Characterization of Zinc Oxide Nanoparticle Cross-Linked Collagen Hydrogels
Abstract
1. Introduction
2. Results and Discussion
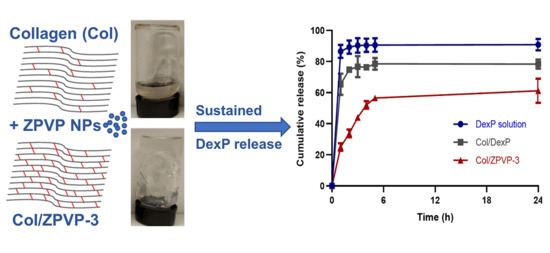
2.1. Visual Appearance
2.2. Texture Analysis
2.3. Syringeability
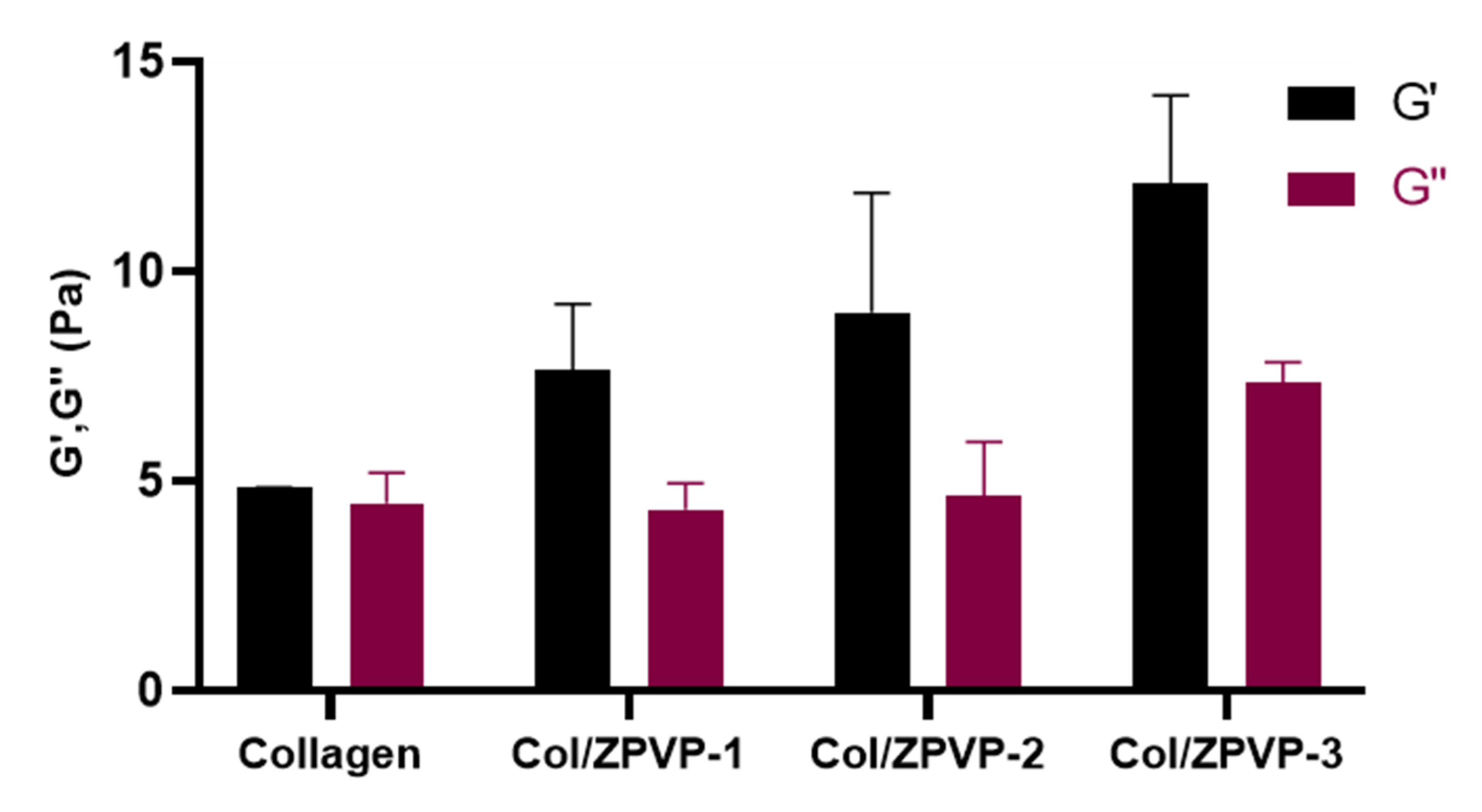
2.4. Rheological Evaluation
2.5. FTIR Spectroscopy
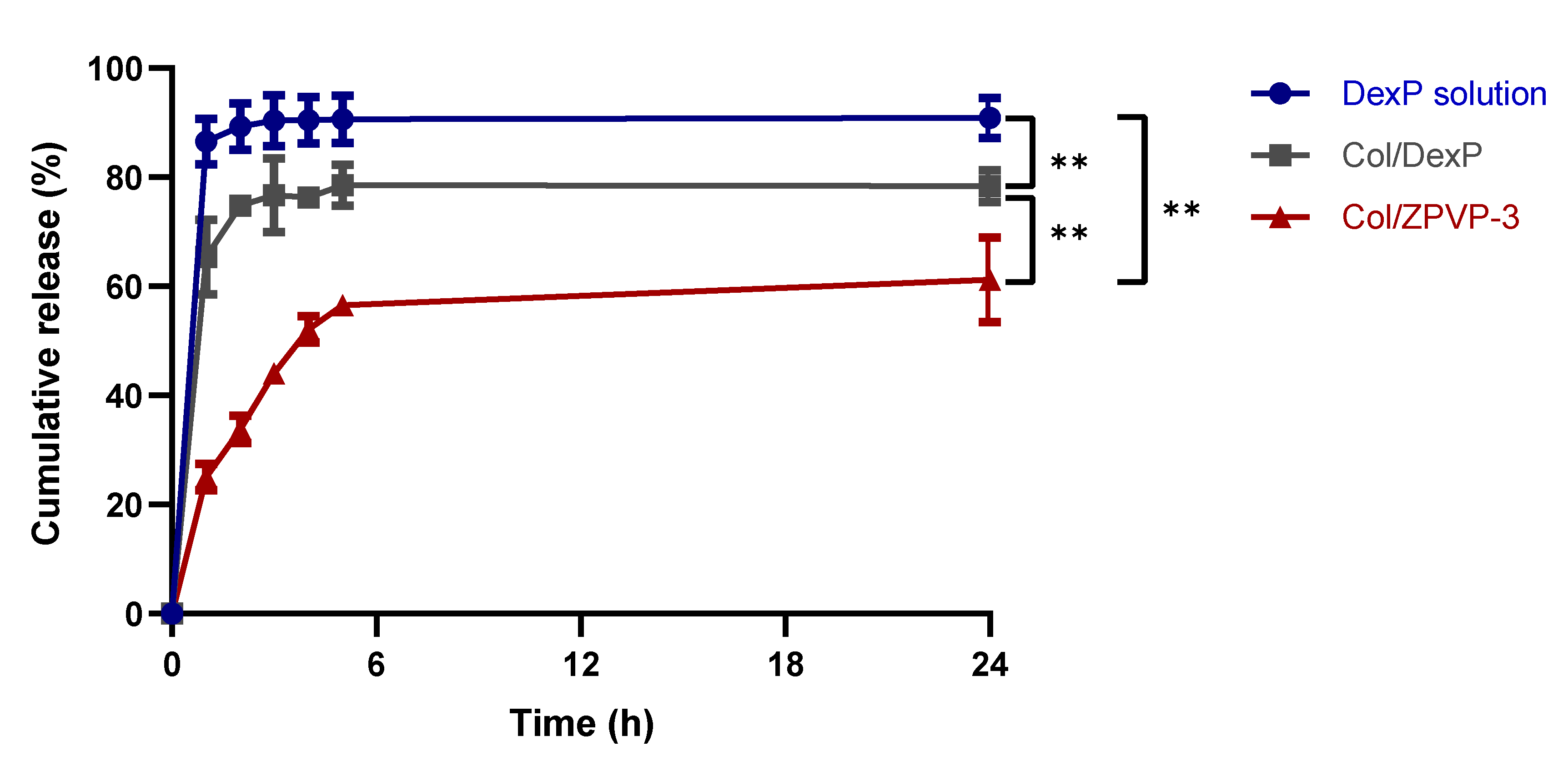
2.6. In-Vitro Drug Release
3. Conclusions
4. Materials and Methods
4.1. Chemicals
4.2. Visual Apperance
4.3. Preparation of Collagen Hydrogels
4.4. Texture Analysis
4.5. Syringeability
4.6. Rheological Evaluations
4.7. FTIR Spectroscopy
4.8. In Vitro Drug Release
4.9. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nair, L.S.; Laurencin, C.T. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007, 32, 762–798. [Google Scholar] [CrossRef]
- Gopi, S.; Amalraj, A. Effective Drug Delivery System of Biopolymers Based On Nanomaterials and Hydrogels—A Review. Drug Des. Open Access 2016, 5. [Google Scholar] [CrossRef]
- Miyata, T.; Taira, T.; Noishiki, Y. Collagen engineering for biomaterial use. Clin. Mater. 1992, 9, 139–148. [Google Scholar] [CrossRef]
- Glowacki, J.; Mizuno, S. Collagen scaffolds for tissue engineering. Biopolymers 2008, 89, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Lv, Y. Application of Collagen Scaffold in Tissue Engineering: Recent Advances and New Perspectives. Polymer 2016, 8, 42. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.; Puetzer, J.L.; Mason, B.N.; Reinhart-King, C.A.; Bonassar, L.J. 3D Bioprinting of Spatially Heterogeneous Collagen Constructs for Cartilage Tissue Engineering. ACS Biomater. Sci. Eng. 2016, 2, 1800–1805. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Raines, R.T. Review collagen-based biomaterials for wound healing. Biopolymers 2014, 101, 821–833. [Google Scholar] [CrossRef]
- Maham, A.; Tang, Z.; Wu, H.; Wang, J.; Lin, Y. Protein-based nanomedicine platforms for drug delivery. Small 2009, 5, 1706–1721. [Google Scholar] [CrossRef]
- Hadassah, J.; Prakash, D.; Sehgal, P.K.; Agarwal, A.; Bhuvaneshwari, N. Clinical evaluation of succinylated collagen bandage lenses for ophthalmic applications. Ophthalmic Res. 2008, 40, 257–266. [Google Scholar] [CrossRef]
- Saettone, M.F.; Salminen, L. Ocular Inserts for Topical Delivery. Adv. Drug Deliv. Rev. 1995, 16, 95–106. [Google Scholar] [CrossRef]
- Willoughby, C.E.; Batterbury, M.; Kaye, S.B. Collagen corneal shields. Surv. Ophthalmol. 2002, 47, 174–182. [Google Scholar] [CrossRef]
- Radu, F.A.; Bause, M.; Knabner, P.; Lee, G.W.; Friess, W.C. Modeling of drug release from collagen matrices. J. Pharm. Sci. 2002, 91, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ren, N.; Qiu, J.; Jiang, H.; Zhao, H.; Wang, G.; Boughton, R.I.; Wang, Y.; Liu, H. Carbodiimide crosslinked collagen from porcine dermal matrix for high-strength tissue engineering scaffold. Int. J. Biol. Macromol. 2013, 61, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Singla, A.; Lee, Y. Biomedical applications of collagen. Int. J. Pharm. 2001, 221, 1–22. [Google Scholar] [CrossRef]
- Pietrucha, K.; Safandowska, M. Dialdehyde cellulose-crosslinked collagen and its physicochemical properties. Process. Biochem. 2015, 50, 2105–2111. [Google Scholar] [CrossRef]
- Andonegi, M.; Penalba, M.; de la Caba, K.; Guerrero, P. ZnO nanoparticle-incorporated native collagen films with electro-conductive properties. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 108, 110394. [Google Scholar] [CrossRef]
- Prasert, A.; Sontikaew, S.; Sriprapai, D.; Chuangchote, S. Polypropylene/ZnO Nanocomposites: Mechanical Properties, Photocatalytic Dye Degradation, and Antibacterial Property. Materials 2020, 13, 914. [Google Scholar] [CrossRef]
- Lian, J.X.; Agban, Y.; Cheong, S.S.; Kuchel, R.P.; Raudsepp, A.; Williams, M.A.K.; Rupenthal, I.D.; Henning, A.; Tilley, R.D.; Holmes, G.; et al. ZnO/PVP nanoparticles induce gelation in type I collagen. Eur. Polym. J. 2016, 75, 399–405. [Google Scholar] [CrossRef]
- Agban, Y.; Lian, J.; Prabakar, S.; Seyfoddin, A.; Rupenthal, I.D. Nanoparticle cross-linked collagen shields for sustained delivery of pilocarpine hydrochloride. Int. J. Pharm. 2016, 501, 96–101. [Google Scholar] [CrossRef]
- Morgen, M.; Tung, D.; Boras, B.; Miller, W.; Malfait, A.M.; Tortorella, M. Nanoparticles for Improved Local Retention after Intra-Articular Injection into the Knee Joint. Pharm. Res. 2013, 30, 257–268. [Google Scholar] [CrossRef]
- Burckbuchler, V.; Mekhloufi, G.; Giteau, A.P.; Grossiord, J.L.; Huille, S.; Agnely, F. Rheological and syringeability properties of highly concentrated human polyclonal immunoglobulin solutions. Eur. J. Pharm. Biopharm. 2010, 76, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Riaz, T.; Zeeshan, R.; Zarif, F.; Ilyas, K.; Muhammad, N.; Safi, S.Z.; Rahim, A.; Rizvi, S.A.A.; Rehman, I.U. FTIR analysis of natural and synthetic collagen. Appl. Spectrosc. Rev. 2018, 53, 703–746. [Google Scholar] [CrossRef]
- Jackson, M.; Mantsch, H.H. The use and misuse of FTIR spectroscopy in the determination of protein structure. Crit. Rev. Biochem. Mol. Biol. 1995, 30, 95–120. [Google Scholar] [CrossRef] [PubMed]
- Koronis, S.; Stavrakas, P.; Balidis, M.; Kozeis, N.; Tranos, P.G. Update in treatment of uveitic macular edema. Drug Des. Dev. 2019, 13, 667–680. [Google Scholar] [CrossRef] [PubMed]
- Abadia, B.; Calvo, P.; Ferreras, A.; Bartol, F.; Verdes, G.; Pablo, L. Clinical Applications of Dexamethasone for Aged Eyes. Drugs Aging 2016, 33, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, J.R.; Villanueva, L.R.; Navarro, M.G. Pharmaceutical technology can turn a traditional drug, dexamethasone into a first-line ocular medicine. A global perspective and future trends. Int. J. Pharm. 2017, 516, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Lei, L.; Song, Q.; Li, X. Calcium ion cross-linking alginate/dexamethasone sodium phosphate hybrid hydrogel for extended drug release. Colloids Surf. B Biointerfaces 2019, 175, 569–575. [Google Scholar] [CrossRef]
- Kim, J.; Peng, C.C.; Chauhan, A. Extended release of dexamethasone from silicone-hydrogel contact lenses containing vitamin E. J. Control. Release 2010, 148, 110–116. [Google Scholar] [CrossRef]
- Yasin, M.N.; Brooke, R.K.; Rudd, S.; Chan, A.; Chen, W.-T.; Waterhouse, G.I.; Evans, D.; Rupenthal, I.D.; Svirskis, D. 3-Dimensionally ordered macroporous PEDOT ion-exchange resins prepared by vapor phase polymerization for triggered drug delivery: Fabrication and characterization. Electrochim. Acta 2018, 269, 560–570. [Google Scholar] [CrossRef]




| Band | Wavelength (cm−1) |
|---|---|
| Amide A | 3305 |
| Amide B | 3018 |
| Amide I | 1633 |
| Amide II | 1552 |
| Amide III | 1239 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agban, Y.; Mugisho, O.O.; Thakur, S.S.; Rupenthal, I.D. Characterization of Zinc Oxide Nanoparticle Cross-Linked Collagen Hydrogels. Gels 2020, 6, 37. https://doi.org/10.3390/gels6040037
Agban Y, Mugisho OO, Thakur SS, Rupenthal ID. Characterization of Zinc Oxide Nanoparticle Cross-Linked Collagen Hydrogels. Gels. 2020; 6(4):37. https://doi.org/10.3390/gels6040037
Chicago/Turabian StyleAgban, Yosra, Odunayo O. Mugisho, Sachin S. Thakur, and Ilva D. Rupenthal. 2020. "Characterization of Zinc Oxide Nanoparticle Cross-Linked Collagen Hydrogels" Gels 6, no. 4: 37. https://doi.org/10.3390/gels6040037
APA StyleAgban, Y., Mugisho, O. O., Thakur, S. S., & Rupenthal, I. D. (2020). Characterization of Zinc Oxide Nanoparticle Cross-Linked Collagen Hydrogels. Gels, 6(4), 37. https://doi.org/10.3390/gels6040037