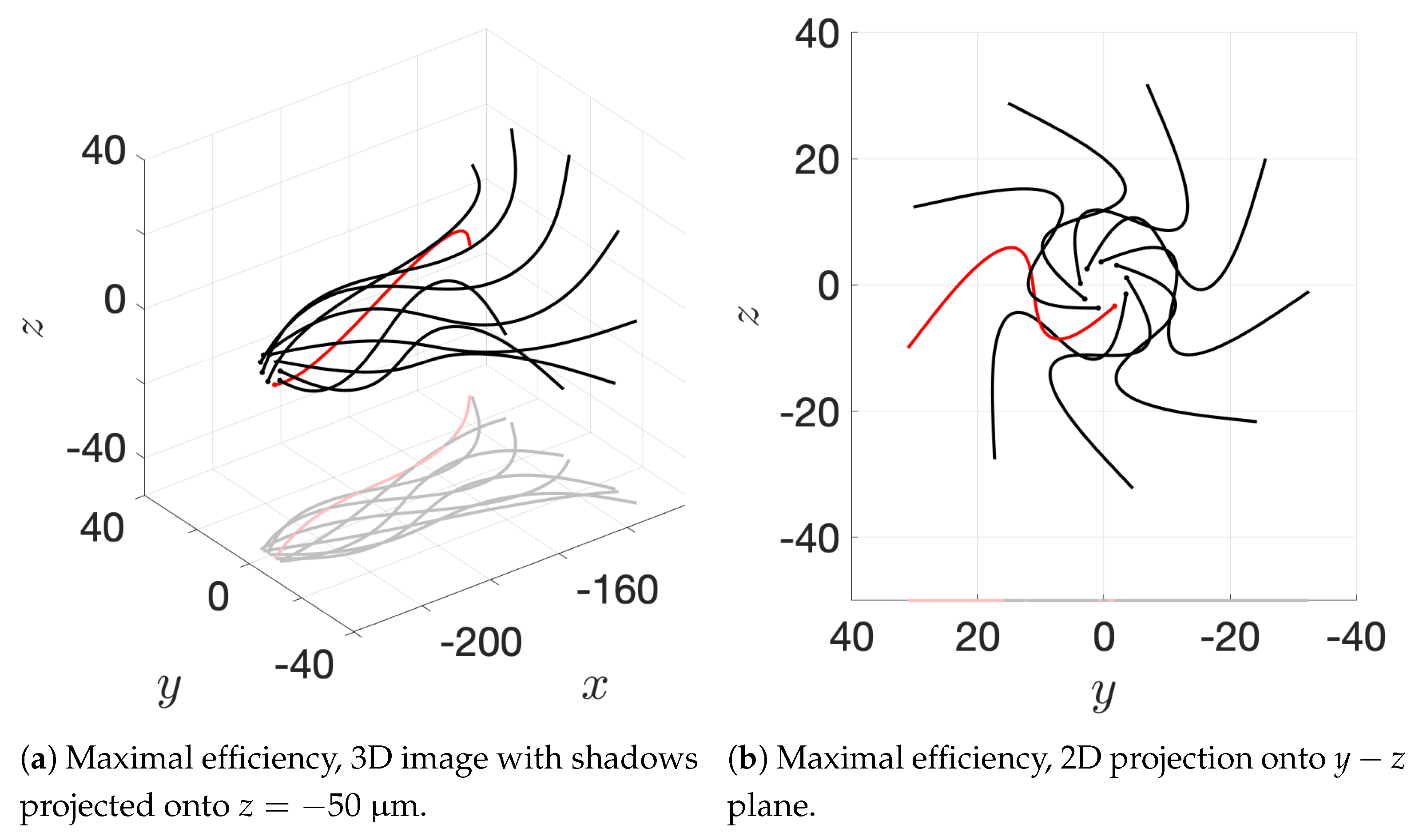
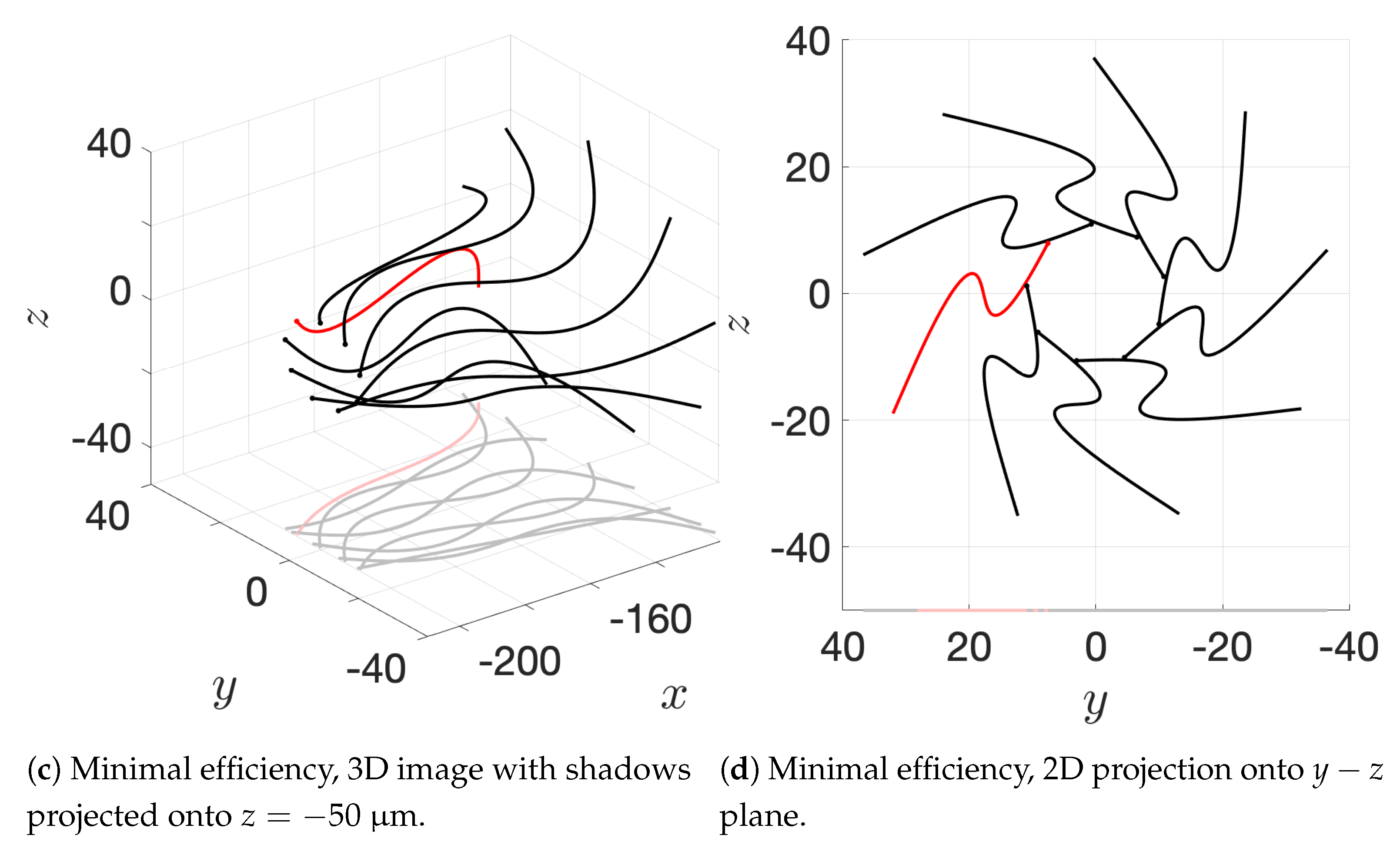
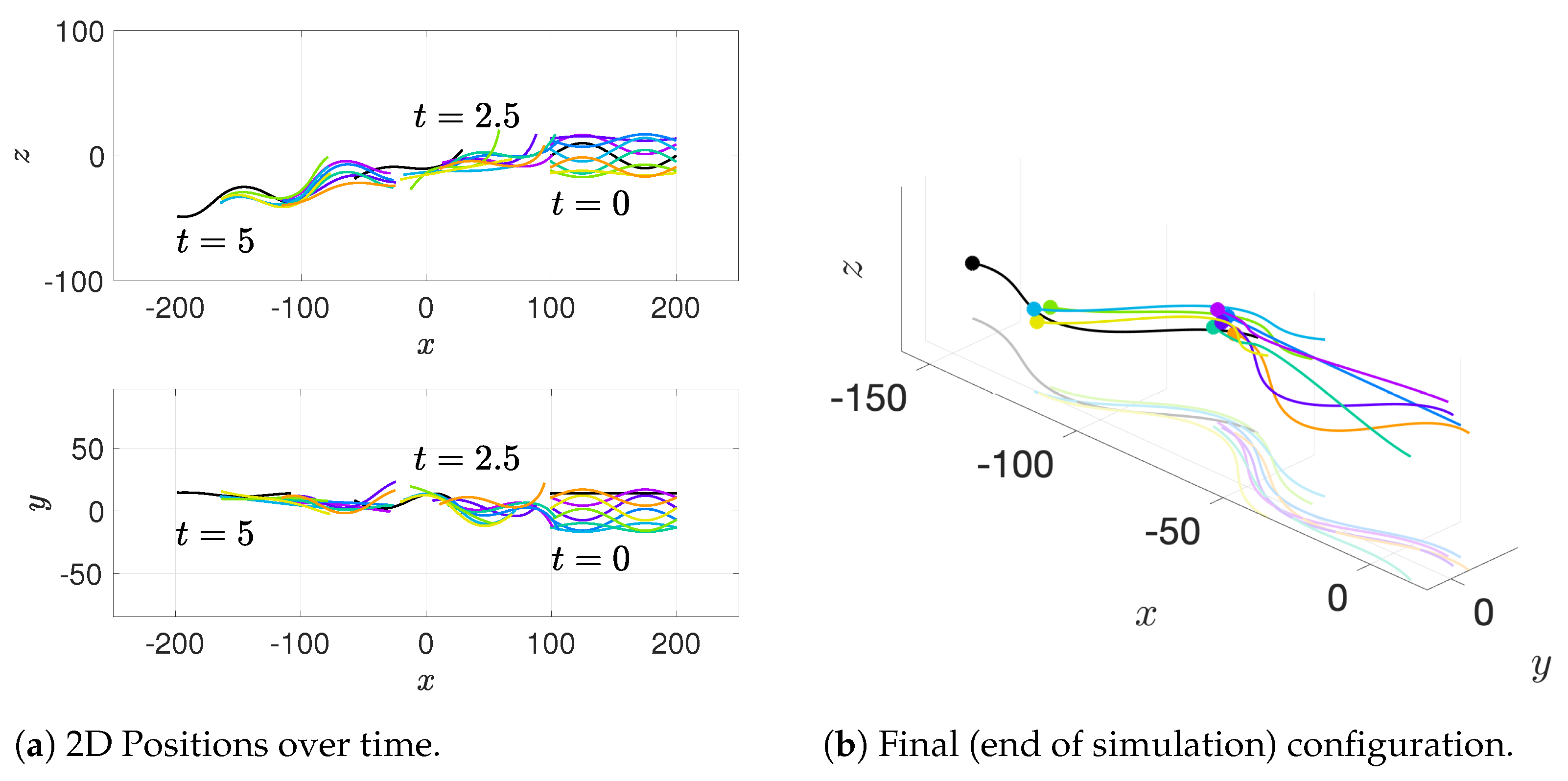
36] observed increases in velocity for sperm that are staggered behind one another. In general, these results are in agreement with our models for flagella that have offset heads (i.e., free-swimming results in from the helical offset configuration shown in
Figure 10c and the apical hook/sperm train results shown in
Figure 18a), though our flagella are swimming in more complex 3D configurations.
Recent, [
35] used 3D simulations to show attraction for a pair of flagella and, intriguingly, a stable steady state configuration for a pair of nearly parallel swimmers using simulation lengths of 20 beats. It is worth noting that in many of our simulations, attraction can be observed in the first 20 beats but the divergent long-term behavior we are presenting here is for beat numbers greater than 50. Moreover, our work goes beyond pairs to consider a larger number of flagella and thus, more degrees of freedom in geometric complexity. Our velocity results are in agreement with [
35]: swimming side-by-side in a plane seems to slow swimmers down, but swimming in parallel or nearly parallel planes can increase velocity.
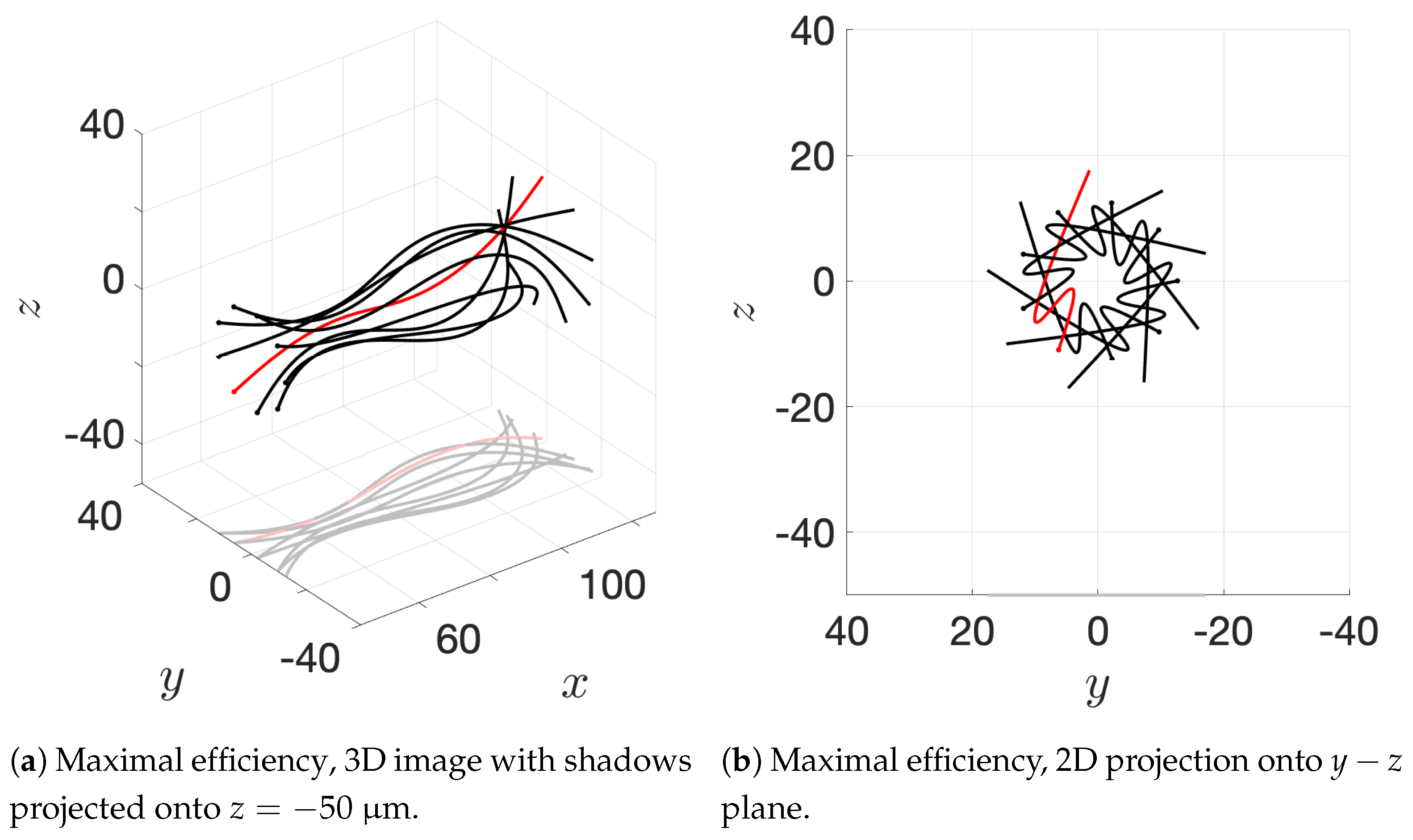
Our results show that flagella swimming in groups will frequently swim in helical or chiral ribbon-like trajectories, despite having a planar flagellar waveform. This is due to hydrodynamic coupling with other nearby flagella and evidence that a non-coplanar waveform is not required for such trajectories. This may provide insight into experimental observations that sperm can swim in helical or chiral ribbon-like trajectories [
8,
9,
10,
11], as some have wondered whether results like these indicate a non-planar waveform is more biologically accurate and whether non-planar waveforms are important to sperm motility and function. This work suggests that chiral ribbon trajectories are a natural result of a planar waveform combined with complex 3D fluid–structure interactions, which includes swimming in groups.
Biological settings for sperm involve interactions with surfaces that are complex and can affect motion significantly [
20,
21,
60,
61]. While we have not done an exhaustive study of the impact of surfaces in populations of flagella here, our model does indicate that swimming with nearby neighbors can enhance surface accumulation. It is known that flagella attract and accumulate near surfaces, and our results add to the complexity of the picture by considering the role of neighboring flagella in this accumulation process. Further exploration is warranted, particularly given results regarding individual flagella near surfaces. In particular, surface accumulation dynamics can depend on wavenumber [
26] and can be modulated by changes in waveform as well as binding dynamics of the sperm heads to epithelial surfaces [
24,
25,
39].
In [
43], the authors found that a pair of flagella attached at the head had a small window of geometrical configurations in which both velocity and efficiency were increased beyond that of an individual swimmer. Ultimately, the maximal efficiency configuration coincided with biologically-observed geometries of fused heads of opossum sperm. This paper extends that work to investigate apical hooks as a means for linking sperm flagella. Sperm trains naturally arise, again matching observations but this time in deer mouse sperm in [
12].
The authors in [
19] found evidence that clustering of sperm is functionally important for fertilization potential and our model demonstrates that there is certainly an advantage to clustering or swimming together in terms of velocity and efficiency. With respect to sperm trains specifically, when we time-average over entire simulations, we observe substantial increases in velocities for sperm trains when compared to individuals. The average velocity of the train of flagella was 15% higher than an individual swimmer with a maximum velocity that was 225% higher than an individual swimmer. Increases of around 50% in velocity were measured in the experimental work [
12], so our results underestimate this value but seem reasonable. One reason why we might not fully capture the increases in velocity observed experimentally is that our flagella are idealized to all start in the same phase and cannot modulate this over time. We plan on exploring this in future work. Finally, our model provides efficiency measures that indicate that over the span of our simulation, a train swims on average 50% more efficiently when compared to an individual swimmer. Thus, our model demonstrates that collective swimming can lead to significant hydrodynamic advantages.
5. Conclusions
Flagellar and ciliary transport is necessary for motion in biological systems well beyond the fertility applications discussed here and can only be understood by using a combination of experimental observation and mathematical modeling. For problems at these scales, laboratory measurements of forces, power, and efficiency are difficult, if not impossible, to obtain. Over the last 20 years, the method of regularized Stokeslets [
45] has enabled us to effectively model and shed light upon flagellar and ciliary mechanics due to its ease of implementation and the ability to extend the method to a variety blob (regularized delta) functions, control for different levels of desired computational accuracy, incorporate nearby surfaces and effectively study velocities, forces, power, and efficiency simultaneously.
In this paper, we used the method of regularized Stokeslets to show that collective swimming appears to not be a stable long-term behavior of populations of free-swimming flagella exhibiting planar waveforms in a viscous fluid, regardless of velocity and efficiency advantages. If the beat patterns of neighboring flagella are in direct opposition, this may drive flagella apart while velocities and efficiencies are enhanced. If beat patterns of neighboring flagella are more aligned or in sync, then flagella cannot push against each other effectively to swim faster or more efficiently though attraction may occur. Somewhere in between these two behaviors may allow for the most effective propulsion. Increases in velocity and efficiency are most dramatic when flagella are allowed to attach to each other via some physical binding mechanism–and in these cases, the flagella tend to align geometrically to synchronize their beat.
This work is the first to examine collective motion of larger groups of free-swimming flagella with a finer grained model across a large breadth of geometrical configurations over such long timescales. This is also the first model for apical hook binding. Our results also provide the first evidence that helical or chiral-ribbon trajectories can arise not from three-dimensional waveforms but as a consequence long-term interactions with other swimmers. We have also demonstrated strong hydrodynamic evidence for advantages to swimming in a sperm train, and the long-range instability of swimming in groups unless a binding mechanism keeps swimmers close to each other.
The hydrodynamic advantages of collective flagellar motion we have shown here may elucidate evolutionary benefits of sperm morphologies and behaviors in some species. In particular, deer mice that exhibit sperm aggregation or train-like behavior are known to be promiscuous and thus sperm competition may be a primary driver for sperm cooperativity [
12]. If competition is high, it makes sense that cooperative mechanisms would arise that enable sperm to swim faster and more efficiently to reach the egg. Even without the complexity of sperm competition, harsh oviductal environments and a short time period of viability for sperm to fertilize the egg also lead to strong evolutionary pressures for sperm to swim quickly and effectively towards the egg.
There are many studies and extensions of this work yet to be done. In particular, there are two fluid assumptions we have made that should be investigated. First, we have assumed a zero Reynolds number, but recent studies of ciliary motion indicate that this simplification may not capture the fluid behavior appropriately [
47]. Second, collective swimming has been observed to be important in fluids that are not only viscous, but also elastic [
18]. There is also potential to extend this work beyond planar flagellar waveforms into the realm of 3D waveforms such as “
Figure 8” deviations or helices observed in some sperm and other flagellated cells. Lastly, understanding flagellar dynamics can provide insight for bioengineered devices–perhaps in the form synthetic cells and devices that can deliver sperm to eggs and aid in overcoming male infertility or, even more broadly, deliver drugs or chemicals to target locations. At the heart of these problems is motion in viscous fluids–a world where inertia has relatively little impact and mathematical models such as the method of regularized Stokeslets may very well hold the key to future developments.