Application of Hydrodynamic Cavitation Reactors for Treatment of Wastewater Containing Organic Pollutants: Intensification Using Hybrid Approaches
Abstract
1. Introduction
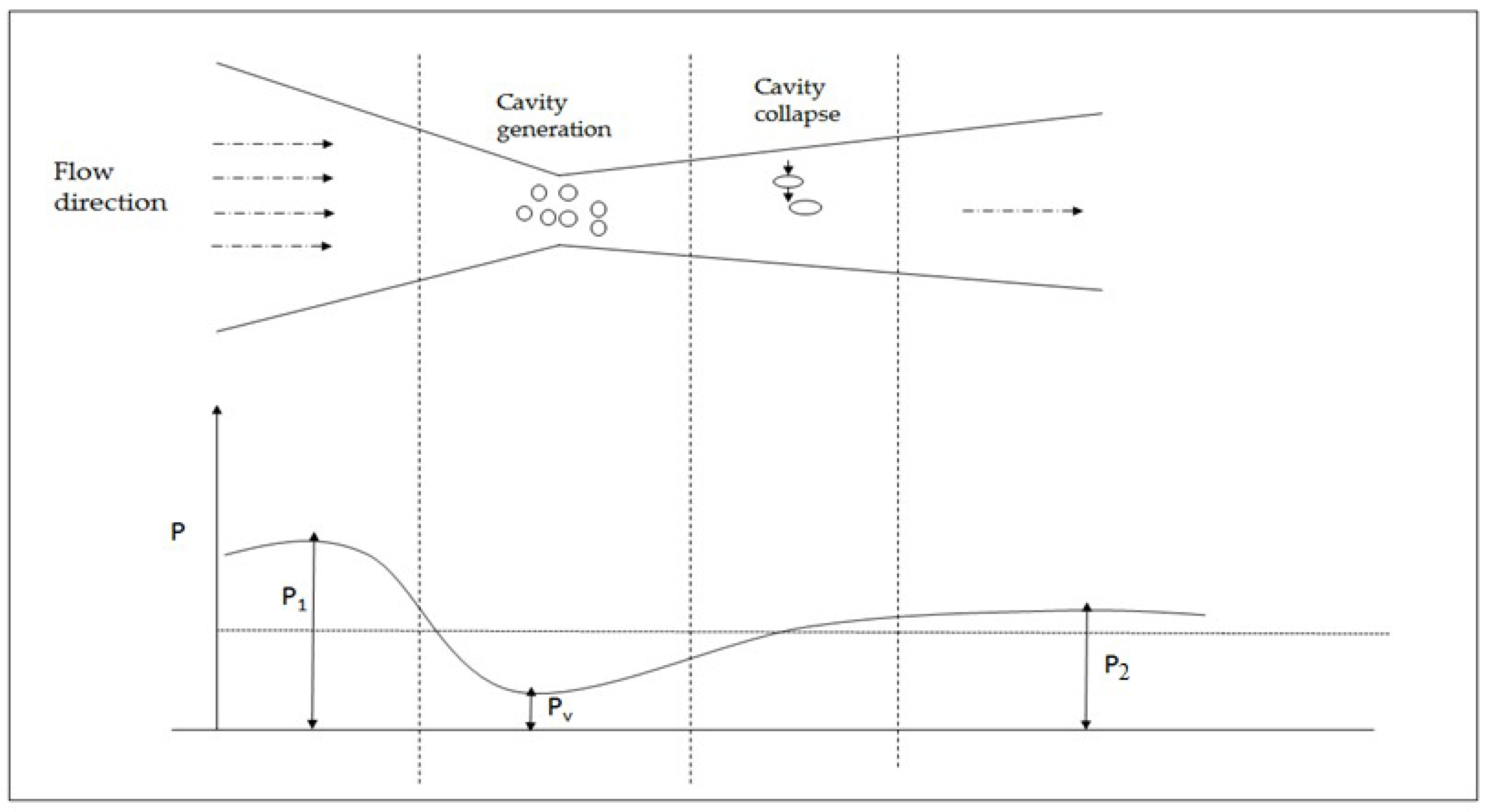
2. Basic Aspects of Cavitation
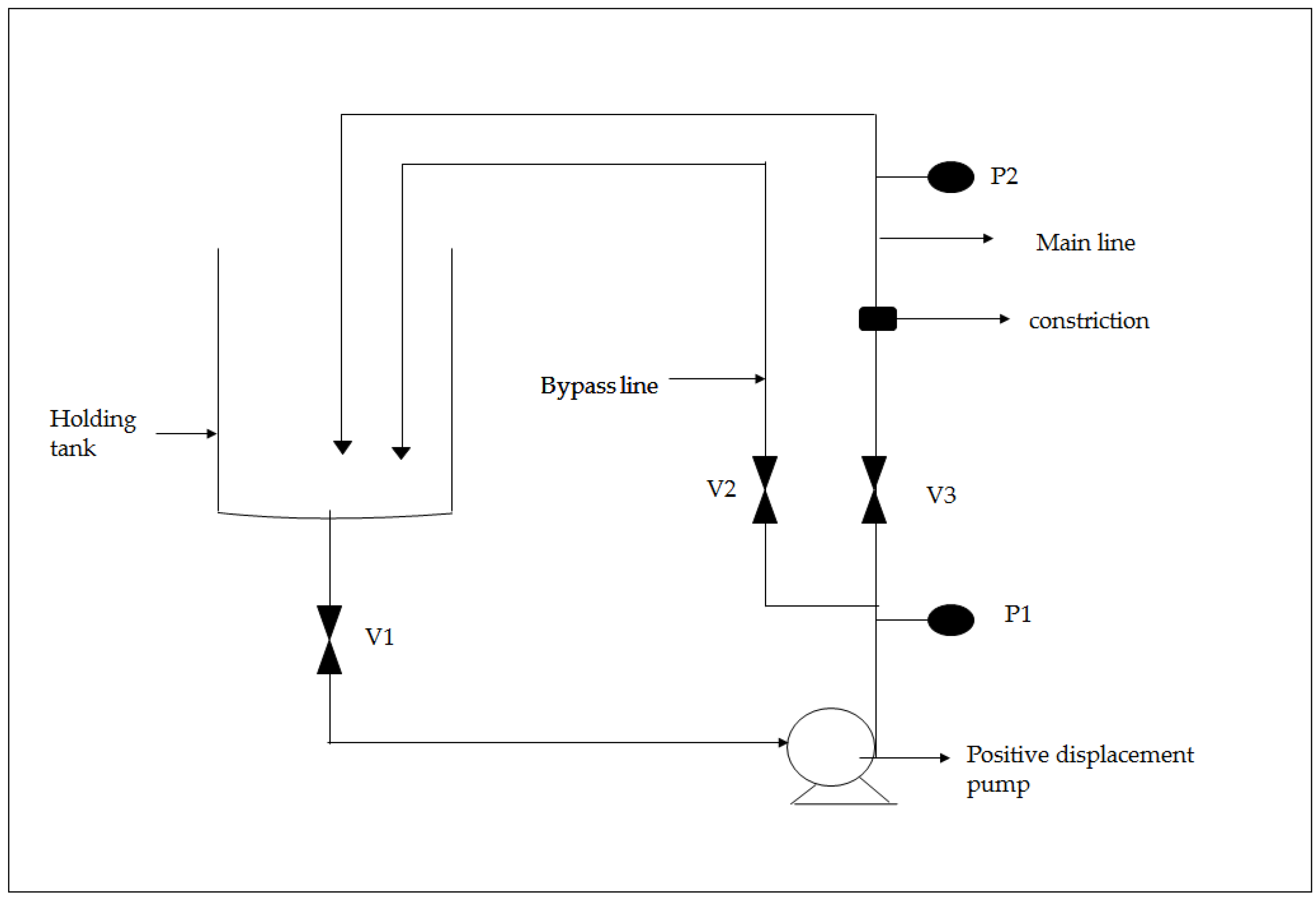
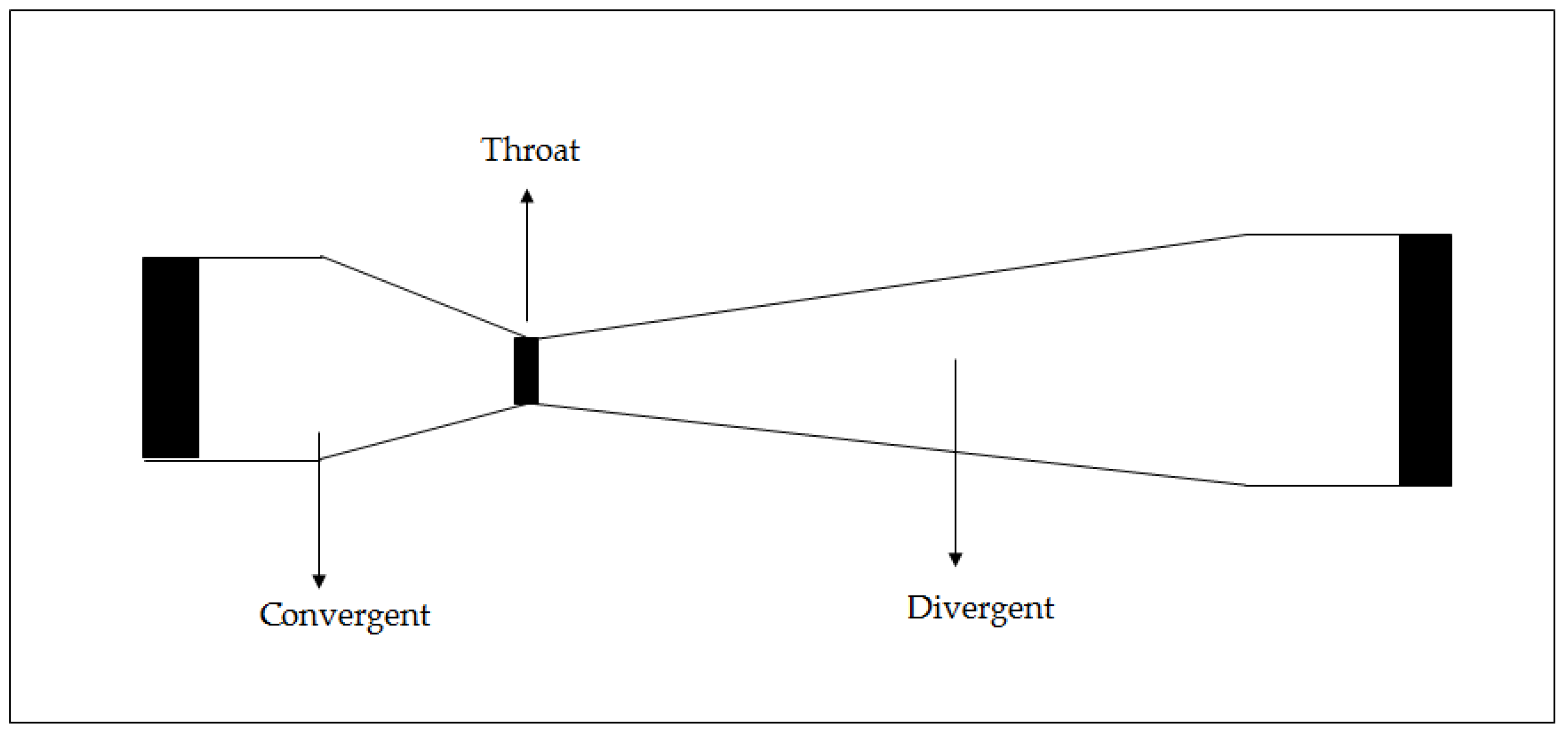
3. Hydrodynamic Cavitation Reactors
4. Review of Hybrid Methods Based on HC for Treatment of Wastewater
4.1. Combination of HC with H2O2
4.2. Combination of HC with O3
4.3. Combination of HC with Fenton
4.4. Combination of HC with UV Photolysis
4.5. Combination of HC with Photocatalysis
5. Review of Hybrid Treatment Approaches Studied Involving HC and Other AOPs
Discussion Related to Selection of Optimum Conditions
6. Conclusions
Funding
Conflicts of Interest
Abbreviations
| COD | Chemical oxidation demand |
| BOD | Biological oxidation demand |
| TOC | Total organic carbon |
| DOC | Dissolved oxygen concentration |
| TDS | Total dissolved solids |
References
- Im, J.-K.; Cho, I.-H.; Kim, S.-K.; Zoh, K.-D. Optimization of carbamazepine removal in O3/UV/H2O2 system using a response surface methodology with central composite design. Desalination 2012, 285, 306–314. [Google Scholar] [CrossRef]
- Zupanc, M.; Kosjek, T.; Petkovsek, M.; Heath, E.; Kompare, B.; Sirok, B. Removal of pharmaceuticals from wastewater by biological processes, hydrodynamic cavitation and UV treatment. Ultrason. Sonochem. 2013, 20, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, T.; Sharma, J.P. Impact of Pesticides Application in Agricultural Industry: An Indian scenario. Int. J. Adv. Res. Eng. Sci. Technol. 2013, 4, 817–822. [Google Scholar]
- Dular, M.; Griessler-Bulc, T.; Gutierrez-Aguirre, I.; Heath, E.; Kosjek, T.; Klemencic, A.K.; Oder, M.; Petkovsek, M.; Racki, N.; Ravnikar, M.; et al. Use of hydrodynamic cavitation in (waste)water treatment. Ultrason. Sonochem. 2016, 29, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Bagal, M.V.; Gogate, P.R. Wastewater treatment using hybrid treatment schemes based on cavitation and Fenton chemistry: A review. Ultrason. Sonochem. 2014, 21, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Gogate, P.R. Cavitational reactors for process intensification of chemical processing applications: A critical review. Chem. Eng. Process. 2008, 47, 515–527. [Google Scholar] [CrossRef]
- Adewuyi, Y.G. Sonochemistry: Environmental Science and Engineering Applications. Ind. Eng. Chem. Res. 2001, 40, 4681–4715. [Google Scholar] [CrossRef]
- Mishra, K.P.; Gogate, P.R. Intensification of degradation of Rhodamine B using hydrodynamic cavitation in the presence of additives. Sep. Purif. Technol. 2010, 75, 385–391. [Google Scholar] [CrossRef]
- Gogate, P.R. Treatment of wastewater streams containing phenolic compounds using hybrid techniques based on cavitation: A review of the current status and the way forward. Ultrason. Sonochem. 2008, 15, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jia, J.; Wang, Y. Combination of photocatalysis with hydrodynamic cavitation for degradation of tetracycline. Chem. Eng. J. 2017, 315, 274–282. [Google Scholar] [CrossRef]
- Saharan, V.K.; Badve, M.P.; Pandit, A.B. Degradation of Reactive Red 120 dye using hydrodynamic cavitation. Chem. Eng. J. 2011, 178, 100–107. [Google Scholar] [CrossRef]
- Thanekar, P.; Murugesan, P.; Gogate, R.P. Improvement in biological oxidation process for the removal of dichlorvos from aqueous solutions using pretreatment based on Hydrodynamic Cavitation. J. Water Process Eng. 2018, 23, 20–26. [Google Scholar] [CrossRef]
- Raut-Jadhav, S.; Saharan, V.K.; Pinjari, D.V.; Saini, D.R.; Sonawane, S.H.; Pandit, A.B. Intensification of degradation of imidacloprid in aqueous solutions by combination of hydrodynamic cavitation with various advanced oxidation processes (AOPs). J. Environ. Chem. Eng. 2013, 1, 850–857. [Google Scholar] [CrossRef]
- Thanekar, P.; Panda, M.; Gogate, P.R. Degradation of carbamazepine using hydrodynamic cavitation combined with advanced oxidation processes. Ultrason. Sonochem. 2018, 40, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Rajoriya, S.; Bargole, S.; Saharan, V.K. Degradation of a cationic dye (Rhodamine 6G) using hydrodynamic cavitation coupled with other oxidative agents: Reaction mechanism and pathway. Ultrason. Sonochem. 2017, 34, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Barik, A.J.; Gogate, P.R. Hybrid treatment strategies for 2,4,6-trichlorophenol degradation based on combination of hydrodynamic cavitation and AOPs. Ultrason. Sonochem. 2018, 40, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Barik, A.J.; Gogate, P.R. Degradation of 4-chloro 2-aminophenol using a novel combined process based on hydrodynamic cavitation, UV photolysis and ozone. Ultrason. Sonochem. 2016, 30, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.S.; Sonawane, S.H.; Bhanvase, B.A.; Bethi, B. Treatment of ternary dye wastewater by hydrodynamic cavitation combined with other advanced oxidation processes (AOP’s). J. Water Process Eng. 2018, 23, 250–256. [Google Scholar] [CrossRef]
- Rajoriya, S.; Bargole, S.; George, S.; Saharan, V.K. Treatment of textile dyeing industry effluent using hydrodynamic cavitation in combination with advanced oxidation reagents. J. Hazard. Mater. 2018, 344, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Ciriminna, R.; Albanese, L.; Meneguzzo, F.; Pagliaro, M. Wastewater remediation via controlled hydrocavitation. Environ. Rev. 2017, 25, 175–183. [Google Scholar] [CrossRef]
- Oller, I.; Malato, S.; Sanchez-Perez, J.A. Combination of Advanced Oxidation Processes and biological treatments for wastewater decontamination-A review. Sci. Total Environ. 2011, 409, 4141–4166. [Google Scholar] [CrossRef] [PubMed]
- Leighton, T.G. Bubble population phenomena in acoustic cavitation. Ultrason. Sonochem. 1995, 2, 123–136. [Google Scholar] [CrossRef]
- Cintas, P.; Luche, J.-L. Green chemistry. A sonochemical approach. Green Chem. 1999, 1, 115–125. [Google Scholar] [CrossRef]
- Gogate, P.R.; Pandit, A.B. Application of Cavitational reactors for cell disruption for recovery of intracellular enzymes. J. Chem. Technol. Biotechnol. 2008, 83, 1083–1093. [Google Scholar] [CrossRef]
- Sancheti, S.V.; Gogate, P.R. A review of engineering aspects of intensification of chemical synthesis using ultrasound. Ultrason. Sonochem. 2017, 36, 527–543. [Google Scholar] [CrossRef] [PubMed]
- Moholkar, V.S.; Pandit, A.B. Bubble behavior in hydrodynamic caviation: Effect of turbulence. AIChE 1997, 43, 1641–1648. [Google Scholar] [CrossRef]
- Gogate, P.R.; Pandit, A.B. Hydrodynamic cavitation reactors: A state of the art review. Rev. Chem. Eng. 2001, 17, 1–85. [Google Scholar] [CrossRef]
- Kanthale, P.M.; Gogate, P.R.; Pandit, A.B.; Wilhelm, A.M. Dynamics of cavitational bubbles and design of a hydrodynamic cavitational reactor: Cluster approach. Ultrason. Sonochem. 2005, 12, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Soyama, H.; Hoshino, J. Enhancing the aggressive intensity of hydrodynamic cavitation through a Venturi tube by increasing the pressure in the region where the bubbles collapse. AIP Adv. 2016, 6, 045113–045126. [Google Scholar] [CrossRef]
- Rajoriya, S.; Bargole, S.; Saharan, V.K. Degradation of reactive blue 13 using hydrodynamic cavitation: Effect of geometrical parameters and different oxidizing additives. Ultrason. Sonochem. 2017, 37, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Moholkar, V.S.; Kumar, P.S.; Pandit, A.B. Hydrodynamic cavitation for sonochemical effects. Chem. Eng. J. 1999, 6, 53–65. [Google Scholar] [CrossRef]
- Gogate, P.R.; Kabadi, A.M. A review of applications of cavitation in biochemical engineering/biotechnology. Biochem. Eng. J. 2009, 44, 60–72. [Google Scholar] [CrossRef]
- Albanese, L.; Ciriminna, R.; Meneguzzo, F.; Pagliaro, M. Energy efficient inactivation of saccharomyces cerevisiae via controlled hydrodynamic cavitation. Energy Sci. Eng. 2015, 3, 221–238. [Google Scholar] [CrossRef]
- Patil, P.N.; Bote, S.D.; Gogate, P.R. Degradation of imidacloprid using combined advanced oxidation processes based on hydrodynamic cavitation. Ultrason. Sonochem. 2014, 21, 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.S.; Sonawane, S.H.; Pandit, A.B. Degradation of methylene blue dye in aqueous solution using hydrodynamic cavitation based hybrid advanced oxidation processes. Chem. Eng. Process. Process Intensif. 2017, 122, 288–295. [Google Scholar] [CrossRef]
- Gogate, P.R.; Patil, P.N. Combined treatment technology based on synergism between hydrodynamic cavitation and advanced oxidation processes. Ultrason. Sonochem. 2015, 25, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.; Su, J.; Zhu, Y.; Wei, X.; Jin, M.; Zhang, H.; Dong, C.; Wei, Z. Decolorization of azo dyes Orange G using hydrodynamic cavitation coupled with heterogeneous Fenton process. Ultrason. Sonochem. 2016, 28, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, Y. Degradation of alachlor in aqueous solution by using hydrodynamic cavitation. J. Hazard. Mater. 2009, 161, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Bagal, M.V.; Gogate, P.R. Degradation of 2, 4-dinitrophenol using a combination of hydrodynamic cavitation, chemical and advanced oxidation processes. Ultrason. Sonochem. 2013, 20, 1226–1235. [Google Scholar] [CrossRef] [PubMed]
- Saharan, V.K.; Pandit, A.B.; Satish Kumar, P.S.; Anandan, S. Hydrodynamic cavitation as an advanced oxidation technique for the degradation of Acid Red 88 dye. Ind. Eng. Chem. Res. 2012, 51, 1981–1989. [Google Scholar] [CrossRef]
- Rajoriya, S.; Carpenter, J.; Saharan, V.K.; Pandit, A.B. Hydrodynamic cavitation: An advanced oxidation process for the degradation of bio-refractory pollutants. Rev. Chem. Eng. 2016, 32, 379–411. [Google Scholar] [CrossRef]
- Sayyaadi, H. Enhanced cavitation—Oxidation process of non-VOC aqueous solution using hydrodynamic cavitation reactor. Chem. Eng. J. 2015, 272, 79–91. [Google Scholar] [CrossRef]
- Raut-Jadhav, S.; Saini, D.; Sonawane, S.; Pandit, A. Effect of process intensifying parameters on the hydrodynamic cavitation based degradation of commercial pesticide (methomyl) in the aqueous solution. Ultrason. Sonochem. 2015, 28, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Jawale, R.H.; Dapurkar, O.; Gogate, P.R. Treatment of atrazine containing wastewater using cavitation based hybrid treatment approaches. Chem. Eng. Process. Process Intensif. 2018, 130, 275–283. [Google Scholar] [CrossRef]
- Bagal, M.V.; Gogate, P.R. Degradation of diclofenac sodium using combined processes based on hydrodynamic cavitation and heterogeneous photocatalysis. Ultrason. Sonochem. 2014, 21, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Pillai, K.C.; Kwon, T.O.; Moon, I.S. Degradation of wastewater from terephthalic acid manufacturing process by ozonation catalyzed with Fe2+, H2O2 and UV light: Direct versus indirect ozonation reactions. Appl. Catal. B Environ. 2009, 91, 319–328. [Google Scholar] [CrossRef]
- Wang, C.; Yediler, A.; Lienert, D.; Wang, Z.; Kettrup, A. Ozonation of an azo dye C.I. Remazol Black 5 and toxicological assessment of its oxidation products. Chemosphere 2003, 52, 1225–1232. [Google Scholar] [CrossRef]
- Maldonado, M.I.; Malato, S.; Perez-Estrada, L.A.; Gernjak, W.; Oller, I.; Domenech, X.; Peral, J. Partial degradation of five pesticides and an industrial pollutant by ozonation in a pilot-plant scale reactor. J. Hazard. Mater. 2006, 138, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Gore, M.M.; Saharan, V.K.; Pinjari, D.V.; Chavan, P.V.; Pandit, A.B. Degradation of reactive orange 4 dye using hydrodynamic cavitation based hybridtechniques. Ultrason. Sonochem. 2014, 21, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Shen, H.; Ondruschka, B.; Zhang, Y.; Wang, W.; Bremner, D.H. Removal of blue-green algae using the hybrid method of hydrodynamic cavitation and ozonation. J. Hazard. Mater. 2012, 235–236, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Gogate, P.R.; Pandit, A.B. A review of imperative technologies for wastewater treatment I: Oxidation technologies at ambient conditions. Adv. Environ. Res. 2004, 8, 501–551. [Google Scholar] [CrossRef]
- Joshi, R.K.; Gogate, P.R. Ultrasonics Sonochemistry Degradation of dichlorvos using hydrodynamic cavitation based treatment strategies. Ultrason. Sonochem. 2012, 19, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Barik, A.J.; Gogate, P.R. Ultrasonics Sonochemistry Degradation of 4-chloro 2-aminophenol using combined strategies based on ultrasound, photolysis and ozone. Ultrason. Sonochem. 2016, 28, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Cehovin, M.; Medic, A.; Scheideler, J.; Mielcke, J.; Ried, A.; Kompare, B.; Gotvajn, A.Z. Hydrodynamic cavitation in combination with the ozone, hydrogen peroxide and the UV-based advanced oxidation processes for the removal of natural organic matter from drinking water. Ultrason. Sonochem. 2017, 37, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Bethi, B.; Sonawane, S.H.; Rohit, G.S.; Holkar, C.R.; Pinjari, D.V.; Bhanvase, B.A.; Pandit, A.B. Ultrasonics Sonochemistry Investigation of TiO2 photocatalyst performance for decolorization in the presence of hydrodynamic cavitation as hybrid AOP. Ultrason. Sonochem. 2016, 28, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Calıskan, Y.; Yatmaz, H.C.; Bektas, N. Photocatalytic oxidation of high concentrated dye solutions enhanced by hydrodynamic cavitation in a pilot reactor. Process Saf. Environ. Prot. 2017, 111, 428–438. [Google Scholar] [CrossRef]
- Montusiewicz, A.; Pasieczna-Patkowska, S.; Lebiocka, M.; Szaja, A.; Szymanska-Chargot, M. Hydrodynamic cavitation of brewery spent grain diluted by wastewater. Chem. Eng. J. 2016, 313, 1–11. [Google Scholar] [CrossRef]
- Jung, K.; Park, D.; Hwang, M.; Ahn, K. Decolorization of Acid Orange 7 by an electric field-assisted modified orifice plate hydrodynamic cavitation system: Optimization of operational parameters. Ultrason. Sonochem. 2015, 26, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Song, Y.; Wang, S.; Tao, Z.; Yu, S.; Liu, Y. Enhanced decolorization of methyl orange using zero-valent copper nanoparticles under assistance of hydrodynamic cavitation. Ultrason. Sonochem. 2015, 22, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Boczkaj, G.; Klein, M.; Przyjazny, A. Effective method of treatment of effluents from production of bitumens under basic pH conditions using hydrodynamic cavitation aided by external oxidants. Ultrason. Sonochem. 2018, 40, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Raut-Jadhav, S.; Badve, M.P.; Pinjari, D.V.; Saini, D.R.; Sonawane, S.H.; Pandit, A.B. Treatment of the pesticide industry effluent using hydrodynamic cavitation and its combination with process intensifying additives (H2O2and ozone). Chem. Eng. J. 2016, 295, 326–335. [Google Scholar] [CrossRef]
- Saxena, S.; Rajoriya, S.; Saharan, V.K.; George, S. Cavitation along with alum coagulation for the mineralization and for the mineralization and biodegradability enhancement of tannery waste effluent. Ultrason. Sonochem. 2018, 44, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Raut-jadhav, S.; Kumar, V.; Pinjari, D.; Sonawane, S.; Saini, D.; Pandit, A. Synergetic effect of combination of AOP’s (hydrodynamic cavitation and H2O2) on the degradation of neonicotinoid class of insecticide. J. Hazard. Mater. 2013, 261, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Chakinala, A.G.; Bremner, D.H.; Gogate, P.R.; Namkung, K.C.; Burgess, A.E. Multivariate analysis of phenol mineralisation by combined hydrodynamic cavitation and heterogeneous advanced Fenton processing. Appl. Catal. B Environ. 2008, 78, 11–18. [Google Scholar] [CrossRef]
- Bremner, D.H.; Carlo, S.D.; Chakinala, A.G.; Cravotto, G. Mineralisation of 2,4-dichlorophenoxyacetic acid by acoustic or hydrodynamic cavitation in conjunction with the advanced Fenton process. Ultrason. Sonochem. 2008, 15, 416–419. [Google Scholar] [CrossRef] [PubMed]
- Chakinala, A.G.; Gogate, P.R.; Burgess, A.E.; Bremner, D.H. Treatment of industrial wastewater effluents using hydrodynamic cavitation and the advanced Fenton process. Ultrason. Sonochem. 2008, 15, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Bethi, B.; Sonawane, S.H.; Potoroko, I.; Bhanvase, B.A.; Sonawane, S.S. Novel hybrid system based on hydrodynamic cavitation for treatment of dye waste water: A first report on bench scale study. J. Environ. Chem. Eng. 2017, 5, 1874–1884. [Google Scholar] [CrossRef]
- Gaekwad, R.R.; Patel, P.R.L. Pesticide wastewater treatment by hydrodynamic cavitation process. Int. J. Adv. Res. Eng. Sci. Technol. 2015, 2, 1–5. [Google Scholar]
- Saharan, V.K.; Rizwani, M.A.; Malani, A.A.; Pandit, A.B. Effect of geometry of hydrodynamically cavitating device on degradation of orange-G. Ultrason. Sonochem. 2013, 20, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Rajoriya, S.; Bargole, S.; George, S.; Kumar, V.; Gogate, P.R. Separation and Purifi cation Technology Synthesis and characterization of samarium and nitrogen doped TiO2 photocatalysts for photo-degradation of 4-acetamidophenol in combination with hydrodynamic and acoustic cavitation. Sep. Purif. Technol. 2019, 209, 254–269. [Google Scholar] [CrossRef]
- Musmarra, D.; Prisciandaro, M.; Capocelli, M.; Karatza, D.; Iovino, P.; Canzano, S.; Lancia, A. Degradation of ibuprofen by hydrodynamic cavitation: Reaction pathways and effect of operational parameters. Ultrason. Sonochem. 2016, 29, 76–83. [Google Scholar] [CrossRef] [PubMed]

| Sr. No. | Type of Wastewater | Treatment Methods | Operating Conditions | Results | Comments | Reference |
|---|---|---|---|---|---|---|
| 1 | Textile dyeing industry effluent | HC, HC + Oxygen, HC + O3, HC + Fenton | HC reactor: Inlet pressure of 5 bar; reaction volume of 6 L; pH as 6.8; treatment time of 120 min HC + Oxygen (2 L/min as O2 flow rate); HC + O3 (3g/h as ozone flow rate); HC + Fenton (molar ratio of FeSO4·7H2O:H2O2 as 1:5) | HC alone resulted in 17% of total organic carbon (TOC)and 12% of chemical oxidation demand (COD)reduction with 25% of decolorization HC + Fenton resulted in 48% of TOC and 38% of COD reduction with 98% of decolorization HC + Oxygen resulted in 48% of TOC, 33% of COD, 62% of decolorization HC + O3 resulted in 48% of TOC, 23% of COD and 88% of decolorization | HC + Fenton’s reagent resulted in highest reduction in TOC, COD, and color as compared to other hybrid processes | [19] |
| 2 | Reactive Orange 4 (RO4) | HC, HC + H2O2, HC + O3 | HC reactor: Inlet pressure of 5 bar; Initial concentration as 40 ppm; pH as 5; Reaction volume of 6 L; Reaction time as 120 min; HC + H2O2 (molar ratio of RO4:H2O2 as 1:1:30); HC + O3 (O3 as 3 g/h) | Extent of mineralization for HC alone as 14.67%; HC + H2O2 as 31.90%; HC + O3 as 76.25% | HC coupled with ozone found to be the most energy efficient method for the degradation of RO4 | [56] |
| 3 | Methomyl | HC, HC + H2O2, HC + Fenton, HC + O3 | HC reactor: Inlet pressure of 5 bar; Initial concentration as 25 ppm; Reaction volume of 5 L; Reaction time as 120 min; pH as 2.5; HC + H2O2 (mole ratio of methomyl:4H2O2 as 1:05); HC + Fe0nton (mole ratio of Fe2+:H2O2 as 1:20; HC + O3 = (2 g/h as ozone flow rate) | Extent of degradation for HC alone as 13.9%, HC + H2O2 as 59.86% in 60 min treatment; HC + Fenton as 100% in 30 min, HC + O3 as 69.87% Extent of TOC reduction for HC alone as 5.45%, HC + H2O2 as 15.4%, HC + Fenton as 35.79% and HC + O3 as 70.79% Synergetic coefficient for HC + H2O2, HC + Fenton, HC + O3 was found to be 5.8, 13.41 and 47.6 respectively | The combined method of HC + O3 found to be the most effective process as it resulted in highest synergetic coefficient, energy efficiency and the extent of mineralization | [43] |
| 4 | Tetracycline | HC, HC + Photocatalysis | HC reactor: Initial concentration as 30 mg/L; Reaction volume of 4 L; Reaction time 90 min; pH as 4.2; TiO2 loading as 100 mg L−1; UV254 lamp | The rate constant obtained for HC alone was 1.4 × 10−3 min−1 and for HC + Photocatalysis as 1.67 × 10−2 min−1 as the rate constant | The rate constant obtained for photocatalysis coupled with HC was found to be 1.5–3.7 times of the sum of the individual processes The degradation of Tetracycline was pH-dependent and favored at alkaline pH | [10] |
| 5 | Imidacloprid | HC, HC + H2O2 | HC reactor: Inlet pressure of 15 bar; Temperature as 32 ± 4 °C; pH as 2.7; Initial concentration as 25 mg/L; Reaction volume of 5 L; HC + H2O2 (Molar ratio of Imidacloprid:H2O2 as 1:40) | HC alone resulted in 26.5% of degradation with rate constant being 2.565 × 10−3 min−1 For HC + H2O2, 100% degradation was achieved in 45 min with rate constant of 0.122216 min−1 and TOC reduction of 36.1% | The combined method of HC + H2O2 was found as an efficient process due to higher synergetic index obtained as 22.79 for degradation of imidacloprid | [63] |
| 6 | Methylene blue (MB) | HC, HC + H2O2, HC + Photocatalysis | HC reactor: Inlet pressure of 5 bar; pH as 2; treatment time of 120 min; initial concentration as 50 ppm HC + H2O2 (MB:H2O2 as 1:20); HC + Photocatalyst (Bismuth doped TiO2 at loading of 200 mg/L) | Extent of decolorization for HC alone as 32.32%, HC + Photocatalyst as 64.58%, HC + H2O2 as 100% Extent of mineralization for HC alone as 9.46%, HC + photocatalysis as 12.68%, HC + H2O2 as 18.41% | The combined method of HC + photocatalytic oxidation shows the synergy but it is less efficient compared to combined method of HC + H2O2 | [35] |
| 7 | Phenol | HC, HC + heterogeneous AFP | HC generated using liquid whistle reactor (LWR): Inlet pressure of 1500 psi; Reaction volume as 4 L; Temperature as 35 ± 3 °C; pH of solution as 2.5; H2O2 concentration as 2000 mg/L; iron 80 g (50 pieces of 1 cm × 2 cm, thickness of 0.10 cm); Orifice as a cavitating device and orifice area 0.012 in.2 | TOC removal of about 50–60% obtained within 105 min of treatment time with the modified AFP combined with HC | The modified AFP combined with HC found be a promising technology for treatment of wastewaters containing high organic content | [64] |
| 8 | Reactive blue 13 (RB13) | HC, HC + Oxygen, HC + H2O2, HC + ferrous sulfate, HC + O3 | HC reactor: Inlet pressure of 0.4 MPa; Solution pH as 2; Initial concentration as 30 ppm; Reaction volume of 6 L; temperature as 30 °C; Reaction time of 120 min; Combined method: HC + H2O2 (molar ratio of RB13:H2O2 as 1:20); HC + Oxygen (oxygen flow rate as 2 L/min); HC + ferrous sulfate (molar ratio of H2O2:FeSO4.7H2O as 1:3); HC + O3 (O3 flow rate as 3 g/h) | Extent of decolorization for HC alone as 47%, HC + Oxygen as 66%, HC + H2O2 as 91%, HC + ferrous sulfate as 66.3%, HC + O3 as 100% Extent of TOC reduction for HC alone as 19.06%, HC + Oxygen as 18.10%, HC + H2O2 as 24.65%, HC + ferrous sulfate as 66.3%, HC + O3 as 71.76% | HC + ozone process established as best treatment approach for the decolorization and mineralization of RB13 | [30] |
| 9 | Tannery dye wastewater (Synthetically prepared mixture of Methylene blue, Methyl orange, Rhodamine-B) | HC, HC + H2O2, HC + Fenton, HC + photo Fenton HC + photolytic, HC + photocatalytic | HC reactor: Inlet pressure of 6 bar; pH as 3; Reaction volume of 5 L; initial concentration as 30 ppm; orifice as a cavitating device; temperature as 30 °C Hybrid methods: HC + H2O2 (Molar ration of dye:H2O2as 1:40); HC + Fenton (Molar ration of FeSO4·7H2O:H2O2 as 1:30); HC + photo Fenton (Ultraviolet irradiations (UV) wavelength 350-750 nm); HC + photocatalytic (TiO2 loading of 200 mg/L) | Extent of TOC removal obtained for HC alone as 8.53%; HC + H2O2 as 16.95%; HC + Fenton as 38.42%; HC + photo Fenton as 41.28%; HC + photolytic as 14.01%; HC + photocatalytic as 15.63% | The method of HC + H2O2 was found as the best combination for the removal of dye from aqueous solution as the complete decolorization was obtained along with higher synergetic index as 28.974 | [18] |
| 10 | 2,4-dichlorophenoxyacetic acid | US, US + AFP HC, HC + AFP | US reactor: Frequency of 20 kHz, operating in a pulse mode of 4 s on and 2 s off; Initial concentration as 0.235 g/L, Temperature as 22 ± 2 °C; Treatment time of 60 min; hydrogen peroxide (30% H2O2 at loading of 1.7 mL); powdered iron (at loading of 0.12 g) HC reactor: orifice as a cavitating device, Inlet pressure of 1500 psi, Initial concentration as 0.235 g/L, Reaction volume of 8 L, Temperature as 18 ± 2 °C, flow rate as 5.2 L/min; Treatment time of 90 min | US alone resulted in 11% of TOC reduction; addition of iron powder (0.12 g) resulted in TOC removal as 18%; Ultrasound + advanced Fenton process resulted in about 60% TOC reduction HC + advanced Fenton process resulted in about 70% TOC removal | HC was found to be more suitable for treating effluent at a much larger scale of operation as compared to acoustic cavitation treatment method | [65] |
| 11 | Industrial effluent 1 containing substituted phenolic compounds Industrial effluent 2 dyestuffs solution | HC + Advanced Fenton process (AFP) | HC generated using liquid whistle reactor (LWR): Reaction volume as 4 L, time 150 min | For industrial effluent 1, 60% TOC reduction; For industrial effluent 2, 70% of TOC reduction was obtained | HC in combination with AFP was found as effective treatment approach reducing TOC by about 60% to 70% | [66] |
| 12 | Crystal violet | HC + hydro gels packed bed adsorption | HC reactor: Inlet pressure of 3 bar, Initial concentration as 500 mg/L, Reaction volume as 2 L, orifice as a cavitating device, Poly-acrylic acid bentonite nano composite hydro gel loading as 0.5 wt % (30 g as the quantity) | HC alone resulted in 20% decolorization and 17% of TOC reduction; HC + hydro gels packed bed adsorption resulted in 96% decolorization and 70% of TOC reduction at pH 2.3; synergetic index for HC + hydro gels packed bed adsorption was found to be 1.2 | HC followed by hydro gel adsorption was found to be an efficient hybrid technology for the treatment of textile industry wastewater containing dyes | [67] |
| 13 | Real pesticide wastewater | HC | HC reactor: Temperature as 28 °C, treatment time of 150 min | About 90.55% COD and 83.21% color removal was achieved | HC was found to be ecofriendly solution for the treatment of the wastewater. Also, generation of sludge after treatment was very less | [68] |
| 14 | 2,4-dinitrophenol (DNP) | HC, HC/H2O2, HC/Na2S2O8/FeSO4, HC/FeSO4/H2O2 HC/Fe/H2O2 HC/CuO/H2O2 | HC reactor: Inlet pressure of 4 bar; pH as 4; Temperature as 35 °C; initial concentration as 20 ppm; treatment time of 120 min; HC/H2O2 (DNP:H2O2 ratio of 1:5); HC/Fenton (0.3 g/L H2O2 + 0.6 g/L FeSO4); HC/advanced Fenton (0.4 g/L H2O2 + 0.6 g/L Fe); HC/CuO/H2O2 (H2O2:CuO ratio of 1:6); HC/Ferrous activated persulfate (0.08 g/L FeSO4 + 0.16 g/L Na2S2O8) | Extent of degradation for HC alone as 12.4%, HC/H2O2 as 21.3%, HC/Fenton as 100%, HC/advanced Fenton as 54.1%, HC/CuO/H2O2 as 29.80%, and HC/Ferrous activated persulfate as 55.30% | The maximum cavitational yield 5.81 × 10−6 min−1 mg/J was obtained for HC/Ferrous activated persulfate approach | [39] |
| 15 | Orange-G | HC | HC reactor: Inlet pressure of 5 bar, Reaction volume as 6 L, time of 2 h, temperature as 32 ± 2 °C Cavitating devices used: Orifice plate, circular venturi and slit venturi | TOC reduction obtained using HC with slit venturi as 37%, circular venturi was 28%, orifice was 14% | The maximum degradation was obtained (almost 50% higher) using HC with slit venturi. Also, the power dissipated into the system and hence the energy transfer efficiency was found to be higher as compared to circular venturi and orifice plate | [69] |
| 16 | 4-acetamidophenol | HC, US, HC + photocatalysis US + Photocatalysis | Photo catalytic reactor: UV lamp of 300 W; Wavelength >400 nm; irradiation intensity as 2.47 W/cm2; TiO2 catalyst loading as 2 g/L HC reactor: inlet pressure of 5 bar; slit venturi as a cavitating device; pH as 6.8; US horn: Frequency as 20 kHz, input power 750 W | Photocatalytic oxidation resulted in almost 50% removal with rate constant of 4.4 × 10−3 min−1; Sm and N doped TiO2 resulted in about 60% and 63% degradation respectively Extent of degradation for US + photocatalysis was 87%, HC + photocatalysis was 91% | HC + photocatalytic oxidation was found to be energy efficient process (9.0 × 10−6 mg/J) with almost 9 fold than the US + photocatalytic process | [70] |
| 17 | Ibuprofen | HC | HC reactor: Inlet pressure of 0.35 MPa; initial concentration as 200 µg/L; pH as 6; Temperature as 25 °C | About 60% of ibuprofen was degraded within 60 min with an electrical energy per order of 10.77 kWhm−3 | HC alone was found as the effective method for degradation of ibuprofen both in the neutral and dissociated form of ibuprofen | [71] |
| 18 | Carbamazepine | HC HC + UV HC + H2O2 HC + O3 HC + H2O2 + O3 | HC reactor: Inlet pressure of 4 bar; pH of solution as 4; Initial concentration as 10 ppm; Reaction volume of 4 L; reaction time as 120 min; HC + UV (UV power as 16 W) HC + H2O2 (molar ratio of CBZ: H2O2 as 1:5); HC + O3 (O3 flow rate as 400 mg/h); | Extent of degradation for HC + H2O2 + O3 was 100%; HC + O3 was 91.4%, HC + H2O2 was 58.3%, and HC + UV was 52.9% Synergetic index obtained for HC + H2O2 + O3 was 3.2, HC + O3 was 2.2, HC + H2O2 was 1.01, and HC + UV was 0.9 | The combined method of HC + H2O2 + O3 established as the most effective treatment for the removal of CBZ from wastewater due to low cost of treatment as 0.29 Rs./L | [14] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thanekar, P.; Gogate, P. Application of Hydrodynamic Cavitation Reactors for Treatment of Wastewater Containing Organic Pollutants: Intensification Using Hybrid Approaches. Fluids 2018, 3, 98. https://doi.org/10.3390/fluids3040098
Thanekar P, Gogate P. Application of Hydrodynamic Cavitation Reactors for Treatment of Wastewater Containing Organic Pollutants: Intensification Using Hybrid Approaches. Fluids. 2018; 3(4):98. https://doi.org/10.3390/fluids3040098
Chicago/Turabian StyleThanekar, Pooja, and Parag Gogate. 2018. "Application of Hydrodynamic Cavitation Reactors for Treatment of Wastewater Containing Organic Pollutants: Intensification Using Hybrid Approaches" Fluids 3, no. 4: 98. https://doi.org/10.3390/fluids3040098
APA StyleThanekar, P., & Gogate, P. (2018). Application of Hydrodynamic Cavitation Reactors for Treatment of Wastewater Containing Organic Pollutants: Intensification Using Hybrid Approaches. Fluids, 3(4), 98. https://doi.org/10.3390/fluids3040098





