Abstract
Pinus bungeana, an endangered and native coniferous tree species in China, has considerable timber and horticulture value. However, little is known about needle diseases in P. bungeana. A needle blight of P. bungeana has been observed in Hebei Province, China. P. bungeana inoculated with mycelial plugs of fungal isolates presented symptoms similar to those observed under field conditions. Ten virulent fungal isolates were identified as a small-spored Alternaria species based on morphological observations. Maximum likelihood and Bayesian phylogenetic analyses carried out with multilocus sequence typing of eight regions (SSU, LSU, ITS, gapdh, tef1, Alt a 1, endoPG, OPA10-2) assigned the pathogen to Alternaria alternata. This is the first report of A. alternata causing needle blight on P. bungeana in China.
1. Introduction
Bunge’s pine (Pinus bungeana Zucc. ex Endl.), a distinctive and evergreen coniferous tree species within the genus Pinus of the family Pinaceae, is mainly distributed across warm temperate areas and the north-subtropical and middle subtropical climatic zones [1]. It is known as an endemic and endangered coniferous tree species in China with high ornamental value, and is widely used in landscaping and afforestation owing to its ability to endure drought and cold climates [2]. Furthermore, the wood of Bunge’s pine is commonly used for construction, furniture and stationery [3]. In addition, P. bungeana plays a key role in local forest ecosystems, with strong resistance to sulphur dioxide, ozone and soot pollution in nature [4]. Due to its ecological and economic value, this species has been the subject of many investigations, mainly on its phylogeny, morphology, genetic diversity and biological characteristics [5,6]. Few diseases of Bunge’s pine have been reported.
The genus Alternaria Nees was described in 1816 [7]. Since then, more than 1100 names have been published, and 275 Alternaria species have been recognised [8,9]. Alternaria is a ubiquitous fungal genus that includes saprophytic, endophytic and pathogenic species [10]. Some Alternaria species are famous as pathogens of plants and animals [11]. In addition, those pathogenic species harm more than 4000 host plants and are distributed worldwide, with a broad host range, including agronomic plants, ornamentals, vegetables, fruit trees and animals [10,12,13]. Leaf blight, leaf spot, black point, stem cancer, fruit rot and mouldy cores are well-known symptoms of infection by Alternaria species [14,15,16].
In the past, Alternaria spp. have been classified based exclusively upon their morphological characteristics, which include cultural morphology, shape and size of conidia, septation, beak formation, branching patterns of conidial chains, and sporulation patterns [17]. This approach is effective when distinguishing large-spored Alternaria spp. from small-spored catenulate species due to conidia that are distinct and easy to recognise. Nevertheless, the identification of small-spored species based on morphological characteristics is challenging due to the overlap of many morphological traits [18]. Therefore, using different molecular tools to support morphological inference for Alternaria taxonomy is essential. These tools include DNA fingerprinting techniques (RAPD, PCR-RFLP, AFLP and ISSR) and sequence analysis of rDNA and protein coding genes, such as nuclear internal transcribed-spacer regions (ITS), the mitochondrial ribosomal large subunit (mtLSU), the mitochondrial small subunit (mtSSU), translation elongation factor (TEF), beta-tubulin, endopolygalacturonase (endoPG) genes, glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene, RNA polymerase second largest subunit (RPB2), plasma membrane ATPase, Alternaria major allergen gene (Alt a 1), calmodulin (CAL) and the anonymous genomic regions OPA1-3, OPA2-1 and OPA10-2 [7,18,19,20,21,22,23]. Among these genes, the plasma membrane ATPase and calmodulin loci were proposed as the most suitable genetic markers for the molecular identification of small-spored Alternaria [7,11]. Furthermore, the histone 3 gene (HIS3) has been used to separate A. alternata from A. tenuissima [24,25].
Alternaria alternata (Fr.) Keissl, the type species for the genus Alternaria, is able to cause diseases in over 100 plants, including vegetables, fruits, herbs and ornamental trees [26,27,28,29]. Additionally, it can cause postharvest disease in various crops and respiratory diseases in humans [20]. It is a causal agent that gives rise to leaf spot, leaf blight and mouldy cores in host plants [15,30,31]. In addition, a serious infection risk was posed to horticultural crops all over the world because of the rapid market globalization of the seeds, long-distance airborne transmission of spores and the influences of changed climate [10].
A few diseases related to P. bungeana have been reported, including needle cast, trunk rot, needle rusts and twig blight [32,33]. However, there are no reports about needle blight in P. bungeana. The aim of this study was to identify the pathogens that cause needle blight in Bunge’s pine using morphological and molecular phylogenetic approaches and lay a theoretical foundation for the control of this pathogen.
2. Materials and Methods
2.1. Disease Investigation and Isolate Collection
In September 2020, leaf spot of Bunge’s pine was found in Hebei Province, China. Thirty symptomatic tissues, the margin between the lesioned and healthy pine leaves, were cut into 3 to 5 mm long pieces. These tissues were surface sterilised for 45 s in ethanol (75%), washed thrice in sterilised distilled water and blotted dry with sterile paper. Pieces were transferred to 2% potato dextrose agar (PDA) in Petri plates, supplemented with ampicillin at 100 μg/mL and incubated at 25 °C (±1 °C) in the dark for 4 days. The single-spore isolation technique was used to obtain purified fungal isolates [34]. Single-spore isolates were cultured on PDA and stored in the Forest Pathology Laboratory of Nanjing Forestry University, Nanjing, China and the representative strain are being deposited to China Center for Type Culture Collection, Wuhan, China (CCTCC).
2.2. Pathogenicity Tests
All isolates were cultured on PDA and used for virulence tests on detached P. bungeana needles under controlled conditions. Asymptomatic needles of P. bungeana were surface disinfected and air-dried. Then, one piercing wound was made on the mid-upper region of each needle with a sterile needle (insect pin, 0.71 mm in diameter). The inoculation was performed by placing mycelial blocks (5 mm in length) from actively growing colony margins onto each stab wound. Needles inoculated with noncolonised PDA blocks were treated as negative controls. Each control and treatment, involving three needles per replicate, was placed into a Petri dish (9 cm) with moist sterile filter paper and sealed with plastic wrap to maintain a high relative humidity. Then, they were incubated at 25 °C in a growth chamber with a 12 h photoperiod. The whole experiment was carried out three times.
Ten isolates that were confirmed to be pathogenic on the detached needles were selected to determine pathogenicity on potted Bunge’s pine. Bunge’s pine needles were disinfected with 75% ethanol and air dried. Then, wound inoculation was conducted on 2-year-old potted, healthy Bunge’s pine with a sterile needle. The blocks (3 mm in length) from colony margins with actively growing mycelia of 3-day-old isolates were placed on each wounded site. Blocks were removed 2 days post-inoculation. PDA discs with no mycelia were used as controls. Three potted plants were treated as one replicate, and three replicates were used. The inoculated plants were placed into a controlled-environment greenhouse. The size of the disease spot was recorded until representative symptoms appeared. The same procedure was carried out on 2-month-old seedlings of Korean pine (P. koraiensis Sieb. et Zucc.).
Re-isolations were performed from the margins of needles inoculated with ten isolates, and morphological and phylogenetic comparisons were conducted to meet Koch’s postulates.
2.3. Morphological Study
Isolates were cultured on PDA for 7 days at 25 °C (±1 °C) to observe the colony morphology [35]. Micromorphological features were observed from those cultured on synthetic nutrient-poor agar plates (SNA) [36]. The characteristics of sporulation formation, including the length of conidial chains, branching patterns of conidial chains and presence of secondary conidiophores, were captured with a Zeiss stereo microscope (SteRo Discovery v20) [35]. A ZEISS Axio Imager A2m microscope (Carl Zeiss, Göttingen, Germany) equipped with differential interference contrast (DIC) optics was used to capture conidial chains and conidia. Fifty mature conidia mounted in sterile water were measured at random under a light microscope at ×100 magnification.
2.4. DNA Extraction and Polymerase Chain Reaction (PCR)
The CTAB method described by Damm et al. [37] was used to extract genomic DNA from isolates that had been cultured on PDA at 25 °C for 5 days. The ITS, tef1, endoPG, OPA10-2, Alt a1, SSU, LSU and gapdh genes were amplified with the primer pairs V9G/ITS4 [38,39], EF1-728F/EF1-986R [40], PG3/PG2b [18], OPA10-2L/OPA10-2R [18], Alt-for/Alt-rev [41], NS1/NS4 [39], LSU1Fd/LR5 [42,43], and gpd1/gpd2 [44], respectively. Polymerase chain reaction (PCR) amplification was conducted in a total reaction volume of 25 μL containing 12.5 μL Taq DNA solution, 1 μL of each primer (10 pmol/μL), 2 μL (100 ng) of genomic DNA and 8.5 µL of double-distilled H2O with a thermal cycler under the conditions listed in Table 1. The PCR products were electrophoresed (160 V for 20 min) on 2% agarose gels and sequenced bidirectionally at the Shanghai Sangon Biological Technology Company (Shanghai, China) using Sanger DNA Sequencing from both directions. The sequenced DNA products were deposited at the National Centre for Biotechnology Information (NCBI) (Table 2).

Table 1.
List of the primers used for PCR and sequencing.

Table 2.
Descriptions and sequence accession numbers obtained from GenBank of Alternaria spp. used in the phylogenetic study.
2.5. DNA Sequencing and Phylogenetic Analysis
The reference sequences of 43 Alternaria spp. described by Woudenberg et al. [20] selected for the phylogenetic analyses are also listed in Table 2 together with their corresponding GenBank accession numbers. Sequences in Table 2 were retrieved from the GenBank database (https://www.ncbi.nlm.nih.gov/ (accessed on 10 February 2021)). A. alternantherae (CBS 124392) was used as the outgroup. The alignments of nucleotide sequences were obtained by using Clustal W in BioEdit software [45]. Treating gaps in the alignment as a fifth character, all of the characters had equal weight [46].
Phylogenetic trees of combined genes were constructed with two independent optimality search criteria, Bayesian inference (BI) phylogenetic analysis and maximum likelihood (ML) analysis. The ML analysis was performed using IQ-TREE [47], choosing the GTR + G + I model, and branch stability was estimated by 1000 bootstrap replicates. The BI analysis was performed in PhyloSuite version 1.2.2. using Mr. Bayes v. 3.2.6. [48] under a partition model (2 parallel runs, 1 × 107 generations), with FigTREE v1.4.4 used to view the phylogenetic trees.
3. Results
3.1. Symptoms in Nature
Symptoms appeared on Bunge’s pine needles and enlarged constantly. The colour of infected needles is off-white at the early stage and then turns to light brown gradually, with dark-brown spots appearing one by one (Figure 1B,C). At the later stage of the disease, a large number of needles are infected, and the growth of the tree is inhibited (Figure 1A). In total, 20 single-spore fungal isolates were collected.

Figure 1.
Symptoms of infection by A. alternata on P. bungeana in the field. (A), Withered tips of the whole tree. (B,C), Magnified image showing symptoms on needles.
3.2. Pathogenicity Tests
Ten isolates were pathogenic, and healthy needles exhibited symptoms similar to those in nature, while mock-inoculated control needles showed no symptoms (Figure 2). Light-brown lesions were first observed at two days after inoculation and then expanded gradually, and dark-brown segments were noticed 14 days after mycelial plug inoculation (Figure 2B). Ten lesions of each strain were counted and there was no significant difference in virulence among the three strains. Symptoms in nature appeared on Korean pine seedlings (Figure 2C). The fungus was re-isolated from inoculated needles, and its colony morphology and molecular sequence were consistent with those of the original isolates.

Figure 2.
Pathogenicity of A. alternata on Bunge’s pine and Korean pine achieved by mycelial discs. (A) Pathogenicity on 2-year-old seedlings of Bunge’s pine. (B) Pathogenicity on detached needles of Bunge’s pine. (C) Pathogenicity on 2-month-old seedlings of Korean pine. Scale bars: (B) = 5 mm.
3.3. Morphology of Fungal Isolates
The virulent isolates shared similar colony morphologies. The colonies, with a regular prominent white margin, were olive green to black 10 d post-incubation. The bottom of the colonies was black surrounded with a light-brown circle. The aerial hyphae were thick and cottony and turned from colourless to pale brown (Figure 3A). Conidiophores arose singly and were separated and pale brown. The conidia were solitary or in chains, the conidial body was 18.09–37.61 μm × 9.15–19.90 μm (average 24 × 14 μm, n = 50), typically obclavate, subglobose and ellipsoid, with 1–5 transverse septa and 1–3 longitudinal septa that slightly constricted near several septa. The conidia were yellow–brown and later turned black–brown (Figure 3B–F). The morphological characterization of ten isolates revealed Alternaria-like morphology.
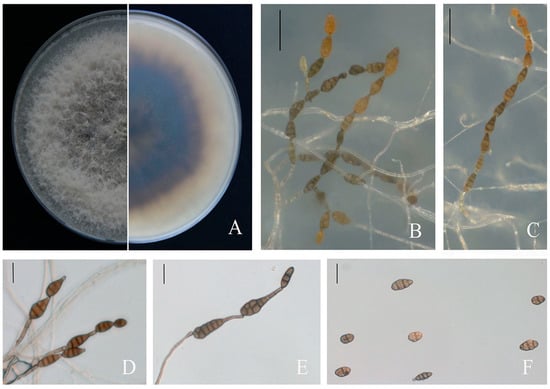
Figure 3.
Morphological characters of A. alternata. (A) Front and back views of colony morphology on PDA after 7 days. (B–E) Conidiophores developed on SNA. (F) Conidia. Scale bars: (B,C) = 50 µm; (D–F) = 20 µm.
3.4. Phylogenetic Analysis
A multilocus phylogenetic analysis was conducted on ten pathogenic isolates based on the sequences from eight genes: SSU, LSU, ITS, gapdh, tef1, Alt a 1, endoPG and OPA10-2 (GenBank accession numbers MZ835355 to MZ835364, MZ835345 to MZ835354, MZ823461 to MZ823470, MZ835385 to MZ835394, MZ835395 to MZ835404, MZ802959 to MZ802968, MZ835375 to MZ835384, MZ835365 to MZ835374). For these ten isolates, the PCR amplification and sequencing of each gene generated product sizes were about 1072, 942, 733, 619, 259, 516, 491 and 753 or 777 bp, respectively. The alignments (including the gaps) for eight genes were 1021, 849, 522, 579, 241, 473, 448 and 634 bp in size, respectively. The ten sequences of isolates along with sequences from 33 Alternaria strains were concatenated for the construction of a phylogenetic tree. The alignment of the eight-locus concatenated dataset consisted of 4767 characters, with 4356 constant characters, 245 parsimony-uninformative characters, and 166 parsimony-informative characters.
ML and BI analyses generated basically the same tree topology, which demonstrated that the evolutionary relationships of the fungus isolates were statistically supported. A single tree with bootstrap proportions (BP) from ML and Bayesian posterior probabilities (BPP) from BI was generated (Figure 4). The phylogenetic analysis showed that all isolates herein clustered into two clades, with a highly supported clade (≥92% BP/0.91 BPP) with A. alternata CBS 121455 and CBS 121336. Two phylogenetic analyses revealed that all isolates with aggressiveness showed >95% similarity to the A. alternata isolates reported previously.
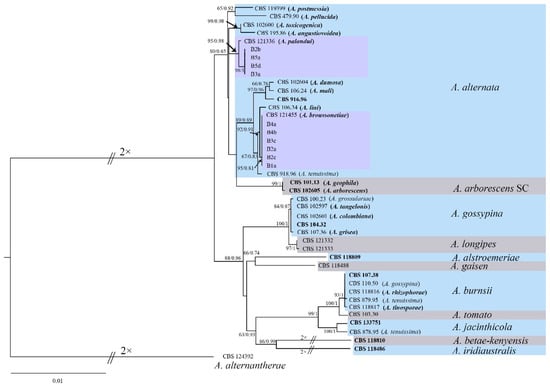
Figure 4.
Maximum likelihood and Bayesian analyses of 43 isolates of the Alternaria species. The tree was generated with concatenated sequences of the SSU, LSU, ITS, gapdh, tef1, Alt a 1, endoPG and OPA10-2 regions or genes. The tree generated by Bayesian inference had a similar topology. Bootstrap support values above 60% (before the slash marks) and Bayesian posterior probability values above 0.75 (after the slash marks) are shown at each node. Species names in parentheses refer to the former species name. Ex-type strains are emphasised in bold. A. alternantherae CBS 124392 was used as an outgroup. The scale bar shows the predicted number of substitutions per nucleotide position.
4. Discussion
Because of its ability to assimilate harmful material in the needles, graceful appearance and fine timber, Bunge’s pine plays an essential role in ecology and the economy. Needle blight disease can not only worsen the pine appearance but also influence apical dominance. The loss of apical dominance reduces wood quality. Moreover, the death of trees can occur in severe cases. Generally, the diseases affecting Bunge’s pine damage the economy and ecology. Based on morphological characteristics and molecular identification with phylogenetic analysis of multiple gene sequences, A. alternata was confirmed to be the causal agent of needle blight on Bunge’s pine in China. This is the first report of A. alternata on P. bungeana.
Several small-spore Alternaria spp. are frequently misidentified due to morphological overlap with A. alternata [35]. The dimensions of conidia in this study were very different from those described by Moumni et al. [49], but were similar to those reported by Gao et al. [50]. This phenomenon could be attributed to the morphological plasticity exhibited by most Alternaria species. Conidial morphology is dependent on culture conditions and conidium age [35]. The number of conidia produced with conidial chains was related to the nutrition that the fungi obtained. In addition, the numbers of longitudinal and transverse septa were variable. It is suggested that morphological characteristics are not stable.
Due to morphological variability and minimal molecular variation, the taxa of Alternaria spp. were reclassified by Woundenberg et al. [51]. Whole-genome sequencing and transcriptome sequencing were used to distinguish 168 Alternaria isolates, and nine gene regions (SSU, LSU, ITS, gapdh, tef1, Alt a 1, endoPG, OPA10-2 and rpb2) were selected to distinguish sect. Alternaria more effectively [20]. Phylogenetic analyses and species identification are challenging in small-spored Alternaria due to lineage sorting, recombination and horizontal transfer [52]. Multilocus species identification was confirmed to be necessary among Alternaria sections for low resolution of species delimitation in small-spored Alternaria [10]. The analysis with a concatenation of six gene regions (ITS, rpb2, endoPG, tef1, Alt a 1 and OPA10-2) was able to separate A. alternata from the A. arborescens species complex [10]. A slowly evolving gene (rpb2) was excluded, while additional molecular markers (gaphd, SSU and LSU) were included in this study as proposed by Woudenberg et al. [20]. The combined phylogenetic tree shows consistency with other studies [10,15,17,20].
Alternaria alternata was reported as a ubiquitous pathogen in the great majority of crops and some broad-leaved trees [17,26,30,31,53,54,55,56]. In particular, A. alternata is the most important mycotoxin-producing genus as a result of the wide reports of TA, AME, AOH, ALT and ATX produced [57]. In addition, A. alternata can not only colonise the phylloplane but also penetrate into living leaves [58]. Nevertheless, A. alternata was reported to be the dominant endophytic fungal taxon in the bark and needles of Chinese oil pine (Pinus tabulaeformis Carr.) and isolated from various plants [59]. In addition, as an endophytic fungus, it showed strong antifungal activity against Raffaelea quercus-mongolicae [60]. When examining the abundance and diversity of fungi on needles of Pinus sylvestris, A. alternata was found to be a common primary or secondary saprotroph [61]. It is difficult for A. alternata to colonise Bunge’s pine needles without wounding, which may be related to plant resistance or pathogenic activity. The result of unwounded inoculation indicated that wounding may play a significant role in the pathogenicity of A. alternata. In nature, needles are prone to chafing, which can induce laceration as a result of the wind. This may provide an opportunity for A. alternata to be virulent. In addition, the virulence of A. alternata may have been obtained horizontally from a recent common saprophytic ancestor [52].
According to previous studies, A. alternata, as a pathogen of pine needles, has never been reported. Although the thicker epidermis and cuticle of needles make it more difficult for fungi to invade plants, it is noteworthy that wounds appearing on needles may lead to disease prevalence. Pathogenicity test results indicate that A. alternata has the ability to infect other Pinus species, and it is necessary to investigate the distribution and propagation of the disease caused by A. alternata. A. alternata may pose a great threat to ecology because the hosts that the pathogen can invade are increasing, especially in Pinus species. Studies on the pathogenicity mechanism of A. alternata and disease management should be conducted in the future.
Author Contributions
Conceptualization, F.-M.C.; methodology, M.-J.Z. and X.-R.Z.; software, X.-R.Z.; validation, M.-J.Z.; formal analysis, M.-J.Z.; investigation, F.-M.C.; resources, F.-M.C.; data curation, M.-J.Z.; writing—original draft preparation, M.-J.Z.; writing—review and editing, X.-R.Z. and H.L.; visualization, M.-J.Z. and X.-R.Z.; supervision, F.-M.C.; project administration, F.-M.C.; funding acquisition, F.-M.C. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by grants from the National Key Research and Development Program of China (2017YFD0600104).
Institutional Review Board Statement
Not applicable for studies not involving humans or animals.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data generated or analyzed during this study are included in this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zhao, Y.; Zhang, X.Z.; Wang, X.A. A study on geographical distribution law of Pinus bungeana natural forests in china. Acta Bot. Boreali-Occident. Sin. 1995, 15, 161–166. [Google Scholar]
- Guo, C.C.; Shen, Y.B.; Shi, F.H. Investigating seed dormancy in Pinus bungeana Zucc. ex Endl.: Understanding the contributions of enclosing tissues and temperature on germination. Forests 2018, 9, 401. [Google Scholar] [CrossRef]
- Zhang, Z.; Ma, J.; Ji, Z.; Xu, F. Comparison of anatomy and composition distribution between normal and compression wood of Pinus bungeana Zucc. revealed by microscopic imaging techniques. Microsc. Microanal. 2012, 18, 1459–1466. [Google Scholar] [CrossRef] [PubMed]
- Duan, D.; Jia, Y.; Yang, J.; Li, Z.H. Comparative transcriptome analysis of male and female conelets and development of microsatellite markers in Pinus bungeana, an endemic conifer in China. Genes 2017, 8, 393. [Google Scholar] [CrossRef]
- Yang, Y.X.; Wang, M.L.; Liu, Z.L.; Zhu, J.; Yan, M.Y.; Li, Z.H. Nucleotide polymorphism and phylogeographic history of an endangered conifer species Pinus bungeana. Biochem. Syst. Ecol. 2016, 64, 89–96. [Google Scholar] [CrossRef]
- Li, Z.H.; Zhu, J.; Yang, Y.X.; Yang, J.; He, J.W.; Zhao, G.F. The complete plastid genome of Bunge’s pine Pinus bungeana (Pinaceae). Mitochondr. DNA 2016, 27, 2971–2972. [Google Scholar] [CrossRef]
- Lawrence, D.P.; Gannibal, P.B.; Peever, T.L.; Pryor, B.M. The sections of Alternaria: Formalizing species-group concepts. Mycologia 2013, 105, 530–546. [Google Scholar] [CrossRef]
- Simmons, E.G. Alternaria. An Identification Manual. CBS Biodiversity Series 6; CBS Fungal Biodiversity Centre: Utrecht, The Netherlands, 2007. [Google Scholar]
- Xu, B.; Song, J.; Xi, P.; Li, M.; Hsiang, T.; Jiang, Z. A destructive leaf spot and blight caused by Alternaria kareliniae sp. nov. on a sand-stabilizing plant, caspian sea karelinia. Plant Dis. 2018, 102, 172–178. [Google Scholar] [CrossRef]
- Matic, S.; Tabone, G.; Garibaldi, A.; Gullino, M.L. Alternaria leaf spot caused by Alternaria species: An emerging problem on ornamental plants in Italy. Plant Dis. 2020, 104, 2275–2287. [Google Scholar] [CrossRef]
- Lawrence, D.P.; Rotondo, F.; Gannibal, P.B. Biodiversity and taxonomy of the pleomorphic genus Alternaria. Mycol. Prog. 2016, 15, 3. [Google Scholar] [CrossRef]
- Thomma, B. Alternaria spp.: From general saprophyte to specific parasite. Mol. Plant Pathol. 2003, 4, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Kahl, S.M.; Ulrich, A.; Kirichenko, A.A.; Muller, M.E.H. Phenotypic and phylogenetic segregation of Alternaria infectoria from small-spored Alternaria species isolated from wheat in Germany and Russia. J. Appl. Microbiol. 2015, 119, 1637–1650. [Google Scholar] [CrossRef] [PubMed]
- Al-Nadabi, H.H.; Maharachchikumbura, S.S.N.; Agrama, H.; Al-Azri, M.; Nasehi, A.; Al-Sadi, A.M. Molecular characterization and pathogenicity of Alternaria species on wheat and date palms in Oman. Eur. J. Plant Pathol. 2018, 152, 577–588. [Google Scholar] [CrossRef]
- Elfar, K.; Zoffoli, J.P.; Latorre, B.A. Identification and characterization of Alternaria species associated with moldy core of apple in Chile. Plant Dis. 2018, 102, 2158–2169. [Google Scholar] [CrossRef] [PubMed]
- Loganathan, M.; Venkataravanappa, V.; Saha, S.; Rai, A.B.; Tripathi, S.; Rai, R.K.; Pandey, A.K. Morphological, pathogenic and molecular characterizations of Alternaria species causing early blight of tomato in Northern India. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2016, 86, 325–330. [Google Scholar] [CrossRef]
- Zhu, Y.; Lujan, P.; Dura, S.; Steiner, R.; Zhang, J.; Sanogo, S. Etiology of Alternaria leaf spot of cotton in Southern New Mexico. Plant Dis. 2019, 103, 1595–1604. [Google Scholar] [CrossRef]
- Andrew, M.; Peever, T.L.; Pryor, B.M. An expanded multilocus phylogeny does not resolve morphological species within the small-spored Alternaria species complex. Mycologia 2009, 101, 95–109. [Google Scholar] [CrossRef]
- Pryor, B.M.; Bigelow, D.M. Molecular characterization of Embellisia and Nimbya species and their relationship to Alternaria, Ulocladium and Stemphylium. Mycologia 2003, 95, 1141–1154. [Google Scholar] [CrossRef]
- Woudenberg, J.H.; Seidl, M.F.; Groenewald, J.Z.; de Vries, M.; Stielow, J.B.; Thomma, B.P.; Crous, P.W. Alternaria section Alternaria: Species, formae speciales or pathotypes? Stud. Mycol. 2015, 82, 1–21. [Google Scholar] [CrossRef]
- Luo, Y.; Hou, L.; Forster, H.; Pryor, B.; Adaskaveg, J.E. Identification of Alternaria species causing heart rot of pomegranates in California. Plant Dis. 2017, 101, 421–427. [Google Scholar] [CrossRef]
- Zhu, X.Q.; Xiao, C.L. Phylogenetic, morphological and pathogenic characterization of Alternaria species Associated with fruit rot of blueberry in California. Phytopathology 2015, 105, 1555–1567. [Google Scholar] [CrossRef] [PubMed]
- Pryor, B.M.; Gilbertson, R.L. Molecular phylogenetic relationships amongst Alternaria species and related fungi based upon analysis of nuclear ITS and mt SSU rDNA sequences. Mycol. Res. 2000, 104, 1312–1321. [Google Scholar] [CrossRef]
- Rang, J.C.; Crous, P.W.; Mchau, G.R.A.; Serdani, M.; Song, S.M. Phylogenetic analysis of Alternaria spp. associated with apple core rot and citrus black rot in South Africa. Mycol. Res. 2002, 106, 1151–1162. [Google Scholar] [CrossRef]
- Sun, X.M.; Huang, J.G. First report of Alternaria tenuissima causing red leaf spot disease on Paeonia lactiflora in China. Plant Dis. 2017, 101, 1322. [Google Scholar] [CrossRef]
- Wang, Q.; Luan, Q.S.; Wang, J.J.; Jiang, X.; Chen, G. First report of Alternaria alternata causing leaf blight on Actinidia arguta in Liaoning, China. Plant Dis. 2020, 104, 2725–2726. [Google Scholar] [CrossRef]
- Mmbaga, M.T.; Shi, A.N.; Kim, M.S. Identification of Alternaria alternata as a causal agent for leaf blight in Syringa species. Plant Pathol. J. 2011, 27, 120–127. [Google Scholar] [CrossRef]
- Kim, H.Y.; Shin, J.H.; Lee, N.H.; Kim, S.; Kim, K. Isolation and evaluation of fungicides for control of Alternaria alternata causing Alternaria leaf spot disease on lettuce. J. Agric. Life Sci. 2021, 55, 89–97. [Google Scholar] [CrossRef]
- Zhang, J.X.; Kang, Y.; Xu, T.T.; Xu, X.Y.; Li, C.X.; Chen, K.F.; Cao, H.Q. First report of leaf spot caused by Alternaria alternata on Sonchus asper in China. Plant Dis. 2021, 105, 503. [Google Scholar] [CrossRef]
- Sun, H.F.; Li, N.; Yan, Y.; Wei, M.Y.; Wang, H.; Yang, H.Y. First report of Alternaria alternata causing leaf blight on Clematis terniflora var. mandshurica in China. Plant Dis. 2022, 106, 2264. [Google Scholar] [CrossRef]
- Long, H.J.; Yang, S.; Yin, X.H.; Zhao, Z.B.; Long, Y.H.; Fan, J.; Shu, R.; Gu, G.F. First report of leaf spot on Prunus salicina caused by Alternaria alternata in China. J. Plant Pathol. 2021, 103, 1021. [Google Scholar] [CrossRef]
- Qi, M.; Xie, C.X.; Chen, Q.W.; Yu, Z.D. Pestalotiopsis trachicarpicola, a novel pathogen causes twig blight of Pinus bungeana (Pinaceae: Pinoideae) in China. Antonie Van Leeuwenhoek. Int. J. Gen. Mol. Microbiol. 2021, 114, 1–9. [Google Scholar] [CrossRef]
- Lin, Y.R.; Tang, Y.P. Seven species of Lophodermium on pinus. Acta Mycol. Sin. 1988, 7, 129–137. [Google Scholar]
- Cai, L.; Hyde, K.D.; Taylor, P.W.J.; Weir, B.S.; Waller, J.M.; Abang, M.M.; Zhang, J.Z.; Yang, Y.L.; Phoulivong, S.; Liu, Z.Y.; et al. A polyphasic approach for studying Colletotrichum. Fungal Divers. 2009, 39, 183–204. [Google Scholar]
- Pryor, B.M.; Michailides, T.J. Morphological, pathogenic, and molecular characterization of Alternaria isolates associated with Alternaria late blight of pistachio. Phytopathology 2002, 92, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Nirenberg, H.I. Untersuchungen uber die morphologische und biologische differenzierung in der Fusaium-Sektion Liseola. In Mitteilungen aus der Biologischen Bundesanstalt für Land- und Forstwirtschaft Berlin-Dahlem; Kommissionsverlag Paul Parey: Berlin, Germany, 1976; Volume 169, pp. 1–117. [Google Scholar]
- Damm, U.; Mostert, L.; Crous, P.W.; Fourie, P.H. Novel Phaeoacremonium species associated with necrotic wood of Prunus trees. Persoonia 2008, 20, 87–102. [Google Scholar] [CrossRef] [PubMed]
- de Hoog, G.S.; Gerrits van den Ende, A.H. Molecular diagnostics of clinical strains of filamentous Basidiomycetes. Mycoses 1998, 41, 183–189. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.B.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Hong, S.G.; Cramer, R.A.; Lawrence, C.B.; Pryor, B.M. Alt a 1 allergen homologs from Alternaria and related taxa: Analysis of phylogenetic content and secondary structure. Fungal Genet. Biol. 2005, 42, 119–129. [Google Scholar] [CrossRef]
- Crous, P.W.; Schoch, C.L.; Hyde, K.D.; Wood, A.R.; Gueidan, C.; Hoog, G.D.; Groenewald, J.Z. Phylogenetic lineages in the Capnodiales. Stud. Mycol. 2009, 64, 17–47. [Google Scholar] [CrossRef]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef]
- Berbee, M.L.; Pirseyedi, M.; Hubbard, S. Cochliobolus phylogenetics and the origin of known, highly virulent pathogens, inferred from ITS and glyceraldehyde-3-phosphate dehydrogenase gene sequences. Mycologia 1999, 91, 964–977. [Google Scholar] [CrossRef]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.R.; Zhang, M.J.; Shang, X.L.; Fang, S.Z.; Chen, F.M. Stem canker on Cyclocarya paliurus is caused by Botryosphaeria dothidea. Plant Dis. 2020, 104, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Hohna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Moumni, M.; Allagui, M.B.; Mancini, V.; Murolo, S.; Tarchoun, N.; Romanazzi, G. Morphological and molecular identification of seedborne fungi in Squash (Cucurbita maxima, Cucurbita moschata). Plant Dis. 2020, 104, 1335–1350. [Google Scholar] [CrossRef]
- Gao, J.; Yang, M.J.; Xie, Z.; Lu, B.H.; Hsiang, T.; Liu, L.P. Morphological and molecular identification and pathogenicity of Alternaria spp. associated with ginseng in Jilin province, China. Can. J. Plant Pathol. 2021, 43, 537–550. [Google Scholar] [CrossRef]
- Woudenberg, J.H.; Groenewald, J.Z.; Binder, M.; Crous, P.W. Alternaria redefined. Stud. Mycol. 2013, 75, 171–212. [Google Scholar] [CrossRef]
- Stewart, J.E.; Timmer, L.W.; Lawrence, C.B.; Pryor, B.M.; Peever, T.L. Discord between morphological and phylogenetic species boundaries: Incomplete lineage sorting and recombination results in fuzzy species boundaries in an asexual fungal pathogen. BMC Evol. Biol. 2014, 14, 38. [Google Scholar] [CrossRef]
- Al-Lami, H.F.D.; You, M.P.; Barbetti, M.J. Incidence, pathogenicity and diversity of Alternaria spp. associated with alternaria leaf spot of canola (Brassica napus) in Australia. Plant Pathol. 2019, 68, 492–503. [Google Scholar] [CrossRef]
- Somma, S.; Amatulli, M.T.; Masiello, M.; Moretti, A.; Logrieco, A.F. Alternaria species associated to wheat black point identified through a multilocus sequence approach. Int. J. Food Microbiol. 2019, 293, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Kant, R.; Joshi, P.; Bhandari, M.S.; Pandey, A.; Pandey, S. Identification and pathogenicity of Alternaria alternata causing leaf spot and blight disease of Ailanthus excelsa in India. Forest Pathol. 2020, 50, e12584. [Google Scholar] [CrossRef]
- Tian, Y.; Qiu, C.D.; Zhang, Y.Y.; Liu, Z.Y. First report of Alternaria alternata causing leaf spot on Chaenomeles cathayensis in Anhui province of China. Plant Dis. 2020, 104, 279–280. [Google Scholar] [CrossRef]
- Logrieco, A.; Moretti, A.; Solfrizzo, M. Alternaria toxins and plant diseases: An overview of origin, occurrence and risks. World Mycotoxin J. 2009, 2, 129–140. [Google Scholar] [CrossRef]
- O’Donnell, J.; Dickinson, C.H. Pathogenicity of Alternaria and Cladosporium isolates on Phaseolus. Trans. Br. Mycol. Soc. 1980, 74, 335–342. [Google Scholar] [CrossRef]
- Wang, Y.; Guo, L.D. A comparative study of endophytic fungi in needles, bark, and xylem of Pinus tabulaeformis. Can. J. Bot. 2007, 85, 911–917. [Google Scholar] [CrossRef]
- Nguyen, M.H.; Yong, J.H.; Sung, H.J.; Lee, J.K. Screening of endophytic fungal isolates dgainst Raffaelea quercus-mongolicae causing oak wilt disease in Korea. Mycobiology 2020, 48, 484–494. [Google Scholar] [CrossRef]
- Behnke-Borowczyk, J.; Kwaśna, H.; Kulawinek, B. Fungi associated with Cyclaneusma needle cast in Scots pine in the west of Poland. Forest Pathol. 2019, 49, e12487. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).