Pharmacodynamics, Mechanisms of Action and Resistance, and Spectrum of Activity of New Antifungal Agents
Abstract
1. Introduction
2. Antifungals with Novel Mechanisms of Action
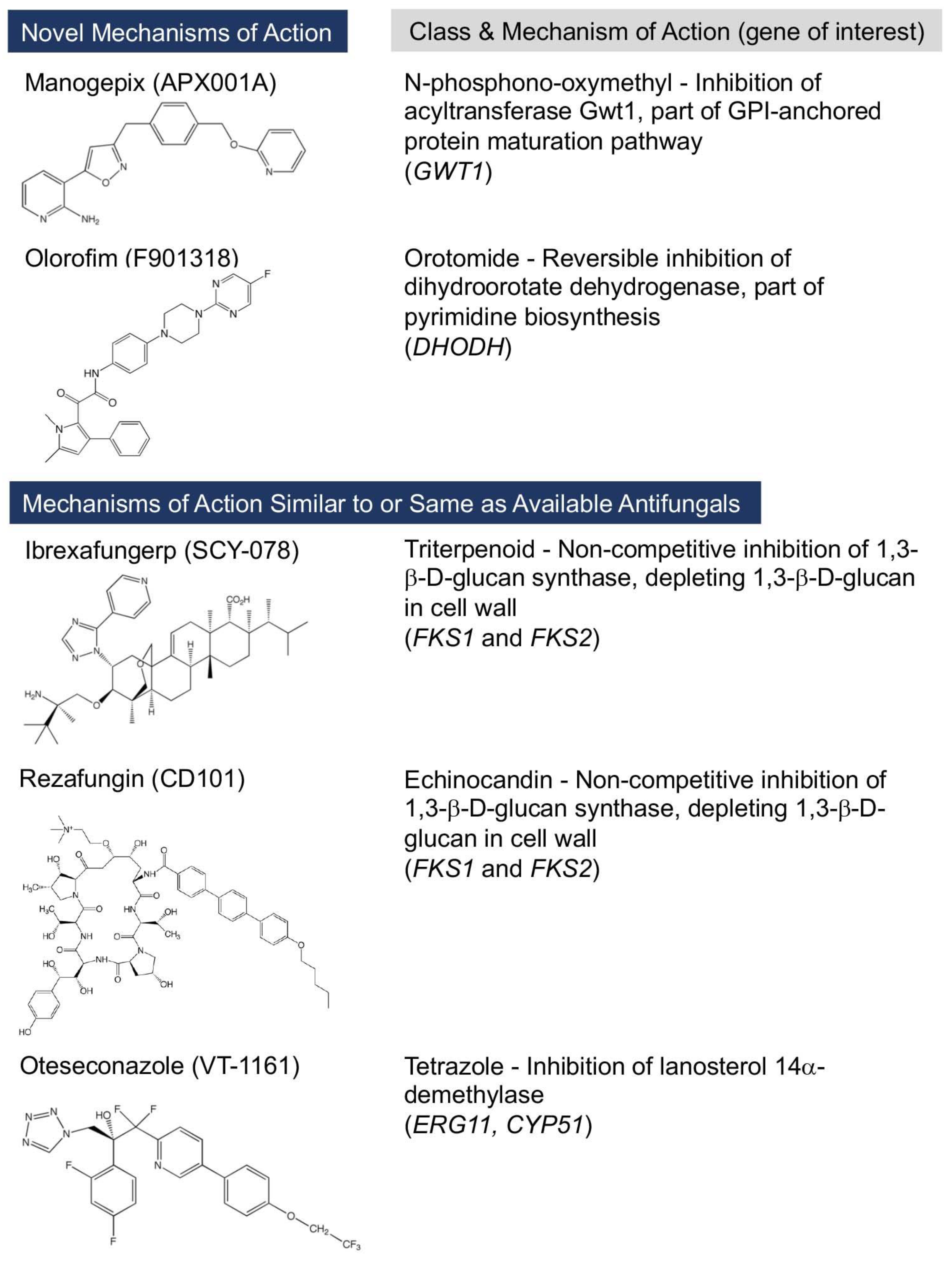
2.1. Manogepix
2.1.1. Mechanism of Action—Pharmacodynamics
2.1.2. Spectrum of Activity and Resistance
2.1.3. In Vivo Efficacy and Pharmacokinetics/Pharmacodynamics
2.1.4. Tolerability and Drug Interactions
2.2. Olorofim
2.2.1. Mechanism of Action—Pharmacodynamics
2.2.2. Spectrum of Activity and Mechanisms of Resistance
2.2.3. In Vivo Efficacy and Pharmacokinetics/Pharmacodynamics
2.2.4. Tolerability and Drug Interactions
2.3. Other Antifungals with Novel Mechanisms of Action
3. New Antifungals That Improve upon Current Classes and Mechanisms of Action
3.1. Ibrexafungerp
3.1.1. Mechanism of Action—Pharmacodynamics
3.1.2. Spectrum of Activity and Mechanisms of Resistance
3.1.3. In Vivo Efficacy and Pharmacokinetics/Pharmacodynamics
3.1.4. Tolerability and Drug Interactions
3.2. Rezafungin
3.2.1. Mechanism of Action—Pharmacodynamics
3.2.2. Spectrum of Activity and Mechanisms of Resistance
3.2.3. In Vivo Efficacy and Pharmacokinetics/Pharmacodynamics
3.2.4. Tolerability and Drug Interactions
3.3. Oteseconazole
3.3.1. Mechanism of Action—Pharmacodynamics
3.3.2. Spectrum of Activity and Mechanisms of Resistance
3.3.3. In Vivo Efficacy and Pharmacokinetics/Pharmacodynamics
3.3.4. Tolerability and Drug Interactions
4. New Routes of Administration
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lewis, R.E. Current concepts in antifungal pharmacology. Mayo Clin. Proc. 2011, 86, 805–817. [Google Scholar] [CrossRef] [PubMed]
- Ashley, E.S.D.; Lewis, R.; Lewis, J.S.; Martin, C.; Andes, D. Pharmacology of systemic antifungal agents. Clin. Infect. Dis. 2006, 43, S28–S29. [Google Scholar] [CrossRef]
- Arastehfar, A.; Gabaldon, T.; Garcia-Rubio, R.; Jenks, J.D.; Hoenigl, M.; Salzer, H.J.F.; Ilkit, M.; Lass-Florl, C.; Perlin, D.S. Drug-Resistant Fungi: An Emerging Challenge Threatening Our Limited Antifungal Armamentarium. Antibiotics 2020, 9, 877. [Google Scholar] [CrossRef] [PubMed]
- Hoenigl, M.; Sprute, R.; Arastehfar, A.; Perfect, J.R.; Lass-Florl, C.; Bellmann, R.; Prattes, J.; Thompson, G.R., 3rd; Wiederhold, N.P.; Al Obaidi, M.M.; et al. Invasive candidiasis: Investigational drugs in the clinical development pipeline and mechanisms of action. Expert Opin. Investig. Drugs 2022, 31, 795–812. [Google Scholar] [CrossRef] [PubMed]
- Hoenigl, M.; Sprute, R.; Egger, M.; Arastehfar, A.; Cornely, O.A.; Krause, R.; Lass-Florl, C.; Prattes, J.; Spec, A.; Thompson, G.R., 3rd; et al. The Antifungal Pipeline: Fosmanogepix, Ibrexafungerp, Olorofim, Opelconazole, and Rezafungin. Drugs 2021, 81, 1703–1729. [Google Scholar] [CrossRef] [PubMed]
- CLSI. Performance Standards for Antifungal Susceptibility Testing of Filamentous Fungi, 2nd ed.; CLSI Supplement M61; CLSI: Wayne, PA, USA, 2020. [Google Scholar]
- CLSI. Performance Standards for Antifungal Susceptibility Testing of Yeasts, 2nd ed.; CLSI Supplement M60; CLSI: Wayne, PA, USA, 2020. [Google Scholar]
- Miyazaki, M.; Horii, T.; Hata, K.; Watanabe, N.A.; Nakamoto, K.; Tanaka, K.; Shirotori, S.; Murai, N.; Inoue, S.; Matsukura, M.; et al. In vitro activity of E1210, a novel antifungal, against clinically important yeasts and molds. Antimicrob. Agents Chemother. 2011, 55, 4652–4658. [Google Scholar] [CrossRef] [PubMed]
- Tsukahara, K.; Hata, K.; Nakamoto, K.; Sagane, K.; Watanabe, N.A.; Kuromitsu, J.; Kai, J.; Tsuchiya, M.; Ohba, F.; Jigami, Y.; et al. Medicinal genetics approach towards identifying the molecular target of a novel inhibitor of fungal cell wall assembly. Mol. Microbiol. 2003, 48, 1029–1042. [Google Scholar] [CrossRef]
- Chaffin, W.L. Candida albicans cell wall proteins. Microbiol. Mol. Biol. Rev. MMBR 2008, 72, 495–544. [Google Scholar] [CrossRef]
- Fu, Y.; Luo, G.; Spellberg, B.J.; Edwards, J.E., Jr.; Ibrahim, A.S. Gene overexpression/suppression analysis of candidate virulence factors of Candida albicans. Eukaryot. Cell 2008, 7, 483–492. [Google Scholar] [CrossRef]
- Hoyer, L.L. The ALS gene family of Candida albicans. Trends Microbiol. 2001, 9, 176–180. [Google Scholar] [CrossRef]
- Kapteyn, J.C.; Hoyer, L.L.; Hecht, J.E.; Muller, W.H.; Andel, A.; Verkleij, A.J.; Makarow, M.; Van Den Ende, H.; Klis, F.M. The cell wall architecture of Candida albicans wild-type cells and cell wall-defective mutants. Mol. Microbiol. 2000, 35, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, D.C.; Yeaman, M.R.; Welch, W.H.; Phan, Q.T.; Fu, Y.; Ibrahim, A.S.; Filler, S.G.; Zhang, M.; Waring, A.J.; Edwards, J.E., Jr. Functional and structural diversity in the Als protein family of Candida albicans. J. Biol. Chem. 2004, 279, 30480–30489. [Google Scholar] [CrossRef] [PubMed]
- Shaw, K.J.; Ibrahim, A.S. Fosmanogepix: A Review of the First-in-Class Broad Spectrum Agent for the Treatment of Invasive Fungal Infections. J. Fungi 2020, 6, 239. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Jorgensen, K.M. Manogepix (APX001A) Displays Potent In Vitro Activity against Human Pathogenic Yeast, but with an Unexpected Correlation to Fluconazole MICs. Antimicrob. Agents Chemother. 2020, 64, e00429-20. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Huband, M.D.; Flamm, R.K.; Bien, P.A.; Castanheira, M. Antimicrobial activity of manogepix, a first-in-class antifungal, and comparator agents tested against contemporary invasive fungal isolates from an international surveillance programme (2018–2019). J. Glob. Antimicrob. Resist. 2021, 26, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Chowdhary, A.; Jorgensen, K.M.; Meletiadis, J. Manogepix (APX001A) In Vitro Activity against Candida auris: Head-to-Head Comparison of EUCAST and CLSI MICs. Antimicrob. Agents Chemother. 2020, 64, e00656-20. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Hata, K.; Jones, R.N.; Messer, S.A.; Moet, G.J.; Castanheira, M. In vitro activity of a novel broad-spectrum antifungal, E1210, tested against Candida spp. as determined by CLSI broth microdilution method. Diagn. Microbiol. Infect. Dis. 2011, 71, 167–170. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Shaw, K.J.; Jaramillo, R.; Patterson, H.; Olivo, M.; Catano, G.; Patterson, T.F. Efficacy of Delayed Therapy with Fosmanogepix (APX001) in a Murine Model of Candida auris Invasive Candidiasis. Antimicrob. Agents Chemother. 2019, 63, e01120-19. [Google Scholar] [CrossRef]
- Shaw, K.J.; Schell, W.A.; Covel, J.; Duboc, G.; Giamberardino, C.; Kapoor, M.; Moloney, M.; Soltow, Q.A.; Tenor, J.L.; Toffaletti, D.L.; et al. In Vitro and In Vivo Evaluation of APX001A/APX001 and Other Gwt1 Inhibitors against Cryptococcus. Antimicrob. Agents Chemother. 2018, 62, e00523-18. [Google Scholar] [CrossRef]
- Hager, C.L.; Larkin, E.L.; Long, L.; Zohra Abidi, F.; Shaw, K.J.; Ghannoum, M.A. In Vitro and In Vivo Evaluation of the Antifungal Activity of APX001A/APX001 against Candida auris. Antimicrob. Agents Chemother. 2018, 62, e02319-17. [Google Scholar] [CrossRef]
- Zhao, M.; Lepak, A.J.; VanScoy, B.; Bader, J.C.; Marchillo, K.; Vanhecker, J.; Ambrose, P.G.; Andes, D.R. In Vivo Pharmacokinetics and Pharmacodynamics of APX001 against Candida spp. in a Neutropenic Disseminated Candidiasis Mouse Model. Antimicrob. Agents Chemother. 2018, 62, e02542-17. [Google Scholar] [CrossRef] [PubMed]
- Alkhazraji, S.; Gebremariam, T.; Alqarihi, A.; Gu, Y.; Mamouei, Z.; Singh, S.; Wiederhold, N.P.; Shaw, K.J.; Ibrahim, A.S. Fosmanogepix (APX001) Is Effective in the Treatment of Immunocompromised Mice Infected with Invasive Pulmonary Scedosporiosis or Disseminated Fusariosis. Antimicrob. Agents Chemother. 2020, 64, e01735-19. [Google Scholar] [CrossRef] [PubMed]
- Castanheira, M.; Duncanson, F.P.; Diekema, D.J.; Guarro, J.; Jones, R.N.; Pfaller, M.A. Activities of E1210 and comparator agents tested by CLSI and EUCAST broth microdilution methods against Fusarium and Scedosporium species identified using molecular methods. Antimicrob. Agents Chemother. 2012, 56, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Gebremariam, T.; Alkhazraji, S.; Alqarihi, A.; Jeon, H.H.; Gu, Y.; Kapoor, M.; Shaw, K.J.; Ibrahim, A.S. APX001 Is Effective in the Treatment of Murine Invasive Pulmonary Aspergillosis. Antimicrob. Agents Chemother. 2019, 63, e01713-18. [Google Scholar] [CrossRef]
- Hata, K.; Horii, T.; Miyazaki, M.; Watanabe, N.A.; Okubo, M.; Sonoda, J.; Nakamoto, K.; Tanaka, K.; Shirotori, S.; Murai, N.; et al. Efficacy of oral E1210, a new broad-spectrum antifungal with a novel mechanism of action, in murine models of candidiasis, aspergillosis, and fusariosis. Antimicrob. Agents Chemother. 2011, 55, 4543–4551. [Google Scholar] [CrossRef]
- Jorgensen, K.M.; Astvad, K.M.T.; Arendrup, M.C. In Vitro Activity of Manogepix (APX001A) and Comparators against Contemporary Molds: MEC Comparison and Preliminary Experience with Colorimetric MIC Determination. Antimicrob. Agents Chemother. 2020, 64, e00730-20. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Duncanson, F.; Messer, S.A.; Moet, G.J.; Jones, R.N.; Castanheira, M. In vitro activity of a novel broad-spectrum antifungal, E1210, tested against Aspergillus spp. determined by CLSI and EUCAST broth microdilution methods. Antimicrob. Agents Chemother. 2011, 55, 5155–5158. [Google Scholar] [CrossRef]
- Arikan, S.; Lozano-Chiu, M.; Paetznick, V.; Rex, J.H. In vitro susceptibility testing methods for caspofungin against Aspergillus and Fusarium isolates. Antimicrob. Agents Chemother. 2001, 45, 327–330. [Google Scholar] [CrossRef]
- Gebremariam, T.; Alkhazraji, S.; Alqarihi, A.; Wiederhold, N.P.; Shaw, K.J.; Patterson, T.F.; Filler, S.G.; Ibrahim, A.S. Fosmanogepix (APX001) Is Effective in the Treatment of Pulmonary Murine Mucormycosis Due to Rhizopus arrhizus. Antimicrob. Agents Chemother. 2020, 64, e00178-20. [Google Scholar] [CrossRef]
- Gebremariam, T.; Gu, Y.; Alkhazraji, S.; Youssef, E.; Shaw, K.J.; Ibrahim, A.S. The Combination Treatment of Fosmanogepix and Liposomal Amphotericin B Is Superior to Monotherapy in Treating Experimental Invasive Mold Infections. Antimicrob. Agents Chemother. 2022, 66, e0038022. [Google Scholar] [CrossRef]
- Kapoor, M.; Moloney, M.; Soltow, Q.A.; Pillar, C.M.; Shaw, K.J. Evaluation of Resistance Development to the Gwt1 Inhibitor Manogepix (APX001A) in Candida Species. Antimicrob. Agents Chemother. 2019, 64, e01387-19. [Google Scholar] [CrossRef] [PubMed]
- Liston, S.D.; Whitesell, L.; Kapoor, M.; Shaw, K.J.; Cowen, L.E. Enhanced Efflux Pump Expression in Candida Mutants Results in Decreased Manogepix Susceptibility. Antimicrob. Agents Chemother. 2020, 64, e00261-20. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Lepak, A.J.; Marchillo, K.; Vanhecker, J.; Sanchez, H.; Ambrose, P.G.; Andes, D.R. APX001 Pharmacokinetic/Pharmacodynamic Target Determination against Aspergillus fumigatus in an In Vivo Model of Invasive Pulmonary Aspergillosis. Antimicrob. Agents Chemother. 2019, 63, e02372-18. [Google Scholar] [CrossRef] [PubMed]
- Viriyakosol, S.; Kapoor, M.; Okamoto, S.; Covel, J.; Soltow, Q.A.; Trzoss, M.; Shaw, K.J.; Fierer, J. APX001 and Other Gwt1 Inhibitor Prodrugs Are Effective in Experimental Coccidioides immitis Pneumonia. Antimicrob. Agents Chemother. 2019, 63, e01715-18. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Lee, M.H.; Paderu, P.; Lee, A.; Jimenez-Ortigosa, C.; Park, S.; Mansbach, R.S.; Shaw, K.J.; Perlin, D.S. Significantly Improved Pharmacokinetics Enhances In Vivo Efficacy of APX001 against Echinocandin- and Multidrug-Resistant Candida Isolates in a Mouse Model of Invasive Candidiasis. Antimicrob. Agents Chemother. 2018, 62, e00425-18. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.A.; Miyazaki, M.; Horii, T.; Sagane, K.; Tsukahara, K.; Hata, K. E1210, a new broad-spectrum antifungal, suppresses Candida albicans hyphal growth through inhibition of glycosylphosphatidylinositol biosynthesis. Antimicrob. Agents Chemother. 2012, 56, 960–971. [Google Scholar] [CrossRef] [PubMed]
- Hodges, M.R.; Ople, E.; Shaw, K.J.; Mansbach, R.; Van Marle, S.J.; Van Hoogdalem, E.; Kramer, W.; Wedel, P. Phase 1 Study to Assess Safety, Tolerability and Pharmacokinetics of Single and Multiple Oral Doses of APX001 and to Investigate the Effect of Food on APX001 Bioavailability. Open Forum Infect. Dis. 2017, 4, S534. [Google Scholar] [CrossRef]
- Hodges, M.R.; Ople, E.; Shaw, K.J.; Mansbach, R.; Van Marle, S.J.; Van Hoogdalem, E.; Wedel, P.; Kramer, W. First-in-Human Study to Assess Safety, Tolerability and Pharmacokinetics of APX001 Administered by Intravenous Infusion to Healthy Subjects. Open Forum Infect. Dis. 2017, 4, S526. [Google Scholar] [CrossRef]
- Pappas, P.G.; Kullberg, B.J.; Vazquez, J.A.; Oren, I.; Rahav, G.; Aoun, M.; Bulpa, P.; Ben-Ami, R.; Ferrer, R.; McCarty, T.; et al. Clinical Safety and Efficacy of Novel Antifungal, Fosmanogepix, in the Treatment of Candidemia: Results from a Phase 2 Proof of Concept Trial. Presented at the Programs and Abstracts of the 55th Annual Infectious Diseases Society of America (IDSA) Meeting, Philadelphia, PA, USA, 21–25 October 2020. IDWeek 2020. [Google Scholar]
- Bulpa, P.; Rahav, G.; Oren, I.; Aoun, M.; Thompson, G.R., 3rd; Pappas, P.G.; Kullberg, B.J.; Vazquez, J.A.; Barbat, S.H.; Wedel, P.; et al. Clinical Safety and Efficacy of Fosmanogepix, a Novel First-in-class Antifungal, in Patients with Renal Insufficiency: Subset Analysis from a Phase 2 Candidemia Trial. In Proceedings of the Programs and Abstracts of the 55th Annual Infectious Diseases Society of America (IDSA) Meeting, Philadelphia, PA, USA, 21–25 October 2020. IDWeek 2020. [Google Scholar]
- Balani, S.K.; Zhu, T.; Yang, T.J.; Liu, Z.; He, B.; Lee, F.W. Effective dosing regimen of 1-aminobenzotriazole for inhibition of antipyrine clearance in rats, dogs, and monkeys. Drug Metab. Dispos. 2002, 30, 1059–1062. [Google Scholar] [CrossRef]
- Oliver, J.D.; Sibley, G.E.; Beckmann, N.; Dobb, K.S.; Slater, M.J.; McEntee, L.; du Pre, S.; Livermore, J.; Bromley, M.J.; Wiederhold, N.P.; et al. F901318 represents a novel class of antifungal drug that inhibits dihydroorotate dehydrogenase. Proc. Natl. Acad. Sci. USA 2016, 113, 12809–12814. [Google Scholar] [CrossRef]
- Gow, N.A.R.; Latge, J.P.; Munro, C.A. The Fungal Cell Wall: Structure, Biosynthesis, and Function. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- du Pre, S.; Beckmann, N.; Almeida, M.C.; Sibley, G.E.M.; Law, D.; Brand, A.C.; Birch, M.; Read, N.D.; Oliver, J.D. Effect of the Novel Antifungal Drug F901318 (Olorofim) on Growth and Viability of Aspergillus fumigatus. Antimicrob. Agents Chemother. 2018, 62, e00231-18. [Google Scholar] [CrossRef] [PubMed]
- du Pre, S.; Birch, M.; Law, D.; Beckmann, N.; Sibley, G.E.M.; Bromley, M.J.; Read, N.D.; Oliver, J.D. The Dynamic Influence of Olorofim (F901318) on the Cell Morphology and Organization of Living Cells of Aspergillus fumigatus. J. Fungi 2020, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Wiederhold, N.P. Review of the Novel Investigational Antifungal Olorofim. J. Fungi 2020, 6, 122. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Law, D.; Birch, M. Dihydroorotate dehydrogenase inhibitor F901318 has potent in vitro activity against Scedosporium species and Lomentospora prolificans. J. Antimicrob. Chemother. 2017, 72, 1977–1980. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Jaramillo, R.; Olivo, M.; Birch, M.; Law, D.; Rex, J.H.; Catano, G.; Patterson, T.F. The Orotomide Olorofim Is Efficacious in an Experimental Model of Central Nervous System Coccidioidomycosis. Antimicrob. Agents Chemother. 2018, 62, e00999-18. [Google Scholar] [CrossRef]
- Biswas, C.; Law, D.; Birch, M.; Halliday, C.; Sorrell, T.C.; Rex, J.; Slavin, M.; Chen, S.C. In vitro activity of the novel antifungal compound F901318 against Australian Scedosporium and Lomentospora fungi. Med. Mycol. 2018, 56, 1050–1054. [Google Scholar] [CrossRef]
- Jorgensen, K.M.; Astvad, K.M.T.; Hare, R.K.; Arendrup, M.C. EUCAST Determination of Olorofim (F901318) Susceptibility of Mold Species, Method Validation, and MICs. Antimicrob. Agents Chemother. 2018, 62, e00487-18. [Google Scholar] [CrossRef]
- Rivero-Menendez, O.; Cuenca-Estrella, M.; Alastruey-Izquierdo, A. In vitro activity of olorofim (F901318) against clinical isolates of cryptic species of Aspergillus by EUCAST and CLSI methodologies. J. Antimicrob. Chemother. 2019, 74, 1586–1590. [Google Scholar] [CrossRef]
- Seyedmousavi, S.; Chang, Y.C.; Law, D.; Birch, M.; Rex, J.H.; Kwon-Chung, K.J. Efficacy of Olorofim (F901318) against Aspergillus fumigatus, A. nidulans, and A. tanneri in Murine Models of Profound Neutropenia and Chronic Granulomatous Disease. Antimicrob. Agents Chemother. 2019, 63, e00129-19. [Google Scholar] [CrossRef]
- Buil, J.B.; Rijs, A.; Meis, J.F.; Birch, M.; Law, D.; Melchers, W.J.G.; Verweij, P.E. In vitro activity of the novel antifungal compound F901318 against difficult-to-treat Aspergillus isolates. J. Antimicrob. Chemother. 2017, 72, 2548–2552. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.; Eadie, K.; Konings, M.; Rijnders, B.; Fahal, A.H.; Oliver, J.D.; Birch, M.; Verbon, A.; van de Sande, W. Madurella mycetomatis, the main causative agent of eumycetoma, is highly susceptible to olorofim. J. Antimicrob. Chemother. 2020, 75, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Buil, J.B.; Oliver, J.D.; Law, D.; Baltussen, T.; Zoll, J.; Hokken, M.W.J.; Tehupeiory-Kooreman, M.; Melchers, W.J.G.; Birch, M.; Verweij, P.E. Resistance profiling of Aspergillus fumigatus to olorofim indicates absence of intrinsic resistance and unveils the molecular mechanisms of acquired olorofim resistance. Emerg. Microbes Infect. 2022, 11, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Hope, W.W.; McEntee, L.; Livermore, J.; Whalley, S.; Johnson, A.; Farrington, N.; Kolamunnage-Dona, R.; Schwartz, J.; Kennedy, A.; Law, D.; et al. Pharmacodynamics of the Orotomides against Aspergillus fumigatus: New Opportunities for Treatment of Multidrug-Resistant Fungal Disease. mBio 2017, 8, e01157-17. [Google Scholar] [CrossRef]
- Negri, C.E.; Johnson, A.; McEntee, L.; Box, H.; Whalley, S.; Schwartz, J.A.; Ramos-Martin, V.; Livermore, J.; Kolamunnage-Dona, R.; Colombo, A.L.; et al. Pharmacodynamics of the Novel Antifungal Agent F901318 for Acute Sinopulmonary Aspergillosis Caused by Aspergillus flavus. J. Infect. Dis. 2018, 217, 1118–1127. [Google Scholar] [CrossRef]
- Seyedmousavi, S.; Chang, Y.C.; Youn, J.H.; Law, D.; Birch, M.; Rex, J.H.; Kwon-Chung, K.J. In Vivo Efficacy of Olorofim against Systemic Scedosporiosis and Lomentosporiosis. Antimicrob. Agents Chemother. 2021, 65, e0043421. [Google Scholar] [CrossRef]
- Kennedy, T.; Allen, G.; Steiner, J.; Heep, M.; Birch, M. Assessment of the duration of infusion on the tolerability and repeat dose pharmacokinetics of F901318 in healthy volunteers (abstr. P1711). In Proceedings of the 27th European Congress of Clinical Microbiology and Infectious Diseases, Vienna, Austria, 22–25 April 2017. [Google Scholar]
- Kennedy, T.; Allen, G.; Steiner, J.; Heep, M.; Oliver, J.; Sibley, G.; Law, D. Multiple dose pharmacokinetics of an immediate-release tablet formulation of F901318 in healthy male and female subjects (abstr. P1710). In Proceedings of the 27th European Congress of Clinical Microbiology and Infectious Diseases, Vienna, Austria, 22–25 April 2017. [Google Scholar]
- Rauseo, A.M.; Coler-Reilly, A.; Larson, L.; Spec, A. Hope on the Horizon: Novel Fungal Treatments in Development. Open Forum Infect. Dis. 2020, 7, ofaa016. [Google Scholar] [CrossRef]
- Kennedy, T.; Allen, G.; Steiner, J.; Oliver, J.; Birch, M.; Sibley, G.; Law, D. An open-label study in healthy volunteers to evaluate the potential for cytochrome P450 3A4 inhibition by F901318 using oral midazolam as a probe (abstr. P1737). In Proceedings of the 27th European Congress of Clinical Microbiology and Infectious Diseases, Vienna, Austria, 22–25 April 2017. [Google Scholar]
- Mitsuyama, J.; Nomura, N.; Hashimoto, K.; Yamada, E.; Nishikawa, H.; Kaeriyama, M.; Kimura, A.; Todo, Y.; Narita, H. In vitro and in vivo antifungal activities of T-2307, a novel arylamidine. Antimicrob. Agents Chemother. 2008, 52, 1318–1324. [Google Scholar] [CrossRef]
- Lanteri, C.A.; Trumpower, B.L.; Tidwell, R.R.; Meshnick, S.R. DB75, a novel trypanocidal agent, disrupts mitochondrial function in Saccharomyces cerevisiae. Antimicrob. Agents Chemother. 2004, 48, 3968–3974. [Google Scholar] [CrossRef]
- Yamada, E.; Nishikawa, H.; Nomura, N.; Mitsuyama, J. T-2307 shows efficacy in a murine model of Candida glabrata infection despite in vitro trailing growth phenomena. Antimicrob. Agents Chemother. 2010, 54, 3630–3634. [Google Scholar] [CrossRef]
- Yamashita, K.; Miyazaki, T.; Fukuda, Y.; Mitsuyama, J.; Saijo, T.; Shimamura, S.; Yamamoto, K.; Imamura, Y.; Izumikawa, K.; Yanagihara, K.; et al. The Novel Arylamidine T-2307 Selectively Disrupts Yeast Mitochondrial Function by Inhibiting Respiratory Chain Complexes. Antimicrob. Agents Chemother. 2019, 63, e00374-19. [Google Scholar] [CrossRef] [PubMed]
- Wiederhold, N.P.; Najvar, L.K.; Jaramillo, R.; Olivo, M.; Patterson, H.; Connell, A.; Fukuda, Y.; Mitsuyama, J.; Catano, G.; Patterson, T.F. The Novel Arylamidine T-2307 Demonstrates In Vitro and In Vivo Activity against Candida auris. Antimicrob. Agents Chemother. 2020, 64, e02198-19. [Google Scholar] [CrossRef] [PubMed]
- Wiederhold, N.P.; Najvar, L.K.; Fothergill, A.W.; Bocanegra, R.; Olivo, M.; McCarthy, D.I.; Fukuda, Y.; Mitsuyama, J.; Patterson, T.F. The novel arylamidine T-2307 demonstrates in vitro and in vivo activity against echinocandin-resistant Candida glabrata. J. Antimicrob. Chemother. 2016, 71, 692–695. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Fothergill, A.W.; Bocanegra, R.; Olivo, M.; McCarthy, D.I.; Kirkpatrick, W.R.; Fukuda, Y.; Mitsuyama, J.; Patterson, T.F. The novel arylamidine T-2307 maintains in vitro and in vivo activity against echinocandin-resistant Candida albicans. Antimicrob. Agents Chemother. 2015, 59, 1341–1343. [Google Scholar] [CrossRef]
- Nishikawa, H.; Fukuda, Y.; Mitsuyama, J.; Tashiro, M.; Tanaka, A.; Takazono, T.; Saijo, T.; Yamamoto, K.; Nakamura, S.; Imamura, Y.; et al. In vitro and in vivo antifungal activities of T-2307, a novel arylamidine, against Cryptococcus gattii: An emerging fungal pathogen. J. Antimicrob. Chemother. 2017, 72, 1709–1713. [Google Scholar] [CrossRef] [PubMed]
- Abe, M.; Nakamura, S.; Kinjo, Y.; Masuyama, Y.; Mitsuyama, J.; Kaku, M.; Miyazaki, Y. Efficacy of T-2307, a novel arylamidine, against ocular complications of disseminated candidiasis in mice. J. Antimicrob. Chemother. 2019, 74, 1327–1332. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Bocanegra, R.; Kirkpatrick, W.R.; Patterson, T.F. Comparison of anidulafungin’s and fluconazole’s in vivo activity in neutropenic and non-neutropenic models of invasive candidiasis. Clin. Microbiol. Infect. 2012, 18, E20–E23. [Google Scholar] [CrossRef][Green Version]
- Nakamura, I.; Yoshimura, S.; Masaki, T.; Takase, S.; Ohsumi, K.; Hashimoto, M.; Furukawa, S.; Fujie, A. ASP2397: A novel antifungal agent produced by Acremonium persicinum MF-347833. J. Antibiot. 2017, 70, 45–51. [Google Scholar] [CrossRef]
- Nakamura, I.; Ohsumi, K.; Takeda, S.; Katsumata, K.; Matsumoto, S.; Akamatsu, S.; Mitori, H.; Nakai, T. ASP2397 Is a Novel Natural Compound That Exhibits Rapid and Potent Fungicidal Activity against Aspergillus Species through a Specific Transporter. Antimicrob. Agents Chemother. 2019, 63, e02689-18. [Google Scholar] [CrossRef]
- Dietl, A.M.; Misslinger, M.; Aguiar, M.M.; Ivashov, V.; Teis, D.; Pfister, J.; Decristoforo, C.; Hermann, M.; Sullivan, S.M.; Smith, L.R.; et al. The Siderophore Transporter Sit1 Determines Susceptibility to the Antifungal VL-2397. Antimicrob. Agents Chemother. 2019, 63, e00807-19. [Google Scholar] [CrossRef]
- Wiederhold, N.P. The antifungal arsenal: Alternative drugs and future targets. Int. J. Antimicrob. Agents 2018, 51, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Jensen, R.H.; Cuenca-Estrella, M. In Vitro Activity of ASP2397 against Aspergillus Isolates with or without Acquired Azole Resistance Mechanisms. Antimicrob. Agents Chemother. 2016, 60, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Rubino, C.M.; Smith, L.R.; Mammen, M.P.; Hopkins, A.M.; Lakota, E.A.; Sullivan, S.M. Pharmacokinetic-Pharmacodynamic Target Attainment Analysis to Support VL-2397 Dose Selection for a Phase 2 Trial in Patients with Invasive Aspergillosis. Open Forum Infect. Dis. 2017, 4, S473. [Google Scholar] [CrossRef][Green Version]
- Pfaller, M.A.; Messer, S.A.; Motyl, M.R.; Jones, R.N.; Castanheira, M. Activity of MK-3118, a new oral glucan synthase inhibitor, tested against Candida spp. by two international methods (CLSI and EUCAST). J. Antimicrob. Chemother. 2013, 68, 858–863. [Google Scholar] [CrossRef]
- Bowman, J.C.; Hicks, P.S.; Kurtz, M.B.; Rosen, H.; Schmatz, D.M.; Liberator, P.A.; Douglas, C.M. The antifungal echinocandin caspofungin acetate kills growing cells of Aspergillus fumigatus in vitro. Antimicrob. Agents Chemother. 2002, 46, 3001–3012. [Google Scholar] [CrossRef]
- Ghannoum, M.; Long, L.; Larkin, E.L.; Isham, N.; Sherif, R.; Borroto-Esoda, K.; Barat, S.; Angulo, D. Evaluation of the Antifungal Activity of the Novel Oral Glucan Synthase Inhibitor SCY-078, Singly and in Combination, for the Treatment of Invasive Aspergillosis. Antimicrob. Agents Chemother. 2018, 62, e00244-18. [Google Scholar] [CrossRef]
- Douglas, C.M.; D’Ippolito, J.A.; Shei, G.J.; Meinz, M.; Onishi, J.; Marrinan, J.A.; Li, W.; Abruzzo, G.K.; Flattery, A.; Bartizal, K.; et al. Identification of the FKS1 gene of Candida albicans as the essential target of 1,3-beta-D-glucan synthase inhibitors. Antimicrob. Agents Chemother. 1997, 41, 2471–2479. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Messer, S.A.; Motyl, M.R.; Jones, R.N.; Castanheira, M. In vitro activity of a new oral glucan synthase inhibitor (MK-3118) tested against Aspergillus spp. by CLSI and EUCAST broth microdilution methods. Antimicrob. Agents Chemother. 2013, 57, 1065–1068. [Google Scholar] [CrossRef]
- Walker, S.S.; Xu, Y.; Triantafyllou, I.; Waldman, M.F.; Mendrick, C.; Brown, N.; Mann, P.; Chau, A.; Patel, R.; Bauman, N.; et al. Discovery of a novel class of orally active antifungal beta-1,3-D-glucan synthase inhibitors. Antimicrob. Agents Chemother. 2011, 55, 5099–5106. [Google Scholar] [CrossRef]
- Jiménez-Ortigosa, C.; Perez, W.B.; Angulo, D.; Borroto-Esoda, K.; Perlin, D.S. De Novo Acquisition of Resistance to SCY-078 in Candida glabrata Involves FKS Mutations That both Overlap and Are Distinct from Those Conferring Echinocandin Resistance. Antimicrob. Agents Chemother. 2017, 61, e00833-17. [Google Scholar] [CrossRef]
- Schell, W.A.; Jones, A.M.; Borroto-Esoda, K.; Alexander, B.D. Antifungal Activity of SCY-078 and Standard Antifungal Agents against 178 Clinical Isolates of Resistant and Susceptible Candida Species. Antimicrob. Agents Chemother. 2017, 61, e01102-17. [Google Scholar] [CrossRef]
- Larkin, E.; Hager, C.; Chandra, J.; Mukherjee, P.K.; Retuerto, M.; Salem, I.; Long, L.; Isham, N.; Kovanda, L.; Borroto-Esoda, K.; et al. The Emerging Pathogen Candida auris: Growth Phenotype, Virulence Factors, Activity of Antifungals, and Effect of SCY-078, a Novel Glucan Synthesis Inhibitor, on Growth Morphology and Biofilm Formation. Antimicrob. Agents Chemother. 2017, 61, e02396-16. [Google Scholar] [CrossRef]
- Berkow, E.L.; Angulo, D.; Lockhart, S.R. In Vitro activity of a novel glucan synthase inhibitor, SCY-078, against clinical isolates of Candida auris. Antimicrob. Agents Chemother. 2017, 61, e00435-17. [Google Scholar] [CrossRef] [PubMed]
- Larkin, E.L.; Long, L.; Isham, N.; Borroto-Esoda, K.; Barat, S.; Angulo, D.; Wring, S.; Ghannoum, M. A Novel 1,3-Beta-d-Glucan Inhibitor, Ibrexafungerp (Formerly SCY-078), Shows Potent Activity in the Lower pH Environment of Vulvovaginitis. Antimicrob. Agents Chemother. 2019, 63, e02611-18. [Google Scholar] [CrossRef] [PubMed]
- Danby, C.S.; Boikov, D.; Rautemaa-Richardson, R.; Sobel, J.D. Effect of pH on in vitro susceptibility of Candida glabrata and Candida albicans to 11 antifungal agents and implications for clinical use. Antimicrob. Agents Chemother. 2012, 56, 1403–1406. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, M.; Wiederhold, N.P. Reduced Antifungal Susceptibility of Vulvovaginal Candida Species at Normal Vaginal pH Levels: Clinical Implications. J. Low. Genit. Tract Dis. 2018, 22, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Rivero-Menendez, O.; Soto-Debran, J.C.; Cuenca-Estrella, M.; Alastruey-Izquierdo, A. In Vitro Activity of Ibrexafungerp against a Collection of Clinical Isolates of Aspergillus, Including Cryptic Species and Cyp51A Mutants, Using EUCAST and CLSI Methodologies. J. Fungi 2021, 7, 232. [Google Scholar] [CrossRef]
- Jiménez-Ortigosa, C.; Paderu, P.; Motyl, M.R.; Perlin, D.S. Enfumafungin derivative MK-3118 shows increased in vitro potency against clinical echinocandin-resistant Candida Species and Aspergillus species isolates. Antimicrob. Agents Chemother. 2014, 58, 1248–1251. [Google Scholar] [CrossRef]
- Petraitis, V.; Petraitiene, R.; Katragkou, A.; Maung, B.B.W.; Naing, E.; Kavaliauskas, P.; Barat, S.; Borroto-Esoda, K.; Azie, N.; Angulo, D.; et al. Combination Therapy with Ibrexafungerp (Formerly SCY-078), a First-in-Class Triterpenoid Inhibitor of (1—>3)-beta-d-Glucan Synthesis, and Isavuconazole for Treatment of Experimental Invasive Pulmonary Aspergillosis. Antimicrob. Agents Chemother. 2020, 64, e02429-19. [Google Scholar] [CrossRef]
- Lamoth, F.; Alexander, B.D. Antifungal activities of SCY-078 (MK-3118) and standard antifungal agents against clinical non-Aspergillus mold isolates. Antimicrob. Agents Chemother. 2015, 59, 4308–4311. [Google Scholar] [CrossRef]
- Park, S.; Kelly, R.; Kahn, J.N.; Robles, J.; Hsu, M.J.; Register, E.; Li, W.; Vyas, V.; Fan, H.; Abruzzo, G.; et al. Specific substitutions in the echinocandin target Fks1p account for reduced susceptibility of rare laboratory and clinical Candida sp. isolates. Antimicrob. Agents Chemother. 2005, 49, 3264–3273. [Google Scholar] [CrossRef] [PubMed]
- Nunnally, N.S.; Etienne, K.A.; Angulo, D.; Lockhart, S.R.; Berkow, E.L. In Vitro Activity of Ibrexafungerp, a Novel Glucan Synthase Inhibitor against Candida glabrata Isolates with FKS Mutations. Antimicrob. Agents Chemother. 2019, 63, e01692-19. [Google Scholar] [CrossRef] [PubMed]
- Jallow, S.; Govender, N.P. Ibrexafungerp: A First-in-Class Oral Triterpenoid Glucan Synthase Inhibitor. J. Fungi 2021, 7, 163. [Google Scholar] [CrossRef] [PubMed]
- Wiederhold, N.P.; Najvar, L.K.; Jaramillo, R.; Olivo, M.; Pizzini, J.; Catano, G.; Patterson, T.F. Oral glucan synthase inhibitor SCY-078 is effective in an experimental murine model of invasive candidiasis caused by WT and echinocandin-resistant Candida glabrata. J. Antimicrob. Chemother. 2018, 73, 448–451. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.C.; Barat, S.A.; Borroto-Esoda, K.; Angulo, D.; Chaturvedi, S.; Chaturvedi, V. Pan-resistant Candida auris isolates from the outbreak in New York are susceptible to ibrexafungerp (a glucan synthase inhibitor). Int. J. Antimicrob. Agents 2020, 55, 105922. [Google Scholar] [CrossRef]
- Ghannoum, M.; Long, L.; Isham, N.; Hager, C.; Wilson, R.; Borroto-Esoda, K.; Barat, S.; Angulo, D. Activity of a novel 1,3-beta-D-glucan Synthase Inhibitor, Ibrexafungerp (formerly SCY-078), Against Candida glabrata. Antimicrob. Agents Chemother. 2019, 63, e01510-19. [Google Scholar] [CrossRef]
- Marcos-Zambrano, L.J.; Gomez-Perosanz, M.; Escribano, P.; Bouza, E.; Guinea, J. The novel oral glucan synthase inhibitor SCY-078 shows in vitro activity against sessile and planktonic Candida spp. J. Antimicrob. Chemother. 2017, 72, 1969–1976. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Messer, S.A.; Rhomberg, P.R.; Borroto-Esoda, K.; Castanheira, M. Differential Activity of the Oral Glucan Synthase Inhibitor SCY-078 against Wild-Type and Echinocandin-Resistant Strains of Candida Species. Antimicrob. Agents Chemother. 2017, 61, e00161-17. [Google Scholar] [CrossRef]
- Mesquida, A.; Diaz-Garcia, J.; Sanchez-Carrillo, C.; Martin-Rabadan, P.; Alcala, L.; Munoz, P.; Escribano, P.; Guinea, J. ΔF659 and F659S substitutions at the HS1 of FKS2 gene, along with E655A and W715L upstream and downstream substitutions, correlate with high ibrexafungerp MICs against Candida glabrata. Clin. Microbiol. Infect. 2022, 28, 1154.e5–1154.e8. [Google Scholar] [CrossRef]
- Lepak, A.J.; Marchillo, K.; Andes, D.R. Pharmacodynamic target evaluation of a novel oral glucan synthase inhibitor, SCY-078 (MK-3118), using an in vivo murine invasive candidiasis model. Antimicrob. Agents Chemother. 2015, 59, 1265–1272. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Olivo, M.; Morris, K.N.; Patterson, H.P.; Catano, G.; Patterson, T.F. Ibrexafungerp Demonstrates In Vitro Activity against Fluconazole-Resistant Candida auris and In Vivo Efficacy with Delayed Initiation of Therapy in an Experimental Model of Invasive Candidiasis. Antimicrob. Agents Chemother. 2021, 65, e02694-20. [Google Scholar] [CrossRef] [PubMed]
- Barat, S.A.; Borroto-Esoda, K.; Angulo, D.; Ashbaugh, A.; Cushion, M. Efficacy of Ibrexafungerp (formerly SCY-078) in a Murine Treatment Model of Pneumocystis Pneumonia. In Proceedings of the ASM Microbe, San Francisco, CA, USA, 20–24 June 2019. [Google Scholar]
- Wring, S.A.; Randolph, R.; Park, S.; Abruzzo, G.; Chen, Q.; Flattery, A.; Garrett, G.; Peel, M.; Outcalt, R.; Powell, K.; et al. Preclinical Pharmacokinetics and Pharmacodynamic Target of SCY-078, a First-in-Class Orally Active Antifungal Glucan Synthesis Inhibitor, in Murine Models of Disseminated Candidiasis. Antimicrob. Agents Chemother. 2017, 61, e02068-16. [Google Scholar] [CrossRef] [PubMed]
- Andes, D.; Diekema, D.J.; Pfaller, M.A.; Bohrmuller, J.; Marchillo, K.; Lepak, A. In vivo comparison of the pharmacodynamic targets for echinocandin drugs against Candida species. Antimicrob. Agents Chemother. 2010, 54, 2497–2506. [Google Scholar] [CrossRef]
- Davis, M.R.; Donnelley, M.A.; Thompson, G.R. Ibrexafungerp: A novel oral glucan synthase inhibitor. Med. Mycol. 2020, 58, 579–592. [Google Scholar] [CrossRef]
- Trucksis, M.; Garrett, G.; Heriman, I. A phase I multiple rising dose study evaluating safety, tolerability, and pharmacokinetics of MK-3118, oral glucan synthase inhibitor, in healthy volunteers. In Proceedings of the 51st Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC), Chicago, IL, USA, 17–20 September 2011. [Google Scholar]
- Alexander, B.D.; Cornely, O.; Pappas, P.; Miller, R.; Vazquez, J.A.; Ostrosky-Zeichner, L.; Spec, A.; Rautemaa-Richardson, R.; Krause, R.; Thompson III, G.R.; et al. 1248. Efficacy and Safety of Oral Ibrexafungerp in 41 Patients with Refractory Fungal Diseases, Interim Analysis of a Phase 3 Open-label Study (FURI). Open Forum Infect. Dis. 2020, 7, S642–S643. [Google Scholar] [CrossRef]
- Spec, A.; Pullman, J.; Thompson, G.R.; Powderly, W.G.; Tobin, E.H.; Vazquez, J.; Wring, S.A.; Angulo, D.; Helou, S.; Pappas, P.G.; et al. MSG-10: A Phase 2 study of oral ibrexafungerp (SCY-078) following initial echinocandin therapy in non-neutropenic patients with invasive candidiasis. J. Antimicrob. Chemother. 2019, 74, 3056–3062. [Google Scholar] [CrossRef]
- Wring, S.; Murphy, G.; Atiee, G.; Corr, C.; Hyman, M.; Willett, M.; Angulo, D. Lack of Impact by SCY-078, a First-in-Class Oral Fungicidal Glucan Synthase Inhibitor, on the Pharmacokinetics of Rosiglitazone, a Substrate for CYP450 2C8, Supports the Low Risk for Clinically Relevant Metabolic Drug-Drug Interactions. J. Clin. Pharmacol. 2018, 58, 1305–1313. [Google Scholar] [CrossRef]
- Garcia-Effron, G. Rezafungin-Mechanisms of Action, Susceptibility and Resistance: Similarities and Differences with the Other Echinocandins. J. Fungi 2020, 6, 262. [Google Scholar] [CrossRef]
- James, K.D.; Laudeman, C.P.; Malkar, N.B.; Krishnan, R.; Polowy, K. Structure-Activity Relationships of a Series of Echinocandins and the Discovery of CD101, a Highly Stable and Soluble Echinocandin with Distinctive Pharmacokinetic Properties. Antimicrob. Agents Chemother. 2017, 61, e01541-16. [Google Scholar] [CrossRef]
- Krishnan, B.R.; James, K.D.; Polowy, K.; Bryant, B.J.; Vaidya, A.; Smith, S.; Laudeman, C.P. CD101, a novel echinocandin with exceptional stability properties and enhanced aqueous solubility. J. Antibiot. 2017, 70, 130–135. [Google Scholar] [CrossRef]
- Ong, V.; James, K.D.; Smith, S.; Krishnan, B.R. Pharmacokinetics of the Novel Echinocandin CD101 in Multiple Animal Species. Antimicrob. Agents Chemother. 2017, 61, e01626-16. [Google Scholar] [CrossRef] [PubMed]
- Sandison, T.; Ong, V.; Lee, J.; Thye, D. Safety and Pharmacokinetics of CD101 IV, a Novel Echinocandin, in Healthy Adults. Antimicrob. Agents Chemother. 2017, 61, e01627-16. [Google Scholar] [CrossRef] [PubMed]
- Krause, D.S.; Reinhardt, J.; Vazquez, J.A.; Reboli, A.; Goldstein, B.P.; Wible, M.; Henkel, T.; Anidulafungin Invasive Candidiasis Study Group. Phase 2, randomized, dose-ranging study evaluating the safety and efficacy of anidulafungin in invasive candidiasis and candidemia. Antimicrob. Agents Chemother. 2004, 48, 2021–2024. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Messer, S.A.; Rhomberg, P.R.; Jones, R.N.; Castanheira, M. Activity of a long-acting echinocandin, CD101, determined using CLSI and EUCAST reference methods, against Candida and Aspergillus spp., including echinocandin- and azole-resistant isolates. J. Antimicrob. Chemother. 2016, 71, 2868–2873. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Messer, S.A.; Rhomberg, P.R.; Castanheira, M. CD101, a long-acting echinocandin, and comparator antifungal agents tested against a global collection of invasive fungal isolates in the SENTRY 2015 Antifungal Surveillance Program. Int. J. Antimicrob. Agents 2017, 50, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Meletiadis, J.; Zaragoza, O.; Jørgensen, K.M.; Marcos-Zambrano, L.J.; Kanioura, L.; Cuenca-Estrella, M.; Mouton, J.W.; Guinea, J. Multicentre determination of rezafungin (CD101) susceptibility of Candida species by the EUCAST method. Clin. Microbiol. Infect. 2018, 24, 1200–1204. [Google Scholar] [CrossRef] [PubMed]
- Tóth, Z.; Forgács, L.; Locke, J.B.; Kardos, G.; Nagy, F.; Kovács, R.; Szekely, A.; Borman, A.M.; Majoros, L. In vitro activity of rezafungin against common and rare Candida species and Saccharomyces cerevisiae. J. Antimicrob. Chemother. 2019, 74, 3505–3510. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Locke, J.B.; Daruwala, P.; Bartizal, K. Rezafungin (CD101) demonstrates potent in vitro activity against Aspergillus, including azole-resistant Aspergillus fumigatus isolates and cryptic species. J. Antimicrob. Chemother. 2018, 73, 3063–3067. [Google Scholar] [CrossRef]
- Dudiuk, C.; Macedo, D.; Leonardelli, F.; Theill, L.; Cabeza, M.S.; Gamarra, S.; Garcia-Effron, G. Molecular Confirmation of the Relationship between Candida guilliermondii Fks1p Naturally Occurring Amino Acid Substitutions and Its Intrinsic Reduced Echinocandin Susceptibility. Antimicrob. Agents Chemother. 2017, 61, e02644-16. [Google Scholar] [CrossRef]
- Garcia-Effron, G.; Katiyar, S.K.; Park, S.; Edlind, T.D.; Perlin, D.S. A naturally occurring proline-to-alanine amino acid change in Fks1p in Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis accounts for reduced echinocandin susceptibility. Antimicrob. Agents Chemother. 2008, 52, 2305–2312. [Google Scholar] [CrossRef]
- Helleberg, M.; Jørgensen, K.M.; Hare, R.K.; Datcu, R.; Chowdhary, A.; Arendrup, M.C. Rezafungin In Vitro Activity against Contemporary Nordic Clinical Candida Isolates and Candida auris Determined by the EUCAST Reference Method. Antimicrob. Agents Chemother. 2020, 64, e02438-19. [Google Scholar] [CrossRef] [PubMed]
- Lepak, A.J.; Zhao, M.; Andes, D.R. Pharmacodynamic Evaluation of Rezafungin (CD101) against Candida auris in the Neutropenic Mouse Invasive Candidiasis Model. Antimicrob. Agents Chemother. 2018, 62, e01572-18. [Google Scholar] [CrossRef] [PubMed]
- Lepak, A.J.; Zhao, M.; Andes, D.R. Determination of Pharmacodynamic Target Exposures for Rezafungin against Candida tropicalis and Candida dubliniensis in the Neutropenic Mouse Disseminated Candidiasis Model. Antimicrob. Agents Chemother. 2019, 63, e01556-19. [Google Scholar] [CrossRef] [PubMed]
- Hager, C.L.; Larkin, E.L.; Long, L.A.; Ghannoum, M.A. Evaluation of the efficacy of rezafungin, a novel echinocandin, in the treatment of disseminated Candida auris infection using an immunocompromised mouse model. J. Antimicrob. Chemother. 2018, 73, 2085–2088. [Google Scholar] [CrossRef]
- Zhao, Y.; Prideaux, B.; Nagasaki, Y.; Lee, M.H.; Chen, P.Y.; Blanc, L.; Ho, H.; Clancy, C.J.; Nguyen, M.H.; Dartois, V.; et al. Unraveling Drug Penetration of Echinocandin Antifungals at the Site of Infection in an Intra-abdominal Abscess Model. Antimicrob. Agents Chemother. 2017, 61, e01009-17. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Perez, W.B.; Jimenez-Ortigosa, C.; Hough, G.; Locke, J.B.; Ong, V.; Bartizal, K.; Perlin, D.S. CD101: A novel long-acting echinocandin. Cell. Microbiol. 2016, 18, 1308–1316. [Google Scholar] [CrossRef]
- Long, L.; Herrada, J.; Caley, D.; Munguba, G.; Sherif, R.; Bartizal, K.; Ghannoum, M.A. Evaluation of the efficacy of rezafungin in the treatment of Candida albicans endophthalmitis using a rabbit model. In Proceedings of the European Congress of Clinical Microbilogy and Infectious Diseases, Paris, France, 18–21 April 2020. [Google Scholar]
- Miesel, L.; Lin, K.Y.; Ong, V. Rezafungin treatment in mouse models of invasive candidiasis and aspergillosis: Insights on the PK/PD pharmacometrics of rezafungin efficacy. Pharmacol. Res. Perspect. 2019, 7, e00546. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Jaramillo, R.; Olivo, M.; Wickes, B.L.; Catano, G.; Patterson, T.F. Extended-Interval Dosing of Rezafungin against Azole-Resistant Aspergillus fumigatus. Antimicrob. Agents Chemother. 2019, 63, e01165-19. [Google Scholar] [CrossRef]
- Cushion, M.T.; Ashbaugh, A. The Long-Acting Echinocandin, Rezafungin, Prevents Pneumocystis Pneumonia and Eliminates Pneumocystis from the Lungs in Prophylaxis and Murine Treatment Models. J. Fungi 2021, 7, 747. [Google Scholar] [CrossRef]
- Lakota, E.A.; Bader, J.C.; Ong, V.; Bartizal, K.; Miesel, L.; Andes, D.R.; Bhavnani, S.M.; Rubino, C.M.; Ambrose, P.G.; Lepak, A.J. Pharmacological Basis of CD101 Efficacy: Exposure Shape Matters. Antimicrob. Agents Chemother. 2017, 61, e00758-17. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Kontoyiannis, D.P.; Chi, J.; Prince, R.A.; Tam, V.H.; Lewis, R.E. Pharmacodynamics of caspofungin in a murine model of invasive pulmonary aspergillosis: Evidence of concentration-dependent activity. J. Infect. Dis. 2004, 190, 1464–1471. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, S.; Goodman, D.B.; Jandourek, A.; O’Reilly, T.; Sandison, T. Lack of Effect of Rezafungin on QT/QTc Interval in Healthy Subjects. Clin. Pharmacol. Drug Dev. 2020, 9, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Thompson, G.R.; Soriano, A.; Skoutelis, A.; Vazquez, J.A.; Honore, P.M.; Horcajada, J.P.; Spapen, H.; Bassetti, M.; Ostrosky-Zeichner, L.; Das, A.F.; et al. Rezafungin versus Caspofungin in a Phase 2, Randomized, Double-Blind Study for the Treatment of Candidemia and Invasive Candidiasis-The STRIVE Trial. Clin. Infect. Dis. 2021, 73, 561–562. [Google Scholar] [CrossRef] [PubMed]
- Ong, V.; Hough, G.; Schlosser, M.; Bartizal, K.; Balkovec, J.M.; James, K.D.; Krishnan, B.R. Preclinical Evaluation of the Stability, Safety, and Efficacy of CD101, a Novel Echinocandin. Antimicrob. Agents Chemother. 2016, 60, 6872–6879. [Google Scholar] [CrossRef] [PubMed]
- Hoekstra, W.J.; Garvey, E.P.; Moore, W.R.; Rafferty, S.W.; Yates, C.M.; Schotzinger, R.J. Design and optimization of highly-selective fungal CYP51 inhibitors. Bioorg. Med. Chem. Lett. 2014, 24, 3455–3458. [Google Scholar] [CrossRef]
- Warrilow, A.G.; Hull, C.M.; Parker, J.E.; Garvey, E.P.; Hoekstra, W.J.; Moore, W.R.; Schotzinger, R.J.; Kelly, D.E.; Kelly, S.L. The clinical candidate VT-1161 is a highly potent inhibitor of Candida albicans CYP51 but fails to bind the human enzyme. Antimicrob. Agents Chemother. 2014, 58, 7121–7127. [Google Scholar] [CrossRef]
- Warrilow, A.G.; Parker, J.E.; Price, C.L.; Nes, W.D.; Garvey, E.P.; Hoekstra, W.J.; Schotzinger, R.J.; Kelly, D.E.; Kelly, S.L. The Investigational Drug VT-1129 Is a Highly Potent Inhibitor of Cryptococcus Species CYP51 but Only Weakly Inhibits the Human Enzyme. Antimicrob. Agents Chemother. 2016, 60, 4530–4538. [Google Scholar] [CrossRef]
- Warrilow, A.G.S.; Parker, J.E.; Price, C.L.; Garvey, E.P.; Hoekstra, W.J.; Schotzinger, R.J.; Wiederhold, N.P.; Nes, W.D.; Kelly, D.E.; Kelly, S.L. The Tetrazole VT-1161 Is a Potent Inhibitor of Trichophyton rubrum through Its Inhibition of T. rubrum CYP51. Antimicrob. Agents Chemother. 2017, 61, e00333-17. [Google Scholar] [CrossRef]
- Shubitz, L.F.; Trinh, H.T.; Galgiani, J.N.; Lewis, M.L.; Fothergill, A.W.; Wiederhold, N.P.; Barker, B.M.; Lewis, E.R.; Doyle, A.L.; Hoekstra, W.J.; et al. Evaluation of VT-1161 for Treatment of Coccidioidomycosis in Murine Infection Models. Antimicrob. Agents Chemother. 2015, 59, 7249–7254. [Google Scholar] [CrossRef]
- Lockhart, S.R.; Fothergill, A.W.; Iqbal, N.; Bolden, C.B.; Grossman, N.T.; Garvey, E.P.; Brand, S.R.; Hoekstra, W.J.; Schotzinger, R.J.; Ottinger, E.; et al. The Investigational Fungal Cyp51 Inhibitor VT-1129 Demonstrates Potent In Vitro Activity against Cryptococcus neoformans and Cryptococcus gattii. Antimicrob. Agents Chemother. 2016, 60, 2528–2531. [Google Scholar] [CrossRef]
- Nishimoto, A.T.; Whaley, S.G.; Wiederhold, N.P.; Zhang, Q.; Yates, C.M.; Hoekstra, W.J.; Schotzinger, R.J.; Garvey, E.P.; Rogers, P.D. Impact of the Major Candida glabrata Triazole Resistance Determinants on the Activity of the Novel Investigational Tetrazoles VT-1598 and VT-1161. Antimicrob. Agents Chemother. 2019, 63, e01304-19. [Google Scholar] [CrossRef]
- Nishimoto, A.T.; Wiederhold, N.P.; Flowers, S.A.; Zhang, Q.; Kelly, S.L.; Morschhauser, J.; Yates, C.M.; Hoekstra, W.J.; Schotzinger, R.J.; Garvey, E.P.; et al. In Vitro Activities of the Novel Investigational Tetrazoles VT-1161 and VT-1598 Compared to the Triazole Antifungals against Azole-Resistant Strains and Clinical Isolates of Candida albicans. Antimicrob. Agents Chemother. 2019, 63, e00341-19. [Google Scholar] [CrossRef] [PubMed]
- Schell, W.A.; Jones, A.M.; Garvey, E.P.; Hoekstra, W.J.; Schotzinger, R.J.; Alexander, B.D. Fungal CYP51 Inhibitors VT-1161 and VT-1129 Exhibit Strong In Vitro Activity against Candida glabrata and C. krusei Isolates Clinically Resistant to Azole and Echinocandin Antifungal Compounds. Antimicrob. Agents Chemother. 2017, 61, e01817-16. [Google Scholar] [CrossRef] [PubMed]
- Monk, B.C.; Keniya, M.V.; Sabherwal, M.; Wilson, R.K.; Graham, D.O.; Hassan, H.F.; Chen, D.; Tyndall, J.D.A. Azole Resistance Reduces Susceptibility to the Tetrazole Antifungal VT-1161. Antimicrob. Agents Chemother. 2019, 63, e02114-18. [Google Scholar] [CrossRef] [PubMed]
- Garvey, E.P.; Hoekstra, W.J.; Schotzinger, R.J.; Sobel, J.D.; Lilly, E.A.; Fidel, P.L., Jr. Efficacy of the clinical agent VT-1161 against fluconazole-sensitive and -resistant Candida albicans in a murine model of vaginal candidiasis. Antimicrob. Agents Chemother. 2015, 59, 5567–5573. [Google Scholar] [CrossRef]
- Garvey, E.P.; Hoekstra, W.J.; Moore, W.R.; Schotzinger, R.J.; Long, L.; Ghannoum, M.A. VT-1161 dosed once daily or once weekly exhibits potent efficacy in treatment of dermatophytosis in a guinea pig model. Antimicrob. Agents Chemother. 2015, 59, 1992–1997. [Google Scholar] [CrossRef]
- Break, T.J.; Desai, J.V.; Natarajan, M.; Ferre, E.M.N.; Henderson, C.; Zelazny, A.M.; Siebenlist, U.; Hoekstra, W.J.; Schotzinger, R.J.; Garvey, E.P.; et al. VT-1161 protects mice against oropharyngeal candidiasis caused by fluconazole-susceptible and -resistant Candida albicans. J. Antimicrob. Chemother. 2018, 73, 151–155. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Shubitz, L.F.; Najvar, L.K.; Jaramillo, R.; Olivo, M.; Catano, G.; Trinh, H.T.; Yates, C.M.; Schotzinger, R.J.; Garvey, E.P.; et al. The Novel Fungal Cyp51 Inhibitor VT-1598 Is Efficacious in Experimental Models of Central Nervous System Coccidioidomycosis Caused by Coccidioides posadasii and Coccidioides immitis. Antimicrob. Agents Chemother. 2018, 62, e02258-17. [Google Scholar] [CrossRef]
- Gebremariam, T.; Wiederhold, N.P.; Fothergill, A.W.; Garvey, E.P.; Hoekstra, W.J.; Schotzinger, R.J.; Patterson, T.F.; Filler, S.G.; Ibrahim, A.S. VT-1161 Protects Immunosuppressed Mice from Rhizopus arrhizus var. arrhizus Infection. Antimicrob. Agents Chemother. 2015, 59, 7815–7817. [Google Scholar] [CrossRef]
- Vivjoa. Vivjoa (Oteseconazole) Package Insert; Vivjoa: Durham, NC, USA, 2022. [Google Scholar]
- Sobel, J.D.; Nyirjesy, P. Oteseconazole: An advance in treatment of recurrent vulvovaginal candidiasis. Future Microbiol. 2021, 16, 1453–1461. [Google Scholar] [CrossRef]
- Brand, S.R.; Degenhardt, T.P.; Person, K.; Sobel, J.D.; Nyirjesy, P.; Schotzinger, R.J.; Tavakkol, A. A phase 2, randomized, double-blind, placebo-controlled, dose-ranging study to evaluate the efficacy and safety of orally administered VT-1161 in the treatment of recurrent vulvovaginal candidiasis. Am. J. Obstet. Gynecol. 2018, 218, e621.e1–e624.e9. [Google Scholar] [CrossRef] [PubMed]
- Brand, S.R.; Sobel, J.D.; Nyirjesy, P.; Ghannoum, M.A.; Schotzinger, R.J.; Degenhardt, T.P. A Randomized Phase 2 Study of VT-1161 for the Treatment of Acute Vulvovaginal Candidiasis. Clin. Infect. Dis. 2021, 73, e1518–e1524. [Google Scholar] [CrossRef] [PubMed]
- Elewski, B.; Brand, S.; Degenhardt, T.; Curelop, S.; Pollak, R.; Schotzinger, R.; Tavakkol, A. A phase II, randomized, double-blind, placebo-controlled, dose-ranging study to evaluate the efficacy and safety of VT-1161 oral tablets in the treatment of patients with distal and lateral subungual onychomycosis of the toenail. Br. J. Dermatol. 2021, 184, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Cass, L.; Murray, A.; Davis, A.; Woodward, K.; Albayaty, M.; Ito, K.; Strong, P.; Ayrton, J.; Brindley, C.; Prosser, J.; et al. Safety and nonclinical and clinical pharmacokinetics of PC945, a novel inhaled triazole antifungal agent. Pharmacol. Res. Perspect. 2021, 9, e00690. [Google Scholar] [CrossRef]
- Colley, T.; Alanio, A.; Kelly, S.L.; Sehra, G.; Kizawa, Y.; Warrilow, A.G.S.; Parker, J.E.; Kelly, D.E.; Kimura, G.; Anderson-Dring, L.; et al. In Vitro and In Vivo Antifungal Profile of a Novel and Long-Acting Inhaled Azole, PC945, on Aspergillus fumigatus Infection. Antimicrob. Agents Chemother. 2017, 61, e02280-16. [Google Scholar] [CrossRef]
- Santangelo, R.; Paderu, P.; Delmas, G.; Chen, Z.W.; Mannino, R.; Zarif, L.; Perlin, D.S. Efficacy of oral cochleate-amphotericin B in a mouse model of systemic candidiasis. Antimicrob. Agents Chemother. 2000, 44, 2356–2360. [Google Scholar] [CrossRef]
- Kibathi, L.; Kumar, P.; Lionakis, M.; Urban, A.; Ferre, E.; McManus, M.; Colton, B.; Lambros, C.; Lu, R.; Mannino, R.; et al. A phase lla efficacy safety, tolerability and pharmacokinetic study of encochleated amphotericin B in patients with mucocutaneous (esophageal, oropharyngeal, vulvovaginal) Candidiasis who are refractory or intolerant to standard non-intravenous therapies. Open Forum Infect. Dis. 2018, 5, S435. [Google Scholar] [CrossRef][Green Version]
| Agent and Company Developing | Routes of Administration | PK/PD Parameter Associated with In Vivo Efficacy | Tolerability/Adverse Effects and Drug Interactions | Current Clinical Trials (Number and Phase) |
|---|---|---|---|---|
| Manogepix (APX001A) Pfizer | Intravenous and oral | AUC/MIC vs. yeasts (fAUC/MIC 1.35–22.54) | Well-tolerated in Phase I and II clinical studies | Candidemia/invasive candidiasis (NCT05421858, Phase III) |
| AUC/MEC vs. molds (fAUC/MEC 89.39) | Drug interaction profile not yet known | |||
| Olorofim (F901318) F2G | Intravenous and oral | Cmin/MIC (Cmin/MIC 3–16.5 vs. A. fumigatus) | Well-tolerated in Phase I studies with no serious adverse effects | Invasive aspergillois (NCT05101187, Phase III) |
| Potential for drug interactions, as it is metabolized by CYP450 enzymes and is a weak inhibitor of CYP3A4 | Aspergillosis, lomentosporiosis, scedosporiosis, and other resistant fungi (NCT03583164, Phase II) | |||
| Ibrexafungerp (SCY-078) Scynexis | Oral (Intravenous formulation under development) | AUC/MIC vs. Candida (fAUC/MIC 0.1–1.7) | Well-tolerated in Phase I and II clinical studies, and no QTc prolongations reported | Complicated vulvovaginal candidiasis (NCT05399641, Phase III) |
| Invasive pulmonary aspergillosis (NCT03672292, Phase III) | ||||
| AUC/MIC Possibly AUC/MEC vs. Aspergillus | Potential for drug interactions, as it is metabolized by CYP3A4 and is also an inhibitor of CYP2C8 and 3A4 | Candida auris candidiasis (NCT03363841, Phase III) | ||
| Invasive mycoses in those who are refractory to or intolerant of other therapies (NCT03059992, Phase III) | ||||
| Rezafungin (CD101) Cidara | Intravenous | AUC/MIC vs. Candida (fAUC/MIC 0.07–11.65) | Well-tolerated in Phase I and II clinical studies; some infusion-related reactions with higher doses | Antifungal prophylaxis in adults undergoing allogeneic stem cell transplantation (NCT04368559, Phase III) |
| AUC/MEC and Cmax/MEC vs. Aspergillus | Low potential for drug interactions | |||
| Oteseconazole (VT-1161) Mycovia | Oral | Undefined, but most likely AUC/MIC (similar to triazoles) | Well-tolerated in Phase I and II clinical studies with mild-to-moderate adverse effects | Recurrent vulvovaginal candidiasis (NCT03562156 and NCT033561701, Phase III—Completed) |
| Drug interactions not observed with agents metabolized by CYP3A4 or p-glycoprotein |
| Antifungal | Manogepix | Olorofim | Ibrexafungerp | Rezafungin | Oteseconazole |
|---|---|---|---|---|---|
| Yeasts | |||||
| C. albicans | + | − | + | + | + |
| C. auris | + | − | + | + | + |
| C. glabrata | + | − | + | + | + |
| C. krusei | − | − | + | + | + |
| C. parapsilosis | + | − | + | + | + |
| C. tropicalis | + | − | + | + | + |
| C. gattii | + | − | − | − | + |
| C. neoformans | + | − | − | − | + |
| Rhodotorula | + | − | − | − | |
| Trichosporon | +/− | − | − | − | |
| Aspergillus | |||||
| A. flavus | + | + | + | + | − |
| A. fumigatus | + | + | + | + | − |
| A. niger | + | + | + | + | − |
| A. terreus | + | + | + | + | − |
| Fusarium | |||||
| F. oxysporum | + | +/− | − | ||
| F. solani | + | − | − | ||
| Scedosporium | |||||
| Scedosporium | + | + | − | ||
| L. prolificans | + | + | − | ||
| Mucorales | |||||
| Mucor | − | − | − | − | |
| Rhizopus | +/− | − | − | − | +/− |
| Other Mucorales | − | − | − | − | |
| Endemic Fungi | |||||
| Blastomyces | + | + | + | ||
| Coccidioides | + | + | + | + | |
| Histoplasma | + | + | + | ||
| Dermatophytes | |||||
| Trichophyton | + | + | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiederhold, N.P. Pharmacodynamics, Mechanisms of Action and Resistance, and Spectrum of Activity of New Antifungal Agents. J. Fungi 2022, 8, 857. https://doi.org/10.3390/jof8080857
Wiederhold NP. Pharmacodynamics, Mechanisms of Action and Resistance, and Spectrum of Activity of New Antifungal Agents. Journal of Fungi. 2022; 8(8):857. https://doi.org/10.3390/jof8080857
Chicago/Turabian StyleWiederhold, Nathan P. 2022. "Pharmacodynamics, Mechanisms of Action and Resistance, and Spectrum of Activity of New Antifungal Agents" Journal of Fungi 8, no. 8: 857. https://doi.org/10.3390/jof8080857
APA StyleWiederhold, N. P. (2022). Pharmacodynamics, Mechanisms of Action and Resistance, and Spectrum of Activity of New Antifungal Agents. Journal of Fungi, 8(8), 857. https://doi.org/10.3390/jof8080857






