Dicarboxylic Amino Acid Permease 7219 Regulates Fruiting Body Type of Auricularia heimuer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strains and Culture Conditions
2.2. Nucleic Acid Extraction
2.3. Primary Mapping of Fruiting Body Type Trait
2.4. Fine Mapping of Fruiting Body Type Trait and SCL-18 Alignment
2.5. Transcriptome Analysis, qRT-PCR Validation, and Candidate Gene Screening
2.6. Polymorphism Analysis for Promoter Base Detection of Candidate Genes
2.7. Statistical Analysis
3. Results
3.1. Classification of Fruiting Body Types of Locational Groups
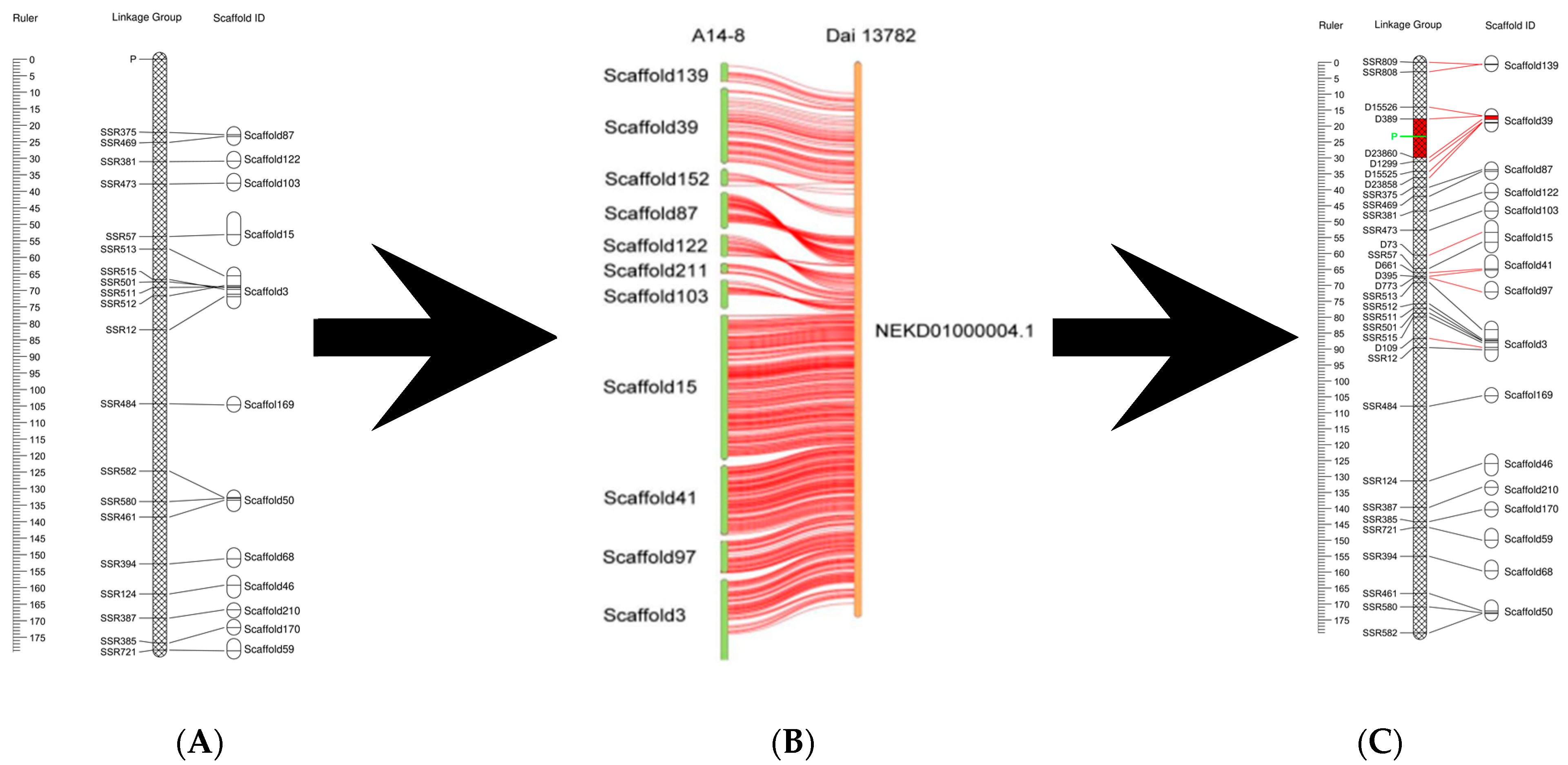
3.2. Localization of the Fruiting Body Type Locus
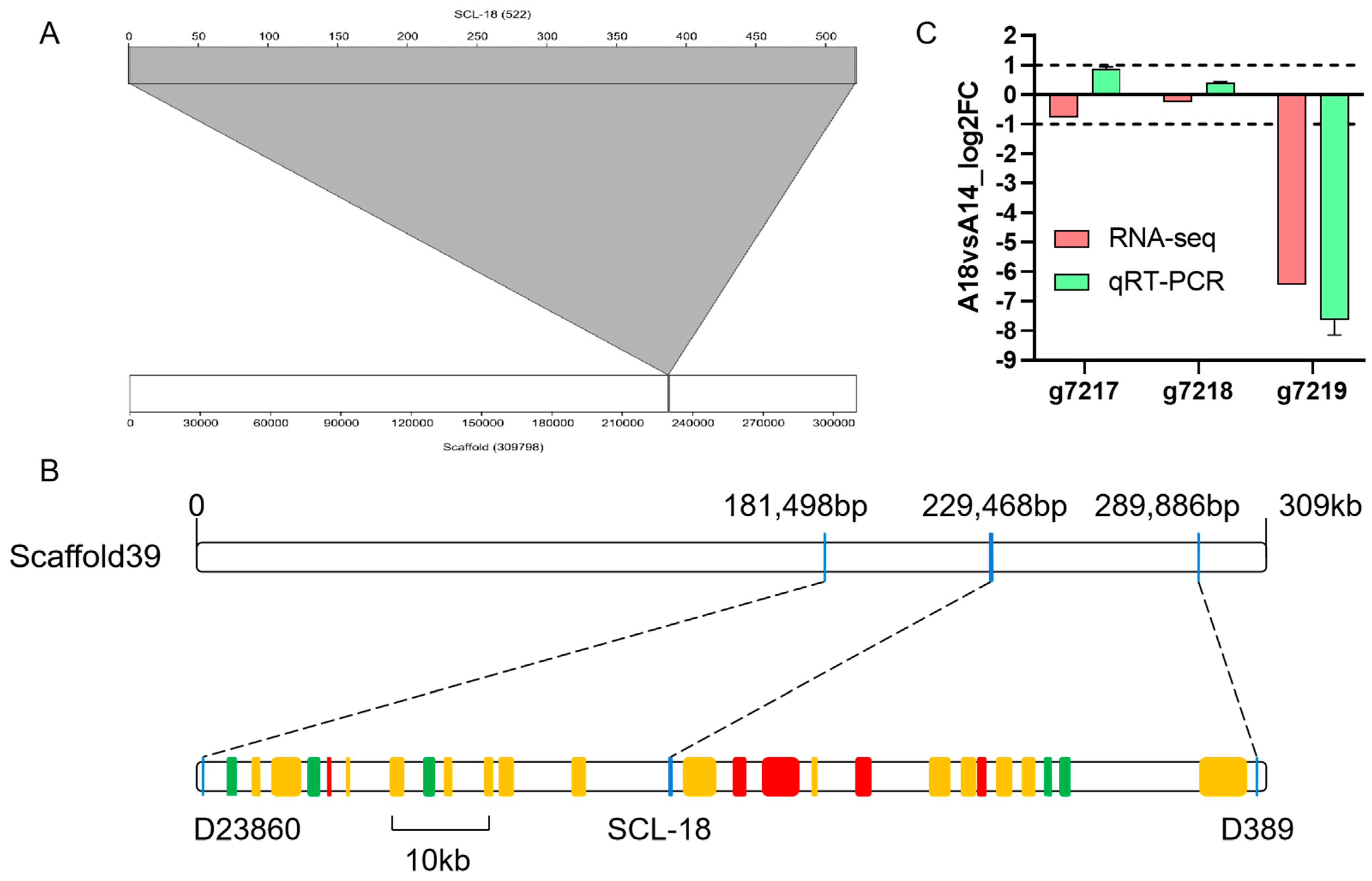
3.3. SCL-18 Alignment and Screening of Candidate Genes
3.4. Nucleotide Sequence of the g7219 Gene
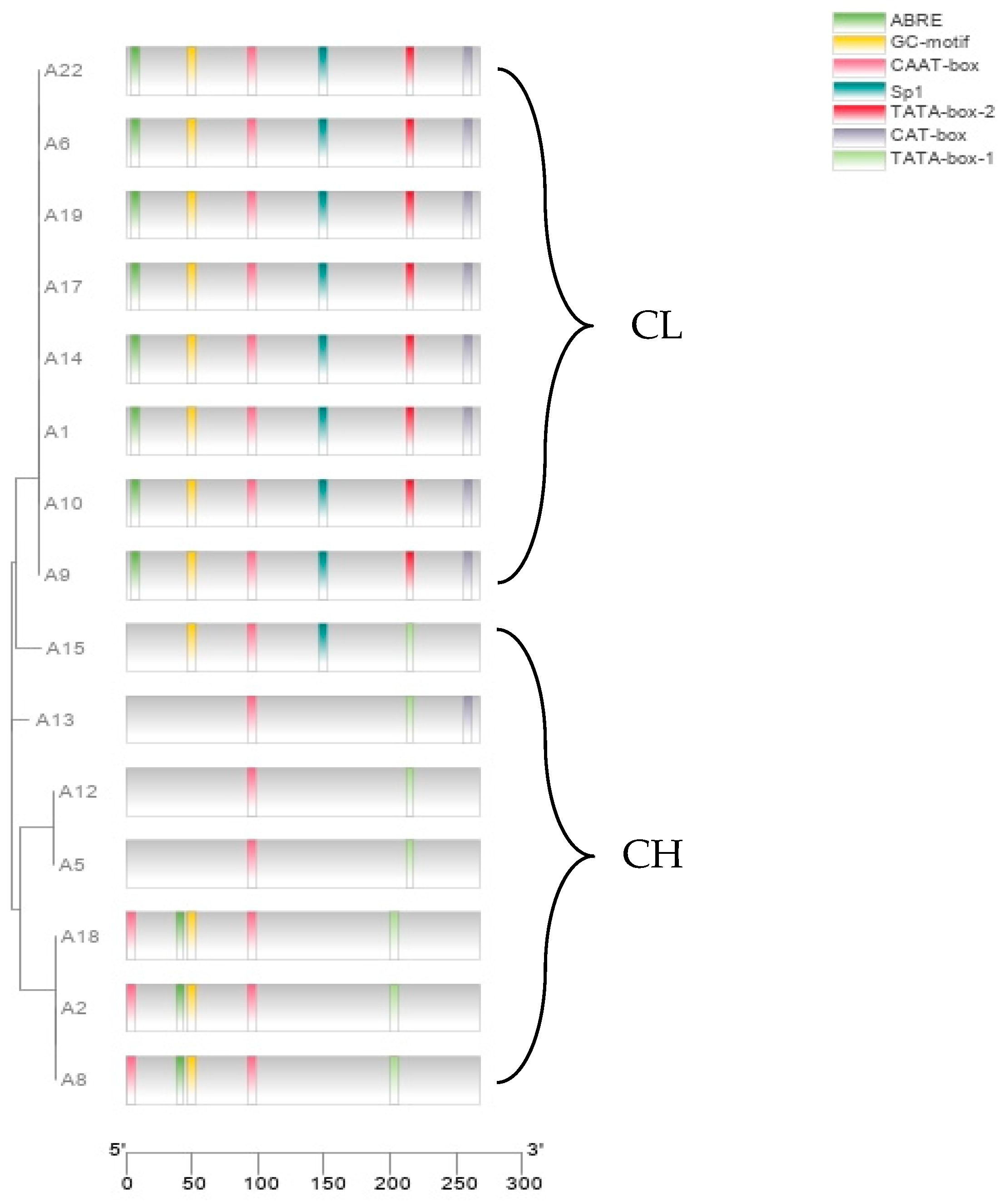
3.5. Polymorphism in the Promoter of the g7219 Gene
4. Discussion
4.1. Primary Positioning
4.2. Increased Density of Genetic Linkage Map
4.3. Fine Positioning of Fruiting Body Type Locus of A. heimuer
4.4. Physical Location of Fruiting Body Type Markers and Functional Prediction of Candidate Genes
4.5. Polymorphism of Candidate Genes in Strains with Different Fruiting Body Types
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dai, Y.C.; Li, Y. Notes on the nomenclature of six important medicinal fungi in China. Mycosystema 2011, 30, 515–518. [Google Scholar] [CrossRef]
- Yao, F.J. The Cultivation Spread of Auricularia auricula from North to South in China. Edible Fungi China 2012, 31, 61–62. [Google Scholar] [CrossRef]
- China Edible Fungi Association. Analysis of the results of the national edible mushroom statistical survey in 2021. Edible Fungi China 2023, 42, 118–127. [Google Scholar] [CrossRef]
- Yao, F.J.; Zhang, Y.M.; Lu, L.X.; Fang, M. Research Progress on Genetics and Breeding of Auricularia auricula-judae. J. Fung. Res. 2015, 13, 125–128. [Google Scholar] [CrossRef]
- Chen, Y.; Yao, F.J.; Zhang, Y.M.; Fang, M. Numerical classification of cultivated germplasm of Auricularia auricula-judae. Mycosystema 2014, 33, 984–996. [Google Scholar] [CrossRef]
- Chen, Y.; Yao, F.J.; Zhang, Y.M.; Chen, L.; Cong, S.; Fang, M. A New Auricularia auricula-judae Cultivar ‘Jihei 3’. Acta Hortic. Sin. 2014, 41, 1751–1752. [Google Scholar] [CrossRef]
- Yao, F.J.; Lu, L.X.; Wang, P.; Fang, M.; Zhang, Y.M.; Chen, Y.; Zhang, W.T.; Kong, X.H.; Lu, J.; Honda, Y. Development of a Molecular Marker for Fruiting Body Pattern in Auricularia auricula-judae. Mycobiology 2018, 46, 72–78. [Google Scholar] [CrossRef]
- Michelmore, R.W.; Paran, I.; Kesseli, R.V. Identification of markers linked to disease-resistance genes by bulked segregant analysis: A rapid method to detect markers in specific genomic regions by using segregating populations. Proc. Natl. Acad. Sci. USA 1991, 88, 9828–9832. [Google Scholar] [CrossRef]
- Li, Z.; Xu, Y. Bulk segregation analysis in the NGS era: A review of its teenage years. Plant J. 2022, 109, 1355–1374. [Google Scholar] [CrossRef]
- Xiao, Y.; Tang, L.H.; Bian, Y.B. Preliminary application of degenerate primers in study on the mating factor of Auricularia auricula. Mycosystema 2006, 25, 682–685. [Google Scholar] [CrossRef]
- Ryu, J.; Kim, M.K.; Ro, H.; Kang, Y.M.; Kwon, J.; Kong, W.; Lee, H. Identification of mating type loci and development of SCAR marker genetically linked to the B3 locus in Pleurotus eryngii. J. Microbiol. Biotechn. 2022, 22, 1177–1184. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, L.; Muez, V.; Alfonso, M.; García Barrenechea, A.; Alfonso, L.; Pisabarro, A.G. Use of molecular markers to differentiate between commercial strains of the button mushroom Agaricus bisporus. FEMS Microbiol. Lett. 2001, 98, 45–48. [Google Scholar] [CrossRef]
- Kong, W.; You, C.; Yoo, Y.; Kim, G.; Kim, K. Molecular marker related to fruitbody color of Flammulina velutipes. Mycobiology 2004, 32, 6–10. [Google Scholar] [CrossRef]
- Sheng, C.G.; Hung, C.Y.; Chen, Q.; Bau, T.; Zhang, J.X. Fruit-Body Color Inheritance of Pleurotus cornucopiae. Sci. Agric. Sin. 2012, 45, 3124–3129. [Google Scholar] [CrossRef]
- Giraudat, J.; Hauge, B.M.; Valon, C.; Smalle, J.; Parcy, F.; Goodman, H.M. Isolation of the Arabidopsis ABI3 gene by positional cloning. Plant Cell 1992, 4, 1251–1261. [Google Scholar] [CrossRef]
- Matsubara, K.; Yamanouchi, U.; Wang, Z.X.; Minobe, Y.; Izawa, T.; Yano, M. Ehd2, a rice ortholog of the maize INDETERMINATE1 gene, promotes flowering by up-regulating Ehd1. Plant Physiol. 2008, 148, 1425–1435. [Google Scholar] [CrossRef]
- Salvi, S.; Tuberosa, R.; Chiapparino, E.; Maccaferri, M.; Veillet, S.; van Beuningen, L.; Isaac, P.; Edwards, K.; Phillips, R.L. Toward positional cloning of Vgt1, a QTL controlling the transition from the vegetative to the reproductive phase in maize. Plant Mol. Biol. 2002, 48, 601–613. [Google Scholar] [CrossRef]
- Watanabe, S.; Hideshima, R.; Xia, Z.J.; Tsubokura, Y.; Sato, S.; Nakamoto, Y.; Yamanaka, N.; Takahashi, R.; Ishimoto, M.; Anai, T.; et al. Map-based cloning of the gene associated with the soybean maturity locus E3. Genetics 2009, 182, 1251–1262. [Google Scholar] [CrossRef]
- Wang, P.; Yao, F.J.; Lu, L.X.; Fang, M.; Zhang, Y.M.; Khan, A.A.; Kong, X.H.; Yu, J.; Jiang, W.Z.; Kitamoto, Y.; et al. Map-based cloning of genes encoding key enzymes for pigment synthesis in Auricularia cornea. Fungal Biol. 2019, 123, 843–853. [Google Scholar] [CrossRef]
- Murray, M.G.; Thompson, W.F. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 1980, 8, 4321–4325. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yao, F.J.; Sun, W.J.; Fang, M.; Wu, C.S. Screening of reference genes for qRT-PCR amplification in Auricularia heimuer. Mycosystema 2020, 39, 1510–1519. [Google Scholar] [CrossRef]
- Van Ooijen, J.W. JoinMap 4, Software for the Calculation of Genetic Linkage Maps in Experimental Populations; Plant Research International BV and Kayazma BV: Wageningen, The Netherlands, 2006. [Google Scholar]
- Lu, L.X.; Yao, F.J.; Wang, P.; Fang, M.; Zhang, Y.M.; Zhang, W.T.; Kong, X.H.; Lu, J. Construction of a genetic linkage map and QTL mapping of agronomic traits in Auricularia auricula-judae. J. Microbiol. Biol. Educ. 2017, 55, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.Z.; Yao, F.J.; Lu, L.X.; Fang, M.; Wang, P.; Zhang, Y.M.; Meng, J.J.; Lu, J.; Ma, X.X.; He, Q.; et al. Affiliations expand. Genetic linkage map construction and quantitative trait loci mapping of agronomic traits in Gloeostereum incarnatum. J. Microbiol. 2021, 59, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.J.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.H.; Xia, R. TBtools: An integrative toolkit developed for interactive analyses of big biological data. Mol. Plant. 2020, 13, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Kofler, R.; Schlötterer, C.; Lelley, T. SciRoKo: A new tool for whole genome microsatellite search and investigation. Bioinformatics 2007, 23, 1683–1685. [Google Scholar] [CrossRef]
- Shi, X.K.; Cai, Z.X.; Guo, Z.J.; Lu, Y.P.; Chen, M.Y.; Liao, J.H. A Preliminary Report on Resequencing 18 Representative Strains of Agaricus bisporus. Fujian J. Agric. Sci. 2009, 34, 1167–1172. [Google Scholar] [CrossRef]
- Wang, L.N.; Zhao, M.R.; Wu, X.L.; Huang, C.Y.; Qu, J.B. Comparative genomic analyses of two Pleurotus ostreatus strains. Mycosystema 2019, 38, 2133–2143. [Google Scholar] [CrossRef]
- Alonge, M.; Soyk, S.; Ramakrishnan, S.; Wang, X.; Goodwin, S.; Sedlazeck, F.J.; Lippman, Z.B.; Schatz, M.C. RaGOO: Fast and accurate reference-guided scaffolding of draft genomes. Genome Biol. 2019, 20, 224. [Google Scholar] [CrossRef]
- Cai, Z.X.; Chen, M.Y.; Liao, J.H.; Li, H.R.; Guo, Z.J.; Cai, D.F.; Wang, Z.S. Primary Screening of Molecular Markers Related to Fruit Body Color in Agaricus bisporus. Acta Edulis Fungi 2014, 21, 1–5. [Google Scholar] [CrossRef]
- Gong, W.B.; Liu, W.; Lu, Y.Y.; Bian, Y.B.; Zhou, Y.; Kwan, H.S.; Cheung, M.K.; Xiao, Y. Constructing a new integrated genetic linkage map and mapping quantitative trait loci for vegetative mycelium growth rate in Lentinula edodes. Fungal Biol. 2014, 118, 295–308. [Google Scholar] [CrossRef]
- Xiao, G.L.; Xu, Z.Q.; Cao, H.J.; Li, J.C.; Xia, J.H.; Song, B.T. Development of SSR Markers Based on Potato Genome Sequence and Construction of a Higher-density Linkage Map. Acta Hortic. Sin. 2018, 45, 1551–1562. [Google Scholar] [CrossRef]
- Wang, J.; Wu, B.W.; Lu, K.; Wei, Q.; Qian, J.J.; Chen, Y.P.; Fang, Z.M. The amino acid permease 5 (OsAAP5) regulates tiller number and grain yield in rice. Plant Physiol. 2019, 180, 1031–1045. [Google Scholar] [CrossRef] [PubMed]
| Number | Name | Fruiting Body Pattern | Number | Name | Fruiting Body Pattern |
|---|---|---|---|---|---|
| A1 | Heiwei 981 | CL | A13 | Heimeur 1 | CH |
| A2 | Hei 793 | CH | A14 | Qihei No. 1 | CL |
| A5 | Zhejiang Ear 1 | CH | A15 | Heier 5 | CH |
| A6 | Zheer 1 | CL | A17 | Heier 6 | CL |
| A8 | H10 | CH | A18 | Qihei No. 2 | CH |
| A9 | Yante 3 | CL | A19 | Fengshou 2 | CL |
| A10 | Yante 5 | CL | A22 | JiAU 2 | CL |
| A12 | Xinke | CH |
| Fruiting Body Pattern | Cluster Type (CL) | Chrysanthemum Type (CH) |
|---|---|---|
| parental strain | A14 | A18 |
| cross population | C3, C6, C7, C8, C11, C21, C22, C23, C25, C27, C28, C29, C30, C33, C34, C35, C37, C38, C39, C40, C42, C45, C55, C58, C60, C63, C68, C72, C77, C79, C80, C85, C87, C89, C91, C95, C96, C106, C107, C109, C112, C119, C127, C129, C133, C139, C149, C153, C157, C160, C161, C163, C164, C166, C167, C169, C171, C174, C175, C183, C184, C185, 186, C188, C193, C199, C201 | C1, C2, C12, C15, C17, C18, C19, C20, C32, C50, C51, C57, C61, C64, C65, C66, C68, C70, C71, C73, C74, C75, C76, C78, C81, C82, C86, C88, C90, C92, C93, C94, C97, C98, C99, C101, C102, C103, C105, C110, C114, C115, C118, C120, C121, C123, C126, C128, C132, C134, C135, C136, C146, C150, C152, C154, C165, C168, C170, C172, C178, C179, C180, C181, C187, C190, C191, C192, C197, C198, C202 |
| Strain | BioProject (PRJNA) | Contingent Count (N50 kb) | Total Length (Mb) | Scaffold Count (N50 Mb) | GC Count (%) |
|---|---|---|---|---|---|
| B14-8 | 382471 | 1071 (121.22) | 43.57 | 534 (0.27) | 57 |
| Dai13782 | 382748 | 114 (1006.44) | 49.76 | 103 (1.35) | 57 |
| Scheme | Characteristics | Secondary Structure | |||||
|---|---|---|---|---|---|---|---|
| Isoelectric Point pI | Molecular Weight Mw(kD) | Instability Index | Hydrophilic | α-Helix (%) | β-Angle of Rotation (%) | Irregular Curl | |
| A14 | 5.90 | 28.35 | 39.43 | 0.904 | 48.03 | 26.38 | 22.83 |
| A18 | 5.37 | 28.34 | 40.05 | 0.911 | 49.61 | 21.65 | 24.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, J.; Lu, L.; Yao, F.; Fang, M.; Ma, X.; Meng, J.; Shao, K. Dicarboxylic Amino Acid Permease 7219 Regulates Fruiting Body Type of Auricularia heimuer. J. Fungi 2023, 9, 876. https://doi.org/10.3390/jof9090876
Lu J, Lu L, Yao F, Fang M, Ma X, Meng J, Shao K. Dicarboxylic Amino Acid Permease 7219 Regulates Fruiting Body Type of Auricularia heimuer. Journal of Fungi. 2023; 9(9):876. https://doi.org/10.3390/jof9090876
Chicago/Turabian StyleLu, Jia, Lixin Lu, Fangjie Yao, Ming Fang, Xiaoxu Ma, Jingjing Meng, and Kaisheng Shao. 2023. "Dicarboxylic Amino Acid Permease 7219 Regulates Fruiting Body Type of Auricularia heimuer" Journal of Fungi 9, no. 9: 876. https://doi.org/10.3390/jof9090876




