Efficient Azo Dye Biodecolorization System Using Lignin-Co-Cultured White-Rot Fungus
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganism and Chemicals
2.2. Decolorization Experiments with Lignin
2.3. Dye Adsorption Assay
2.4. Effect of Cell-Free Extracellular Liquid on Dye Decolorization
2.5. Metabolites Analysis Using Gas Chromatography/Mass Spectrometry
2.6. Effect of the Lignin-Derived Compound on DR5B Decolorization and Laccase Induction
2.7. Analytical Methods
3. Results and Discussion
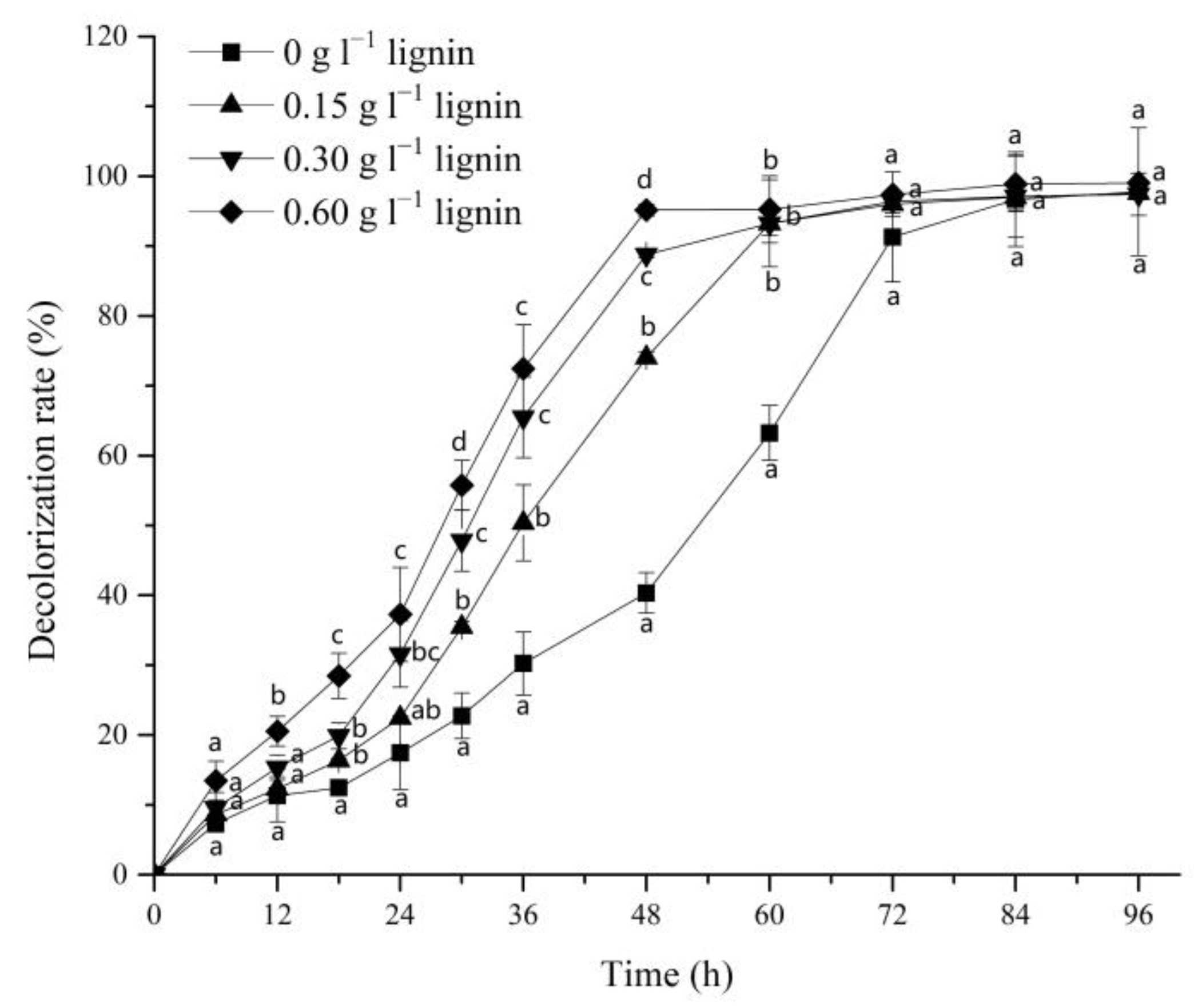
3.1. Effect of Alkali Lignin on Dye Decolorization
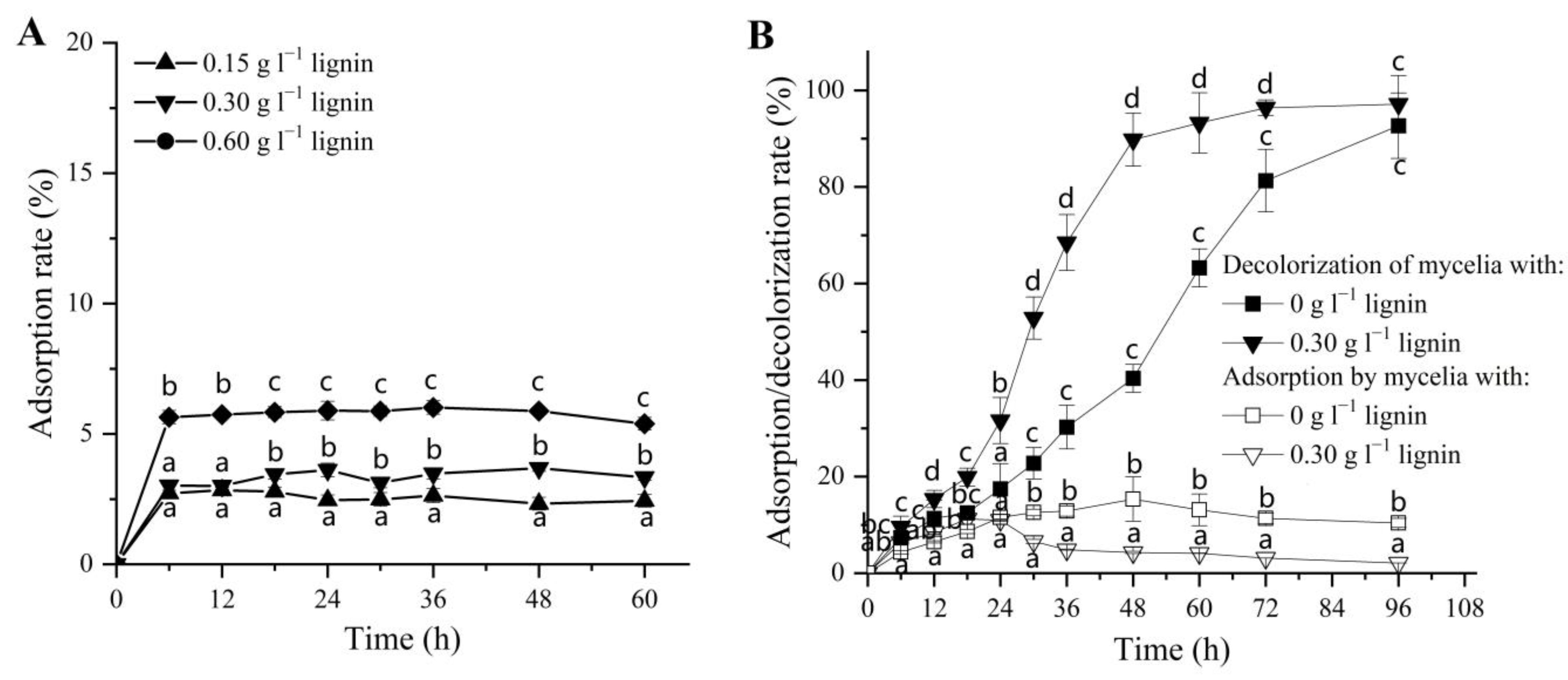
3.2. Physical Adsorption of DR5B by Alkali Lignin and Fungal Mycelia
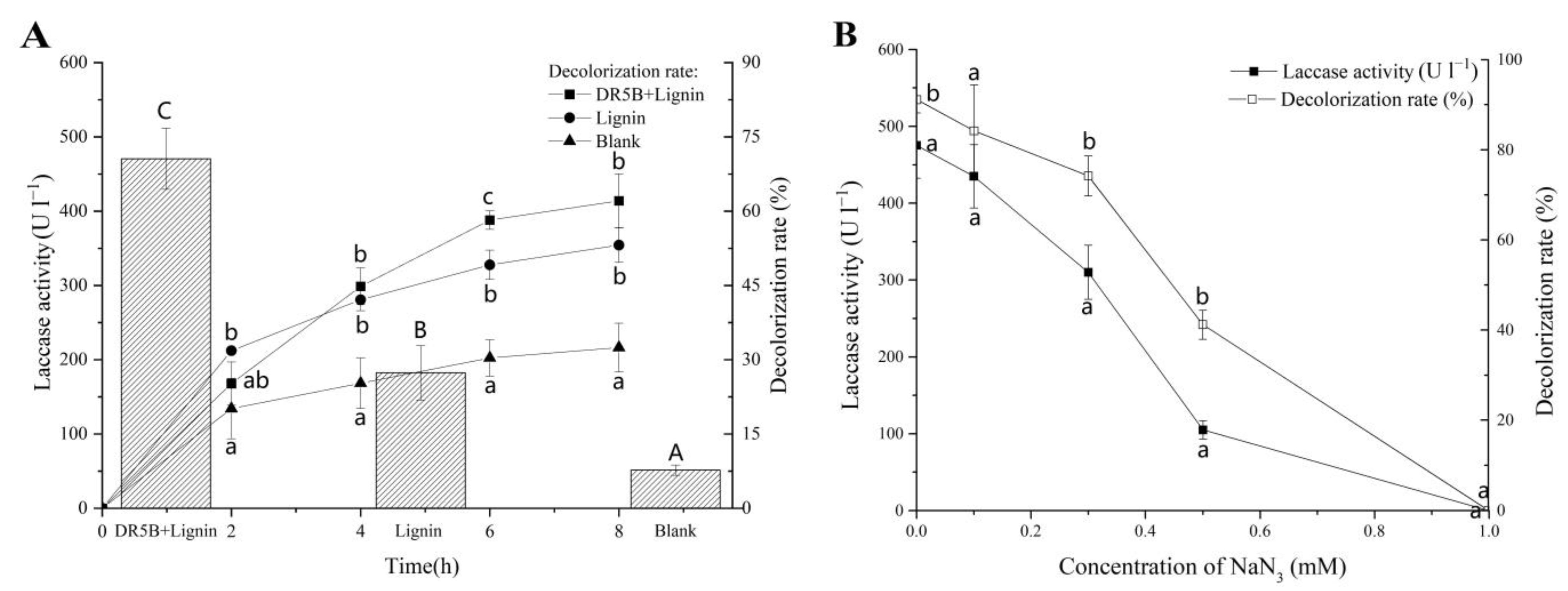
3.3. Effect of Alkali Lignin on Laccase Activities during the Degradation of DR5B by G. lucidum EN2
3.4. Effect of Alkali Lignin Degradation of Extracellular Liquid on DR5B Removal
3.5. Characterization of Metabolites Resulting from Degradation of Both DR5B and Alkali Lignin by G. lucidum EN2
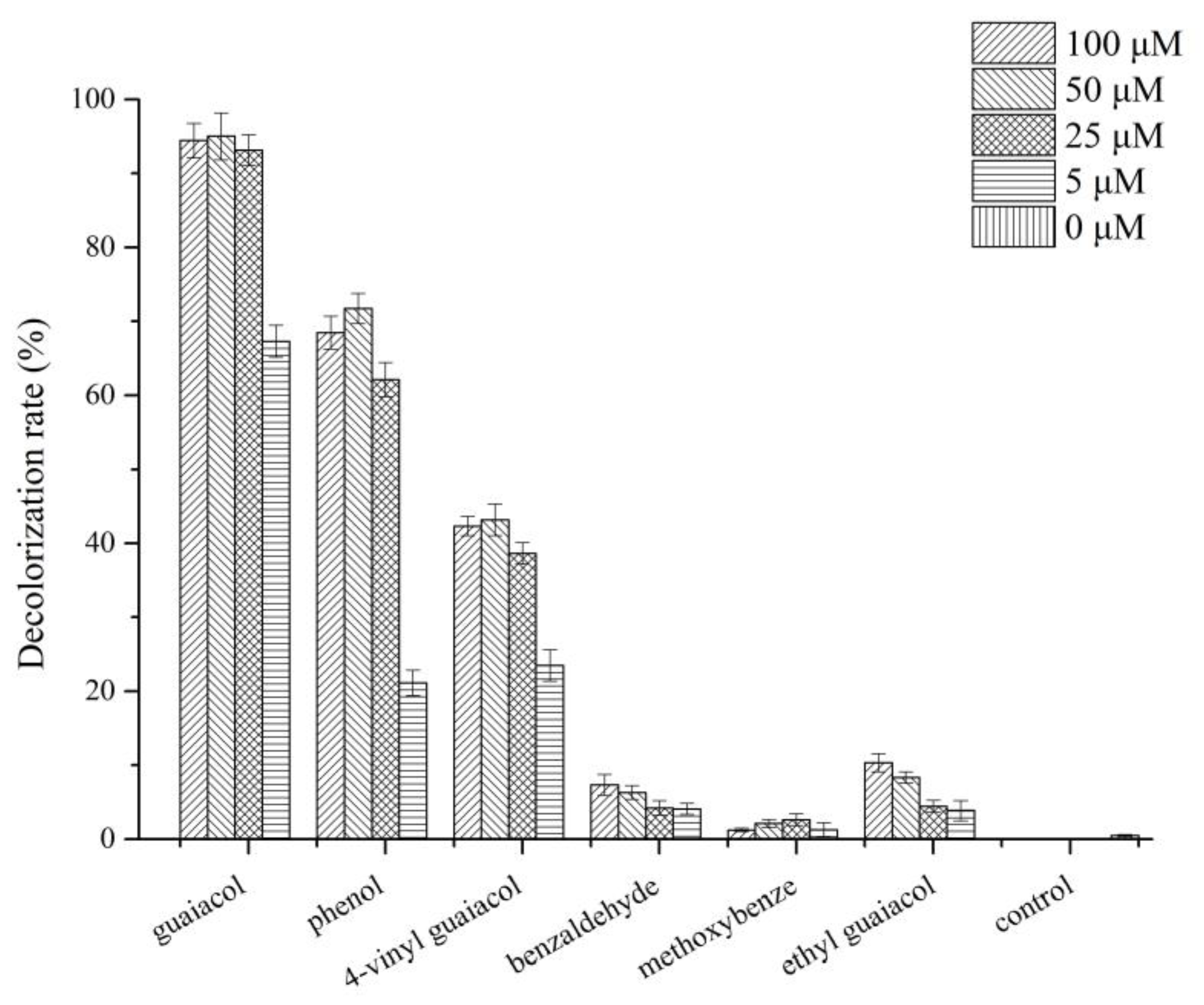
3.6. Effect of Different Lignin-Derived Aromatic Compounds on Dye Decolorization and Laccase Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Haarstrick, A.; Bahadir, M. Water and its Global Meaning. In Water and Wastewater Management; Bahadir, M., Haarstrick, A., Eds.; Springer: Cham, Switzerland, 2022; pp. 3–14. ISBN 978-3-030-95288-4. [Google Scholar]
- Boretti, A.; Rosa, L. Reassessing the Projections of the World Water Development Report. npj Clean Water 2019, 2, 15–19. [Google Scholar] [CrossRef]
- Al-Tohamy, R.; Ali, S.S.; Li, F.; Okasha, K.M.; Mahmoud, Y.A.-G.; Elsamahy, T.; Jiao, H.; Fu, Y.; Sun, J. A Critical Review on the Treatment of Dye-Containing Wastewater: Ecotoxicological and Health Concerns of Textile Dyes and Possible Remediation Approaches for Environmental Safety. Ecotoxicol. Environ. Saf. 2022, 231, 113160–113176. [Google Scholar] [CrossRef]
- Dhruv Patel, D.; Bhatt, S. Environmental Pollution, Toxicity Profile, and Physico-Chemical and Biotechnological Approaches for Treatment of Textile Wastewater. Biotechnol. Genet. Eng. Rev. 2022, 38, 33–86. [Google Scholar] [CrossRef] [PubMed]
- Kuppusamy, S.; Sethurajan, M.; Kadarkarai, M.; Aruliah, R. Biodecolourization of Textile Dyes by Novel, Indigenous Pseudomonas Stutzeri MN1 and Acinetobacter Baumannii MN3. J. Environ. Chem. Eng. 2017, 5, 716–724. [Google Scholar] [CrossRef]
- Yusuf, M. Synthetic Dyes: A Threat to the Environment and Water Ecosystem. In Textiles and Clothing: Environmental Concerns and Solutions, 1st ed.; Shabbir, M., Ed.; Scrivener Publishing LLC: New York, NY, USA, 2019; pp. 11–26. ISBN 9781119526599. [Google Scholar]
- Pinheiro, L.R.; Gradíssimo, D.G.; Xavier, L.P.; Santos, A.V. Degradation of Azo Dyes: Bacterial Potential for Bioremediation. Sustainability 2022, 14, 1510. [Google Scholar] [CrossRef]
- Blánquez, A.; Rodríguez, J.; Brissos, V.; Mendes, S.; Martins, L.O.; Ball, A.S.; Arias, M.E.; Hernández, M. Decolorization and Detoxification of Textile Dyes Using a Versatile Streptomyces Laccase-Natural Mediator System. Saudi J. Biol. Sci. 2019, 26, 913–920. [Google Scholar] [CrossRef]
- Solís, M.; Solís, A.; Pérez, H.I.; Manjarrez, N.; Flores, M. Microbial Decolouration of Azo Dyes: A Review. Process Biochem. 2012, 47, 1723–1748. [Google Scholar] [CrossRef]
- Deng, D.; Guo, J.; Zeng, G.; Sun, G. Decolorization of Anthraquinone, Triphenylmethane and Azo Dyes by a New Isolated Bacillus Cereus Strain DC11. Int. Biodeterior. Biodegrad. 2008, 62, 263–269. [Google Scholar] [CrossRef]
- Caritá, R.; Marin-Morales, M.A. Induction of Chromosome Aberrations in the Allium Cepa Test System Caused by the Exposure of Seeds to Industrial Effluents Contaminated with Azo Dyes. Chemosphere 2008, 72, 722–725. [Google Scholar] [CrossRef]
- Kishor, R.; Purchase, D.; Saratale, G.D.; Saratale, R.G.; Ferreira, L.F.R.; Bilal, M.; Chandra, R.; Bharagava, R.N. Ecotoxicological and Health Concerns of Persistent Coloring Pollutants of Textile Industry Wastewater and Treatment Approaches for Environmental Safety. J. Environ. Chem. Eng. 2021, 9, 105012–105029. [Google Scholar] [CrossRef]
- Zhuo, R.; Fan, F. A Comprehensive Insight into the Application of White Rot Fungi and Their Lignocellulolytic Enzymes in the Removal of Organic Pollutants. Sci. Total Environ. 2021, 778, 146132–146149. [Google Scholar] [CrossRef]
- Kunjadia, P.D.; Sanghvi, G.V.; Kunjadia, A.P.; Mukhopadhyay, P.N.; Dave, G.S. Role of Ligninolytic Enzymes of White Rot Fungi (Pleurotus Spp.) Grown with Azo Dyes. Springerplus 2016, 5, 1487. [Google Scholar] [CrossRef]
- Han, Y.; Shi, L.; Meng, J.; Yu, H.; Zhang, X. Azo Dye Biodecolorization Enhanced by Echinodontium Taxodii Cultured with Lignin. PLoS ONE 2014, 9, e109786. [Google Scholar] [CrossRef]
- Rekik, H.; Zaraî Jaouadi, N.; Bouacem, K.; Zenati, B.; Kourdali, S.; Badis, A.; Annane, R.; Bouanane-Darenfed, A.; Bejar, S.; Jaouadi, B. Physical and Enzymatic Properties of a New Manganese Peroxidase from the White-Rot Fungus Trametes Pubescens Strain I8 for Lignin Biodegradation and Textile-Dyes Biodecolorization. Int. J. Biol. Macromol. 2019, 125, 514–525. [Google Scholar] [CrossRef]
- Koyani, R.D.; Sanghvi, G.V.; Sharma, R.K.; Rajput, K.S. Contribution of Lignin Degrading Enzymes in Decolourisation and Degradation of Reactive Textile Dyes. Int. Biodeterior. Biodegrad. 2013, 77, 1–9. [Google Scholar] [CrossRef]
- Samir Ali, S.; Al-Tohamy, R.; Khalil, M.A.; Ho, S.-H.; Fu, Y.; Sun, J. Exploring the Potential of a Newly Constructed Manganese Peroxidase-Producing Yeast Consortium for Tolerating Lignin Degradation Inhibitors While Simultaneously Decolorizing and Detoxifying Textile Azo Dye Wastewater. Bioresour. Technol. 2022, 351, 126861–126874. [Google Scholar] [CrossRef] [PubMed]
- Al-Tohamy, R.; Sun, J.; Fareed, M.F.; Kenawy, E.-R.; Ali, S.S. Ecofriendly Biodegradation of Reactive Black 5 by Newly Isolated Sterigmatomyces Halophilus SSA1575, Valued for Textile Azo Dye Wastewater Processing and Detoxification. Sci. Rep. 2020, 10, 12370–12386. [Google Scholar] [CrossRef]
- Mir-Tutusaus, J.A.; Baccar, R.; Caminal, G.; Sarrà, M. Can White-Rot Fungi Be a Real Wastewater Treatment Alternative for Organic Micropollutants Removal? A Review. Water Res. 2018, 138, 137–151. [Google Scholar] [CrossRef] [PubMed]
- Shi, K.; Liu, Y.; Chen, P.; Li, Y. Contribution of Lignin Peroxidase, Manganese Peroxidase, and Laccase in Lignite Degradation by Mixed White-Rot Fungi. Waste Biomass Valorization 2021, 12, 3753–3763. [Google Scholar] [CrossRef]
- Othmani, A.; John, J.; Rajendran, H.; Mansouri, A.; Sillanpää, M.; Velayudhaperumal Chellam, P. Biochar and Activated Carbon Derivatives of Lignocellulosic Fibers towards Adsorptive Removal of Pollutants from Aqueous Systems: Critical Study and Future Insight. Sep. Purif. Technol. 2021, 274, 119062–119074. [Google Scholar] [CrossRef]
- Robinson, T.; Nigam, P.S. Remediation of Textile Dye Waste Water Using a White-Rot Fungus Bjerkandera Adusta Through Solid-State Fermentation (SSF). Appl. Biochem. Biotechnol. 2008, 151, 618. [Google Scholar] [CrossRef]
- Yan, K.; Wang, H.; Zhang, X. Biodegradation of Crystal Violet by Low Molecular Mass Fraction Secreted by Fungus. J. Biosci. Bioeng. 2009, 108, 421–424. [Google Scholar] [CrossRef]
- Han, M.-L.; Yang, J.; Liu, Z.-Y.; Wang, C.-R.; Chen, S.-Y.; Han, N.; Hao, W.-Y.; An, Q.; Dai, Y.-C. Evaluation of Laccase Activities by Three Newly Isolated Fungal Species in Submerged Fermentation with Single or Mixed Lignocellulosic Wastes. Front. Microbiol. 2021, 12, 682679–682689. [Google Scholar] [CrossRef]
- Raj, T.; Chandrasekhar, K.; Naresh Kumar, A.; Rajesh Banu, J.; Yoon, J.-J.; Kant Bhatia, S.; Yang, Y.-H.; Varjani, S.; Kim, S.-H. Recent Advances in Commercial Biorefineries for Lignocellulosic Ethanol Production: Current Status, Challenges and Future Perspectives. Bioresour. Technol. 2022, 344, 126292–126303. [Google Scholar] [CrossRef]
- Intasit, R.; Khunrae, P.; Meeinkuirt, W.; Soontorngun, N. Fungal Pretreatments of Napier Grass and Sugarcane Leaves for High Recovery of Lignocellulosic Enzymes and Methane Production. Ind. Crops Prod. 2022, 180, 114706–114715. [Google Scholar] [CrossRef]
- dos Santos Bazanella, G.C.; de Souza, D.F.; Castoldi, R.; Oliveira, R.F.; Bracht, A.; Peralta, R.M. Production of Laccase and Manganese Peroxidase by Pleurotus Pulmonarius in Solid-State Cultures and Application in Dye Decolorization. Folia Microbiol. (Praha.) 2013, 58, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Dashtban, M.; Schraft, H.; Qin, W. Fungal Bioconversion of Lignocellulosic Residues; Opportunities & Perspectives. Int. J. Biol. Sci. 2009, 5, 578–595. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Raquet, G.; Durand, E.; Braun, F.; Cravo-Laureau, C.; Godon, J.-J. Impact of Microbial Diversity Depletion on Xenobiotic Degradation by Sewage-Activated Sludge. Environ. Microbiol. Rep. 2013, 5, 588–594. [Google Scholar] [CrossRef]
- Mohapatra, B.; Phale, P.S. Microbial Degradation of Naphthalene and Substituted Naphthalenes: Metabolic Diversity and Genomic Insight for Bioremediation. Front. Bioeng. Biotechnol. 2021, 9, 602445–602473. [Google Scholar] [CrossRef]
- Elisashvili, V.; Kachlishvili, E.; Khardziani, T.; Agathos, S.N. Effect of Aromatic Compounds on the Production of Laccase and Manganese Peroxidase by White-Rot Basidiomycetes. J. Ind. Microbiol. Biotechnol. 2010, 37, 1091–1096. [Google Scholar] [CrossRef]
- Susana, C.; David, I.; Jesús, M.M.; Martínez, Á.T. Lignin-Derived Compounds as Efficient Laccase Mediators for Decolorization of Different Types of Recalcitrant Dyes. Appl. Environ. Microbiol. 2005, 71, 1775–1784. [Google Scholar] [CrossRef]
- Ralph, J.; Lapierre, C.; Boerjan, W. Lignin Structure and Its Engineering. Curr. Opin. Biotechnol. 2019, 56, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Cline, S.P.; Smith, P.M. Opportunities for Lignin Valorization: An Exploratory Process. Energy Sustain. Soc. 2017, 7, 26–37. [Google Scholar] [CrossRef]
- Zheng, L.; Lu, G.; Pei, W.; Yan, W.; Li, Y.; Zhang, L.; Huang, C.; Jiang, Q. Understanding the Relationship between the Structural Properties of Lignin and Their Biological Activities. Int. J. Biol. Macromol. 2021, 190, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Melro, E.; Filipe, A.; Sousa, D.; Medronho, B.; Romano, A. Revisiting Lignin: A Tour through Its Structural Features, Characterization Methods and Applications. New J. Chem. 2021, 45, 6986–7013. [Google Scholar] [CrossRef]
- Yuan, Y.; Jiang, B.; Chen, H.; Wu, W.; Wu, S.; Jin, Y.; Xiao, H. Recent Advances in Understanding the Effects of Lignin Structural Characteristics on Enzymatic Hydrolysis. Biotechnol. Biofuels 2021, 14, 205–224. [Google Scholar] [CrossRef]
- Huang, J.; Fu, S.; Gan, L. Structure and Characteristics of Lignin. In Lignin Chemistry and Applications; Huang, J., Fu, S., Gan, L., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Chapter 2; pp. 25–50. ISBN 978-0-12-813941-7. [Google Scholar]
- del Cerro, C.; Erickson, E.; Dong, T.; Wong, A.R.; Eder, E.K.; Purvine, S.O.; Mitchell, H.D.; Weitz, K.K.; Markillie, L.M.; Burnet, M.C.; et al. Intracellular Pathways for Lignin Catabolism in White-Rot Fungi. Proc. Natl. Acad. Sci. USA 2021, 118, e2017381118. [Google Scholar] [CrossRef]
- Ilić Đurđić, K.; Ostafe, R.; Prodanović, O.; Đurđević Đelmaš, A.; Popović, N.; Fischer, R.; Schillberg, S.; Prodanović, R. Improved Degradation of Azo Dyes by Lignin Peroxidase Following Mutagenesis at Two Sites near the Catalytic Pocket and the Application of Peroxidase-Coated Yeast Cell Walls. Front. Environ. Sci. Eng. 2020, 15, 21419–21428. [Google Scholar] [CrossRef]
- Meng, X.; Scheidemantle, B.; Li, M.; Wang, Y.; Zhao, X.; Toro-González, M.; Singh, P.; Pu, Y.; Wyman, C.E.; Ozcan, S.; et al. Synthesis, Characterization, and Utilization of a Lignin-Based Adsorbent for Effective Removal of Azo Dye from Aqueous Solution. ACS Omega 2020, 5, 2865–2877. [Google Scholar] [CrossRef]
- Kirk, T.K.; Schultz, E.; Connors, W.J.; Lorenz, L.F.; Zeikus, J.G. Influence of Culture Parameters on Lignin Metabolism by Phanerochaete Chrysosporium. Arch. Microbiol. 1978, 117, 277–285. [Google Scholar] [CrossRef]
- Yan, K.; Wang, H.; Zhang, X.; Yu, H. Bioprocess of Triphenylmethane Dyes Decolorization by Pleurotus Ostreatus BP under Solid-State Cultivation. J. Microbiol. Biotechnol. 2009, 19, 1421–1430. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Yu, H.; Dong, T.; Kong, W.; Ke, M.; Ma, F.; Zhang, X. Biochemical and Molecular Characterization of a Novel Laccase from Selective Lignin-Degrading White-Rot Fungus Echinodontium Taxodii 2538. Process Biochem. 2014, 49, 1097–1106. [Google Scholar] [CrossRef]
- Hakala, T.K.; Lundell, T.; Galkin, S.; Maijala, P.; Kalkkinen, N.; Hatakka, A. Manganese Peroxidases, Laccases and Oxalic Acid from the Selective White-Rot Fungus Physisporinus Rivulosus Grown on Spruce Wood Chips. Enzyme Microb. Technol. 2005, 36, 461–468. [Google Scholar] [CrossRef]
- Qin, X.; Su, X.; Luo, H.; Ma, R.; Yao, B.; Ma, F. Deciphering Lignocellulose Deconstruction by the White Rot Fungus Irpex Lacteus Based on Genomic and Transcriptomic Analyses. Biotechnol. Biofuels 2018, 11, 58–71. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Song, C.; Li, Y.; Wang, N.; Xu, L.; Han, X.; Wei, D. Efficient Degradation of Azo Dyes by a Newly Isolated Fungus Trichoderma Tomentosum under Non-Sterile Conditions. Ecotoxicol. Environ. Saf. 2018, 150, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Jonstrup, M.; Kumar, N.; Guieysse, B.; Murto, M.; Mattiasson, B. Decolorization of Textile Dyes by Bjerkandera Sp. BOL 13 Using Waste Biomass as Carbon Source. J. Chem. Technol. Biotechnol. 2013, 88, 388–394. [Google Scholar] [CrossRef]
- Sen, S.K.; Raut, S.; Bandyopadhyay, P.; Raut, S. Fungal Decolouration and Degradation of Azo Dyes: A Review. Fungal Biol. Rev. 2016, 30, 112–133. [Google Scholar] [CrossRef]
- Rybczyńska-Tkaczyk, K.; Korniłłowicz-Kowalska, T. Biosorption Optimization and Equilibrium Isotherm of Industrial Dye Compounds in Novel Strains of Microscopic Fungi. Int. J. Environ. Sci. Technol. 2016, 13, 2837–2846. [Google Scholar] [CrossRef]
- Kaushik, P.; Malik, A. Fungal Dye Decolourization: Recent Advances and Future Potential. Environ. Int. 2009, 35, 127–141. [Google Scholar] [CrossRef]
- Abdel-Hamid, A.M.; Solbiati, J.O.; Cann, I.K.O. Insights into Lignin Degradation and Its Potential Industrial Applications. Adv. Appl. Microbiol. 2013, 82, 1–28. [Google Scholar] [CrossRef]
- Rodrigues, E.M.; Karp, S.G.; Malucelli, L.C.; Helm, C.V.; Alvarez, T.M. Evaluation of Laccase Production by Ganoderma Lucidum in Submerged and Solid-State Fermentation Using Different Inducers. J. Basic Microbiol. 2019, 59, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Couto, S.R.; Sanromán, M.A.; Guebitz, G.M. Influence of Redox Mediators and Metal Ions on Synthetic Acid Dye Decolourization by Crude Laccase from Trametes Hirsuta. Chemosphere 2005, 58, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, R.; Yuan, P.; Yang, Y.; Zhang, S.; Ma, F.; Zhang, X. Induction of Laccase by Metal Ions and Aromatic Compounds in Pleurotus Ostreatus HAUCC 162 and Decolorization of Different Synthetic Dyes by the Extracellular Laccase. Biochem. Eng. J. 2017, 117, 62–72. [Google Scholar] [CrossRef]
- Yang, S.; Hai, F.I.; Nghiem, L.D.; Price, W.E.; Roddick, F.; Moreira, M.T.; Magram, S.F. Understanding the Factors Controlling the Removal of Trace Organic Contaminants by White-Rot Fungi and Their Lignin Modifying Enzymes: A Critical Review. Bioresour. Technol. 2013, 141, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Zilly, A.; da Silva Coelho-Moreira, J.; Bracht, A.; Marques de Souza, C.G.; Carvajal, A.E.; Koehnlein, E.A.; Peralta, R.M. Influence of NaCl and Na2SO4 on the Kinetics and Dye Decolorization Ability of Crude Laccase from Ganoderma Lucidum. Int. Biodeterior. Biodegrad. 2011, 65, 340–344. [Google Scholar] [CrossRef]
- Liu, S.-H.; Tsai, S.-L.; Guo, P.-Y.; Lin, C.-W. Inducing Laccase Activity in White Rot Fungi Using Copper Ions and Improving the Efficiency of Azo Dye Treatment with Electricity Generation Using Microbial Fuel Cells. Chemosphere 2020, 243, 125304–125310. [Google Scholar] [CrossRef]
- Liu, J.; Sun, S.; Han, Y.; Meng, J.; Chen, Y.; Yu, H.; Zhang, X.; Ma, F. Lignin Waste as Co-Substrate on Decolorization of Azo Dyes by Ganoderma Lucidum. J. Taiwan Inst. Chem. Eng. 2021, 122, 85–92. [Google Scholar] [CrossRef]
- Cancel, A.M.; Orth, A.B.; Tien, M. Lignin and Veratryl Alcohol Are Not Inducers of the Ligninolytic System of Phanerochaete Chrysosporium. Appl. Environ. Microbiol. 1993, 59, 2909–2913. [Google Scholar] [CrossRef]
- Faison, B.D.; Kirk, T.K. Factors Involved in the Regulation of a Ligninase Activity in Phanerochaete Chrysosporium. Appl. Environ. Microbiol. 1985, 49, 299–304. [Google Scholar] [CrossRef]
- Piscitelli, A.; Giardina, P.; Lettera, V.; Pezzella, C.; Sannia, G.; Faraco, V. Induction and Transcriptional Regulation of Laccases in Fungi. Curr. Genom. 2011, 12, 104–112. [Google Scholar] [CrossRef]
- Giatti Marques De Souza, C.; Kirst Tychanowicz, G.; Farani De Souza, D.; Peralta, R.M. Production of Laccase Isoforms by Pleurotus Pulmonarius in Response to Presence of Phenolic and Aromatic Compounds. J. Basic Microbiol. An Int. J. Biochem. Physiol. Genet. Morphol. Ecol. Microorg. 2004, 44, 129–136. [Google Scholar] [CrossRef]
- Unuofin, J.O.; Okoh, A.I.; Nwodo, U.U. Aptitude of Oxidative Enzymes for Treatment of Wastewater Pollutants: A Laccase Perspective. Molecules 2019, 24, 2064. [Google Scholar] [CrossRef]
- Mäkelä, M.R.; Tuomela, M.; Hatakka, A.; Hildén, K. Fungal laccases and their potential in bioremediation applications. In Microbiology Monographs: Laccases in Bioremediation and Waste Valorisation; Schlosser, D., Ed.; Springer International Publishing: Cham, Germany, 2020; pp. 1–25. ISBN 978-3-030-47906-0. [Google Scholar]
- Raj, A.; Reddy, M.M.K.; Chandra, R. Identification of Low Molecular Weight Aromatic Compounds by Gas Chromatography–Mass Spectrometry (GC–MS) from Kraft Lignin Degradation by Three Bacillus sp. Int. Biodeterior. Biodegrad. 2007, 59, 292–296. [Google Scholar] [CrossRef]
- Gnanamani, A.; Jayaprakashvel, M.; Arulmani, M.; Sadulla, S. Effect of Inducers and Culturing Processes on Laccase Synthesis in Phanerochaete Chrysosporium NCIM 1197 and the Constitutive Expression of Laccase Isozymes. Enzyme Microb. Technol. 2006, 38, 1017–1021. [Google Scholar] [CrossRef]
- Manavalan, A.; Manavalan, T.; Murugesan, K.; Kutzner, A.; Thangavelu, K.P.; Heese, K. Characterization of a Solvent, Surfactant and Temperature-Tolerant Laccase from Pleurotus Sp. MAK-II and Its Dye Decolorizing Property. Biotechnol. Lett. 2015, 37, 2403–2409. [Google Scholar] [CrossRef]
- Hu, M.R.; Chao, Y.P.; Zhang, G.Q.; Xue, Z.Q.; Qian, S. Laccase-Mediator System in the Decolorization of Different Types of Recalcitrant Dyes. J. Ind. Microbiol. Biotechnol. 2009, 36, 45–51. [Google Scholar] [CrossRef]
- Azubuike, C.C.; Allemann, M.N.; Michener, J.K. Microbial Assimilation of Lignin-Derived Aromatic Compounds and Conversion to Value-Added Products. Curr. Opin. Microbiol. 2022, 65, 64–72. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Phan, D.-P.; Sarwar, A.; Tran, M.H.; Lee, O.K.; Lee, E.Y. Valorization of Industrial Lignin to Value-Added Chemicals by Chemical Depolymerization and Biological Conversion. Ind. Crops Prod. 2021, 161, 113219–113233. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, S.; Liu, P.; Ullah, M. Efficient Azo Dye Biodecolorization System Using Lignin-Co-Cultured White-Rot Fungus. J. Fungi 2023, 9, 91. https://doi.org/10.3390/jof9010091
Sun S, Liu P, Ullah M. Efficient Azo Dye Biodecolorization System Using Lignin-Co-Cultured White-Rot Fungus. Journal of Fungi. 2023; 9(1):91. https://doi.org/10.3390/jof9010091
Chicago/Turabian StyleSun, Su, Pengyang Liu, and Mati Ullah. 2023. "Efficient Azo Dye Biodecolorization System Using Lignin-Co-Cultured White-Rot Fungus" Journal of Fungi 9, no. 1: 91. https://doi.org/10.3390/jof9010091
APA StyleSun, S., Liu, P., & Ullah, M. (2023). Efficient Azo Dye Biodecolorization System Using Lignin-Co-Cultured White-Rot Fungus. Journal of Fungi, 9(1), 91. https://doi.org/10.3390/jof9010091





