Antibacterial Properties of Silver Nanoparticles Embedded on Polyelectrolyte Hydrogels Based on α-Amino Acid Residues
Abstract
:1. Introduction
2. Results and Discussion
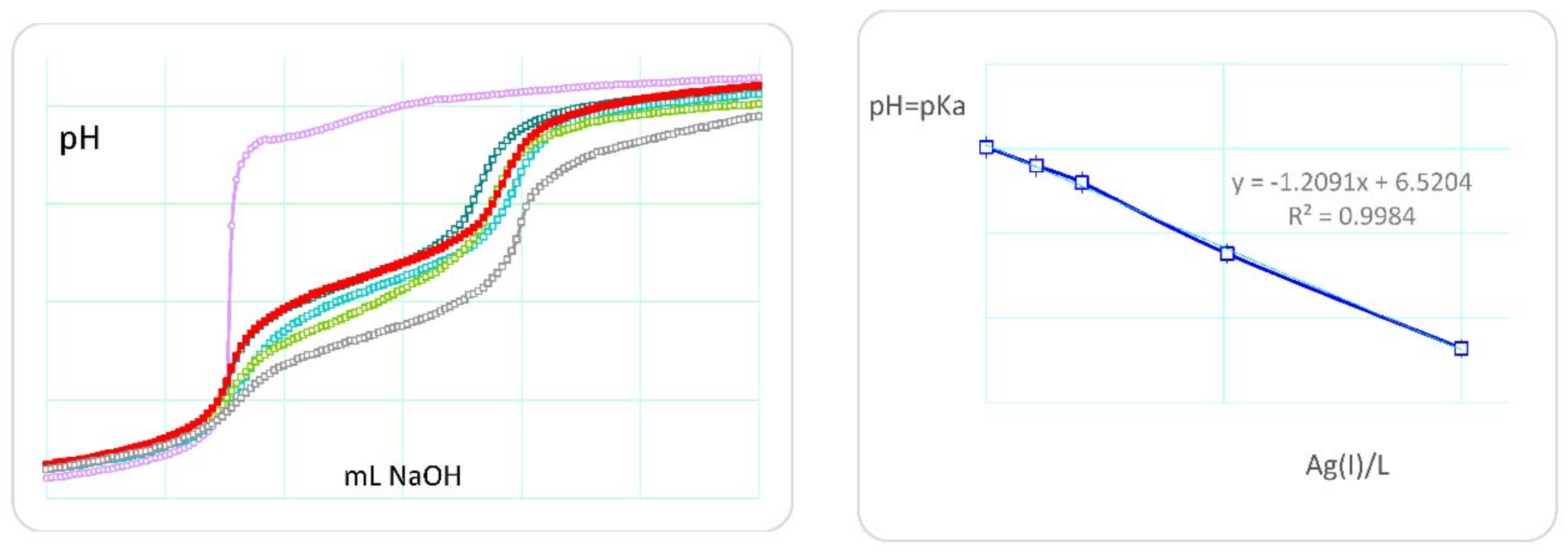
2.1. Protonation and Complex Formation Thermodynamics
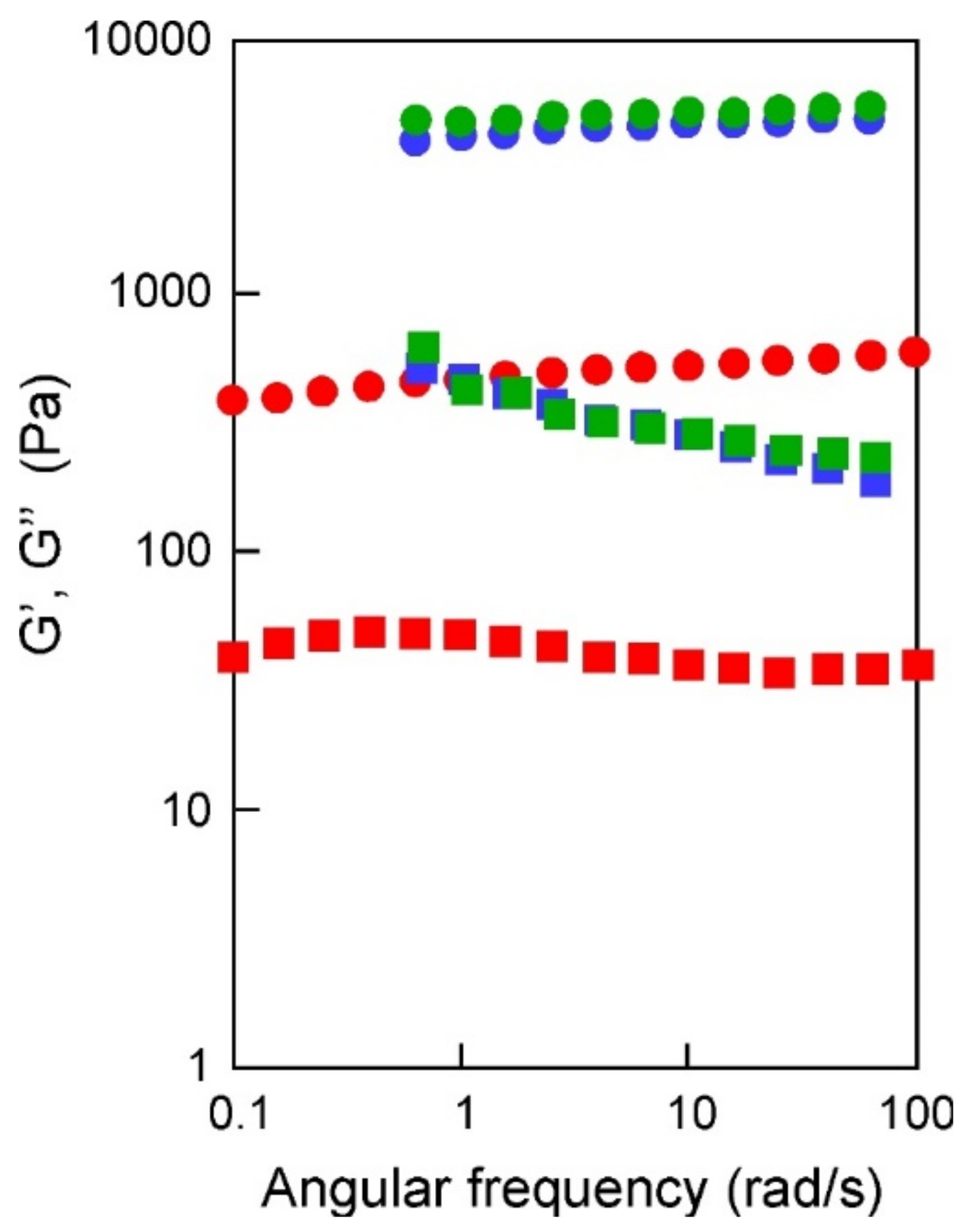
2.2. Syntheses and Characterization
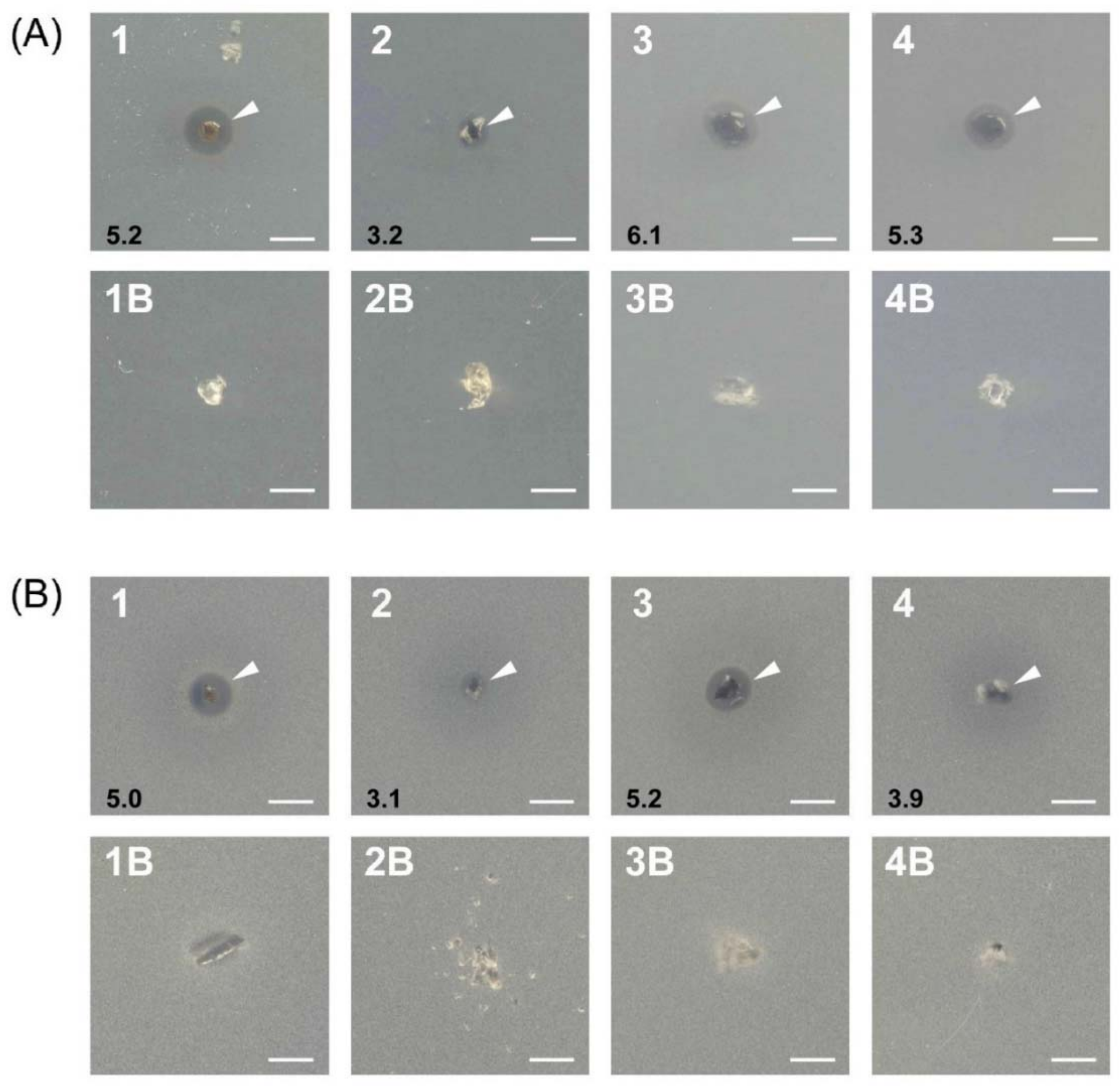
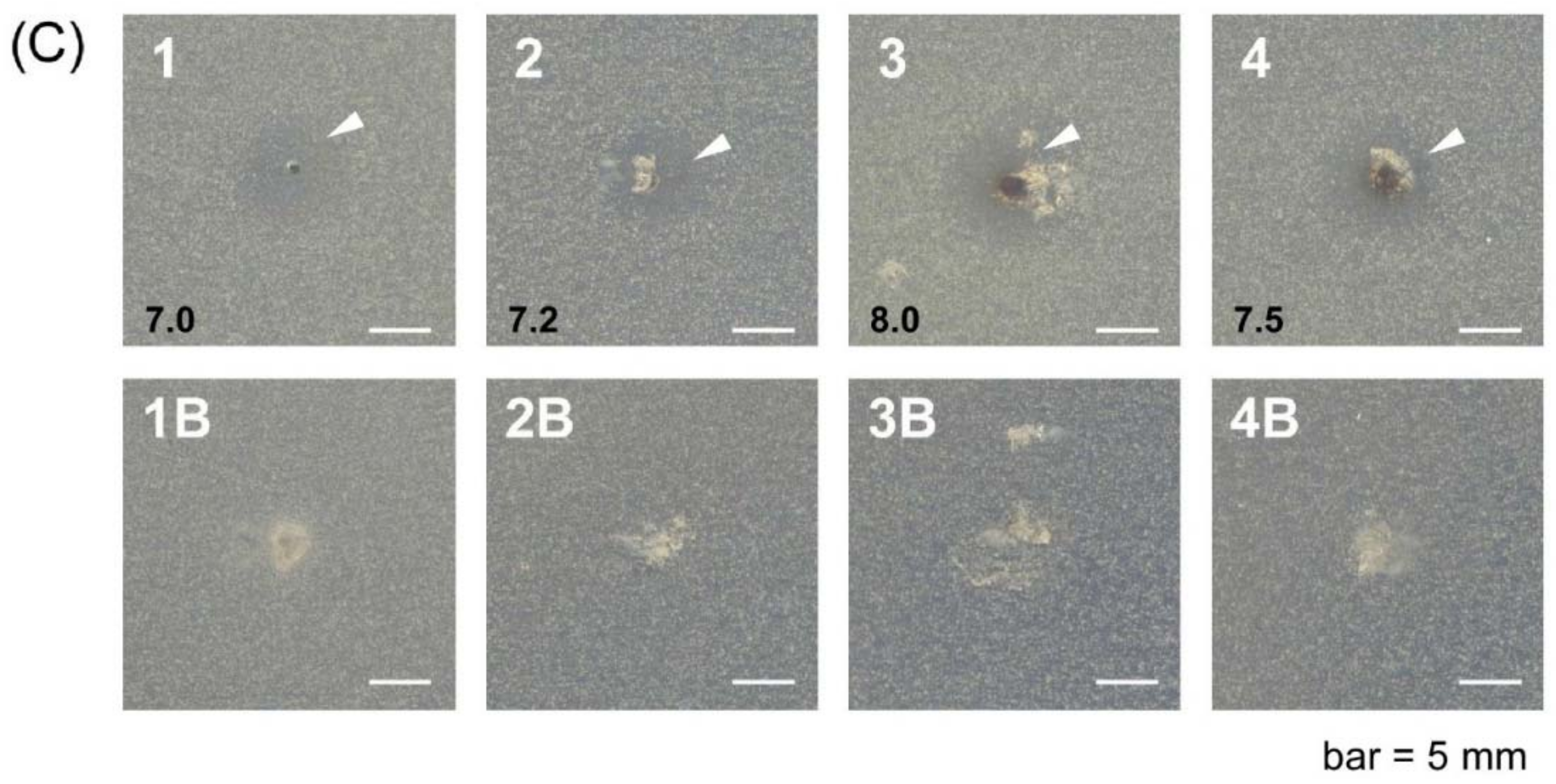
2.3. In Vitro Antimicrobial Activity
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Synthesis of Hydrogels
4.3. Swelling
4.4. Potentiometric Titrations
4.5. Spectroscopic Characterization
4.6. Rheological Measurements
4.7. Antibacterial and Antifungal Activity Studies
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Knipe, J.M.; Peppas, N.A. Multi-responsive hydrogels for drug delivery and tissue engineering applications. Regen. Biomater. 2014, 1, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Lorenzo, C.; Concheiro, A. (Eds.) Smart Materials for Drug Delivery; RSC Publishing: Cambridge, UK, 2013. [Google Scholar]
- Bauri, K.; Nandi, M.; De, P. Amino acid-derived stimuli-responsive polymers and their applications. Polym. Chem. 2018. [Google Scholar] [CrossRef]
- Casolaro, M.; Casolaro, I. Stimuli-responsive hydrogels bearing-aminoacid residues: A potential platform for future therapies. Biomed. Eng. Med. Dev. 2016, 1, 1. [Google Scholar]
- Casolaro, M.; Casolaro, I. Multiple stimuli-responsive hydrogels for metal-based drug therapy. Polymers 2012, 4, 964–985. [Google Scholar] [CrossRef]
- Casolaro, M.; Cini, R.; Del Bello, B.; Ferrali, M.; Maellaro, E. Cisplatin/hydrogel complex in cancer therapy. Biomacromolecules 2009, 10, 944–949. [Google Scholar] [CrossRef] [PubMed]
- Casolaro, M.; Del Bello, B.; Maellaro, E. Hydrogel containing l-valine residues as a platform for cisplatin chemotherapy. Coll. Surf. B Biointerfaces 2011, 88, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Tamasi, G.; Serinelli, F.; Consumi, M.; Magnani, A.; Casolaro, M.; Cini, R. Release studies from smart hydrogels as carriers for piroxicam and copper(II)-oxicam complexes as anti-inflammatory and anti-cancer drugs. X-ray structures of new copper(II)-piroxicam and isoxicam complex molecules. J. Inorg. Biochem. 2008, 102, 1862–1873. [Google Scholar] [CrossRef] [PubMed]
- Tamasi, G.; Casolaro, M.; Magnani, A.; Sega, A.; Chiasserini, L.; Messori, L.; Gabbiani, C.; Valiahdi, S.M.; Jakupec, M.; Keppler, B.K. New platinum-oxicam complexes as anti-cancer drugs. Synthesis, characterization, release studies from smart hydrogels, evaluation of reactivity with selected proteins and cytotoxic activity in vitro. J. Inorg. Biochem. 2010, 104, 799–814. [Google Scholar] [CrossRef] [PubMed]
- Casolaro, M.; Casolaro, I.; Bottari, S.; Del Bello, B.; Maellaro, E.; Demadis, K.D. Long-term doxorubicin release from multiple stimuli-responsive hydrogels based on α-amino-acid residues. Eur. J. Pharm. Biopharm. 2014, 88, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Casolaro, M.; Casolaro, I.; Lamponi, S. Stimuli-responsive hydrogels for controlled pilocarpine ocular delivery. Eur. J. Pharm. Biopharm. 2012, 80, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Casolaro, M.; Casolaro, I. Controlled release of antidepressant drugs by multiple stimuli-sensitive hydrogels based on α-aminoacid residues. J. Drug Deliv. Sci. Technol. 2015, 30, 82–89. [Google Scholar] [CrossRef]
- Casolaro, M.; Casolaro, I. Polyelectrolyte hydrogel platforms for the delivery of antidepressant drugs. Gels 2016, 2, 24. [Google Scholar] [CrossRef]
- Casolaro, M.; Casolaro, I. Pulsed release of antidepressants from nanocomposite hydrogels. Biol. Eng. Med. 2017, 2, 1–8. [Google Scholar] [CrossRef]
- Zhang, X.-F.; Liu, Z.-G.; Shen, W.; Gurunathan, S. Silver nanoparticles: Synthesis, characterization, properties, applications, and therapeutic approaches. Int. J. Mol. Sci. 2016, 17, 1534. [Google Scholar] [CrossRef] [PubMed]
- Kędziora, A.; Speruda, M.; Krzyżewska, E.; Rybka, J.; Łukowiak, A.; Bugla-Płoskońska, G. Similarities and differences between silver ions and silver in nanoforms as antibacterial agents. Int. J. Mol. Sci. 2018, 19, 444. [Google Scholar] [CrossRef] [PubMed]
- Li, W.-R.; Xie, X.-B.; Shi, Q.-S.; Zheng, H.-Y.; OU-Yang, Y.-S.; Chen, Y.-B. Antibacterial activity and mechanism of silver nanoparticles on Escherichia coli. Appl. Microbiol. Biotechnol. 2010, 85, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Liu, W.; Luo, B.; Tian, J.; Wen, W.; Liu, M.; Zhou, C. Antibacterial activity and cytocompatibility of chitooligosaccharide-modified polyurethane membrane via polydopamine adhesive layer. Carbohydr. Polym. 2017, 156, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Ma, J.; Tang, C.Y.; Wang, Z.; Ng, H.Y.; Wu, Z. Antibiofouling polyvinylidene fluoride membrane modified by quaternary ammonium compound: Direct contact-killing versus induced indirect contact killing. Environ. Sci. Technol. 2016, 50, 5086–5093. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Wada, A.; Tsuzuki, S.; Casolaro, M.; Ito, Y. Copolymers including L-histidine and hydrophobic moiety for preparation of nonbiofouling surface. Biomacromolecules 2007, 8, 3340–3344. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Song, X.; Xiang, T.; Liu, Q.; Su, B.; Zhao, W.; Zhao, C. Mussel-inspired chitosan-polyurethane coatings for improving the antifouling and antibacterial properties of polyethersulfone membranes. Carbohydr. Polym. 2017, 168, 310–319. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Wang, Q.; Zhang, J.; Zhao, W.; Zhao, C. Substrate-independent Ag-nanoparticle-loaded hydrogel coating with regenerable bactericidal and thermoresponsive antibacterial properties. ACS Appl. Mater. Interfaces 2017, 9, 44782–44791. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Xie, Y.; Xiao, T.; Zhao, W.; Li, J.; Zhao, C. Tannic acd-inspiration and post-crosslinking of zwitterionic polymer as a universal approach towards antifouling surface. Chem. Eng. J. 2018, 337, 122–132. [Google Scholar] [CrossRef]
- Huang, J.-F.; Zhong, J.; Chen, G.-P.; Lin, Z.-T.; Deng, Y.; Liu, Y.-L.; Cao, P.-Y.; Wang, B.; Wei, Y.; Wu, T.; et al. A hydrogel-based hybrid theranostic contact lens for fungal keratitis. ACS Nano 2016, 10, 6464–6473. [Google Scholar] [CrossRef] [PubMed]
- Lengert, E.; Saveleva, M.; Abalymov, A.; Atkin, V.; Wuytens, P.C.; Kamyshinsky, R.; Vasiliev, A.L.; Gorin, D.A.; Sukhorukov, G.B.; Skirtach, A.G.; et al. Silver alginate hydrogel micro- and nanocontainers for theranostics: Synthesis, encapsulation, remote release, and detection. Appl. Mater. Interfaces 2017, 9, 21949–21958. [Google Scholar] [CrossRef] [PubMed]
- Gonzàles-Sànchez, M.I.; Perni, S.; Tommasi, G.; Morris, N.G.; Hawkins, K.; Lòpez-Cabarcos, E.; Prokopovich, P. Silver nanoparticle based antibacterial methacrylate hydrogels potential for bone graft applications. Mater. Sci. Eng. C 2015, 50, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Varaprasad, K.; Mohan, Y.M.; Vimala, K.; Raju, K.M. Synthesis and characterization of hydrogel-silver nanoparticle-curcumin composites for wound dressing and antibacterial application. J. Appl. Polym. Sci. 2011, 121, 784–796. [Google Scholar] [CrossRef]
- Kim, Y.; Babu, V.R.; Thangadurai, D.T.; Rao, K.S.V.K.; Cha, H.; Kim, C.; Joo, W.; Lee, Y.-I. Synthesis, characterization, and antibacterial applications of novel copolymeric silver nanocomposite hydrogels. Bull. Korean Chem. Soc. 2011, 32, 553–558. [Google Scholar] [CrossRef]
- Smith, R.M.; Martell, A.E. Critical Stability Constants; Plenum Press: New York, NY, USA, 1975. [Google Scholar]
- Bauman, J.E.; Wang, J.C. Imidazole Complexes of Nickel(II), Copper(II), Zinc(II), and Silver(I). Inorg. Chem. 1964, 3, 368–373. [Google Scholar] [CrossRef]
- Czoik, R.; Heintz, A.; John, E.; Marczak, W. Complexes of silver with histidine and imidazole investigated by the calorimetric and potentiometric methods. Acta Phys. Pol. A 2008, 114, A51–A56. [Google Scholar] [CrossRef]
- Krot, K.A.; Danil de Namor, A.F.; Aguilar-Cornejo, A.; Nolan, K.B. Speciation, stability constants and structures of complexes of copper(II), nickel(II), silver(I) and mercury(II) with PAMAM dentrimer and related tetraamide ligands. Inorg. Chim. Acta 2005, 358, 3497–3505. [Google Scholar] [CrossRef]
- Solomon, S.D.; Bahadory, M.; Jeyarajasingam, A.V.; Rutkowsky, S.A.; Boritz, C.; Mulfinger, L. Synthesis and study of silver nanoparticles. J. Chem. Educ. 2007, 84, 322. [Google Scholar]
- Rao, K.M.; Kumar, A.; Haider, A.; Han, S.S. Polysaccharides based antibacterial polyelectrolyte hydrogels with silver nanoparticles. Mater. Lett. 2016, 184, 189–192. [Google Scholar] [CrossRef]
- Caldera-Villalobos, M.; Garcia-Serrano, J.; Pelaez-Cid, A.A.; Herrera-Gonzalez, A.M. Polyelectrolytes with sulfonate groups obtained by chemical modification of chitosan useful in green synthesis of Au and Ag nanoparticles. J. Appl. Polym. Sci. 2017, 134. [Google Scholar] [CrossRef]
- He, Y.; Huang, G.; Pan, Z.; Liu, Y.; Gong, Q.; Yao, C.; Gao, J. Polyelectrolyte induced formation of silver nanoparticles in copolymer hydrogel and their application as catalyst. Mater. Res. Bull. 2015, 70, 263–271. [Google Scholar] [CrossRef]
- Endo, T.; Ikeda, R.; Yanagida, Y.; Hatsuzawa, T. Stimuli-responsive hydrogel-silver nanoparticles composite for development of localized surface plasmon resonance-based optical biosensor. Anal. Chim. Acta 2008, 611, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.F.; Du, Y.M.; Hu, X.W.; Shi, X.W.; Kennedy, J.F. Rheological characterization of novel thermosensitive chitosan/poly(vinyl alcohol) blend hydrogel. Carbohyd. Polym. 2007, 67, 491–499. [Google Scholar] [CrossRef]
- Lubick, N. Nanosilver toxicity: Ions, nanoparticles-or both? Environ. Sci. Technol. 2008, 42, 8617. [Google Scholar] [CrossRef] [PubMed]
- Casolaro, M. Thermodynamics of multiple stimuli-responsive polyelectrolytes with complexing ability towards the copper(II) ion. React. Polym. 1994, 23, 71–83. [Google Scholar] [CrossRef]
- Casolaro, M.; Paccagnini, E.; Mendichi, R. Stimuli-responsive polymers based on L-phenylalanine residues: Protonation thermodynamics of free polymers and cross-linked hydrogels. Macromolecules 2005, 38, 2460–2468. [Google Scholar] [CrossRef]
- Casolaro, M.; Bottari, S.; Cappelli, A.; Mendichi, R.; Ito, Y. Vinyl polymers based on L-histidine residues. Part 1. The thermodynamics of poly(ampholyte)s in the free and in the cross-linked gel form. Biomacromolecules 2004, 5, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Casolaro, M.; Ito, Y.; Ishii, T.; Bottari, S.; Samperi, F.; Mendichi, R. Stimuli-responsive poly(ampholyte)s containing L-histidine residues: Synthesis and protonation thermodynamics of methacrylic polymers in the free and in the cross-linked gel forms. eXPRESS Polym. Lett. 2008, 2, 165–183. [Google Scholar] [CrossRef]
- Amendola, V.; Bakr, O.M.; Stellacci, F. A study of the surface plasmon resonance of silver nanoparticles by the discrete dipole approximation method: Effect of shape, size, structure, and assembly. Plasmonics 2010, 5, 85–97. [Google Scholar] [CrossRef]
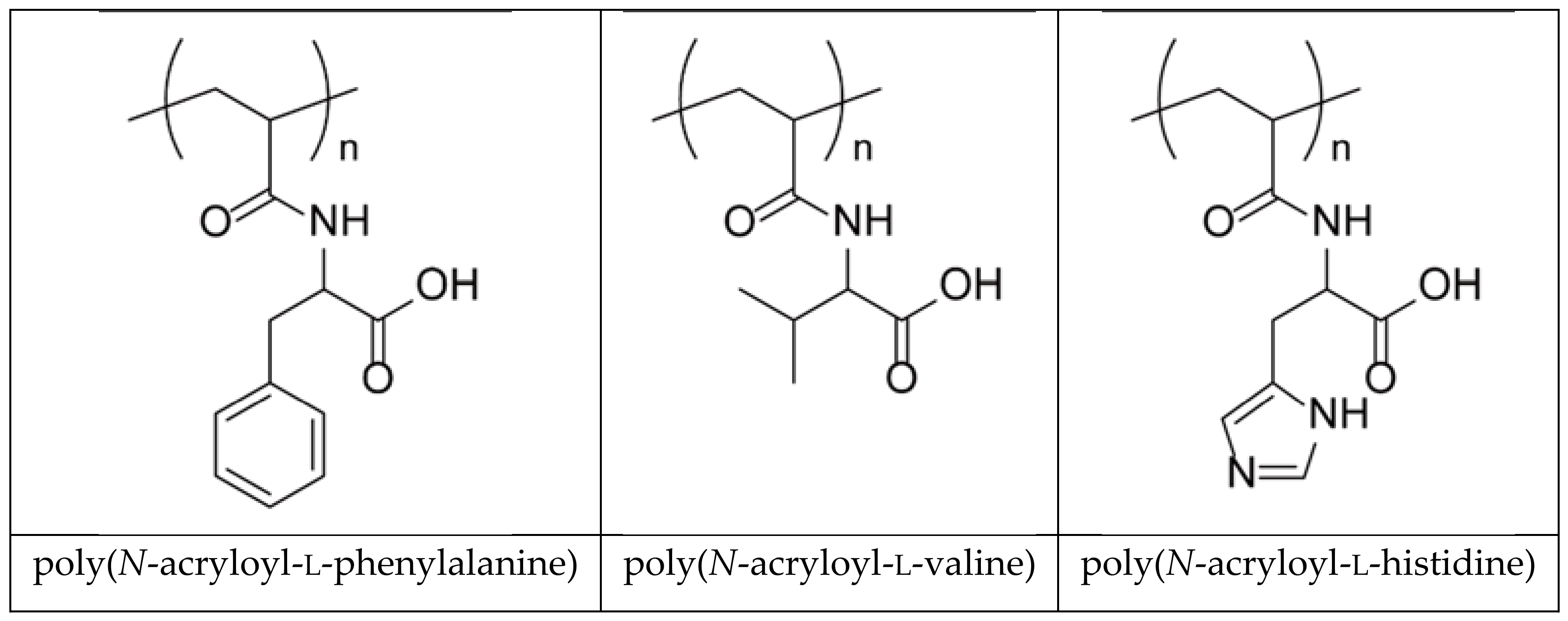
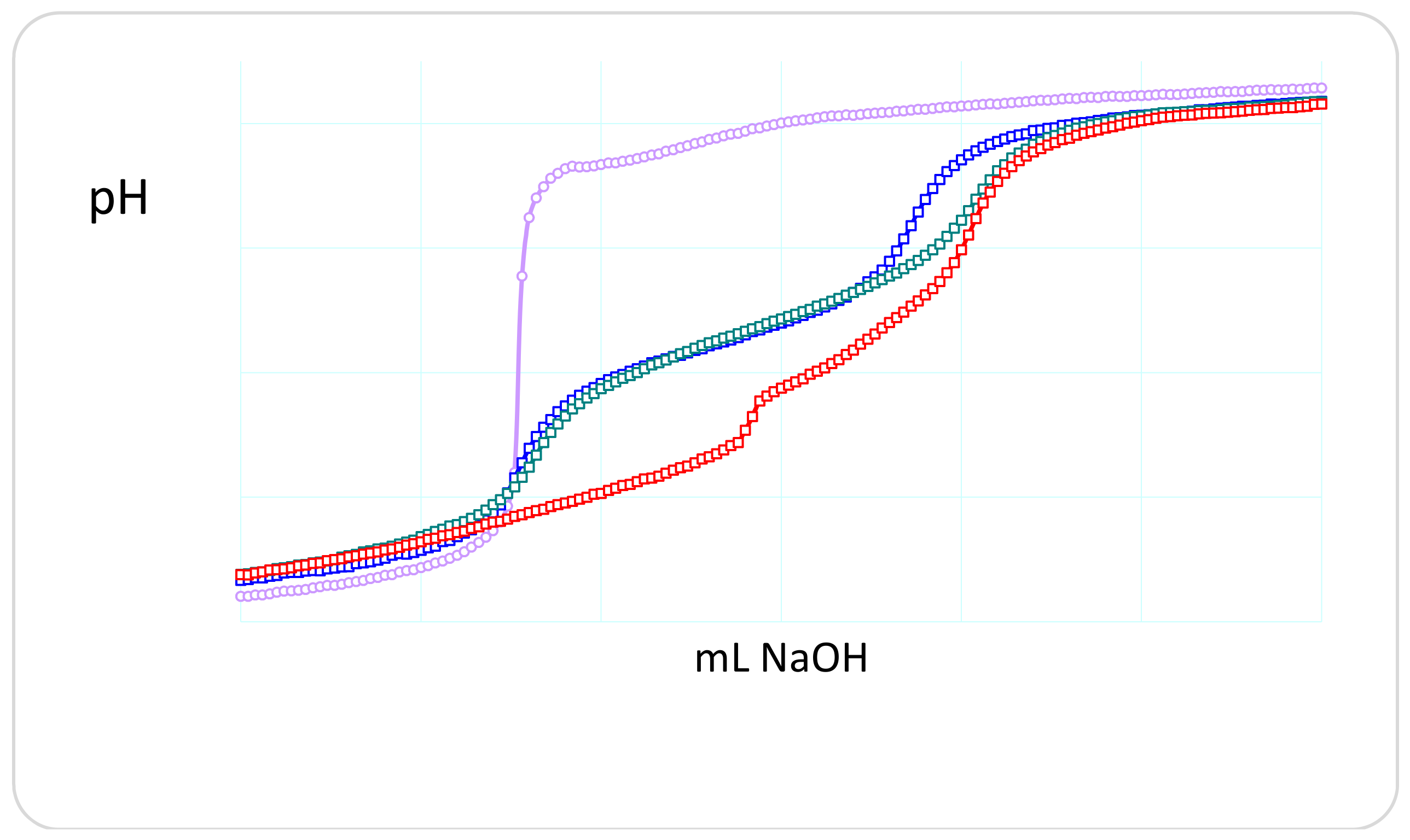

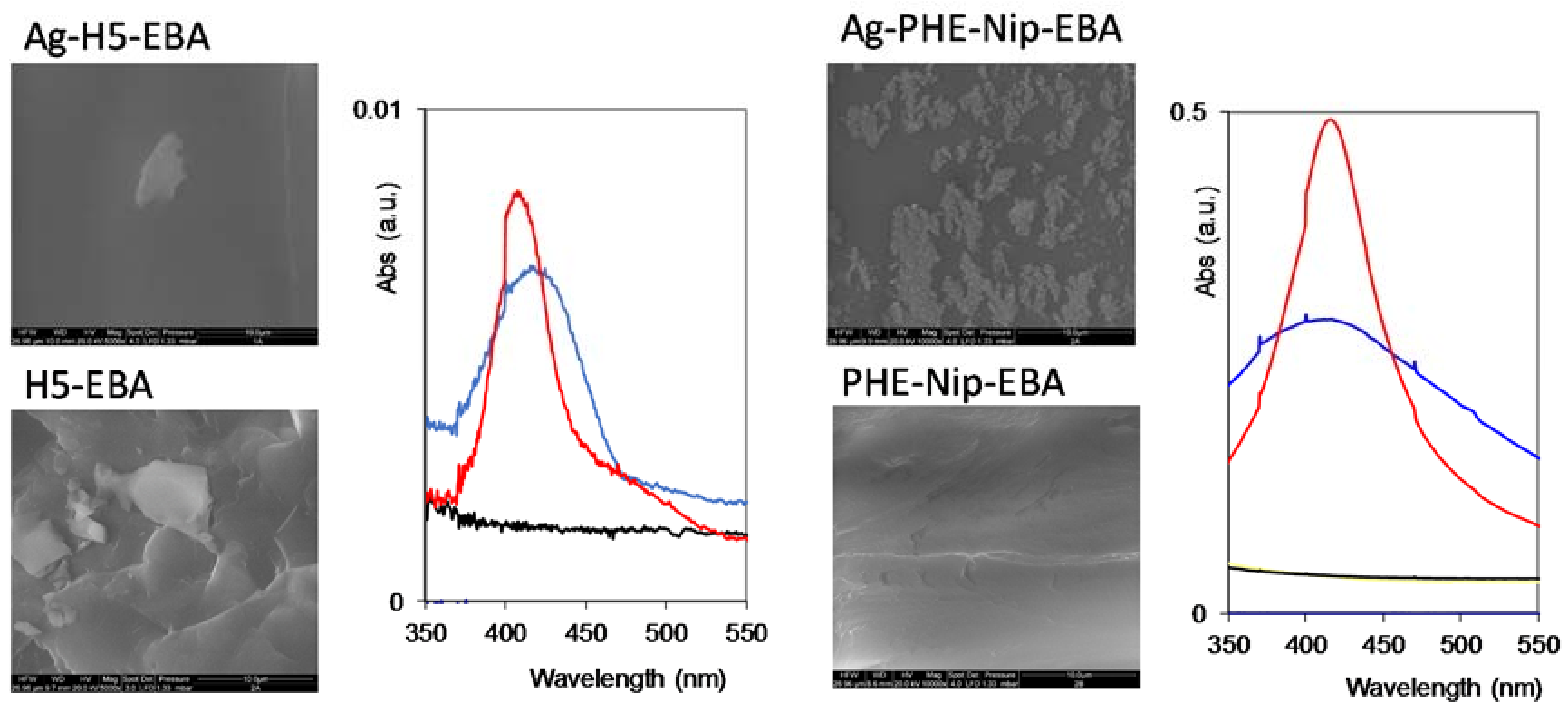
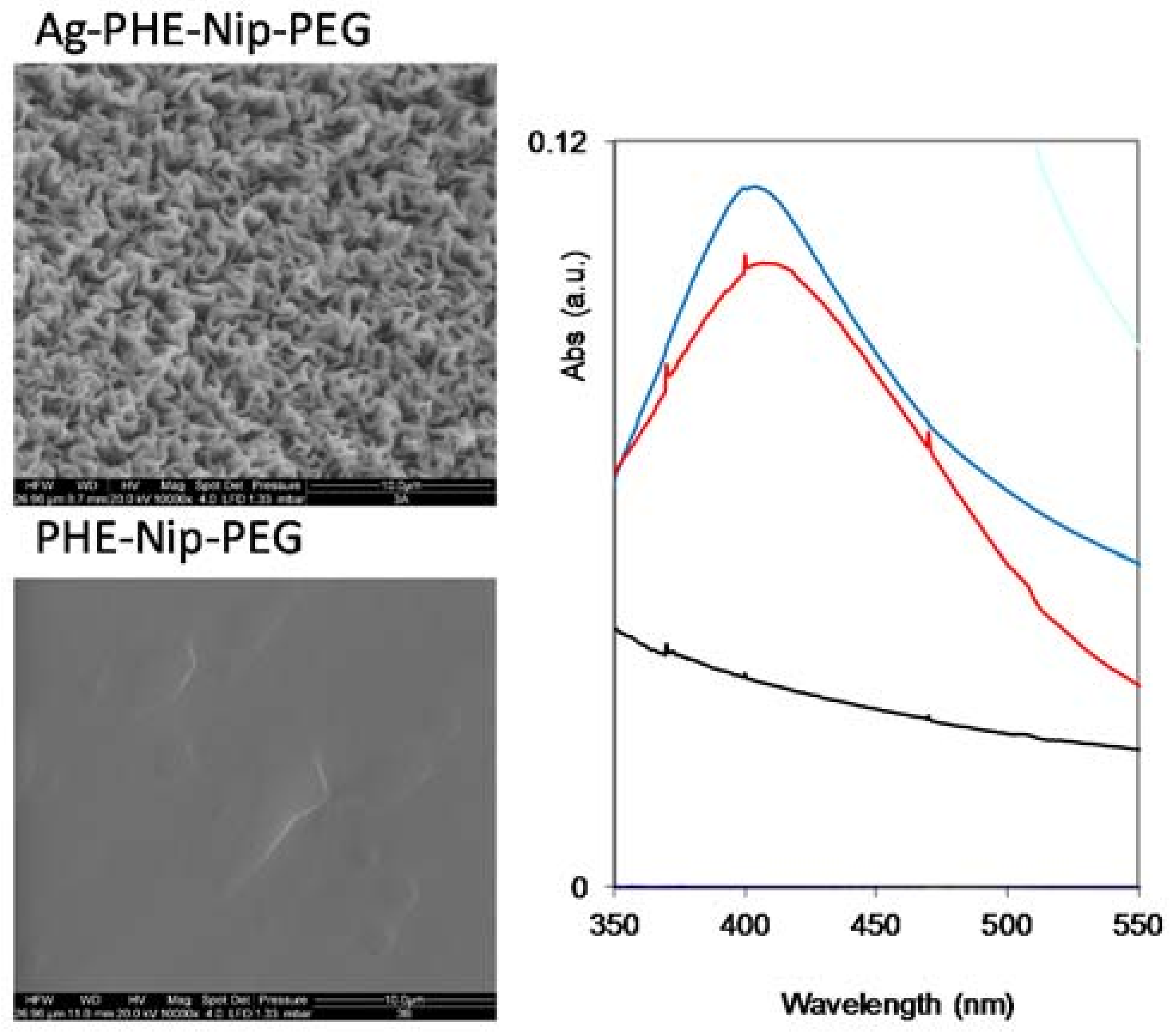

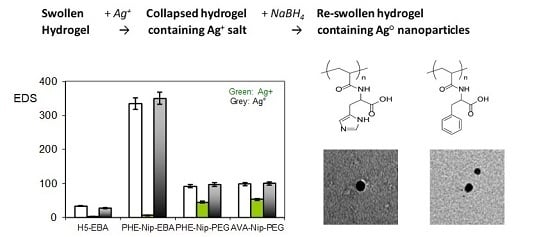
| Hydrogel | Monomer Composition (mol %) | Cross-Linking Composition (mol %) | ||||
|---|---|---|---|---|---|---|
| Hist | PHE | AVA | Nip | EBA | PEG-DA258 | |
| H5-EBA | 100 | - | - | - | 5 | - |
| PHE-Nip-EBA | - | 50 | - | 50 | 2 | - |
| PHE-Nip-PEG | - | 10 | - | 90 | - | 12 |
| AVA-Nip-PEG | - | - | 10 | 90 | - | 12 |
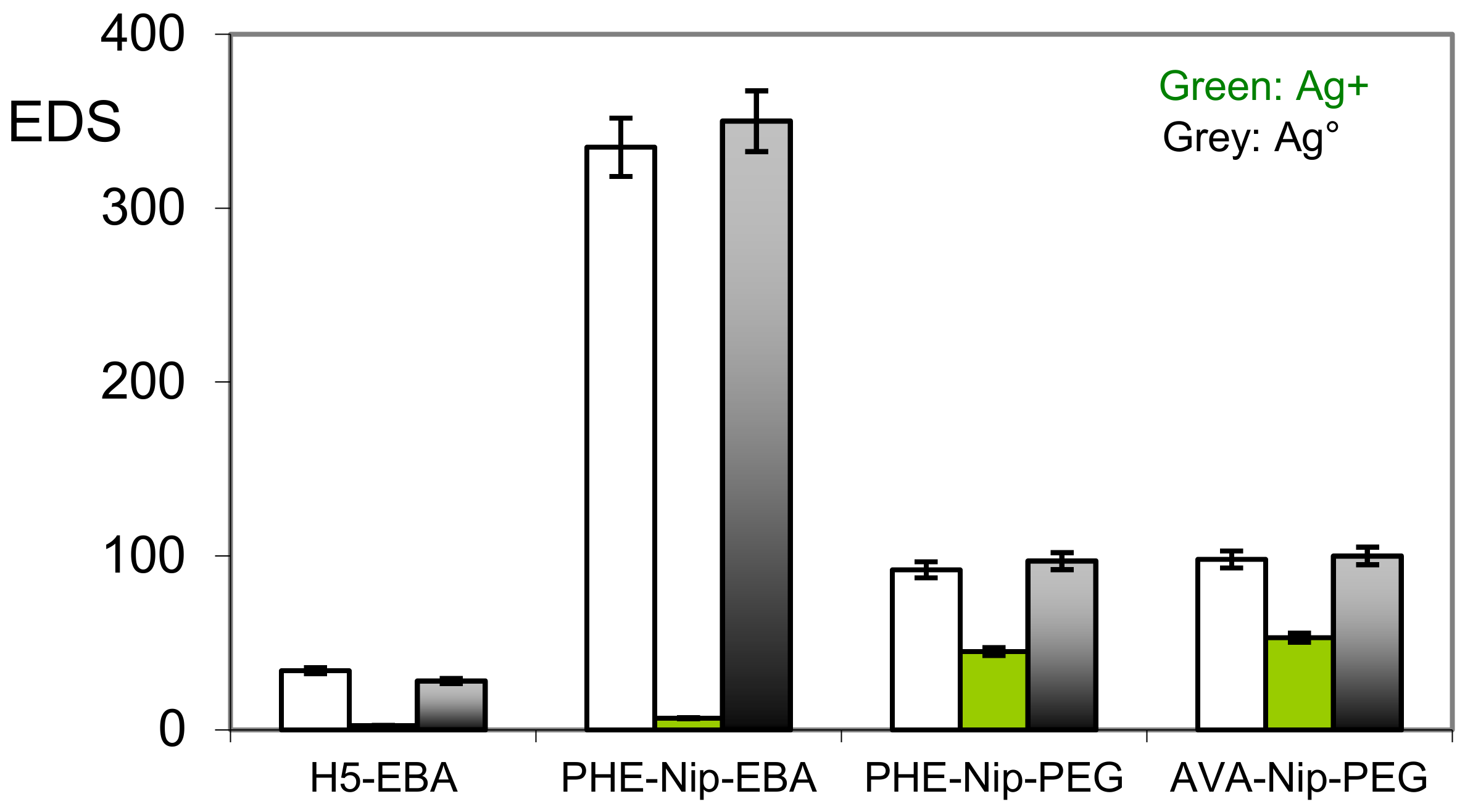
| Hydrogel | Dry Sample | EDS 1 (Swollen) | Conc. Ag+ (mM) | Residence Time (h) | EDS 2 | Conc. NaBH4 (mM) | EDS 3 | Ag° Increase (wt %) |
|---|---|---|---|---|---|---|---|---|
| H5-EBA | 10.5 | 34.3 | 13.5 | 24 | 2.5 | 40.9 | 27.7 | - |
| PHE-Nip-EBA | 9.2 | 335.0 | 6.5 | 24 | 6.7 | 38.3 | 350.0 | - |
| PHE-Nip-PEG | 126.3 | 91.4 | 10.6 | 24 | 45.2 | 34.0 | 97.9 | 4.2 |
| AVA-Nip-PEG | 98.1 | 98.5 | 10.6 | 24 | 53.4 | 34.0 | 100.3 | 7.4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casolaro, M.; Casolaro, I.; Akimoto, J.; Ueda, M.; Ueki, M.; Ito, Y. Antibacterial Properties of Silver Nanoparticles Embedded on Polyelectrolyte Hydrogels Based on α-Amino Acid Residues. Gels 2018, 4, 42. https://doi.org/10.3390/gels4020042
Casolaro M, Casolaro I, Akimoto J, Ueda M, Ueki M, Ito Y. Antibacterial Properties of Silver Nanoparticles Embedded on Polyelectrolyte Hydrogels Based on α-Amino Acid Residues. Gels. 2018; 4(2):42. https://doi.org/10.3390/gels4020042
Chicago/Turabian StyleCasolaro, Mario, Ilaria Casolaro, Jun Akimoto, Motoki Ueda, Masashi Ueki, and Yoshihiro Ito. 2018. "Antibacterial Properties of Silver Nanoparticles Embedded on Polyelectrolyte Hydrogels Based on α-Amino Acid Residues" Gels 4, no. 2: 42. https://doi.org/10.3390/gels4020042
APA StyleCasolaro, M., Casolaro, I., Akimoto, J., Ueda, M., Ueki, M., & Ito, Y. (2018). Antibacterial Properties of Silver Nanoparticles Embedded on Polyelectrolyte Hydrogels Based on α-Amino Acid Residues. Gels, 4(2), 42. https://doi.org/10.3390/gels4020042