Species Identification and In Vitro Antifungal Susceptibility of Paecilomyces/Purpureocillium Species Isolated from Clinical Respiratory Samples: A Multicenter Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungal Isolates
2.2. Matrix-Assisted Laser Desorption Ionization-Time of Flight (MALDI-TOF) Mass Spectrometry Identification
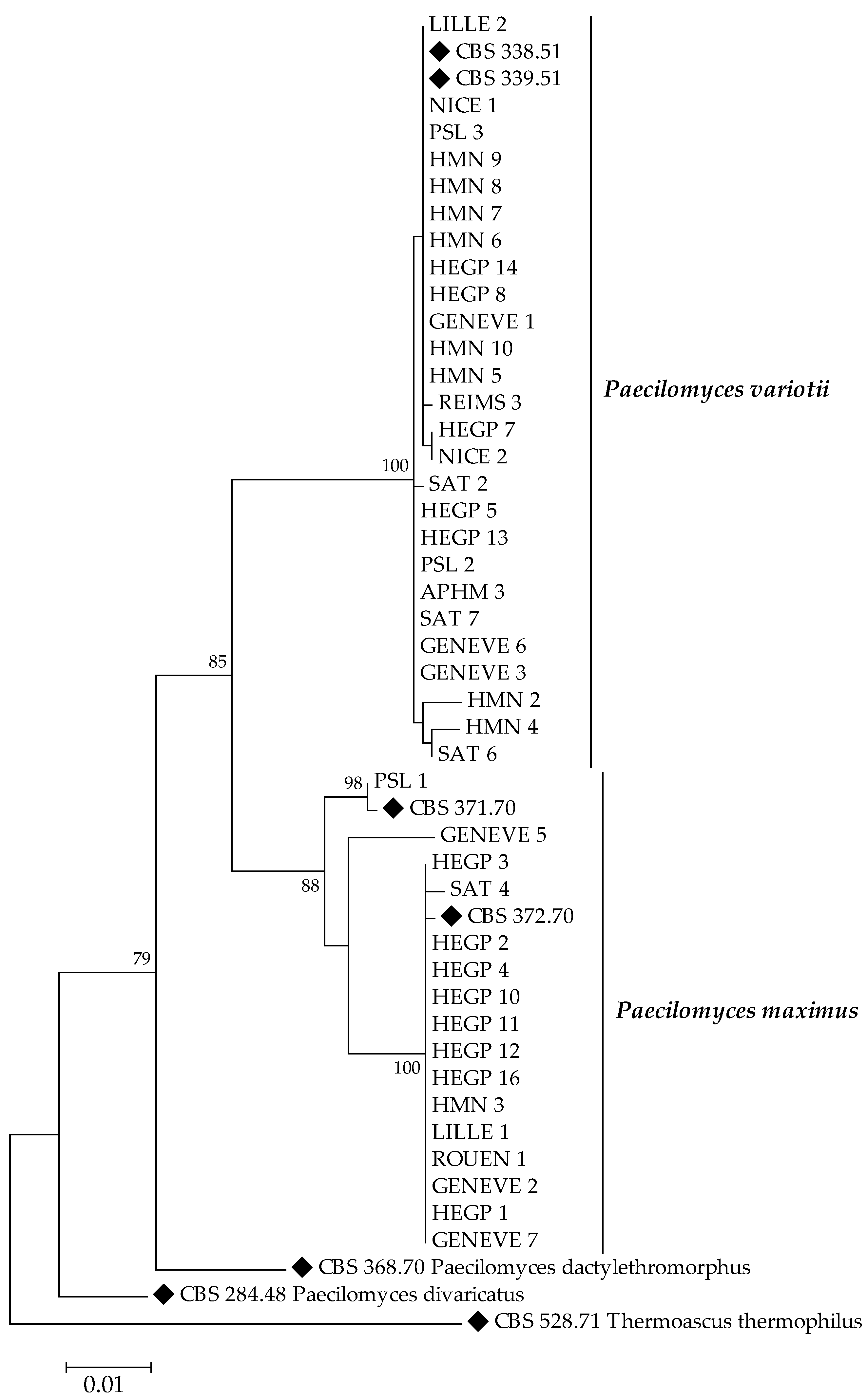
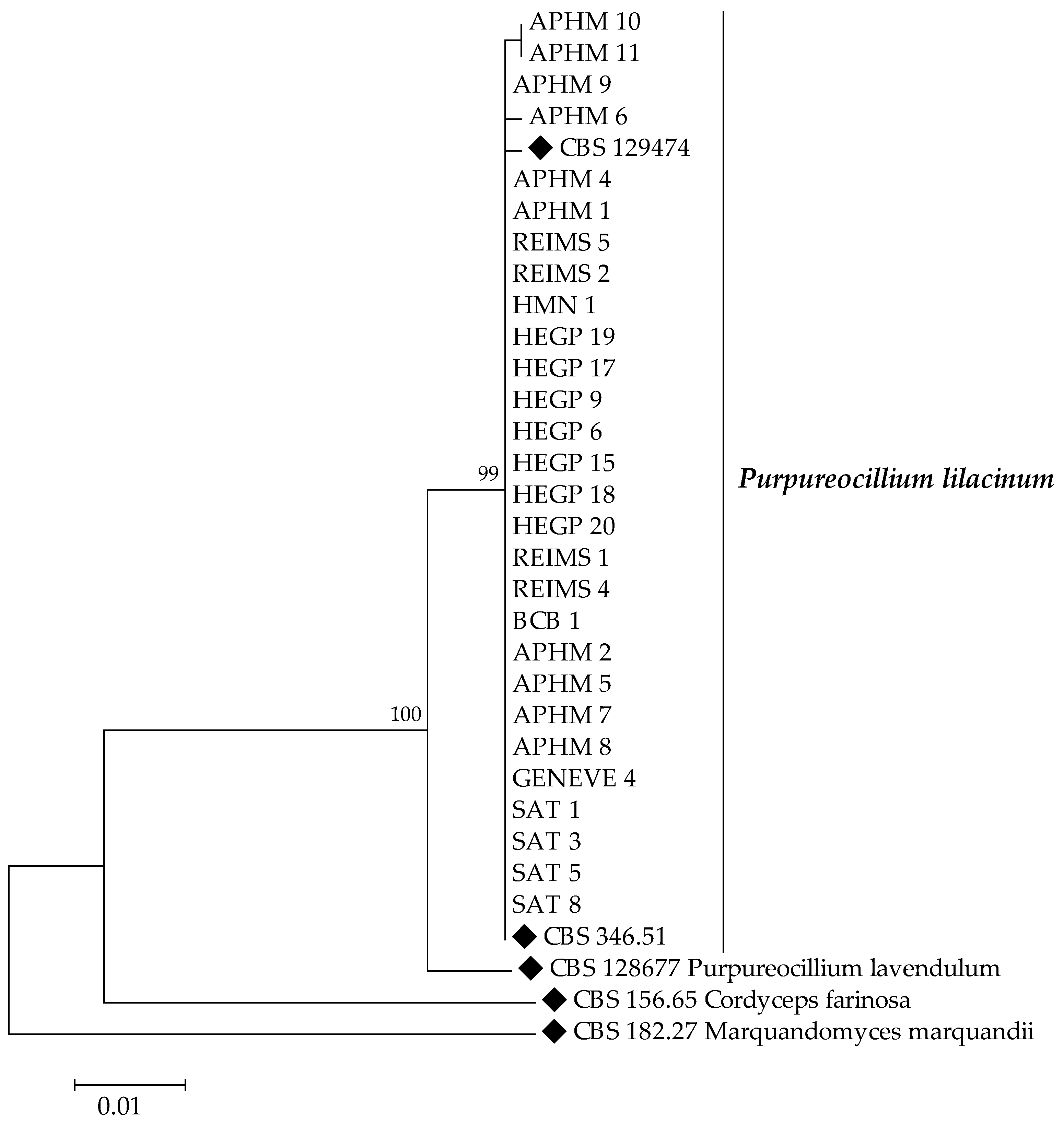
2.3. Molecular Identification and Phylogenetic Analysis
2.4. Antifungal Susceptibility Testing
3. Results
3.1. Isolates and Samples
3.2. Species Identification
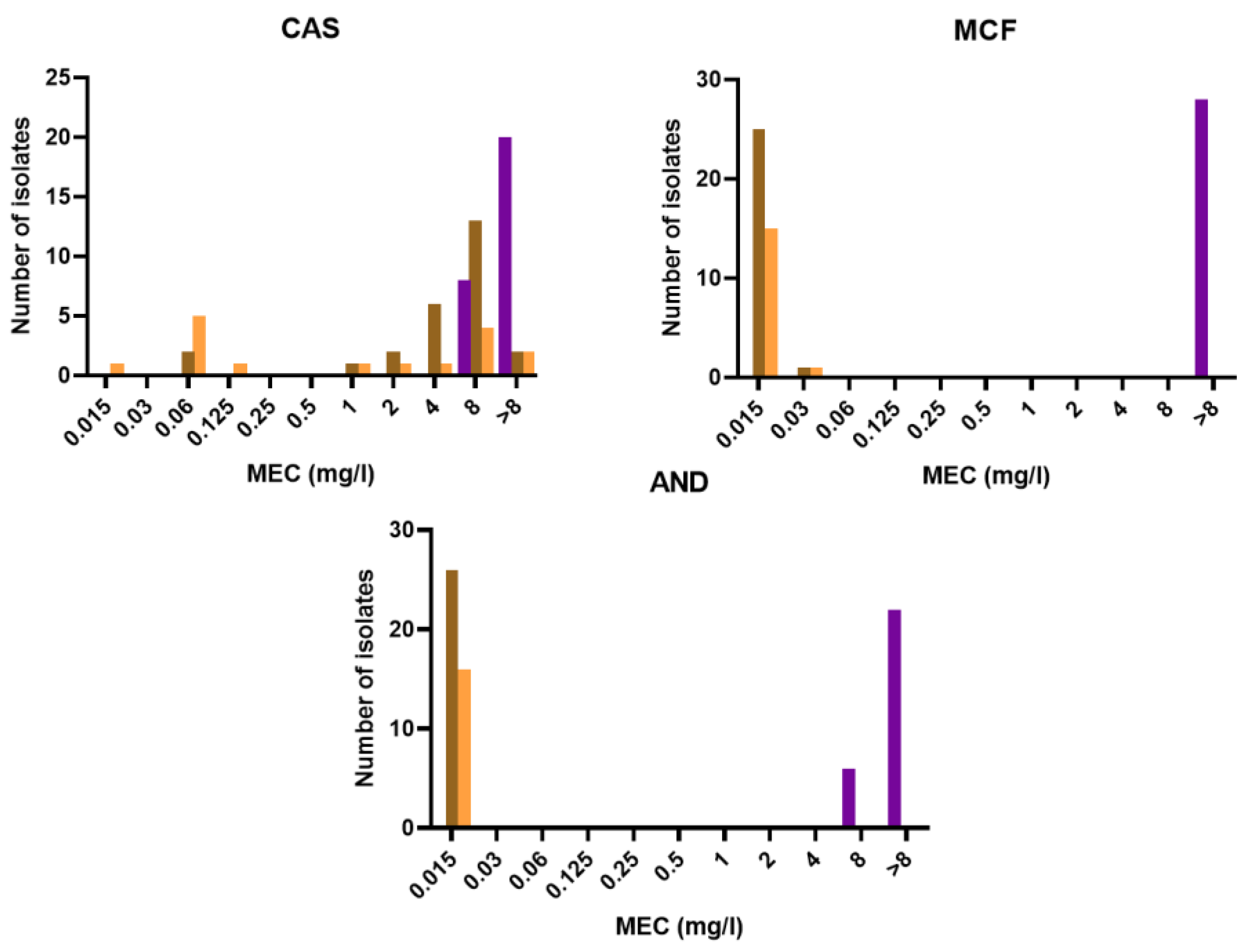
3.3. Antifungal Susceptibility
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Samson, R. Paecilomyces and some allied hyphomycetes. Stud. Mycol. 1974, 6, 1–119. [Google Scholar]
- Hoenigl, M.; Salmanton-García, J.; Walsh, T.J.; Nucci, M.; Neoh, C.F.; Jenks, J.D.; Lackner, M.; Sprute, R.; Al-Hatmi, A.M.S.; Bassetti, M.; et al. Global guideline for the diagnosis and management of rare mould infections: An initiative of the European Confederation of Medical Mycology in cooperation with the Tnternational Society for Human and Animal Mycology and the American Society for Microbiology. Lancet Infect. Dis. 2021, 21, e246–e257. [Google Scholar] [CrossRef] [PubMed]
- Eren, D.; Eroglu, E.; Ulu Kilic, A.; Atalay, M.A.; Mumcu, N.; Sipahioglu, M.H.; Canoz, O.; Koc, A.N.; Oymak, O. Cutaneous clcerations caused by Paecilomyces variotii in a renal transplant recipient. Transpl. Infect. Dis. 2018, 20, e12871. [Google Scholar] [CrossRef] [PubMed]
- Sprute, R.; Salmanton-García, J.; Sal, E.; Malaj, X.; Falces-Romero, I.; Hatvani, L.; Heinemann, M.; Klimko, N.; López-Soria, L.; Meletiadis, J.; et al. Characterization and outcome of invasive infections due to Paecilomyces variotii: Analysis of patients from the FungiScope® registry and literature reports. J. Antimicrob. Chemother. 2021, 76, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Sprute, R.; Salmanton-García, J.; Sal, E.; Malaj, X.; Ráčil, Z.; Ruiz de Alegría Puig, C.; Falces-Romero, I.; Barać, A.; Desoubeaux, G.; Kindo, A.J.; et al. Invasive infections with Purpureocillium lilacinum: Clinical characteristics and outcome of 101 cases from FungiScope® and the literature. J. Antimicrob. Chemother. 2021, 76, 1593–1603. [Google Scholar] [CrossRef]
- Pastor, F.J.; Guarro, J. Clinical manifestations, treatment and outcome of Paecilomyces lilacinus infections. Clin. Microbiol. Infect. 2006, 12, 948–960. [Google Scholar] [CrossRef]
- Chen, Y.-T.; Yeh, L.-K.; Ma, D.H.K.; Lin, H.-C.; Sun, C.-C.; Tan, H.-Y.; Chen, H.-C.; Chen, S.-Y.; Sun, P.-L.; Hsiao, C.-H. Paecilomyces/Purpureocillium keratitis: A consecutive study with a case series and literature review. Med. Mycol. 2020, 58, 293–299. [Google Scholar] [CrossRef]
- Feldman, R.; Cockerham, L.; Buchan, B.W.; Lu, Z.; Huang, A.M. Treatment of Paecilomyces variotii pneumonia with posaconazole: Case report and literature review. Mycoses 2016, 59, 746–750. [Google Scholar] [CrossRef]
- Luangsa-ard, J.J.; Hywel-Jones, N.L.; Samson, R.A. The polyphyletic nature of Paecilomyces sensu lato based on 18S-Generated rDNA phylogeny. Mycologia 2004, 96, 773–780. [Google Scholar] [CrossRef]
- Samson, R.A.; Houbraken, J.; Varga, J.; Frisvad, J.C. Polyphasic taxonomy of the heat resistant ascomycete genus Byssochlamys and its Paecilomyces anamorphs. Persoonia 2009, 22, 14–27. [Google Scholar] [CrossRef]
- Luangsa-Ard, J.; Houbraken, J.; van Doorn, T.; Hong, S.-B.; Borman, A.M.; Hywel-Jones, N.L.; Samson, R.A. Purpureocillium, a new genus for the medically important Paecilomyces lilacinus. FEMS Microbiol. Lett. 2011, 321, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Tortorano, A.M.; Richardson, M.; Roilides, E.; van Diepeningen, A.; Caira, M.; Munoz, P.; Johnson, E.; Meletiadis, J.; Pana, Z.-D.; Lackner, M.; et al. ESCMID and ECMM joint guidelines on diagnosis and management of hyalohyphomycosis: Fusarium spp., Scedosporium spp. and others. Clin. Microbiol. Infect. 2014, 20, 27–46. [Google Scholar] [CrossRef] [PubMed]
- Castelli, M.V.; Alastruey-Izquierdo, A.; Cuesta, I.; Monzon, A.; Mellado, E.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M. Susceptibility testing and molecular classification of Paecilomyces spp. Antimicrob. Agents Chemother. 2008, 52, 2926–2928. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, C.; Pujol, I.; Sala, J.; Guarro, J. Antifungal susceptibilities of Paecilomyces species. Antimicrob. Agents Chemother. 1998, 42, 1601–1604. [Google Scholar] [CrossRef] [PubMed]
- Houbraken, J.; Verweij, P.E.; Rijs, A.J.M.M.; Borman, A.M.; Samson, R.A. Identification of Paecilomyces variotii in clinical samples and settings. J. Clin. Microbiol. 2010, 48, 2754–2761. [Google Scholar] [CrossRef]
- MSI 2 Platform Database. Available online: https://msi.happy-dev.fr/ (accessed on 18 March 2022).
- Imbert, S.; Normand, A.C.; Gabriel, F.; Cassaing, S.; Bonnal, C.; Costa, D.; Lachaud, L.; Hasseine, L.; Kristensen, L.; Schuttler, C.; et al. Multi-centric evaluation of the online MSI Platform for the identification of cryptic and rare species of Aspergillus by MALDI-TOF. Med. Mycol. 2019, 57, 962–968. [Google Scholar] [CrossRef]
- Barker, A.P.; Horan, J.L.; Slechta, E.S.; Alexander, B.D.; Hanson, K.E. Complexities associated with the molecular and proteomic identification of Paecilomyces species in the clinical mycology laboratory. Med. Mycol. 2014, 52, 537–545. [Google Scholar] [CrossRef][Green Version]
- White, T. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc. A Guide Methods Appl. 1990, 315–322. [Google Scholar] [CrossRef]
- Vu, D.; Groenewald, M.; de Vries, M.; Gehrmann, T.; Stielow, B.; Eberhardt, U.; Al-Hatmi, A.; Groenewald, J.Z.; Cardinali, G.; Houbraken, J.; et al. Large-scale generation and analysis of filamentous fungal DNA barcodes boosts coverage for kingdom fungi and reveals thresholds for fungal species and higher taxon delimitation. Stud. Mycol. 2019, 92, 135–154. [Google Scholar] [CrossRef]
- EUCAST Method for Susceptibility Testing of Moulds. Available online: https://www.eucast.org/astoffungi/methodsinantifungalsusceptibilitytesting/ast_of_moulds/ (accessed on 10 October 2021).
- Jacobs, S.E.; Wengenack, N.L.; Walsh, T.J. Non-Aspergillus Hyaline Molds: Emerging causes of sino-pulmonary fungal infections and other invasive mycoses. Semin. Respir. Crit. Care Med. 2020, 41, 115–130. [Google Scholar] [CrossRef]
- Houbraken, J.; Kocsubé, S.; Visagie, C.M.; Yilmaz, N.; Wang, X.-C.; Meijer, M.; Kraak, B.; Hubka, V.; Bensch, K.; Samson, R.A.; et al. Classification of Aspergillus, Penicillium, Talaromyces and related genera (Eurotiales): An overview of families, genera, subgenera, sections, series and species. Stud. Mycol. 2020, 95, 5–169. [Google Scholar] [CrossRef] [PubMed]
- Ban, S.; Azuma, Y.; Sato, H.; Suzuki, K.-I.; Nakagiri, A. Isaria takamizusanensis is the anamorph of Cordyceps ryogamimontana, warranting a new combination, Purpureocillium takamizusanense Comb. Nov. Int. J. Syst. Evol. Microbiol. 2015, 65, 2459–2465. [Google Scholar] [CrossRef] [PubMed]
- Perdomo, H.; Cano, J.; Gené, J.; García, D.; Hernández, M.; Guarro, J. Polyphasic analysis of Purpureocillium lilacinum isolates from different origins and proposal of the new species Purpureocillium lavendulum. Mycologia 2013, 105, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Calvillo-Medina, R.P.; Ponce-Angulo, D.G.; Raymundo, T.; Müller-Morales, C.A.; Escudero-Leyva, E.; Campos Guillén, J.; Bautista-de Lucio, V.M. Purpureocillium roseum sp. Nov. A new ocular pathogen for Humans and mice resistant to antifungals. Mycoses 2021, 64, 162–173. [Google Scholar] [CrossRef]
- Chen, Y.-S.; Liu, Y.-H.; Teng, S.-H.; Liao, C.-H.; Hung, C.-C.; Sheng, W.-H.; Teng, L.-J.; Hsueh, P.-R. Evaluation of the Matrix-Assisted Laser Desorption/Ionization Time-of-Flight mass spectrometry Bruker Biotyper for identification of Penicillium marneffei, Paecilomyces species, Fusarium solani, Rhizopus species, and Pseudallescheria boydii. Front. Microbiol. 2015, 6, 679. [Google Scholar] [CrossRef]
- Carolus, H.; Pierson, S.; Lagrou, K.; Van Dijck, P. Amphotericin B and other polyenes-discovery, clinical use, mode of action and drug resistance. J. Fungi 2020, 6, 321. [Google Scholar] [CrossRef] [PubMed]
- Blum, G.; Hörtnagl, C.; Jukic, E.; Erbeznik, T.; Pümpel, T.; Dietrich, H.; Nagl, M.; Speth, C.; Rambach, G.; Lass-Flörl, C. New insight into amphotericin B resistance in Aspergillus terreus. Antimicrob. Agents Chemother. 2013, 57, 1583–1588. [Google Scholar] [CrossRef] [PubMed]
- Jukic, E.; Blatzer, M.; Posch, W.; Steger, M.; Binder, U.; Lass-Flörl, C.; Wilflingseder, D. Oxidative stress response tips the balance in Aspergillus terreus amphotericin B resistance. Antimicrob. Agents Chemother. 2017, 61, e00670-17. [Google Scholar] [CrossRef]
- Posch, W.; Blatzer, M.; Wilflingseder, D.; Lass-Flörl, C. Aspergillus terreus: Novel lessons learned on amphotericin B resistance. Med. Mycol. 2018, 56, S73–S82. [Google Scholar] [CrossRef]
- Sharma, C.; Chowdhary, A. Molecular bases of antifungal resistance in filamentous fungi. Int. J. Antimicrob. Agents 2017, 50, 607–616. [Google Scholar] [CrossRef]

| CBS No | Gene Bank Accession N° | ||
|---|---|---|---|
| Species | ITS [20] | D1D2 [20] | |
| CBS 372.70 T | Paecilomyces maximus | MH859719.1 | MH871470.1 |
| CBS 371.70 T | Paecilomyces maximus | MH859718.1 | MH871469.1 |
| CBS 339.51 | Paecilomyces variotii | MH856887.1 | MH868409.1 |
| CBS 338.51 | Paecilomyces variotii | MH856886.1 | MH868408.1 |
| CBS 284.48 T | Paecilomyces divaricatus | MH856344.1 | MH867896.1 |
| CBS 368.70 | Paecilomyces dactylethromorphus | MH859715.1 | MH871467.1 |
| CBS 129474 | Purpureocillium lilacinum | MH865347.1 | MH876802.1 |
| CBS 346.51 | Purpureocillium lilacinum | MH856891.1 | MH868413.1 |
| CBS 528.71 T | Thermoascus thermophilus | MH860254.1 | MH872018.1 |
| CBS 128677 T | Purpureocillium lavendulum | MH864976.1 | MH876429.1 |
| CBS 182.27 T | Marquandomyces marquandii | MH854923.1 | MH866418.1 |
| CBS 156.65 | Cordyceps farinosa | MH858528.1 | MH870163.1 |
| Species and Drug | Range | GM | MIC50/MEC50 | MIC90/MEC90 |
|---|---|---|---|---|
| (mg/L) | (mg/L) | (mg/L) | (mg/L) | |
| Purpureocillium lilacinum (n = 28) | ||||
| VRC | 0.06–4 | 0.320 | 0.25 | 0.5 |
| ITC | 0.5–8 | 1.414 | 1 | 2 |
| PCZ | 0.06–0.5 | 0.262 | 0.25 | 0.5 |
| ISA | 0.125–4 | 0.305 | 0.25 | 1 |
| CAS | 8–16 | 13.125 | 16 | 16 |
| MCF | 16–16 | 16 | 16 | 16 |
| AND | 8–16 | 13.792 | 16 | 16 |
| AMB | 16–16 | 16 | 16 | 16 |
| Paecilomyces variotii stricto sensu (n = 26) | ||||
| VRC | 0.25–16 | 4.108 | 4 | 16 |
| ITC | 0.015–0.5 | 0.130 | 0.125 | 0.25 |
| PCZ | 0.015–0.25 | 0.080 | 0.06 | 0.125 |
| ISA | 0.06–16 | 5.499 | 8 | 16 |
| CAS | 0.06–16 | 4.095 | 8 | 8 |
| MCF | 0.015–0.03 | 0.015 | 0.015 | 0.015 |
| AND | 0.015–0.015 | 0.015 | 0.015 | 0.015 |
| AMB | 0.06–0.5 | 0.171 | 0.25 | 0.25 |
| Paecilomyces maximus (n = 16) | ||||
| VRC | 0.5–16 | 8.775 | 8 | 16 |
| ITC | 0.125–1 | 0.379 | 0.5 | 0.5 |
| PCZ | 0.125–0.5 | 0.228 | 0.25 | 0.25 |
| ISA | 4–16 | 12.699 | 16 | 16 |
| CAS | 0.015–16 | 0.746 | 1 | 8 |
| MCF | 0.015–0.06 | 0.016 | 0.015 | 0.015 |
| AND | 0.015–0.015 | 0.015 | 0.015 | 0.015 |
| AMB | 0.125–0.5 | 0.262 | 0.25 | 0.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monpierre, L.; Aït-Ammar, N.; Valsecchi, I.; Normand, A.-C.; Guitard, J.; Riat, A.; Huguenin, A.; Bonnal, C.; Sendid, B.; Hasseine, L.; et al. Species Identification and In Vitro Antifungal Susceptibility of Paecilomyces/Purpureocillium Species Isolated from Clinical Respiratory Samples: A Multicenter Study. J. Fungi 2022, 8, 684. https://doi.org/10.3390/jof8070684
Monpierre L, Aït-Ammar N, Valsecchi I, Normand A-C, Guitard J, Riat A, Huguenin A, Bonnal C, Sendid B, Hasseine L, et al. Species Identification and In Vitro Antifungal Susceptibility of Paecilomyces/Purpureocillium Species Isolated from Clinical Respiratory Samples: A Multicenter Study. Journal of Fungi. 2022; 8(7):684. https://doi.org/10.3390/jof8070684
Chicago/Turabian StyleMonpierre, Lorra, Nawel Aït-Ammar, Isabel Valsecchi, Anne-Cécile Normand, Juliette Guitard, Arnaud Riat, Antoine Huguenin, Christine Bonnal, Boualem Sendid, Lilia Hasseine, and et al. 2022. "Species Identification and In Vitro Antifungal Susceptibility of Paecilomyces/Purpureocillium Species Isolated from Clinical Respiratory Samples: A Multicenter Study" Journal of Fungi 8, no. 7: 684. https://doi.org/10.3390/jof8070684
APA StyleMonpierre, L., Aït-Ammar, N., Valsecchi, I., Normand, A.-C., Guitard, J., Riat, A., Huguenin, A., Bonnal, C., Sendid, B., Hasseine, L., Raberin, H., Dehais, M., Ranque, S., Hennequin, C., Piarroux, R., Dannaoui, E., & Botterel, F. (2022). Species Identification and In Vitro Antifungal Susceptibility of Paecilomyces/Purpureocillium Species Isolated from Clinical Respiratory Samples: A Multicenter Study. Journal of Fungi, 8(7), 684. https://doi.org/10.3390/jof8070684








