Predicting Species Boundaries and Assessing Undescribed Diversity in Pneumocystis, an Obligate Lung Symbiont
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| mtSSU | Mitochondrial small subunit |
| mtLSU | Mitochondrial large subunit |
| ABGD | Automatic Barcode Gap Discovery |
| GMYC | Generalized Mixed Yule Coalescent model |
| PTP | Poisson Tree Processes model |
| BPP | Bayesian Phylogenetics and Phylogeography |
References
- Kew Royal Botanic Gardens. State of the World’s Fungi; Technical Report; Royal Botanic Gardens: Kew, UK, 2018. [Google Scholar]
- Hawksworth, D.L.; Lücking, R. Fungal Diversity Revisited: 2.2 to 3.8 Million Species. Microbiol. Spectr. 2017, 5, 5–14. [Google Scholar] [CrossRef]
- Yoshida, Y.; Matsumoto, Y.; Yamada, M.; Okabayashi, K.; Yoshikawa, H.; Nakazawa, M. Pneumocystis carinii: Electron microscopic investigation on the interaction of trophozoite and alveolar lining cell. Zentralblatt fur Bakteriologie Mikrobiologie Hygiene Ser. A Med Microbiol. Infect. Dis. Virol. Parasitol. 1984, 256, 390–399. [Google Scholar] [CrossRef]
- Kelly, M.N.; Shellito, J.E. Current understanding of pneumocystis immunology. Future Microbiol. 2010, 5, 43–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aliouat-Denis, C.M.; Chabé, M.; Demanche, C.; Aliouat, E.M.; Viscogliosi, E.; Guillot, J.; Delhaes, L.; Dei-Cas, E. Pneumocystis species, co-evolution and pathogenic power. Infect. Genet. Evol. 2008, 8, 708–726. [Google Scholar] [CrossRef] [PubMed]
- Aliouat, E.; Mazars, E.; Dei-Cas, E.; Cesbron, J.; Camus, D. Intranasal Inoculation of Mouse, Rat or Rabbit-Derived Pneumocystis to SCID Mice. J. Protozool. Res. 1993, 3, 94–98. [Google Scholar]
- Durand-Joly, I.; Aliouat, E.M.; Recourt, C.; Guyot, K.; François, N.; Wauquier, M.; Camus, D.; Dei-Cas, E. Pneumocystis carinii f. sp. hominis is not infectious for SCID mice. J. Clin. Microbiol. 2002, 40, 1862–1865. [Google Scholar] [CrossRef] [Green Version]
- Walzer, P.D. The ecology of Pneumocystis: Perspectives, personal recollections, and future research opportunities. J. Eukaryot. Microbiol. 2013, 60, 634–645. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hauser, P.M. Is the unique camouflage strategy of Pneumocystis associated with its particular niche within host lungs? PLoS Pathog. 2019, 15, e1007480. [Google Scholar] [CrossRef] [PubMed]
- Frenkel, J. Pneumocystis jiroveci n. sp. From Man: Morphology, Physiology, and Immunology in Relation to Pathology. Natl. Cancer Inst. Monogr. 1976, 43, 13–30. [Google Scholar] [PubMed]
- Keely, S.P.; Fischer, J.M.; Cushion, M.T.; Stringer, J.R.; Scott Keely, C.P. Phylogenetic identification of Pneumocystis murina sp. nov., a new species in laboratory mice. Microbiology 2004, 150, 1153–1165. [Google Scholar] [CrossRef] [Green Version]
- Cushion, M.T.; Keely, S.P.; Stringer, J.R. Molecular and Phenotypic Description of Pneumocystis wakefieldiae sp. nov., a New Species in Rats. Mycologia 2004, 96, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Dei-Cas, E.; Chabé, M.; Moukhlis, R.; Durand-Joly, I.; Aliouat, E.M.; Stringer, J.R.; Cushion, M.; Noël, C.; Sybren De Hoog, G.; Guillot, J.; et al. Pneumocystis oryctolagi sp. nov., an uncultured fungus causing pneumonia in rabbits at weaning: Review of current knowledge, and description of a new taxon on genotypic, phylogenetic and phenotypic bases. FEMS Microbiol. Rev. 2006, 30. [Google Scholar] [CrossRef] [Green Version]
- Cissé, O.H.; Ma, L.; Dekker, J.P.; Khil, P.P.; Youn, J.H.; Brenchley, J.M.; Blair, R.; Pahar, B.; Chabé, M.; Van Rompay, K.K.; et al. Genomic insights into the host specific adaptation of the Pneumocystis genus. Commun. Biol. 2021, 4, 305. [Google Scholar] [CrossRef] [PubMed]
- Burgin, C.J.; Colella, J.P.; Kahn, P.L.; Upham, N.S. How many species of mammals are there? J. Mammal. 2018, 99, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Mammal Diversity Database. 2022. Available online: https://www.mammaldiversity.org/ (accessed on 25 July 2022).
- Hugot, J.P.; Demanche, C.; Barriel, V.; Dei-Cas, E.; Guillot, J. Phylogenetic Systematics and Evolution of Primate-Derived Pneumocystis Based on Mitochondrial or Nuclear DNA Sequence Comparison. Syst. Biol. 2003, 52, 735–744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chabé, M.; Aliouat-Denis, C.M.; Delhaes, L.; Aliouat, E.M.; Viscogliosi, E.; Dei-Cas, E. Pneumocystis: From a doubtful unique entity to a group of highly diversified fungal species. FEMS Yeast Res. 2011, 11, 2–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Danesi, P.; da Rold, G.; Rizzoli, A.; Hauffe, H.C.; Marangon, S.; Samerpitak, K.; Demanche, C.; Guillot, J.; Capelli, G.; de Hoog, S.G. Barcoding markers for Pneumocystis species in wildlife. Fungal Biology 2016, 120, 191–206. [Google Scholar] [CrossRef]
- Latinne, A.; Bezé, F.; Delhaes, L.; Pottier, M.; Gantois, N.; Nguyen, J.; Blasdell, K.; Dei-Cas, E.; Morand, S.; Chabé, M. Genetic diversity and evolution of Pneumocystis fungi infecting wild Southeast Asian murid rodents. Parasitology 2018, 145. [Google Scholar] [CrossRef] [PubMed]
- Demanche, C.; Guillot, J.; Chabé, M. Pneumocystis Species Co-evolution: State-of-the-Art Review. OBM Genet. 2019, 3, 1. [Google Scholar] [CrossRef]
- Babb-Biernacki, S.J.; Esselstyn, J.A.; Doyle, V.P. Rethinking host range in Pneumocystis. PLoS Pathog. 2020, 16, e1008824. [Google Scholar] [CrossRef] [PubMed]
- Latinne, A.; Chen, H.W.; Kuo, C.C.; Lorica, R.; Singleton, G.; Stuart, A.; Malbas, F.F.; Demanche, C.; Chabé, M.; Michaux, J.; et al. Revisiting the Pneumocystis host specificity paradigm and transmission ecology in wild Southeast Asian rodents. Infect. Genet. Evol. 2021, 93, 104978. [Google Scholar] [CrossRef] [PubMed]
- Demanche, C.; Berthelemy, M.; Petit, T.; Polack, B.; Wakefield, A.E.; Dei-Cas, E.; Guillot, J. Phylogeny of Pneumocystis carinii from 18 primate species confirms host specificity and suggests coevolution. J. Clin. Microbiol. 2001, 39, 2126–2133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akbar, H.; Pinçon, C.; Aliouat-Denis, C.M.; Derouiche, S.; Taylor, M.L.; Pottier, M.; Carreto-Binaghi, L.H.; González-González, A.E.; Courpon, A.; Barriel, V.; et al. Characterizing pneumocystis in the lungs of bats: Understanding pneumocystis evolution and the spread of pneumocystis organisms in mammal populations. Appl. Environ. Microbiol. 2012, 78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Redhead, S.A.; Cushion, M.T.; Frenkel, J.K.; Stringer, J.R. Pneumocystis and Trypanosoma cruzi: Nomenclature and typifications. J. Eukaryot. Microbiol. 2006, 53, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Britz, R.; Hundsdörfer, A.; Fritz, U. Funding, training, permits—The three big challenges of taxonomy. Megataxa 2020, 1. [Google Scholar] [CrossRef] [Green Version]
- Stringer, J.R.; Cushion, M.; Wakefielp, A.E. New nomenclature for the genus Pneumocystis. J. Eukaryot. Microbiol. 2001, 184S–189S. [Google Scholar] [CrossRef]
- Beard, C.B.; Roux, P.; Nevez, G.; Hauser, P.M.; Kovacs, J.A.; Unnasch, T.R.; Lundgren, B. Strain Typing Methods and Molecular Epidemiology of Pneumocystis Pneumonia. Emerg. Infect. Dis. 2004, 10, 1729–1735. [Google Scholar] [PubMed]
- Zamora, J.C.; Svensson, M.; Kirschner, R.; Olariaga, I.; Ryman, S.; Parra, L.A.; Geml, J.; Rosling, A.; Adamčík, S.; Ahti, T.; et al. Considerations and consequences of allowing DNA sequence data as types of fungal taxa. IMA Fungus 2018, 9, 167–175. [Google Scholar] [CrossRef]
- Pons, J.; Barraclough, T.G.; Gomez-Zurita, J.; Cardoso, A.; Duran, D.P.; Hazell, S.; Kamoun, S.; Sumlin, W.D.; Vogler, A.P. Sequence-based species delimitation for the DNA taxonomy of undescribed insects. Syst. Biol. 2006, 55, 595–609. [Google Scholar] [CrossRef] [Green Version]
- Puillandre, N.; Lambert, A.; Brouillet, S.; Achaz, G. ABGD, Automatic Barcode Gap Discovery for primary species delimitation. Mol. Ecol. 2012, 21, 1864–1877. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Kapli, P.; Pavlidis, P.; Stamatakis, A. A general species delimitation method with applications to phylogenetic placements. Bioinformatics 2013, 29, 2869–2876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, A.; Ling, C.; Ho, S.Y.; Zhu, C.D. Comparison of methods for molecular species delimitation across a range of speciation scenarios. Syst. Biol. 2018, 67, 830–846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boluda, C.G.; Rico, V.J.; Divakar, P.K.; Nadyeina, O.; Myllys, L.; McMullin, R.T.; Zamora, J.C.; Scheidegger, C.; Hawksworth, D.L. Evaluating methodologies for species delimitation: The mismatch between phenotypes and genotypes in lichenized fungi (Bryoria sect. implexae, Parmeliaceae). Persoonia Mol. Phylogeny Evol. Fungi 2019, 42, 75–100. [Google Scholar] [CrossRef] [Green Version]
- Lima, N.B.; Lima, W.G.; Tovar-Pedraza, J.M.; Michereff, S.J.; Câmara, M.P. Comparative epidemiology of Colletotrichum species from mango in northeastern Brazil. Eur. J. Plant Pathol. 2015, 141, 679–688. [Google Scholar] [CrossRef]
- Veloso, J.S.; Câmara, M.P.; Lima, W.G.; Michereff, S.J.; Doyle, V.P. Why species delimitation matters for fungal ecology: Colletotrichum diversity on wild and cultivated cashew in Brazil. Fungal Biol. 2018, 122, 677–691. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z. The BPP program for species tree estimation and species delimitation. Curr. Zool. 2015, 61, 854–865. [Google Scholar] [CrossRef]
- Millanes, A.M.; Truong, C.; Westberg, M.; Diederich, P.; Wedin, M. Host switching promotes diversity in host-specialized mycoparasitic fungi: Uncoupled evolution in the biatoropsis-usnea system. Evolution 2014, 68, 1576–1593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sela, I.; Ashkenazy, H.; Katoh, K.; Pupko, T. GUIDANCE2: Accurate detection of unreliable alignment regions accounting for the uncertainty of multiple parameters. Nucleic Acids Res. 2015, 43, 7–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, G.; Muffato, M.; Ledergerber, C.; Herrero, J.; Goldman, N.; Gil, M.; Dessimoz, C. Current methods for automated filtering of multiple sequence alignments frequently worsen single-gene phylogenetic inference. Syst. Biol. 2015, 64, 778–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.; Von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bouckaert, R.; Heled, J.; Kühnert, D.; Vaughan, T.; Wu, C.H.; Xie, D.; Suchard, M.A.; Rambaut, A.; Drummond, A.J. BEAST 2: A Software Platform for Bayesian Evolutionary Analysis. PLoS Comput. Biol. 2014, 10, e1003537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior summarization in Bayesian phylogenetics using Tracer 1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef] [Green Version]
- Hebert, P.D.N.; Cywinska, A.; Ball, S.L.; DeWaard, J.R. Biological identifications through DNA barcodes. Proc. R. Soc. Biol. Sci. 2003, 270, 313–321. [Google Scholar] [CrossRef] [Green Version]
- Fujisawa, T.; Barraclough, T.G. Delimiting species using single-locus data and the generalized mixed yule coalescent approach: A revised method and evaluation on simulated data sets. Syst. Biol. 2013, 62, 707–724. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahrens, D.; Fujisawa, T.; Krammer, H.J.; Eberle, J.; Fabrizi, S.; Vogler, A.P. Rarity and incomplete sampling in DNA-based species delimitation. Syst. Biol. 2016, 65, 478–494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flouri, T.; Jiao, X.; Rannala, B.; Yang, Z. Species tree inference with BPP using genomic sequences and the multispecies coalescent. Mol. Biol. Evol. 2018, 35, 2585–2593. [Google Scholar] [CrossRef] [PubMed]
- Leaché, A.D.; Fujita, M.K. Bayesian species delimitation in West African forest geckos (Hemidactylus fasciatus). Proc. R. Soc. B Biol. Sci. (R. Soc.) 2010, 277, 3071–3077. [Google Scholar] [CrossRef] [Green Version]
- Chabé, M.; Hugot, J.P.; Dei-Cas, E. Pneumocystis molecular phylogeny: A way to understand both pneumocystosis natural history and host taxonomy. In New Frontiers of Molecular Epidemiology of Infectious Diseases; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar] [CrossRef]
- Dos Reis, M.; Inoue, J.; Hasegawa, M.; Asher, R.J.; Donoghue, P.C.; Yang, Z. Phylogenomic datasets provide both precision and accuracy in estimating the timescale of placental mammal phylogeny. Proc. R. Soc. B Biol. Sci. 2012, 279, 3491–3500. [Google Scholar] [CrossRef] [PubMed]
- Esselstyn, J.A.; Oliveros, C.H.; Swanson, M.T.; Faircloth, B.C. Investigating difficult nodes in the placental mammal tree with expanded taxon sampling and thousands of ultraconserved elements. Genome Biol. Evol. 2017, 9, 2308–2321. [Google Scholar] [CrossRef]
- Guillot, J.; Demanche, C.; Hugot, J.P.; Berthelemy, M.; Wakefield, A.E.; Dei-Cas, E.; Chermette, R. Parallel Phylogenies of Pneumocystis Species and their Mammalian Hosts. J. Eukaryot. Microbiol. 2001, 48, 113–115. [Google Scholar] [CrossRef]
- Pollock, D.D.; Zwickl, D.J.; McGuire, J.A.; Hillis, D.M. Increased taxon sampling is advantageous for phylogenetic inference. Syst. Biol. 2002, 51, 664–671. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolaczkowski, B.; Thornton, J.W. Long-branch attraction bias and inconsistency in bayesian phylogenetics. PLoS ONE 2009, 4, e7891. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nantarat, N.; Sutcharit, C.; Tongkerd, P.; Wade, C.M.; Naggs, F.; Panha, S. Phylogenetics and species delimitations of the operculated land snail Cyclophorus volvulus (Gastropoda: Cyclophoridae) reveal cryptic diversity and new species in Thailand. Sci. Rep. 2019, 9, 7041. [Google Scholar] [CrossRef]
- Stringer, J.R.; Beard, C.B.; Miller, R.F.; Wakefield, A.E. A new name (Pneumocystis jiroveci) for Pneumocystis from humans. Emerg. Infect. Dis. 2002, 8, 891–896. [Google Scholar] [CrossRef]
- Esselstyn, J.A.; Evans, B.J.; Sedlock, J.L.; Khan, F.A.A.; Heaney, L.R. Single-locus species delimitation: A test of the mixed yule-coalescent model, with an empirical application to Philippine round-leaf bats. Proc. R. Soc. B Biol. Sci. 2012, 279, 3678–3686. [Google Scholar] [CrossRef]
- García-Melo, J.E.; Oliveira, C.; Da Costa Silva, G.J.; Ochoa-Orrego, L.E.; Garcia Pereira, L.H.; Maldonado-Ocampo, J.A. Species delimitation of neotropical Characins (Stevardiinae): Implications for taxonomy of complex groups. PLoS ONE 2019, 14, e0216786. [Google Scholar] [CrossRef]
- Herrera, C.S.; Hirooka, Y.; Chaverri, P. Pseudocospeciation of the mycoparasite Cosmospora with their fungal hosts. Ecol. Evol. 2016, 6, 1504–1514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vallejos-Garrido, P.; Rivera, R.; Inostroza-Michae, O.; Rodríguez-Serrano, E.; Hernández, C.E. Historical dynamics and current environmental effects explain the spatial distribution of species richness patterns of New World monkeys. PeerJ 2017, 2017, e3850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Refrégier, G.; Le Gac, M.; Jabbour, F.; Widmer, A.; Shykoff, J.A.; Yockteng, R.; Hood, M.E.; Giraud, T. Cophylogeny of the anther smut fungi and their caryophyllaceous hosts: Prevalence of host shifts and importance of delimiting parasite species for inferring cospeciation. BMC Evol. Biol. 2008, 8, 100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging fungal threats to animal, plant and ecosystem health. Nature 2012, 484, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Danesi, P.; Corrò, M.; Corrò, C.; Falcaro, C.; Carminato, A.; Furlanello, T.; Cocchi, M.; Krockenberger, M.B.; Meyer, W.; Capelli, G.; et al. Molecular detection of Pneumocystis in the lungs of cats. Med. Mycol. 2018, 57, 813–824. [Google Scholar] [CrossRef]
- Doyle, V.P.; Oudemans, P.V.; Rehner, S.A.; Litt, A. Habitat and Host Indicate Lineage Identity in Colletotrichum gloeosporioides s.l. from Wild and Agricultural Landscapes in North America. PLoS ONE 2013, 8, e62394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freudenstein, J.V.; Barrett, C.F. Fungal host utilization helps circumscribe leafless Coralroot orchid species: An integrative analysis of Corallorhiza odontorhiza and C. wisteriana. Taxon 2014, 63, 759–772. [Google Scholar] [CrossRef]

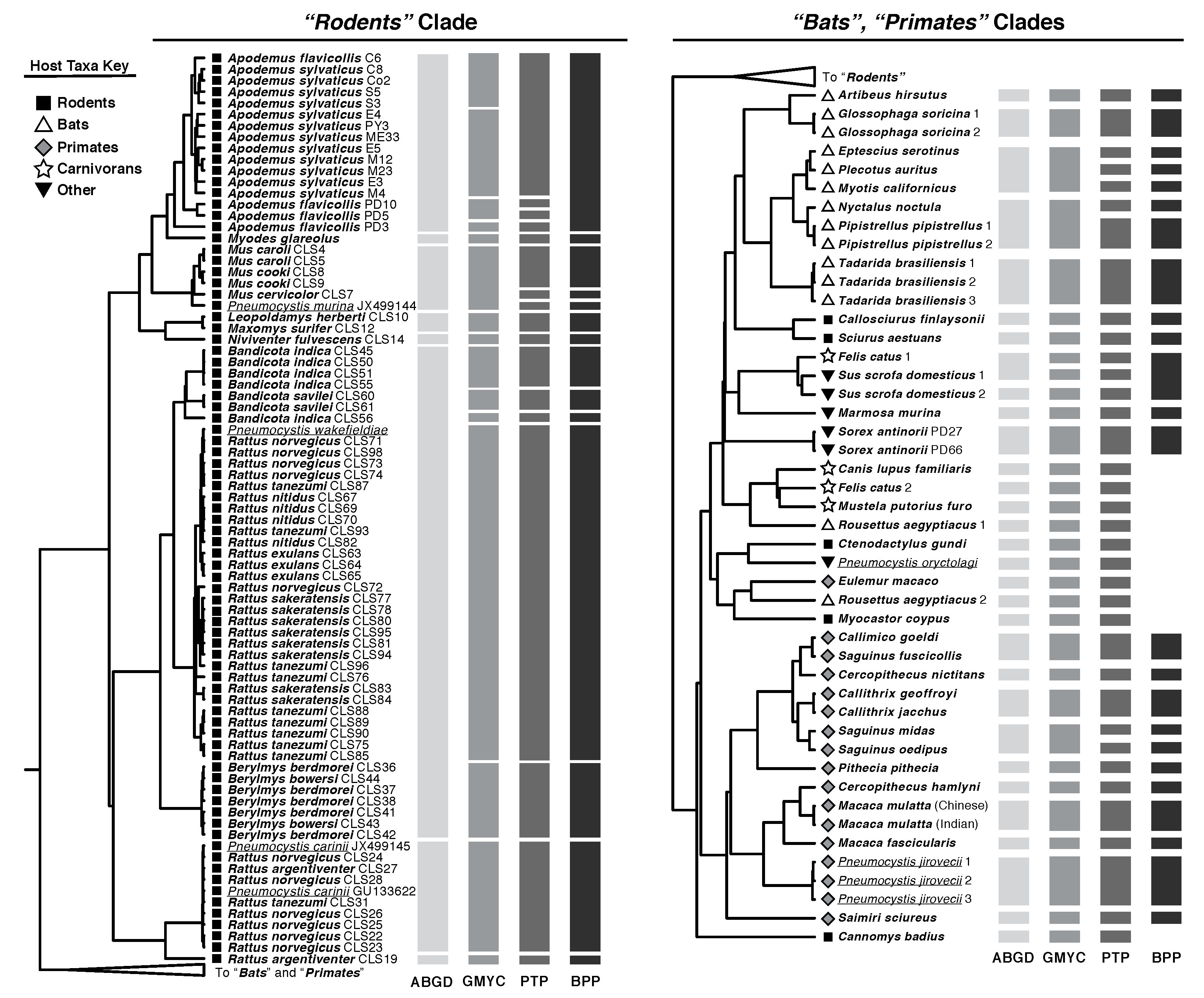
| Delimitation Method | Inferred Pneumocystis Species | Host: Symbiont Ratio | Predicted Global Pneumocystis Species |
|---|---|---|---|
| ABGD | 39 | 1:0.71 | 4606 |
| GMYC | 47 | 1:0.85 | 5550 |
| PTP | 53 | 1:0.96 | 6259 |
| BPP | 48 | 1:0.87 | 5668 |
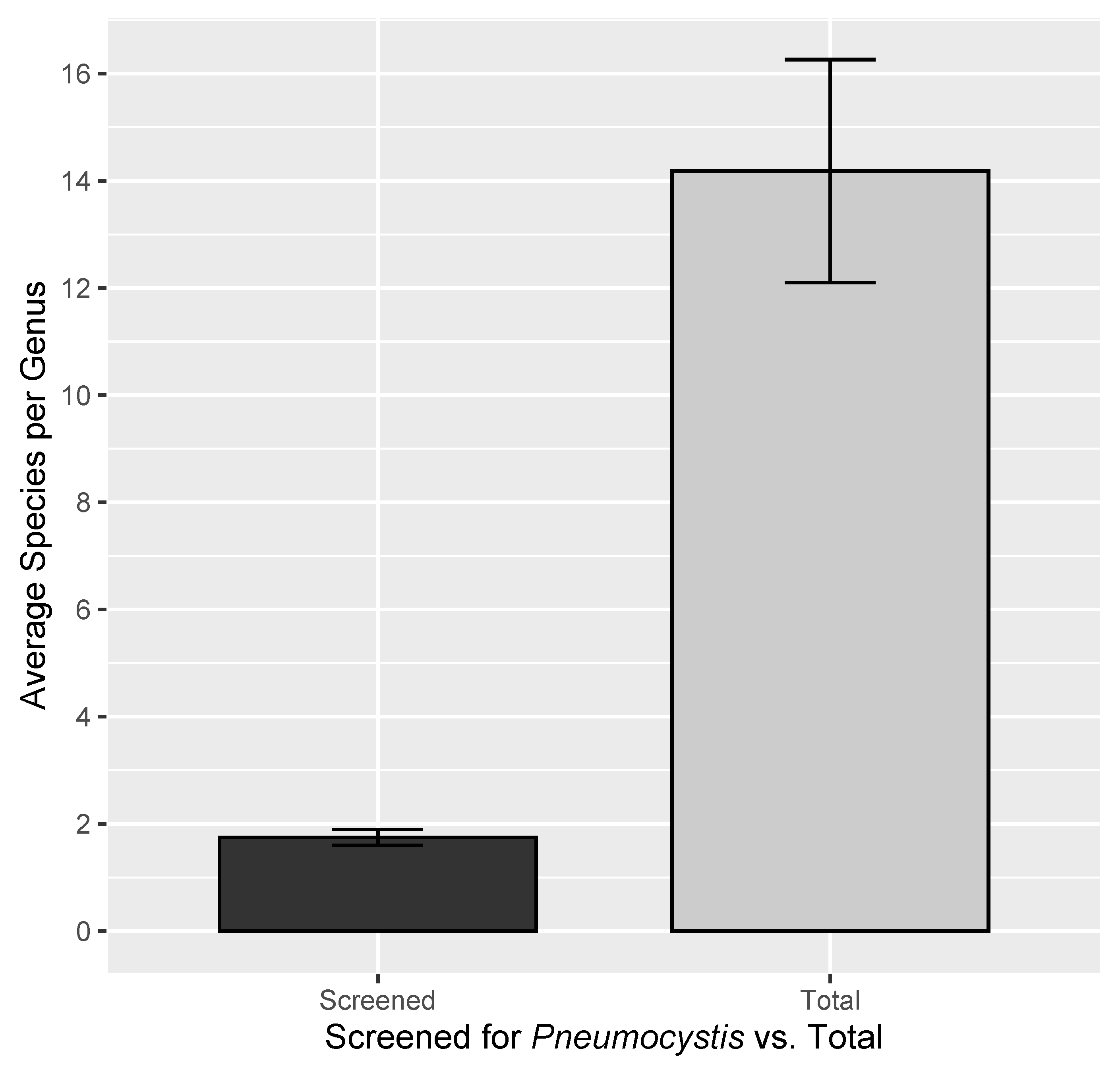
| Taxonomic Rank | Number Screened for Pneumocystis | Total in Mammalia | Percent Sampled | Pneumocystis Genetic Data Available | Percent Screened with Genetic Data | Percent Total with Genetic Data |
|---|---|---|---|---|---|---|
| Mammal Species | 240 | 6495 | 3.7 | 129 | 54 | 1.99 |
| Mammal Genera | 142 | 1342 | 10.58 | - | - | - |
| Mammal Orders | 15 | 27 | 55.56 | - | - | - |
| Mammal Order | Species Screened for Pneumocystis | Total Species in Order | Percent Screened |
|---|---|---|---|
| Afrosoricida | 1 | 55 | 1.82 |
| Artiodactyla | 7 | 359 | 1.95 |
| Carnivora | 14 | 307 | 4.56 |
| Chiroptera | 37 | 1447 | 2.56 |
| Diprotodontia | 1 | 150 | 0.67 |
| Didelphimorphia | 2 | 129 | 1.56 |
| Eulipotyphlya | 17 | 564 | 3.01 |
| Hyracoidea | 1 | 6 | 16.67 |
| Lagomorpha | 3 | 106 | 2.83 |
| Monotremata | 1 | 5 | 20 |
| Perissodactyla | 1 | 18 | 5.56 |
| Pilosa | 1 | 16 | 6.25 |
| Primates | 35 | 516 | 6.78 |
| Rodentia | 116 | 2623 | 4.42 |
| Scandentia | 1 | 23 | 4.35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Babb-Biernacki, S.J.; Esselstyn, J.A.; Doyle, V.P. Predicting Species Boundaries and Assessing Undescribed Diversity in Pneumocystis, an Obligate Lung Symbiont. J. Fungi 2022, 8, 799. https://doi.org/10.3390/jof8080799
Babb-Biernacki SJ, Esselstyn JA, Doyle VP. Predicting Species Boundaries and Assessing Undescribed Diversity in Pneumocystis, an Obligate Lung Symbiont. Journal of Fungi. 2022; 8(8):799. https://doi.org/10.3390/jof8080799
Chicago/Turabian StyleBabb-Biernacki, Spenser J., Jacob A. Esselstyn, and Vinson P. Doyle. 2022. "Predicting Species Boundaries and Assessing Undescribed Diversity in Pneumocystis, an Obligate Lung Symbiont" Journal of Fungi 8, no. 8: 799. https://doi.org/10.3390/jof8080799
APA StyleBabb-Biernacki, S. J., Esselstyn, J. A., & Doyle, V. P. (2022). Predicting Species Boundaries and Assessing Undescribed Diversity in Pneumocystis, an Obligate Lung Symbiont. Journal of Fungi, 8(8), 799. https://doi.org/10.3390/jof8080799






