Fusarium spp. Associated with Dendrobium officinale Dieback Disease in China
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection, Fungal Isolation, and Morphological Characterization
2.2. DNA Sequencing and Molecular Phylogeny
2.3. Pathogenicity Studies
3. Results
3.1. Field Survey, Disease Symptoms, and Pathogen Isolations
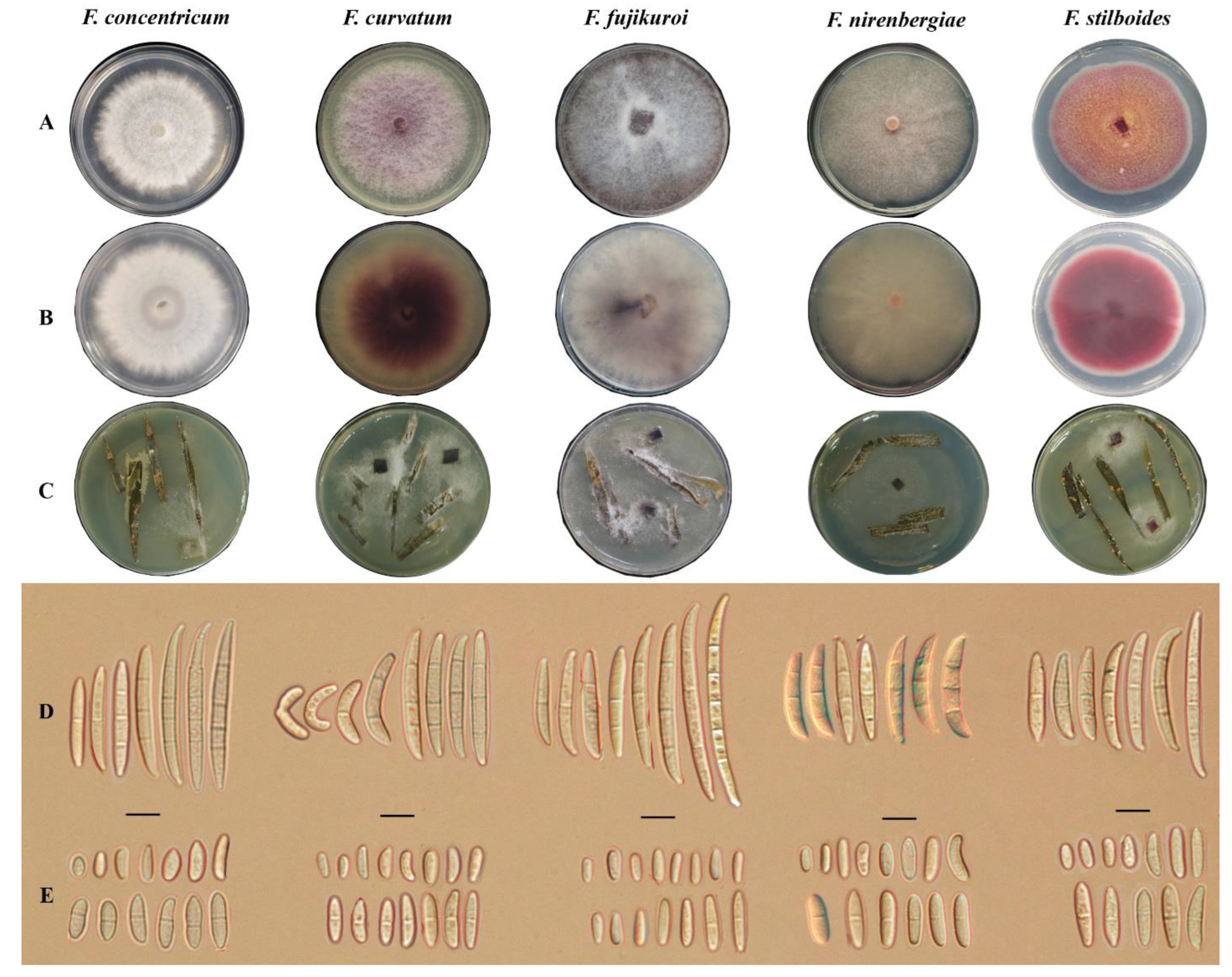
3.2. Morphological Identification
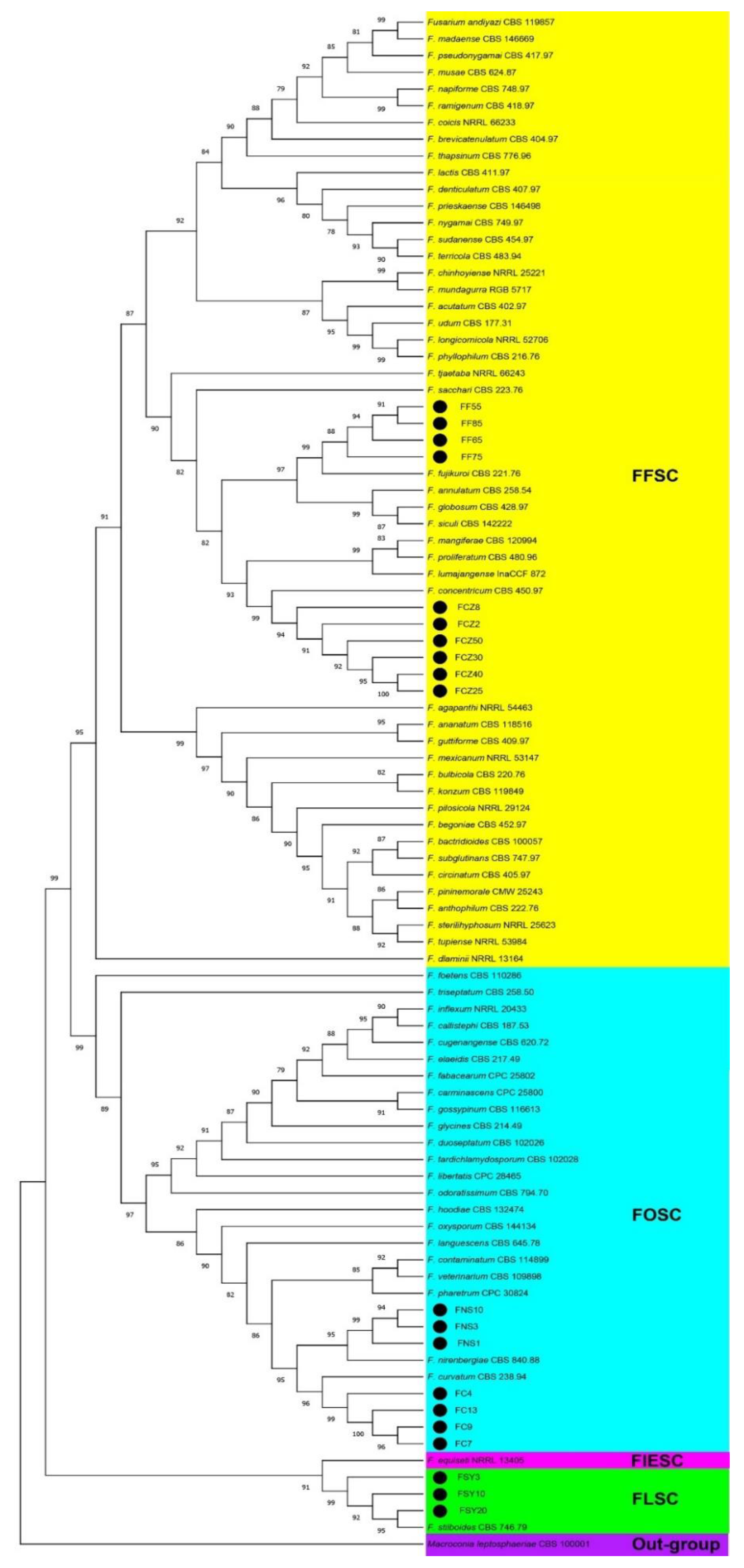
3.3. Molecular Identification and Phylogenetic Analyses
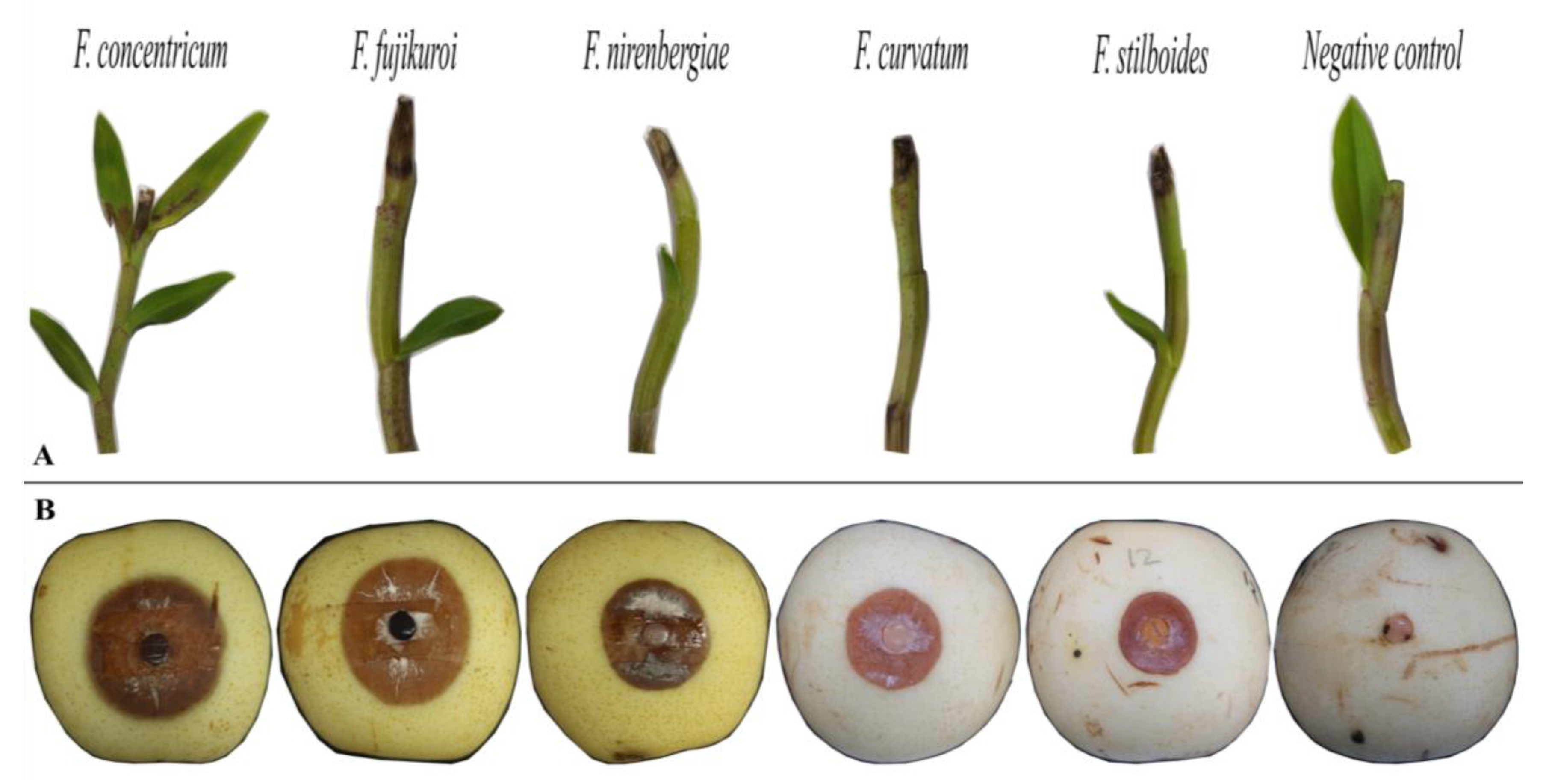
3.4. Pathogenicity Assays
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nie, S.; Cui, S.W.; Xie, M. Glucomannans from Dendrobium officinale and aloe. Bio. Poly. 2018, 6, 295–347. [Google Scholar] [CrossRef]
- Cheng, J.; Dang, P.P.; Zhao, Z.; Yuan, L.C.; Zhou, Z.H.; Wolf, D. An assessment of the Chinese medicinal Dendrobium industry: Supply, demand and sustainability. J. Ethnopharma. 2019, 229, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Sarsaiya, S.; Jain, A.; Jia, Q.; Fan, X.; Shu, F.; Chen, Z. Molecular identification of endophytic fungi and their pathogenicity evaluation against Dendrobium nobile and Dendrobium officinale. Inter. J. Mole. Sci. 2020, 21, 316. [Google Scholar] [CrossRef]
- Shan, T.; Zhou, L.; Li, B.; Chen, X.; Guo, S.; Wang, A. The plant growth-promoting fungus MF23 (Mycena sp.) increases production of Dendrobium officinale (Orchidaceae) by affecting nitrogen uptake and NH4+ assimilation. Front. Plant Sci. 2021, 12, 693561. [Google Scholar] [CrossRef] [PubMed]
- Teoh, E.S. Dwelling on rocks (Shihu). In Orchids as Aphrodisiac, Medicine or Food; Springer: Berlin/Heidelberg, Germany, 2019; Volume 4, pp. 69–86. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Lin, B.Y.; Zou, M.Y.; Liang, J.X.; Hu, H.Q. First report of Fusarium wilt of Dendrobium officinale caused by Fusarium oxysporum in China. Plant Dis. 2017, 101, 1039. [Google Scholar] [CrossRef]
- Xie, Y.Y.; Wang, L.P.; Fang, L.; Wang, H.R. First report of leaf spot caused by Phoma multirostrata var. microspora on Dendrobium officinale in Zhejiang Province of China. Plant Dis. 2018, 102, 1655. [Google Scholar] [CrossRef]
- Guo, M.; Li, B.; Wang, R.; Liu, P.; Chen, Q. Occurrence of dieback disease caused by Fusarium equiseti on Dendrobium officinale in China. Crop Prot. 2020, 137, 105209. [Google Scholar] [CrossRef]
- Xiao, C.; Li, R.; Song, X.; Tian, X.; Zhao, Q. First report of soft rot on Dendrobium officinale caused by Epicoccum sorghinum in China. Plant Dis. 2021, 1063. [Google Scholar] [CrossRef]
- Xiao, C.; Li, R. Detection and control of Fusarium oxysporum from soft rot in Dendrobium officinale by Loop-Mediated isothermal amplification assays. Biology 2021, 10, 1136. [Google Scholar] [CrossRef]
- Cao, P.; Zheng, Z.; Fang, Y.; Han, X.; Zou, H.; Yan, X. First report of stem rot caused by Fusarium kyushuense on Dendrobium officinale in China. Plant Dis. 2022, 31, 2257. [Google Scholar] [CrossRef]
- Cao, P.; Zheng, Z.; Han, X.; Zou, H.; Yan, X. Occurrence of Neopestalotiopsis clavispora causing leaf spot on Dendrobium officinale in China. Plant Dis. 2022, 12, 1761. [Google Scholar] [CrossRef] [PubMed]
- Crous, P.W.; Lombard, L.; Sandoval-Denis, M.; Seifert, K.A.; Schroers, H.J.; Chaverri, P.; Gené, J.; Guarro, J.; Hirooka, Y.; Bensch, K.; et al. Fusarium: More than a node or a foot-shaped basal cell. Stud. Mycol. 2021, 98, 100116. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S. Molecular taxonomy, diversity, and potential applications of genus Fusarium. In Industrially Important Fungi for Sustainable Development; Springer: Berlin/Heidelberg, Germany, 2021; pp. 277–293. [Google Scholar] [CrossRef]
- O’Donnell, K.; Whitaker, B.; Laraba, I.; Proctor, R.; Brown, D.; Broders, K.; Kim, H.S.; McCormick, S.P.; Busman, M.; Aoki, T.; et al. DNA sequence-based identification of Fusarium: A work in progress. Plant Dis. 2021, 106, 1597–1609. [Google Scholar] [CrossRef] [PubMed]
- Lombard, L.; Sandoval-Denis, M.; Lamprecht, S.C.; Crous, P.W. Epitypification of Fusarium oxysporum: Clearing the taxonomic chaos. Persoonia 2019, 43, 1–47. [Google Scholar] [CrossRef] [PubMed]
- Crous, P.W.; Hernández-Restrepo, M.; van Iperen, A.L.; Starink-Willemse, M.; Sandoval-Denis, M.; Groenewald, J.Z. Citizen science project reveals novel Fusarioid fungi (Nectriaceae, Sordariomycetes) from urban soils. Fungal Sys. Evol. 2021, 8, 101–127. [Google Scholar] [CrossRef] [PubMed]
- Nucci, M.; Ramos, J.F. Fusarium and Fusariosis, reference module in biomedical sciences. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar] [CrossRef]
- Yilmaz, N.; Sandoval-Denis, M.; Lombard, L.; Visagie1, C.M.; Wingfield1, B.D.; Crous, P.W. Redefining species limits in the Fusarium fujikuroi species complex. Persoonia 2021, 46, 129–162. [Google Scholar] [CrossRef] [PubMed]
- McTaggart, A.R.; James, T.Y.; Shivas, R.G.; Drenth, A.; Wingfield, B.D.; Summerell, B.A. Population genomics reveals historical and ongoing recombination in the Fusarium oxysporum species complex. Stud. Mycol. 2021, 99, 100132. [Google Scholar] [CrossRef]
- Hay, F.; Stricker, S.; Gossen, B.D.; McDonald, M.R.; Heck, D.; Hoepting, C.; Sharma, S.; Pethybridge, S. Stemphylium Leaf blight: A re-emerging threat to onion production in eastern north America. Plant Dis. 2021, 12, 3780–3794. [Google Scholar] [CrossRef]
- Riseh, R.S.; Skorik, Y.A.; Thakur, V.K.; Pour, M.M.; Tamanadar, E.; Noghabi, S.S. Encapsulation of plant biocontrol bacteria with alginate as a main polymer material. Int. J. Mol. Sci. 2021, 22, 11165. [Google Scholar] [CrossRef]
- Copes, W.E.; Ojiambo, P.S. Efficacy of hypochlorite in disinfesting nonfungal plant pathogens in agricultural and horticultural plant production: A Meta-Analysis. Plant Dis. 2021, 105, 4084–4094. [Google Scholar] [CrossRef]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Knyaz, M.; Li, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Raza, M.; Zhang, Z.F.; Hyde, K.D.; Diao, Y.Z.; Cai, L. Culturable plant pathogenic fungi associated with sugarcane in southern China. Fungal Divers. 2019, 99, 1–104. [Google Scholar] [CrossRef]
- Liu, F.; Tang, G.; Zheng, X.; Li, Y.; Sun, X.; Qi, X. Molecular and phenotypic characterization of Colletotrichum species associated with anthracnose disease in peppers from Sichuan Province, China. Sci. Rep. 2016, 6, 32761. [Google Scholar] [CrossRef] [PubMed]
- Nirenberg, H.I.; O’Donnell, K. New Fusarium species and combinations within the Gibberella fujikuroi species complex. Mycologia 1998, 90, 434–458. [Google Scholar] [CrossRef]
- Du, Q.; Duan, C.X.; Li, S.C.; Tang, Z.L.; Luo, J.Y. First report of maize ear rot caused by Fusarium concentricum in China. Plant Dis. 2020, 104, 1539. [Google Scholar] [CrossRef]
- Huda-Shakirah, A.R.; Nur-Salsabila, K.; Mohd, M.H. First report of Fusarium concentricum causing fruit blotch on roselle (Hibiscus sabdariffa). Aust. Plant Dis. 2020, 15, 15. [Google Scholar] [CrossRef]
- Qin, D.; Jiang, Y.; Zhang, R.; Ali, E.; Huo, J.; Li, Y. First report of Fusarium concentricum causing wilt on Podocarpus macrophyllus in China. Plant Dis. 2021, 18. [Google Scholar] [CrossRef]
- Dongzhen, F.; Xilin, L.; Xiaorong, C.; Wenwu, Y.; Yunlu, H.; Yi, C.; Jia, C.; Zhimin, L.; Litao, G.; Tuhong, W.; et al. Fusarium species and Fusarium oxysporum species complex genotypes associated with yam wilt in south-central China. Front. Microbiol. 2020, 11, 1964. [Google Scholar] [CrossRef]
- Zhu, Y.; Abdelraheem, A.; Wedegaertner, T.; Nichols, R.; Zhang, J.F. First report of Fusarium fujikuroi causing wilt on Pima cotton (Gossypium barbadense) seedlings in New Mexico, USA. Plant Dis. 2021, 105, 228. [Google Scholar] [CrossRef]
- Mirghasempour, S.A.; Studholme, D.J.; Chen, W.; Cui, D.; Mao, B. Identification and characterization of Fusarium nirenbergiae associated with saffron corm rot disease. Plant Dis. 2022, 106, 486–495. [Google Scholar] [CrossRef] [PubMed]
- Aiello, D.; Fiorenza, A.; Leonardi, G.R.; Vitale, A.; Polizzi, G. Fusarium nirenbergiae (Fusarium oxysporum species complex) causing the wilting of passion fruit in Italy. Plants 2021, 10, 2011. [Google Scholar] [CrossRef]
- Aiello, D.; Gusella, G.; Vitale, A.; Polizzi, G. Characterization of Fusarium nirenbergiae and F. elaeidis causing diseases on Dipladenia and Grevillea plants. Eur. J. Plant Pathol. 2022, 14, 885–896. [Google Scholar] [CrossRef]
- Mirkova, E.; Karadjova, Y. New species of Fusarium in glasshouse carnations in Bulgaria. Biotechnol. Biotechnol. Equip. 1994, 8, 8–12. [Google Scholar] [CrossRef][Green Version]
- Hurley, B.P.; Govender, P.; Coutinho, T.A.; Wingfield, B.D.; Wingfield, M.J. Fungus gnats and other Diptera in South African forestry nurseries and their possible association with the pitch canker fungus. S. Afr. J. Sci. 2007, 103, 43–46. [Google Scholar]
- Sandoval-Chávez, R.A.; Martínez-Peniche, R.Á.; Hernández-Iturriaga, M.; Fernández-Escartín, E.; Arvizu-Medrano, S.; Soto-Muñoz, L. Postharvest biological and chemical control of Fusarium stilboides on bell pepper (Capsicum annuum L.) Revista Chapingo. Serie Horticul. 2011, 17, 161–172. [Google Scholar] [CrossRef][Green Version]
- Tshilenge-Djim, P.; Muengula-Manyi, M.; Kingunza-Mawanga, K.; Ngombo-Nzokwani, A.; Tshilenge-Lukanda, L.; Kalonji-Mbuyi, A. Field assessment of th e potential role of Fusarium species in the pathogenesis of coffee wilt diseas e in democratic republic of congo. Asian Res. J. Agric. 2016, 2, 1–7. [Google Scholar] [CrossRef]
- López-Moral, A.; Lovera, M.; Raya, M.D.C.; Cortés-Cosano, N.; Arquero, O.; Trapero, A.; Agustí-Brisach, C. Etiology of branch bieback and shoot blight of english walnut caused by Botryosphaeriaceae and Diaporthe species in southern Spain. Plant Dis. 2020, 104, 533–550. [Google Scholar] [CrossRef]
- Guo, Z.; Yu, Z.; Li, Q.; Tang, L.; Guo, T.; Huang, S.; Mo, J.; Hsiang, T.; Luo, S. Fusarium species associated with leaf spots of mango in China. Microb. Pathog. 2021, 150, 104736. [Google Scholar] [CrossRef]
- Wang, W.; Wang, B.; Sun, X.; Qi, X.; Zhao, C.; Chang, X. Symptoms and pathogens diversity of corn Fusarium sheath rot in Sichuan Province, China. Sci. Rep. 2021, 11, 2835. [Google Scholar] [CrossRef]
- Yu, J.; Babadoost, M. Occurrence of Fusarium commune and F. oxysporum in horseradish roots. Plant Dis. 2013, 97, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Asselin, J.; Bonasera, J.M.; Beer, S.V. PCR primers for detection of Pantoea ananatis, Burkholderia spp., and Enterobacter sp. from onion. Plant Dis. 2016, 100, 836–846. [Google Scholar] [CrossRef] [PubMed]
- Swett, C.L.; Science, P.; Architecture, L.; Park, C.; Sharon, C. Evidence for a hemibiotrophic association of the pitch canker pathogen Fusarium circinatum with Pinus radiata. Plant Dis. 2016, 100, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Degani, O.; Movshowitz, D.; Dor, S.; Meerson, A.; Goldblat, Y.; Rabinovitz, O. Evaluating azoxystrobin seed coating against maize late wilt disease using a sensitive qPCR-based method. Plant Dis. 2019, 103, 238–248. [Google Scholar] [CrossRef]
- Mayorquin, J.S.; Nouri, M.T.; Peacock, B.B.; Trouillas, F.P.; Douhan, G.W.; Kallsen, C. Identification, pathogenicity, and spore trapping of Colletotrichum karstii associated with twig and shoot dieback in California. Plant Dis. 2019, 103, 1464–1473. [Google Scholar] [CrossRef] [PubMed]
- Bock, C.H.; Barbedo, J.G.A.; Del Ponte, E.M.; Bohnenkamp, D.; Mahlein, A.K. From visual estimates to fully automated sensor-based measurements of plant disease severity: Status and challenges for improving accuracy. Phytopathol. Res. 2020, 2, 9. [Google Scholar] [CrossRef]

| Species | Culture Collection No./Isolate | GenBank Accession | ||
|---|---|---|---|---|
| rpb2 | tef1 | tub1 | ||
| Fusarium carminascens | CPC 144738 T | MH484937 | MH485028 | MH485119 |
| F. contaminatum | CBS 111552 T | MH484901 | MH484992 | MH485083 |
| F. pharetrum | CBS 144751 T | MH484952 | MH485043 | MH485134 |
| F. veterinarium | CBS 109898 T | MH484899 | MH484990 | MH485081 |
| F. cugenangense | CBS 620.72 | MH484879 | MH484970 | MH485061 |
| F. curvatum | CBS 238.94 T | MH484893 | MH484984 | MH485075 |
| FC4 | ON137565 | ON137544 | ON137586 | |
| FC6 | ON137566 | ON137545 | ON137587 | |
| FC7 | ON137567 | ON137546 | ON137588 | |
| FC9 | ON137568 | ON137547 | ON137589 | |
| FC13 | ON137568 | ON137548 | ON137590 | |
| F. fabacearum | CPC 25802 T | MH484939 | MH485030 | MH485121 |
| F. glycines | CBS 144746 T | MH484942 | MH485033 | MH485124 |
| F. gossypinum | CBS 116613 T | MH484909 | MH485000 | MH485091 |
| F. languescens | CBS 645.78 T | MH484880 | MH484971 | MH485062 |
| F. libertatis | CPC 28465 T | MH484944 | MH485035 | MH485126 |
| F. nirenbergiae | CBS 840.88 T | MH484887 | MH484978 | MH485069 |
| FNS1 | ON137574 | ON137553 | ON137595 | |
| FNS3 | ON137575 | ON137553 | ON137596 | |
| FNS10 | ON137576 | ON137553 | ON137597 | |
| F. oxysporum | CBS 144134 ET | MH484953 | MH485044 | MH485135 |
| F. hoodiae | CBS 132474 T | MH484929 | MH485020 | MH485111 |
| F. duoseptatum | CBS 102026 T | MH484896 | MH484987 | MH485078 |
| F. callistephi | CBS 187.53 T | MH484875 | MH484966 | MH485057 |
| F. triseptatum | CBS 258.50 T | MH484910 | MH485001 | MH485055 |
| F. languescens | CBS 645.78 T | MH484880 | MH484971 | MH485062 |
| F. elaeidis | CBS 217.49 | MH484870 | MH484961 | MH485052 |
| F.acutatum | CBS 402.97 T | MW402768 | MW402125 | MW402323 |
| F. agapanthi | NRRL 54463 T | KU900625 | KU900630 | KU900635 |
| F. ananatum | CBS 118516 T | LT996137 | LT996091 | MN534089 |
| F. andiyazi | CBS 119857 T | LT996138 | MN193854 | LT996113 |
| F. annulatum | CBS 258.54 T | MT010983 | MT010994 | MT011041 |
| F. anthophilum | CBS 222.76 ET | MW402811 | MW402114 | MW402312 |
| F.bactridioides | CBS 100057 T | MN534235 | MN533993 | MN534112 |
| F. begoniae | CBS 452.97 T | MN534243 | MN533994 | MN534101 |
| F. brevicatenulatum | CBS 404.97 T | MN534295 | MN533995 | MN534063 |
| F. bulbicola | CBS 220.76 T | MW402767 | KF466415 | KF466437 |
| F. chinhoyiense | NRRL 25221 T | MN534262 | MN534050 | MN534082 |
| F. subglutinans | CBS 747.97 NT | MW402773 | MW402150 | MW402351 |
| F. circinatum | CBS 405.97 T | MN534252 | MN533997 | MN534097 |
| F. coicis | NRRL 66233 T | KP083274 | KP083251 | LT996115 |
| F. concentricum | CBS 450.97 T | JF741086 | AF160282 | MW402334 |
| FCZ2 | ON137559 | ON107278 | ON137580 | |
| FCZ8 | ON137560 | ON107279 | ON137581 | |
| FCZ25 | ON137561 | ON107280 | ON137582 | |
| FCZ30 | ON137562 | ON107281 | ON137583 | |
| FCZ40 | ON137563 | ON107282 | ON137584 | |
| FCZ50 | ON137564 | ON107283 | ON137585 | |
| F. globosum | CBS 428.97 T | KF466406 | KF466417 | MN534124 |
| F. guttiforme | CBS 409.97 T | MT010967 | MT010999 | MT011048 |
| F. konzum | CBS 119849 T | MW402733 | LT996098 | MN534095 |
| F. lactis | CBS 411.97 ET | MN534275 | MN193862 | MN534077 |
| F. longicornicola | NRRL 52706 T | JF741114 | JF740788 | MW402360 |
| F. denticulatum | CBS 407.97 T | MN534274 | MN534000 | MN534068 |
| F. dlaminii | CBS 119860 T | KU171701 | MW401995 | MW402195 |
| F. fujikuroi | CBS 221.76 T | KU604255 | MN534010 | MN534130 |
| FF55 | ON137570 | ON137549 | ON137591 | |
| FF65 | ON137571 | ON137550 | ON137592 | |
| FF75 | ON137572 | ON137551 | ON137593 | |
| FF85 | ON137573 | ON137552 | ON137594 | |
| F.madaense | CBS 146669 T | MW402764 | MW402098 | MW402297 |
| F.mangiferae | CBS 120994 T | MN534271 | MN534017 | MN534128 |
| F.mexicanum | NRRL 53147 T | MN724973 | GU737282 | GU737494 |
| F.mundagurra | RGB5717 T | KP083276 | KP083256 | MN534146 |
| F.musae | CBS 624.87 T | MW402772 | FN552086 | FN545368 |
| F. napiforme | CBS 748.97 T | MN534291 | MN193863 | MN534085 |
| F. nygamai | CBS 749.97 T | EF470114 | MW402151 | MW402352 |
| F. phyllophilum | CBS 216.76 T | KF466410 | MN193864 | KF466443 |
| F. pilosicola | NRRL 29124 T | MN534248 | MN534055 | MN534099 |
| F. proliferatum | CBS 480.96 ET | MN534272 | MN534059 | MN534129 |
| F. pseudonygamai | CBS 417.97 T | MN534285 | AF160263 | MN534066 |
| F. ramigenum | CBS 418.97 T | KF466412 | KF466423 | MN534145 |
| F. sacchari | CBS 223.76 ET | JX171580 | MW402115 | MW402313 |
| F.siculi | CBS 142222 T | LT746327 | LT746214 | LT746346 |
| F.succisae | CBS 219.76 ET | MW402766 | AF160291 | U34419 |
| F.sudanense | CBS 454.97 T | MN534278 | MN534037 | MN534073 |
| F.terricola | CBS 483.94 T | LT996156 | MN534042 | MN534076 |
| F.thapsinum | CBS 776.96 T | MN534289 | MN534044 | MN534080 |
| F.tjaetaba | NRRL 66243 T | KP083275 | KP083263 | GU737296 |
| F. tupiense | NRRL 53984 | LR792619 | GU737404 | GU737296 |
| F. prieskaense | CBS 146498 T | MW834006 | MW834274 | MW834302 |
| F. foetens | CBS 110286 T | MW928825 | MT011001 | MT011049 |
| F. hostae | NRRL 29889 T | MT409446 | MT409456 | AY329042 |
| F. udum | NRRL 25199 ET | KY498875 | KY498862 | KY498892 |
| F. stilboides | CBS 746.79 T | MW928832 | MW928843 | - |
| FSY3 | ON137577 | ON137556 | ON137598 | |
| FSY10 | ON137578 | ON137557 | ON137599 | |
| FSY20 | ON137579 | ON137558 | ON137600 | |
| F. buharicum | NRRL 25488 | KX302928 | KX302912 | - |
| F. equiseti | NRRL 26419 | - | GQ505599 | - |
| NRRL 20697 | - | GQ505594 | - | |
| NRRL 13405 T | GQ915491 | GQ915507 | GQ915441 | |
| DFS | - | MN823983 | - | |
| F.peruvianum | CBS 511.75 | - | MN120767 | - |
| F. sarcochroum | CBS 745.79 | JX171586 | MW834278 | - |
| F. inflexum | NRRL 20433 | JX171583 | AF008479 | U34435 |
| F. sublunatum | NRRL 20897 | KX302935 | KX302919 | - |
| F. convolutans | CBS 144207 T | LT996141 | LT996094 | - |
| F. sarcochroum | CBS 745.79 | - | MW834278 | - |
| Macroconia leptosphaeriae | CBS 100001 | HQ728164 | KM231959 | KM232097 |
| Geographic Origin | Species | No. Isolate | Isolation Frequency (%) |
|---|---|---|---|
| Zhejiang Province (Yueqing City) | F. concentricum | 53 | 34.6 |
| F. curvatum | 34 | 22.3 | |
| F. fujikuroi | 28 | 18.4 | |
| F. nirenbergiae | 21 | 13.8 | |
| F. stilboides | 16 | 10.5 | |
| Total | 152 | - |
| Species | Mean Lesion Size (mm) ± SD |
|---|---|
| F. concentricum | 45.30 a ± 3.19 |
| F. fujikuroi | 41.83 b ± 1.84 |
| F. nirenbergiae | 39.85 c ± 1.64 |
| F. curvatum | 34.8 d ± 1.66 |
| F. stilboides | 28.92 e ± 2.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mirghasempour, S.A.; Michailides, T.; Chen, W.; Mao, B. Fusarium spp. Associated with Dendrobium officinale Dieback Disease in China. J. Fungi 2022, 8, 919. https://doi.org/10.3390/jof8090919
Mirghasempour SA, Michailides T, Chen W, Mao B. Fusarium spp. Associated with Dendrobium officinale Dieback Disease in China. Journal of Fungi. 2022; 8(9):919. https://doi.org/10.3390/jof8090919
Chicago/Turabian StyleMirghasempour, Seyed Ali, Themis Michailides, Weiliang Chen, and Bizeng Mao. 2022. "Fusarium spp. Associated with Dendrobium officinale Dieback Disease in China" Journal of Fungi 8, no. 9: 919. https://doi.org/10.3390/jof8090919
APA StyleMirghasempour, S. A., Michailides, T., Chen, W., & Mao, B. (2022). Fusarium spp. Associated with Dendrobium officinale Dieback Disease in China. Journal of Fungi, 8(9), 919. https://doi.org/10.3390/jof8090919







