Protective Efficacy of Anti-Hyr1p Monoclonal Antibody against Systemic Candidiasis Due to Multi-Drug-Resistant Candida auris
Abstract
1. Introduction
2. Materials and Methods
2.1. Candida Culture and Strains
2.2. In Silico Analysis
2.3. Monoclonal Antibody Generation
2.4. Binding Affinity Determination by Microscale Thermophoresis (MST)
2.5. Flow Cytometry
2.6. Biofilm Formation Assay
2.7. Opsonophagocytic Killing (OPK) Assay
2.8. Mice Infection and Treatment
2.9. Statistical Analysis
3. Results
3.1. C. auris Has Orthologs of Cal-Hyr1p
3.2. Anti-Cal-Hyr1p#5 Monoclonal Antibody Development
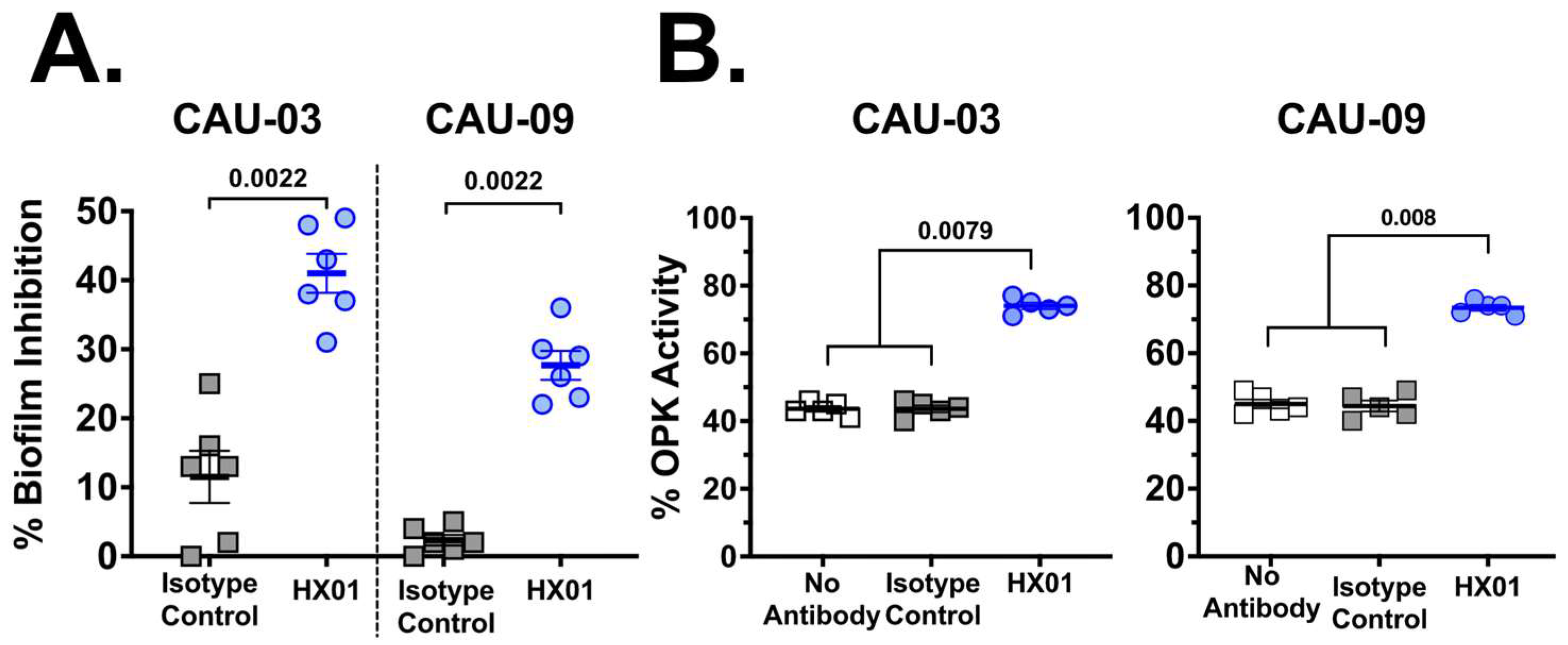
3.3. HX01 MAb Inhibits C. auris Biofilm Formation In Vitro and Augments Macrophage Opsonophagocytic Killing (OPK) of C. auris
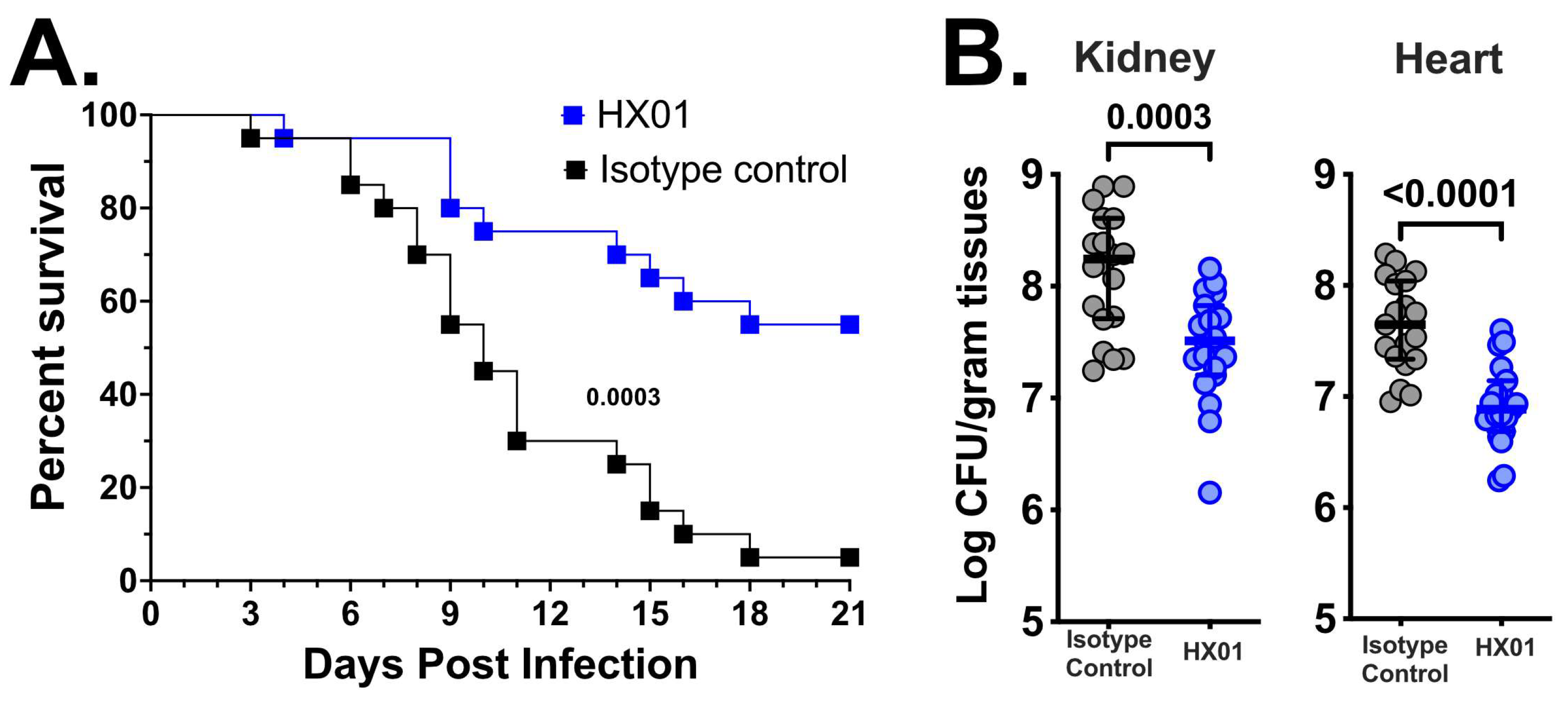
3.4. HX01 MAb Protects Mice from Hematogenously Disseminated Candidiasis Due to C. auris Infection
3.5. HX01 Does Not Protect Mice from Hematogenously Disseminated Candidiasis Due to C. albicans Infection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lockhart, S.R.; Etienne, K.A.; Vallabhaneni, S.; Farooqi, J.; Chowdhary, A.; Govender, N.P.; Colombo, A.L.; Calvo, B.; Cuomo, C.A.; Desjardins, C.A.; et al. Simultaneous Emergence of Multidrug-Resistant Candida auris on 3 Continents Confirmed by Whole-Genome Sequencing and Epidemiological Analyses. Clin. Infect. Dis. 2017, 64, 134–140. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention Screening for Candida auris Colonization|Candida auris|Fungal Diseases|CDC. Available online: https://www.cdc.gov/fungal/candida-auris/c-auris-screening.html (accessed on 10 December 2022).
- CDC Tracking Candida auris. Available online: https://www.cdc.gov/fungal/candida-auris/tracking-c-auris.html (accessed on 10 December 2022).
- Chakrabarti, A.; Sood, P.; Rudramurthy, S.M.; Chen, S.; Kaur, H.; Capoor, M.; Chhina, D.; Rao, R.; Eshwara, V.K.; Xess, I.; et al. Incidence, Characteristics and Outcome of ICU-Acquired Candidemia in India. Intensive Care Med. 2015, 41, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.G.; Shin, J.H.; Uh, Y.; Kang, M.G.; Kim, S.H.; Park, K.H.; Jang, H.C. First Three Reported Cases of Nosocomial Fungemia Caused by Candida auris. J. Clin. Microbiol. 2011, 49, 3139–3142. [Google Scholar] [CrossRef] [PubMed]
- Sarma, S.; Upadhyay, S. Current Perspective on Emergence, Diagnosis and Drug Resistance in Candida auris. Infect. Drug Resist. 2017, 10, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Adams, E.; Quinn, M.; Tsay, S.; Poirot, E.; Chaturvedi, S.; Southwick, K.; Greenko, J.; Fernandez, R.; Kallen, A.; Vallabhaneni, S.; et al. Candida auris in Healthcare Facilities, New York, USA, 2013–2017. Emerg. Infect. Dis. 2018, 24, 1816–1824. [Google Scholar] [CrossRef]
- National Center for Emerging and Zoonotic Infectious Diseases (NCEZID); Center for Disease Control and Prevention (CDC). Candida auris: A Drug-Resistant Germ That Spreads in Healthcare Facilities. Available online: https://www.cdc.gov/fungal/diseases/candidiasis/pdf/Candida_auris_508.pdf (accessed on 10 December 2022).
- Piedrahita, C.T.; Cadnum, J.L.; Jencson, A.L.; Shaikh, A.A.; Ghannoum, M.A.; Donskey, C.J. Environmental Surfaces in Healthcare Facilities Are a Potential Source for Transmission of Candida auris and Other Candida Species. Infect. Control Hosp. Epidemiol. 2017, 38, 1107–1109. [Google Scholar] [CrossRef]
- Vallabhaneni, S.; Kallen, A.; Tsay, S.; Chow, N.; Welsh, R.; Kerins, J.; Kemble, S.K.; Pacilli, M.; Black, S.R.; Landon, E.; et al. Investigation of the First Seven Reported Cases of Candida auris, a Globally Emerging Invasive, Multidrug-Resistant Fungus—United States, May 2013–August 2016. Am. J. Transplant. 2017, 17, 296–299. [Google Scholar] [CrossRef]
- Mathur, P.; Hasan, F.; Singh, P.K.; Malhotra, R.; Walia, K.; Chowdhary, A. Five-Year Profile of Candidaemia at an Indian Trauma Centre: High Rates of Candida auris Blood Stream Infections. Mycoses 2018, 61, 674–680. [Google Scholar] [CrossRef]
- Jeffery-Smith, A.; Taori, S.K.; Schelenz, S.; Jeffery, K.; Johnson, E.M.; Borman, A.; Manuel, R.; Browna, C.S. Candida auris: A Review of the Literature. Clin. Microbiol. Rev. 2018, 31, e00029-17. [Google Scholar] [CrossRef]
- Chowdhary, A.; Voss, A.; Meis, J.F. Multidrug-Resistant Candida auris: ‘New Kid on the Block’ in Hospital-Associated Infections? J. Hosp. Infect. 2016, 94, 209–212. [Google Scholar] [CrossRef]
- Horton, M.V.; Nett, J.E. Candida auris infection and biofilm formation: Going beyond the surface. Curr. Clin. Microbiol. Rep. 2020, 7, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Sherry, L.; Ramage, G.; Kean, R.; Borman, A.; Johnson, E.M.; Richardson, M.D.; Rautemaa-Richardson, R. Biofilm-Forming Capability of Highly Virulent, Multidrug-Resistant Candida auris. Emerg. Infect. Dis. 2017, 23, 328–331. [Google Scholar] [CrossRef] [PubMed]
- Morales-López, S.E.; Parra-Giraldo, C.M.; Ceballos-Garzón, A.; Martínez, H.P.; Rodríguez, G.J.; Álvarez-Moreno, C.A.; Rodríguez, J.Y. Invasive Infections with Multidrug-Resistant Yeast Candida auris, Colombia. Emerg. Infect. Dis. 2017, 23, 162–164. [Google Scholar] [CrossRef] [PubMed]
- Abdoli, A.; Falahi, S.; Kenarkoohi, A. COVID-19-Associated Opportunistic Infections: A Snapshot on the Current Reports. Clin. Exp. Med. 2022, 22, 327–346. [Google Scholar] [CrossRef] [PubMed]
- Arastehfar, A.; Carvalho, A.; Nguyen, M.H.; Hedayati, M.T.; Netea, M.G.; Perlin, D.S.; Hoenigl, M. COVID-19-Associated Candidiasis (Cac): An Underestimated Complication in the Absence of Immunological Predispositions? J. Fungi 2020, 6, 211. [Google Scholar] [CrossRef]
- Chowdhary, A.; Sharma, A. The Lurking Scourge of Multidrug Resistant Candida auris in Times of COVID-19 Pandemic. J. Glob. Antimicrob. Resist. 2020, 22, 175–176. [Google Scholar] [CrossRef]
- Pemán, J.; Ruiz-Gaitán, A.; García-Vidal, C.; Salavert, M.; Ramírez, P.; Puchades, F.; García-Hita, M.; Alastruey-Izquierdo, A.; Quindós, G. Fungal Co-Infection in COVID-19 Patients: Should We Be Concerned? Rev. Iberoam. Micol. 2020, 37, 41–46. [Google Scholar] [CrossRef]
- CDC 2019 Antibiotic Resistance Threats Report|CDC. Available online: https://www.cdc.gov/drugresistance/Biggest-Threats.html%0Ahttps://www.cdc.gov/drugresistance//biggest-threats.html (accessed on 9 November 2022).
- Kean, R.; Ramage, G. Combined Antifungal Resistance and Biofilm Tolerance: The Global Threat of Candida auris. mSphere 2019, 4, e00458-19. [Google Scholar] [CrossRef]
- Meis, J.F.; Chowdhary, A. Candida auris: A Global Fungal Public Health Threat. Lancet Infect. Dis. 2018, 18, 1298–1299. [Google Scholar] [CrossRef]
- Healey, K.R.; Kordalewska, M.; Ortigosa, C.J.; Singh, A.; Berrío, I.; Chowdhary, A.; Perlin, D.S. Limited ERG11 Mutations Identified in Isolates of Candida auris Directly Contribute to Reduced Azole Susceptibility. Antimicrob. Agents Chemother. 2018, 62, e01427-18. [Google Scholar] [CrossRef]
- Chatterjee, S.; Alampalli, S.V.; Nageshan, R.K.; Chettiar, S.T.; Joshi, S.; Tatu, U.S. Draft Genome of a Commonly Misdiagnosed Multidrug Resistant Pathogen Candida auris. BMC Genom. 2015, 16, 686. [Google Scholar] [CrossRef] [PubMed]
- Klis, F.M.; Sosinska, G.J.; de Groot, P.W.J.; Brul, S. Covalently Linked Cell Wall Proteins of Candida albicans and Their Role in Fitness and Virulence. FEMS Yeast Res. 2009, 9, 1013–1028. [Google Scholar] [CrossRef] [PubMed]
- de Groot, P.W.J.; de Boer, A.D.; Cunningham, J.; Dekker, H.L.; de Jong, L.; Hellingwerf, K.J.; de Koster, C.; Klis, F.M. Proteomic Analysis of Candida albicans Cell Walls Reveals Covalently Bound Carbohydrate-Active Enzymes and Adhesins. Eukaryot. Cell 2004, 3, 955–965. [Google Scholar] [CrossRef]
- Bailey, D.A.; Feldmann, P.J.F.; Bovey, M.; Gow, N.A.R.; Brown, A.J.P. The Candida albicans HYR1 Gene, Which Is Activated in Response to Hyphal Development, Belongs to a Gene Family Encoding Yeast Cell Wall Proteins. J. Bacteriol. 1996, 178, 5353–5360. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, L.L. The ALS Gene Family of Candida albicans. Trends Microbiol. 2001, 9, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Lipke, P.N. What We Do Not Know about Fungal Cell Adhesion Molecules. J. Fungi 2018, 4, 59. [Google Scholar] [CrossRef]
- Luo, G.; Ibrahim, A.S.; Spellberg, B.; Nobile, C.J.; Mitchell, A.P.; Fu, Y. Candida albicans Hyr1p Confers Resistance to Neutrophil Killing and Is a Potential Vaccine Target. J. Infect. Dis. 2010, 201, 1718–1728. [Google Scholar] [CrossRef]
- Wang, L.; Che, X.; Jiang, J.; Qian, Y.; Wang, Z. New Insights into Ocular Complications of Human Immunodeficiency Virus Infection. Curr. HIV Res. 2021, 19, 476–487. [Google Scholar] [CrossRef]
- Reithofer, V.; Fernández-Pereira, J.; Alvarado, M.; de Groot, P.; Essen, L.O. A Novel Class of Candida glabrata Cell Wall Proteins with β-Helix Fold Mediates Adhesion in Clinical Isolates. PLoS Pathog. 2021, 17, e1009980. [Google Scholar] [CrossRef]
- Koteiche, H.A.; Mchaourab, H.S. Folding Pattern of the α-Crystallin Domain in AA-Crystallin Determined by Site-Directed Spin Labeling. J. Mol. Biol. 1999, 294, 561–577. [Google Scholar] [CrossRef]
- Liu, Y.; Filler, S.G. Candida albicans Als3, a Multifunctional Adhesin and Invasin. Eukaryot. Cell 2011, 10, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Phan, Q.T.; Luo, G.; Solis, N.v.; Liu, Y.; Cormack, B.P.; Edwards, J.E.; Ibrahim, A.S.; Filler, S.G. Investigation of the Function of Candida albicans Als3 by Heterologous Expression in Candida glabrata. Infect. Immun. 2013, 81, 2528–2535. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.S.; Spellberg, B.J.; Avanesian, V.; Fu, Y.; Edwards, J.E. The Anti-Candida Vaccine Based on the Recombinant N-Terminal Domain of Als1p Is Broadly Active against Disseminated Candidiasis. Infect. Immun. 2006, 74, 3039–3041. [Google Scholar] [CrossRef]
- Spellberg, B.J.; Ibrahim, A.S.; Avenissian, V.; Filler, S.G.; Myers, C.L.; Fu, Y.; Edwards, J.E. The Anti-Candida albicans Vaccine Composed of the Recombinant N Terminus of Als1p Reduces Fungal Burden and Improves Survival in Both Immunocompetent and Immunocompromised Mice. Infect. Immun. 2005, 73, 6191–6193. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.S.; White, C.J.; Ibrahim, A.S.; Filler, S.G.; Fu, Y.; Yeaman, M.R.; Edwards, J.E.; Hennessey, J.P. NDV-3, a Recombinant Alum-Adjuvanted Vaccine for Candida and Staphylococcus aureus, Is Safe and Immunogenic in Healthy Adults. Vaccine 2012, 30, 7594–7600. [Google Scholar] [CrossRef] [PubMed]
- Spellberg, B.J.; Ibrahim, A.S.; Avanesian, V.; Fu, Y.; Myers, C.; Phan, Q.T.; Filler, S.G.; Yeaman, M.R.; Edwards, J.E. Efficacy of the Anti-Candida RAls3p-N or RAls1p-N Vaccines against Disseminated and Mucosal Candidiasis. J. Infect. Dis. 2006, 194, 256–260. [Google Scholar] [CrossRef]
- Edwards, J.E.; Schwartz, M.M.; Schmidt, C.S.; Sobel, J.D.; Nyirjesy, P.; Schodel, F.; Marchus, E.; Lizakowski, M.; Demontigny, E.A.; Hoeg, J.; et al. A Fungal Immunotherapeutic Vaccine (NDV-3A) for Treatment of Recurrent Vulvovaginal Candidiasis-A Phase 2 Randomized, Double-Blind, Placebo-Controlled Trial. Clin. Infect. Dis. 2018, 66, 1928–1936. [Google Scholar] [CrossRef]
- Singh, S.; Uppuluri, P.; Mamouei, Z.; Alqarihi, A.; Elhassan, H.; French, S.; Lockhart, S.R.; Chiller, T.; Edwards, J.E.; Ibrahim, A.S. The NDV-3A Vaccine Protects Mice from Multidrug Resistant Candida auris Infection. PLoS Pathog. 2019, 15, e1007460. [Google Scholar] [CrossRef]
- Spellberg, B.; Ibrahim, A.S.; Yeaman, M.R.; Lin, L.; Fu, Y.; Avanesian, V.; Bayer, A.S.; Filler, S.G.; Lipke, P.; Otoo, H.; et al. The Antifungal Vaccine Derived from the Recombinant N Terminus of Als3p Protects Mice against the Bacterium Staphylococcus aureus. Infect. Immun. 2008, 76, 4574–4580. [Google Scholar] [CrossRef]
- Youssef, E.G.; Zhang, L.; Alkhazraji, S.; Gebremariam, T.; Singh, S.; Yount, N.Y.; Yeaman, M.R.; Uppuluri, P.; Ibrahim, A.S. Monoclonal IgM Antibodies Targeting Candida albicans Hyr1 Provide Cross-Kingdom Protection Against Gram-Negative Bacteria. Front. Immunol. 2020, 11, 76. [Google Scholar] [CrossRef]
- Uppuluri, P.; Lin, L.; Alqarihi, A.; Luo, G.; Youssef, E.G.; Alkhazraji, S.; Yount, N.Y.; Ibrahim, B.A.; Bolaris, M.A.; Edwards, J.E.; et al. The Hyr1 Protein from the Fungus Candida albicans Is a Cross Kingdom Immunotherapeutic Target for Acinetobacter Bacterial Infection. PLoS Pathog. 2018, 14, e1007056. [Google Scholar] [CrossRef]
- Smoak, R.A.; Snyder, L.F.; Fassler, J.S.; He, B.Z. Parallel Expansion and Divergence of the Hyr/Iff-like (Hil) Adhesin Family in Pathogenic Yeasts Including Candida auris. bioRxiv 2022. [Google Scholar] [CrossRef]
- Kelley, L.A.; Mezulis, S.; Yates, C.M.; Wass, M.N.; Sternberg, M.J.E. The Phyre2 Web Portal for Protein Modeling, Prediction and Analysis. Nat. Protoc. 2015, 10, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yan, R.; Roy, A.; Xu, D.; Poisson, J.; Zhang, Y. The I-TASSER Suite: Protein Structure and Function Prediction. Nat. Methods 2014, 12, 7–8. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Zhang, Y. Ab Initio Protein Structure Assembly Using Continuous Structure Fragments and Optimized Knowledge-Based Force Field. Proteins Struct. Funct. Bioinform. 2012, 80, 1715–1735. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.F.; Waterman, M.S. Identification of Common Molecular Subsequences. J. Mol. Biol. 1981, 147, 195–197. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A Visualization System for Exploratory Research and Analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Zhang, Y.; Skolnick, J. TM-Align: A Protein Structure Alignment Algorithm Based on the TM-Score. Nucleic Acids Res. 2005, 33, 2302–2309. [Google Scholar] [CrossRef]
- Uppuluri, P.; Singh, S.; Alqarihi, A.; Schmidt, C.S.; Hennessey, J.P.; Yeaman, M.R.; Filler, S.G.; Edwards, J.E.; Ibrahim, A.S. Human Anti-Als3p Antibodies Are Surrogate Markers of NDV-3A Vaccine Efficacy Against Recurrent Vulvovaginal Candidiasis. Front. Immunol. 2018, 9, 1349. [Google Scholar] [CrossRef]
- Waldmann, T.A.; Strober, W. Metabolism of Immunoglobulins. Prog. Allergy 1969, 13, 1–110. [Google Scholar] [CrossRef]
- Seijsing, J.; Yu, S.; Frejd, F.Y.; Höiden-Guthenberg, I.; Gräslund, T. In Vivo Depletion of Serum IgG by an Affibody Molecule Binding the Neonatal Fc Receptor. Sci. Rep. 2018, 8, 5141. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Ibrahim, A.S.; French, S.W.; Edwards, J.E.; Fu, Y. Active and Passive Immunization with RHyr1p-N Protects Mice against Hematogenously Disseminated Candidiasis. PLoS ONE 2011, 6, e25909. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Nabeela, S.; Barbarino, A.; Ibrahim, A.S.; Uppuluri, P. Antibodies Targeting Candida albicans Als3 and Hyr1 Antigens Protect Neonatal Mice from Candidiasis. Front. Immunol. 2022, 13, 925821. [Google Scholar] [CrossRef] [PubMed]
- Mansour, S.C.; Pletzer, D.; de la Fuente-Núñez, C.; Kim, P.; Cheung, G.Y.C.; Joo, H.S.; Otto, M.; Hancock, R.E.W. Bacterial Abscess Formation Is Controlled by the Stringent Stress Response and Can Be Targeted Therapeutically. EBioMedicine 2016, 12, 219–226. [Google Scholar] [CrossRef]
- Heilmann, C.J.; Sorgo, A.G.; Siliakus, A.R.; Dekker, H.L.; Brul, S.; Koster, C.G.; de Koning, L.J.; Klis, F.M. Hyphal Induction in the Human Fungal Pathogen Candida albicans Reveals a Characteristic Wall Protein Profile. Microbiology 2011, 157, 2297–2307. [Google Scholar] [CrossRef]
- Muñoz, J.F.; Gade, L.; Chow, N.A.; Loparev, V.N.; Juieng, P.; Berkow, E.L.; Farrer, R.A.; Litvintseva, A.P.; Cuomo, C.A. Genomic Insights into Multidrug-Resistance, Mating and Virulence in Candida auris and Related Emerging Species. Nat. Commun. 2018, 9, 5346. [Google Scholar] [CrossRef]
- Yona, S.; Kim, K.W.; Wolf, Y.; Mildner, A.; Varol, D.; Breker, M.; Strauss-Ayali, D.; Viukov, S.; Guilliams, M.; Misharin, A.; et al. Fate Mapping Reveals Origins and Dynamics of Monocytes and Tissue Macrophages under Homeostasis. Immunity 2013, 38, 79–91. [Google Scholar] [CrossRef]
- Rudkin, F.M.; Raziunaite, I.; Workman, H.; Essono, S.; Belmonte, R.; MacCallum, D.M.; Johnson, E.M.; Silva, L.M.; Palma, A.S.; Feizi, T.; et al. Single Human B Cell-Derived Monoclonal Anti-Candida Antibodies Enhance Phagocytosis and Protect against Disseminated Candidiasis. Nat. Commun. 2018, 9, 5288. [Google Scholar] [CrossRef]
- di Mambro, T.; Vanzolini, T.; Bruscolini, P.; Perez-Gaviro, S.; Marra, E.; Roscilli, G.; Bianchi, M.; Fraternale, A.; Schiavano, G.F.; Canonico, B.; et al. A New Humanized Antibody Is Effective against Pathogenic Fungi In Vitro. Sci. Rep. 2021, 11, 19500. [Google Scholar] [CrossRef]
- Rosario-colon, J.; Eberle, K.; Adams, A.; Courville, E.; Xin, H. Candida Cell-Surface-Specific Monoclonal Antibodies Protect Mice against Candida auris Invasive Infection. Int. J. Mol. Sci. 2021, 22, 6162. [Google Scholar] [CrossRef]
- Lin, L.; Ibrahim, A.S.; Xu, X.; Farber, J.M.; Avanesian, V.; Baquir, B.; Fu, Y.; French, S.W.; Edwards, J.E.; Spellberg, B. Th1-Th17 Cells Mediate Protective Adaptive Immunity against Staphylococcus aureus and Candida albicans Infection in Mice. PLoS Pathog. 2009, 5, e1000703. [Google Scholar] [CrossRef] [PubMed]
- Torosantucci, A.; Chiani, P.; Bromuro, C.; De Bernardis, F.; Palma, A.S.; Liu, Y.; Mignogna, G.; Maras, B.; Colone, M.; Stringaro, A.; et al. Protection by Anti-b-Glucan Antibodies Is Associated with Restricted b-1,3 Glucan Binding Specificity and Inhibition of Fungal Growth and Adherence. PLoS ONE 2009, 4, e5392. [Google Scholar] [CrossRef] [PubMed]
- Han, Y. Efficacy of Combination Immunotherapy of IgM MAb B6.1 and Amphotericin B against Disseminated Candidiasis. Int. Immunopharmacol. 2010, 10, 1526–1531. [Google Scholar] [CrossRef]
- Vilanova, M.; Teixeira, L.; Caramalho, I.; Torrado, E.; Marques, A.; Madureira, P.; Ribeiro, A.; Ferreira, P.; Gama, M.; Demengeot, J. Protection against systemic candidiasis in mice immunized with secreted aspartic proteinase 2. Immunology 2004, 111, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Hodgetts, S.; Nooney, L.; Al-Akeel, R.; Curry, A.; Awad, S.; Matthews, R.; Burnie, J. Efungumab and Caspofungin: Pre-Clinical Data Supporting Synergy. J. Antimicrob. Chemother. 2008, 61, 1132–1139. [Google Scholar] [CrossRef]
- Dromer, F.; Charreire, J.; Contrepois, A.; Carbon, C.; Yeni, P. Protection of Mice against Experimental Cryptococcosis by Anti-Cryptococcus neoformans Monoclonal Antibody. Infect. Immun. 1987, 55, 749–752. [Google Scholar] [CrossRef]
- Larsen, R.A.; Pappas, P.G.; Perfect, J.; Aberg, J.A.; Casadevall, A.; Cloud, G.A.; James, R.; Filler, S.; Dismukes, W.E. Phase I Evaluation of the Safety and Pharmacokinetics of Murine-Derived Anticryptococcal Antibody 18B7 in Subjects with Treated Cryptococcal Meningitis. Antimicrob. Agents Chemother. 2005, 49, 952–958. [Google Scholar] [CrossRef]
- Nascimento, R.C.; Espíndola, N.M.; Castro, R.A.; Teixeira, P.A.C.; Penha, C.V.L.; Lopes-Bezerra, L.M.; Almeida, S.R. Passive Immunization with Monoclonal Antibody against a 70-KDa Putative Adhesin of Sporothrix schenckii Induces Protection in Murine Sporotrichosis. Eur. J. Immunol. 2008, 38, 3080–3089. [Google Scholar] [CrossRef]
- de Mattos Grosso, D.; de Almeida, S.R.; Mariano, M.; Lopes, J.D. Characterization of Gp70 and Anti-Gp70 Monoclonal Antibodies in Paracoccidioides brasiliensis Pathogenesis. Infect. Immun. 2003, 71, 6534–6542. [Google Scholar] [CrossRef]
- Nosanchuk, J.D.; Steenbergen, J.N.; Shi, L.; Deepe, G.S.; Casadevall, A. Antibodies to a Cell Surface Histone-like Protein Protect against Histoplasma capsulatum. J. Clin. Investig. 2003, 112, 1164–1175. [Google Scholar] [CrossRef]
- Yadav, R.K.; Shukla, P.K. A Novel Monoclonal Antibody against Enolase Antigen of Aspergillus fumigatus Protects Experimental Aspergillosis in Mice. FEMS Microbiol. Lett. 2019, 366, fnz015. [Google Scholar] [CrossRef]
- Gebremariam, T.; Alkhazraji, S.; Soliman, S.S.M.; Gu, Y.; Jeon, H.H.; Zhang, L.; French, S.W.; Stevens, D.A.; Edwards, J.E., Jr.; Filler, S.G.; et al. Anti-CotH3 antibodies protect mice from mucormycosis by prevention of invasion and augmenting opsonophagocytosis. Sci. Adv. 2019, 5, eaaw1327. [Google Scholar] [CrossRef]
- Keizer, R.J.; Huitema, A.D.R.; Schellens, J.H.M.; Beijnen, J.H. Clinical Pharmacokinetics of Therapeutic Monoclonal Antibodies. Clin. Pharmacokinet. 2010, 49, 493–507. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, S.; Barbarino, A.; Youssef, E.G.; Coleman, D.; Gebremariam, T.; Ibrahim, A.S. Protective Efficacy of Anti-Hyr1p Monoclonal Antibody against Systemic Candidiasis Due to Multi-Drug-Resistant Candida auris. J. Fungi 2023, 9, 103. https://doi.org/10.3390/jof9010103
Singh S, Barbarino A, Youssef EG, Coleman D, Gebremariam T, Ibrahim AS. Protective Efficacy of Anti-Hyr1p Monoclonal Antibody against Systemic Candidiasis Due to Multi-Drug-Resistant Candida auris. Journal of Fungi. 2023; 9(1):103. https://doi.org/10.3390/jof9010103
Chicago/Turabian StyleSingh, Shakti, Ashley Barbarino, Eman G. Youssef, Declan Coleman, Teclegiorgis Gebremariam, and Ashraf S. Ibrahim. 2023. "Protective Efficacy of Anti-Hyr1p Monoclonal Antibody against Systemic Candidiasis Due to Multi-Drug-Resistant Candida auris" Journal of Fungi 9, no. 1: 103. https://doi.org/10.3390/jof9010103
APA StyleSingh, S., Barbarino, A., Youssef, E. G., Coleman, D., Gebremariam, T., & Ibrahim, A. S. (2023). Protective Efficacy of Anti-Hyr1p Monoclonal Antibody against Systemic Candidiasis Due to Multi-Drug-Resistant Candida auris. Journal of Fungi, 9(1), 103. https://doi.org/10.3390/jof9010103









