Mechanical, Physical, and Chemical Properties of Mycelium-Based Composites Produced from Various Lignocellulosic Residues and Fungal Species
Abstract
1. Introduction
2. Materials and Methods
2.1. Sources of Fungal Strains and Culture Conditions
2.2. Sources of Lignocellulosic Residues and Preparation
2.3. Preparation of Mycelium-Based Composites and Mould Design
2.3.1. Inoculum Preparation of Fungal Mycelium
2.3.2. Mycelial Growth on Substrates
2.3.3. Mold Preparation
2.3.4. Mycelium-Based Composite Fabrication, Preparation for Testing, and Moisture Content
2.4. Scanning Electron Microscope Observations
2.5. Determination of Physical Properties
2.5.1. Density
2.5.2. Water Absorption
2.5.3. Shrinkage
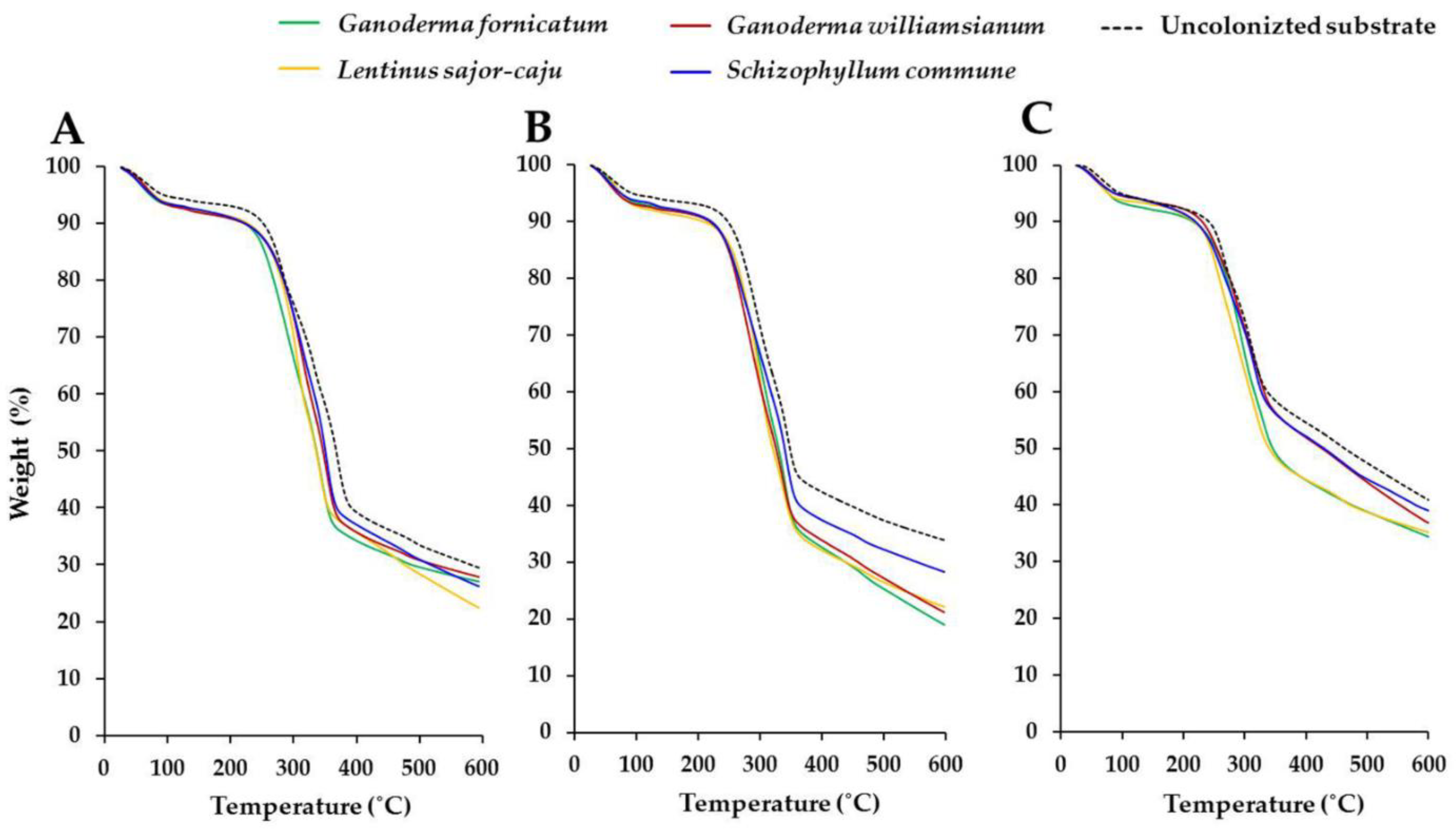
2.5.4. Thermal Degradation
2.6. Determination of Mechanical Properties
2.6.1. Compression Strength
2.6.2. Tensile Strength
2.6.3. Flexural Strength
2.6.4. Impact Strength
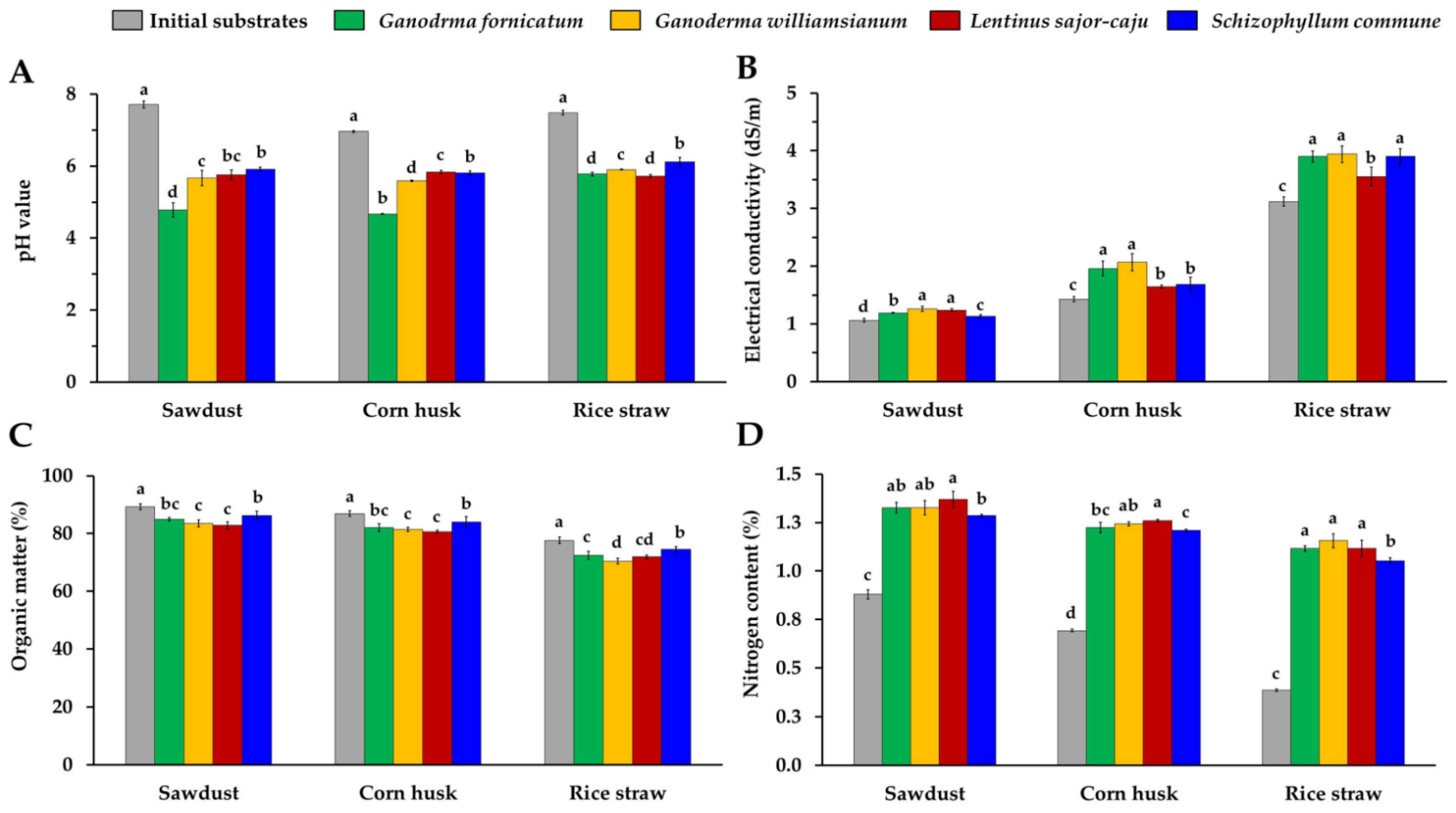
2.7. Determination of Chemical Properties
2.8. Statistical Analysis
3. Results and Discussion
3.1. Scanning Electron Microscope Observations and Moisture Content
3.2. Determination of Physical Properties
3.2.1. Density
3.2.2. Water Absorption
3.2.3. Shrinkage
3.2.4. Thermal Degradation
3.3. Determination of Mechanical Properties
3.3.1. Compression Strength
3.3.2. Tensile Strength
3.3.3. Flexural Strength
3.3.4. Impact Strength
3.4. Determination of Chemical Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Acevedo, M.D.; Urena, L.J.B.; García, F.J.C.; Ferre, F.C.F. Agricultural waste: Review of the evolution, approaches and perspectives on alternative uses. Glob. Ecol. Conserv. 2020, 22, 00902. [Google Scholar]
- Santana-Méridas, O.; González-Coloma, A.; Sánchez-Vioque, R. Agricultural residues as a source of bioactive natural products. Phytochem. Rev. 2012, 11, 447–466. [Google Scholar] [CrossRef]
- Cherubin, M.R.; Oliveira, D.M.D.S.; Feigl, B.J.; Pimentel, L.G.; Lisboa, I.P.; Gmach, M.R.; Varanda, L.L.; Morais, M.C.; Satiro, L.S.; Popin, G.V.; et al. Crop residue harvest for bioenergy production and its implications on soil functioning and plant growth: A review. Sci. Agric. 2018, 75, 255–272. [Google Scholar] [CrossRef]
- Tassie, K.; Endalew, B. Willingness to pay for improved solid waste management services and associated factors among urban households: One and one half bounded contingent valuation study in Bahir Dar city, Ethiopia. Cogent Environ. Sci. 2020, 6, 1807275. [Google Scholar] [CrossRef]
- Pandey, S. Wood waste utilization and associated product development from under-utilized low-quality wood and its prospects in Nepal. SN Appl. Sci. 2022, 4, 168. [Google Scholar] [CrossRef]
- Launio, C.C.; Asis, C.A., Jr.; Manalili, R.G.; Javier, E.F.; Belizario, A.F. What factors influence choice of waste management practice? Evidence from rice straw management in the Philippines. Waste Manag. Res. 2014, 32, 140–148. [Google Scholar] [CrossRef]
- Arunrat, N.; Pumijumnong, N.; Sereenonchai, S. Air-Pollutant Emissions from Agricultural Burning in Mae Chaem Basin, Chiang Mai Province, Thailand. Atmosphere 2018, 9, 145. [Google Scholar] [CrossRef]
- Sereenonchai, S.; Arunrat, N.; Kamnoonwatana, D. Risk perception on haze pollution and willingness to pay for self-protection and haze management in Chiang Mai Province, northern Thailand. Atmosphere 2020, 11, 600. [Google Scholar] [CrossRef]
- Sereenonchai, S.; Arunrat, N. Farmers’ perceptions, insight behavior and communication strategies for rice straw and stubble management in Thailand. Agronomy 2022, 12, 200. [Google Scholar] [CrossRef]
- Donner, M.; Gohier, R.; de Vries, H. A new circular business model typology for creating value from agro-waste. Sci. Total Environ. 2020, 716, 137065. [Google Scholar] [CrossRef]
- Guerriero, G.; Hausman, J.F.; Strauss, J.; Ertan, H.; Siddiqui, K.S. Lignocellulosic biomass: Biosynthesis, degradation, and industrial utilization. Eng. Life Sci. 2016, 16, 1–16. [Google Scholar] [CrossRef]
- Eriksen, M.; Lebreton, L.C.M.; Carson, H.S.; Thiel, M.; Moore, C.J.; Borerro, J.C.; Reisser, J. Plastic pollution in the world’s oceans: More than 5 trillion plastic pieces weighing over 250,000 tons afloat at sea. PLoS ONE 2014, 9, e111913. [Google Scholar] [CrossRef]
- Li, W.C.; Tse, H.F.; Fok, L. Plastic waste in the marine environment: A review of sources, occurrence and effects. Sci. Total Environ. 2016, 566, 333–349. [Google Scholar] [CrossRef]
- Sadh, P.K.; Duhan, S.; Duhan, J.S. Agro-industrial wastes and their utilization using solid state fermentation: A review. Bioresour. Bioprocess. 2018, 5, 1. [Google Scholar] [CrossRef]
- Santolini, E.; Bovo, M.; Barbaresi, A.; Torreggiani, D.; Tassinari, P. Turning agricultural wastes into biomaterials: Assessing the sustainability of scenarios of circular valorization of corn cob in a life-cycle perspective. Appl. Sci. 2021, 11, 6281. [Google Scholar] [CrossRef]
- Song, J.H.; Murphy, R.J.; Narayan, R.; Davies, G.B.H. Biodegradable and compostable alternatives to conventional plastics. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2127–2139. [Google Scholar] [CrossRef]
- Pohl, C.; Schmidt, B.; Nunez Guitar, T.; Klemm, S.; Gusovius, H.J.; Platzk, S.; Kruggel-Emden, H.; Klunker, A.; Völlmecke, C.; Fleck, C.; et al. Establishment of the basidiomycete Fomes fomentarius for the production of composite materials. Fungal Biol. Biotechnol. 2022, 9, 4. [Google Scholar] [CrossRef]
- Alemu, D.; Tafesse, M.; Mondal, A.K. Mycelium-based composite: The future sustainable biomaterial. Int. J. Biomater. 2022, 2022, 173–182. [Google Scholar] [CrossRef]
- Aiduang, W.; Chanthaluck, A.; Kumla, J.; Jatuwong, K.; Srinuanpan, S.; Waroonkun, T.; Oranratmanee, R.; Suwannarach, N.; Lumyong, S. Amazing Fungi for Eco-Friendly Composite Materials: A Comprehensive Review. J. Fungi 2022, 8, 842. [Google Scholar] [CrossRef]
- Butu, A.; Rodino, S.; Miu, B.; Butu, M. Mycelium-based materials for the ecodesign of bioeconomy. Dig. J. Nanomater. Biostruct. 2020, 15, 1129–1140. [Google Scholar]
- Jones, M.; Mautner, A.; Luenco, S.; Bismarck, A.; John, S. Engineered mycelium composite construction materials from fungal biorefineries: A critical review. Mater. Des. 2020, 187, 108397. [Google Scholar] [CrossRef]
- Manan, S.; Ullah, M.W.; Ul-Islam, M.; Atta, O.M.; Yang, G. Synthesis and applications of fungal mycelium-based advanced functional materials. J. Bioresour. Bioprod. 2021, 6, 1–10. [Google Scholar] [CrossRef]
- Yang, L.; Park, D.; Qin, Z. Material function of mycelium-based bio-composite: A review. Front. Mater. 2021, 8, 374. [Google Scholar] [CrossRef]
- Fairus, M.J.M.; Bahrin, E.K.; Natasha, E.; Arbaain, N.; Ramli, N. Mycelium-based composite: A way forward for renewable material. J. Sustain. Sci. Manag. 2022, 17, 271–280. [Google Scholar] [CrossRef]
- Haneef, M.; Ceseracciu, L.; Canale, C.; Bayer, I.S.; Heredia-Guerrero, J.A.; Athanassiou, A. Advanced materials from fungal mycelium: Fabrication and tuning of physical properties. Sci. Rep. 2017, 7, 41292. [Google Scholar] [CrossRef]
- Appels, F.V.W.; Camere, S.; Montalti, M.; Karana, E.; Jansen, K.M.B.; Dijksterhuis, J.; Krijgsheld, P.; Wosten, H.A.B. Fabrication factors influencing mechanical, moisture and water related properties of mycelium-based composites. Mater. Des. 2019, 161, 64–71. [Google Scholar] [CrossRef]
- Soh, E.; Chew, Z.Y.; Saeidi, N.; Javadian, A.; Hebel, D.; Le Ferrand, H. Development of an extrudable paste to build mycelium-bound composites. Mater. Des. 2020, 195, 109058. [Google Scholar] [CrossRef]
- Dias, P.P.; Jayasinghe, L.B.; Waldmann, D. Investigation of mycelium-miscanthus composites as building insulation material. Results Mater. 2021, 10, 100189. [Google Scholar] [CrossRef]
- Lee, T.; Choi, J. Mycelium-composite panels for atmospheric particulate matter adsorption. Results Mater. 2021, 11, 100208. [Google Scholar] [CrossRef]
- Pelletier, M.G.; Holt, G.A.; Wanjura, J.D.; Lara, A.J.; Tapia-Carillo, A.; McIntyre, G.; Bayer, E. An evaluation study of pressurecompressed acoustic absorbers grown on agricultural by-products. Ind. Crops Prod. 2017, 95, 342–347. [Google Scholar] [CrossRef]
- Pelletier, M.G.; Holt, G.A.; Wanjura, J.D.; Greetham, L.; McIntyre, G.; Bayer, E.; Kaplan-Bie, J. Acoustic evaluation of mycological biopolymer, an all-natural closed cell foam alternative. Ind. Crops Prod. 2019, 139, 111533. [Google Scholar] [CrossRef]
- De Lima, G.G.; Schoenherr, Z.C.P.; Magalhães, W.L.E.; Tavares, L.B.B.; Helm, C.V. Enzymatic activities and analysis of a mycelium-based composite formation using peach palm (Bactris gasipaes) residues on Lentinula edodes. Bioresour. Bioprocess. 2020, 7, 58. [Google Scholar] [CrossRef]
- Salami, A.O.; Bankole, F.A.; Olawole, O.I. Effect of different substrates on the growth and protein content of oyster mushroom (Pleurotus florida). Int. J. Biol. Chem. Sci. 2016, 10, 475–485. [Google Scholar] [CrossRef]
- Kupradi, C.; Khongla, C.; Musika, S.; Ranok, A.; Tamaruay, K.; Woraratphoka, J.; Mangkalanan, S. Cultivation of Lentinus squarrosulus and Pleurotus ostreatus on cassava bagasse based substrates. Int. J. Agric. Technol. 2017, 13, 883–892. [Google Scholar]
- Xing, Y.; Brewer, M.; El-Gharabawy, H.; Griffith, G.; Jones, P. Growing and testing mycelium bricks as building insulation materials. In Proceedings of the EEEP2017: International Conference on Energy Engineering and Environmental Protection, Sanya, China, 20–22 November 2017; IOP Conference Series: Earth and Environmental Science. p. 022032. [Google Scholar]
- Elsacker, E.; Vandelook, S.; Brancart, J.; Peeters, E.; Laet, L.D. Mechanical, physical and chemical characterisation of mycelium–based composites with different types of lignocellulosic substrates. PLoS ONE 2019, 7, e0213954. [Google Scholar] [CrossRef]
- Venkatasubramanian, H.; Chaithanyan, C.; Raghuraman, S.; Panneerselvam, T. Evaluation of mechanical properties of abaca-glass-banana fiber reinforced hybrid composites. Int. J. Innov. Res. Technol. Sci. Eng. 2014, 3, 8169–8177. [Google Scholar]
- Ilyas, R.A.; Sapuan, S.M.; Atiqah, A.; Ibrahim, R.; Abral, H.; Ishak, M.R.; Zainudin, E.S.; Nurazzi, N.M.; Atikah, M.S.N.; Ansari, M.N.M.; et al. Sugar palm (Arenga pinnata [Wurmb.] Merr) starch films containing sugar palm nanofibrillated cellulose as reinforcement: Water barrier properties. Polym. Compos. 2020, 41, 459–467. [Google Scholar] [CrossRef]
- Attias, N.; Danai, O.; Tarazi, E.; Pereman, I.; Grobman, Y.J. Implementing bio-design tools to develop mycelium-based products. Des. J. 2019, 22, 1647–1657. [Google Scholar] [CrossRef]
- Sánchez, Ó.J.; Montoya, S. Assessment of polysaccharide and biomass production from three white-rot fungi by solid-state fermentation using wood and agro-industrial residues: A kinetic approach. Forests 2020, 11, 1055. [Google Scholar] [CrossRef]
- Attias, N.; Danai, O.; Ezov, N.; Tarazi, E.; Grobman, Y.J. Developing novel applications of mycelium based bio-composite materials for design and architecture. In Proceedings of the Building with Bio-based Materials: Best Practice and Performance Specification, Zagreb, Croatia, 9–16 September 2017; pp. 1–10. [Google Scholar]
- Gou, L.; Li, S.; Yin, J.; Li, T.; Liu, X. Morphological and physico-mechanical properties of mycelium biocomposites with natural reinforcement particles. Constr. Build. Mater. 2021, 304, 124656. [Google Scholar] [CrossRef]
- Deacon, J.W. Fungal Biology; Blackwell-Wiley: Oxford, UK, 2006; 95p. [Google Scholar]
- Velasco, P.M.; Ortiz, M.P.M.; Giro, M.A.M.; Castelló, M.C.J.; Velasco, L.M. Development of better insulation bricks by adding mushroom compost wastes. Energy Build. 2014, 80, 17–22. [Google Scholar] [CrossRef]
- Girometta, C.; Picco, A.M.; Baiguera, R.M.; Dondi, D.; Babbini, S.; Cartabia, M.; Pellegrini, M.; Savino, E. Physico-mechanical and thermodynamic properties of mycelium-based biocomposites: A review. Sustainability 2019, 11, 281. [Google Scholar] [CrossRef]
- Chan, X.Y.; Saeidi, N.; Javadian, A.; Hebel, D.E.; Gupta, M. Mechanical properties of dense mycelium-bound composites under accelerated tropical weathering conditions. Sci. Rep. 2021, 11, 22112. [Google Scholar] [CrossRef]
- Tacer-Caba, Z.; Varis, J.J.; Lankinen, P.; Mikkonen, K.S. Comparison of novel fungal mycelia strains and sustainable growth substrates to produce humidity-resistant biocomposites. Mater. Des. 2020, 192, 108728. [Google Scholar] [CrossRef]
- Bruscato, C.; Malvessi, E.; Brandalise, R.N.; Camassola, M. High performance of macrofungi in the production of mycelium-based biofoams using sawdust—Sustainable technology for waste reduction. J. Clean. Prod. 2019, 234, 225–232. [Google Scholar] [CrossRef]
- Monteiro, S.N.; Lopes, F.P.D.; Barbosa, A.P.; Bevitori, A.B.; Silva, I.L.A.D.; Costa, L.L.D. Natural lignocellulosic fibers as engineering materials—An overview. Metall. Mater. Trans. A 2011, 42, 2963–2974. [Google Scholar] [CrossRef]
- Ferraz, P.F.P.; Mendes, R.F.; Marin, D.B.; Paes, J.L.; Cecchin, D.; Barbari, M. Agricultural residues of lignocellulosic materials in cement composites. Appl. Sci. 2020, 10, 8019. [Google Scholar] [CrossRef]
- Yang, S.Y. (Ed.) Mechanical properties of polyimide foams with different densities. In Advanced Polyimide Materials: Synthesis, Characterization, and Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–498. [Google Scholar]
- Omnexus. Mechanical Properties of Plastics. Available online: https://omnexus.specialchem.com/polymer-properties/properties/toughness (accessed on 20 August 2022).
- Du, B.X.; He, Z.Y.; Du, Q.; Guo, Y.G. Effects of water absorption on surface charge and dielectric breakdown of polyimide/Al2O3 nanocomposite films. IEEE Trans. Dielectr. Electr. Insul. 2016, 23, 134–141. [Google Scholar] [CrossRef]
- Shi, Y.; Hu, A.; Wang, Z.; Li, K.; Yang, S. Closed-cell rigid polyimide foams for high-temperature applications: The effect of structure on combined properties. Polymers 2021, 13, 4434. [Google Scholar] [CrossRef]
- Wang, L.; Hu, A.; Fan, L.; Yang, S. Structures and properties of closed-cell polyimide rigid foams. J. Appl. Polym. Sci. 2013, 130, 3282–3291. [Google Scholar] [CrossRef]
- Wei, J.; Wei, C.; Su, L.; Fu, J.; Lv, J. Synergistic reinforcement of phenol-formaldehyde resin composites by poly (hexanedithiol)/graphene oxide. J. Mater. Sci. Chem. Eng. 2015, 3, 56. [Google Scholar] [CrossRef]
- Li, Y.C.; Tang, K.J.; Jin, F.L.; Park, S.J. Enhanced thermal stability and impact strength of phenolic formaldehyde resin using acid-treated basalt scales. J. Appl. Polym. Sci. 2022, 139, e52827. [Google Scholar] [CrossRef]
- Smirnov, K.V.; Chuklanov, V.Y.; Smirnova, N.N. Thermal insulation materials based on polyurethane foam modified by the polymethylphenylsiloxane. In Proceedings of the International Conference on Materials Physics, Building Structures and Technologies in Construction, Industrial and Production Engineering, Vladimir, Russia, 27–28 April 2020; IOP Conference Series: Materials Science and Engineering. IOP Publishing: Bristol, UK, 2020; p. 012104. [Google Scholar]
- Dizon, J.R.C.; Valino, A.D.; Souza, L.R.; Espera, A.H.; Chen, Q.; Advincula, R.C. Three-dimensional-printed molds and materials for injection molding and rapid tooling applications. MRS Commun. 2019, 9, 1267–1283. [Google Scholar] [CrossRef]
- Forest Products Laboratory, Forest Service, U.S. Department of Agriculture. Manufacture and General Characteristics of Flat Plywood; Forest Service, U.S. Department of Agriculture: Madison, WI, USA, 1964; pp. 1–15.
- Stark, N.; Cai, Z.; Carll, C. Wood-based composite materials: Panel products, glued laminated timber, structural composite lumber, and wood-nonwood composites. In Wood Handbook: Wood as an Engineering Material; Ross, R.J., Ed.; U.S. Department of Agriculture: Madison, WI, USA, 2010; pp. 1–508. [Google Scholar]
- Ashby, M.F. Materials and the Environment: Eco-Informed Material Choice; Butterworth Heinemann: Oxford, UK, 2012. [Google Scholar]
- MatWeb LLC. Material Property Data. Available online: http://www.matweb.com/2019 (accessed on 20 August 2022).
- Azahari, M.S.M.; Rus, A.Z.M.; Kormin, S.; Zaliran, M.T. An acoustic study of Shorea leprosula wood fiber filled polyurethane composite foam. Malays. J. Anal. Sci. 2018, 22, 1031–1039. [Google Scholar]
- Filip, D.; Macocinschi, D.; Vlad, S. Thermogravimetric study for polyurethane materials for biomedical applications. Compos. B Eng. 2011, 42, 1474–1479. [Google Scholar] [CrossRef]
- NPCS Board of Consultants & Engineers. How to Start a Phenolic Resin Production Business. In Phenolic Resins Technology Handbook; Niir Project Consultancy Services: Delhi, India, 2019; 624p. [Google Scholar]
- Niu, M.; Wang, G. The preparation and performance of phenolic foams modified by active polypropylene glycol. Cell. Polym. 2013, 32, 155–172. [Google Scholar] [CrossRef]
- Jalalian, M.; Jiang, Q.; Coulon, A.; Storb, M.; Woodward, R.; Bismarck, A. Mechanically whipped phenolic froths as versatile templates for manufacturing phenolic and carbon foams. Mater. Des. 2019, 168, 107658. [Google Scholar] [CrossRef]
- Papadopoulou, E.; Chrissafis, K. Thermal study of phenol–formaldehyde resin modified with cashew nut shell liquid. Thermochim. Acta. 2011, 512, 105–109. [Google Scholar] [CrossRef]
- Tailor, R.B.; Ramachandran, M.; Raichurkar, P.P. Review on non-woven polymeric gaskets their characteristics and applications. Int. J. Text. Eng. Process. 2017, 3, 14–21. [Google Scholar]
- Deng, H.; Reynolds, C.T.; Cabrera, N.O.; Barkoula, N.M.; Alcock, B.; Peijs, T. The water absorption behaviour of all-polypropylene composites and its effect on mechanical properties. Compos. Part B Eng. 2010, 41, 268–275. [Google Scholar] [CrossRef]
- Dou, Y.; Rodrigue, D. Morphological, thermal and mechanical properties of polypropylene foams via rotational molding. Cell. Polym. 2021, 40, 198–211. [Google Scholar] [CrossRef]
- Zhu, L.H.; Sheng, J.F.; Guo, Z.F.; Ju, X.S.; Li, S.; Chen, Y.F.; Luo, J. Properties of polypropylene and surface modified glass-fibre composites. Polym. Polym. Compos. 2014, 22, 381–386. [Google Scholar] [CrossRef]
- Shen, J.; Li, X.; Yan, X. Mechanical and acoustic properties of jute fiber-reinforced polypropylene composites. ACS Omega 2021, 6, 31154–31160. [Google Scholar] [CrossRef]
- Da Costa Castro, C.D.P.; Dias, C.G.B.T.; de Assis Fonseca Faria, J. Production and evaluation of recycled polymers from açaí fibers. Mater. Res. 2010, 13, 159–163. [Google Scholar] [CrossRef]
- Handayani, S.U.; Fahrudin, M.; Mangestiyono, W.; Muhamad, A.F.H. Mechanical properties of commercial recycled polypropylene from plastic waste. J. Vocat. Stud. Appl. Res. 2021, 3, 1–4. [Google Scholar] [CrossRef]
- Goulart, S.A.S.; Oliveira, T.A.; Teixeira, A.; Miléo, P.C.; Mulinari, D.R. Mechanical behavior of polypropylene reinforced palm fibers composites. Procedia Eng. 2011, 10, 2034–2039. [Google Scholar] [CrossRef]
- Del Menezzi, C.H.S. New approaches for production of laminated wood products from tropical woods. In Proceedings of the World Conference on Timber Engineering, Vienna, Austria, 22–25 August 2016; pp. 1–7. [Google Scholar]
- Çolakoğlu, G.; Colak, S. The effects of waiting time of alder (Alnus glutinosa subsp. barbata) veneers before drying on shear and bending strength of plywood. Holz Roh Werkst. 2002, 60, 127–129. [Google Scholar] [CrossRef]
- Jivkov, V.; Simeonova, R.; Marinova, A. Influence of the veneer quality and load direction on the strength properties of beech plywood as structural material for furniture. Innov. Woodwork. Ind. Eng. 2013, 2, 86–92. [Google Scholar]
- Sinha, A.; Nairn, J.A.; Gupta, R. Thermal degradation of bending strength of plywood and oriented strand board: A kinetics approach. Wood Sci. Technol. 2011, 45, 315–330. [Google Scholar] [CrossRef]
- Jamalirad, L.; Doosthoseini, K.; Koch, G.; Mirshokraie, S.A.; Hedjazi, S. Physical and mechanical properties of plywood manufactured from treated red-heart beech (Fagus orientalis L.) wood veneers. BioResources 2011, 6, 3973–3986. [Google Scholar]
- Engineering Toolbox. Wood Beams-Strength of Material. 2009. Available online: https://www.engineeringtoolbox.com/woodbeams-strength-d_1480.html (accessed on 20 August 2022).
- Fateh, T.; Rogaume, T.; Luche, J.; Richard, F.; Jabouille, F. Kinetic and mechanism of the thermal degradation of a plywood by using thermogravimetry and Fourier-transformed infrared spectroscopy analysis in nitrogen and air atmosphere. Fire Saf. J. 2013, 58, 25–37. [Google Scholar] [CrossRef]
- Zabihzadeh, S.M. Water uptake and flexural properties of natural filler/HDPE composites. BioResources 2009, 5, 316–323. [Google Scholar]
- STRUCTAflor. Particleboard Flooring Fact Sheet Shrinkage of Laid Floors. Available online: https://www.timbertrading.com.au/wp-content/uploads/2020/11/STRUCTAFlor-particleboard-flooring-fact-sheet-shrinking.pdf (accessed on 12 October 2022).
- Mawardi, I.; Aprilia, S.; Faisal, M.; Rizal, S. Characterization of thermal bio-insulation materials based on oil palm wood: The effect of hybridization and particle size. Polymers 2021, 13, 3287. [Google Scholar] [CrossRef]
- Acda, M.N.; Cabangon, R.J. Termite resistance and physico-mechanical properties of particleboard using waste tobacco stalk and wood particles. Int. Biodeterior. Biodegrad. 2013, 85, 354–358. [Google Scholar] [CrossRef]
- Gößwald, J.; Barbu, M.C.; Petutschnigg, A.; Tudor, E.M. Binderless Thermal insulation panels made of spruce bark fibres. Polymers 2021, 13, 1799. [Google Scholar] [CrossRef]
- Ge, J.; Wang, R.Q.; Liu, L. Study on the thermal degradation kinetics of the common wooden boards. Procedia Eng. 2016, 135, 72–82. [Google Scholar] [CrossRef]
- Segovia, F.; Blanchet, P.; Auclair, N.; Essoua Essoua, G.G. Thermo-mechanical properties of a wood fiber insulation board using a bio-based adhesive as a binder. Buildings 2020, 10, 152. [Google Scholar] [CrossRef]
- Kallakas, H.; Närep, M.; Närep, A.; Poltimäe, T.; Kers, J. Mechanical and physical properties of industrial hemp-based insulation materials. Proc. Est. Acad. Sci. 2018, 67, 183–192. [Google Scholar] [CrossRef]
- Harshavardhan, A.; Muruganandam, L. Preparation and characteristic study of particle board from solid waste. In Proceedings of the 14th International Conference on Science, Engineering the Technology 14th (ICSET2017), Vellore, India, 2–3 May 2017; IOP Conference Series: Materials Science and Engineering. p. 032005. [Google Scholar]
- Muthuraj, R.; Lacoste, C.; Lacroix, P.; Bergeret, A. Sustainable thermal insulation biocomposites from rice husk, wheat husk, wood fibers and textile waste fibers: Elaboration and performances evaluation. Ind. Crops Prod. 2019, 135, 238–245. [Google Scholar] [CrossRef]
- Shakir, M.A.; Azahari, B.; Yusup, Y.; Yhaya, M.F.; Salehabadi, A.; Ahmad, M.I. Preparation and characterization of mycelium as a bio-matrix in fabrication of bio-composite. J. Adv. Res. Fluid Mech. Therm. Sci. 2020, 65, 253–263. [Google Scholar]
- Kuribayashi, T.; Lankinen, P.; Hietala, S.; Mikkonen, K.S. Dense and continuous networks of aerial hyphae improve flexibility and shape retention of mycelium composite in the wet state. Compos. Part A Appl. Sci. Manuf. 2022, 152, 106688. [Google Scholar] [CrossRef]
- Jones, M.; Bhat, T.; Wang, C.H.; Moinuddin, K.; John, S. Thermal degradation and fire reaction properties of mycelium composites. In Proceedings of the 21st International Conference on Composite Materials, Xi’an, China, 20–25 August 2017; pp. 1–9. [Google Scholar]
- Kumla, J.; Suwannarach, N.; Sujarit, K.; Penkhrue, W.; Kakumyan, P.; Jatuwong, K.; Vadthanarat, S.; Lumyong, S. Cultivation of mushrooms and their lignocellulolytic enzyme production through the utilization of agro-industrial waste. Molecules 2020, 25, 2811. [Google Scholar] [CrossRef]
- Song, S.T.; Saman, N.; Johari, K.; Mat, H.B. Removal of mercury (II) from aqueous solution by using rice residues. J. Teknol. 2013, 63, 67–73. [Google Scholar] [CrossRef]
- Pattanayak, S.S.; Laskar, S.H.; Sahoo, S. Investigation of organic corn husk-based flat microwave absorber. Int. J. Microw. Wirel. Technol. 2021, 13, 779–788. [Google Scholar] [CrossRef]
- Hassan, S.A.M.; Ahmad, M.S.; Samat, A.F.; Zakaria, N.Z.I.; Sohaimi, K.S.A.; Nordin, N. Comparison of glucose yield from rubberwood sawdust (RSD), growth medium (GM), and mushroom spent medium (MSM) under different sodium hydroxide pretreatment techniques. In Proceedings of the MATEC Web of Conferences, Penang, Malaysia, 6–7 December 2017; p. 06023. [Google Scholar]
- Robertson, O.; Høgdal, F.; Mckay, L.; Lenau, T. Fungal Future: A review of mycelium biocomposites as an ecological alter-native insulation material. In Proceedings of the Nord Design 2020, Lyngby, Denmark, 12–14 August 2020; pp. 1–13. [Google Scholar]
- Gumowska, A.; Robles, E.; Kowaluk, G. Evaluation of functional features of lignocellulosic particle composites containing biopolymer binders. Materials 2021, 14, 7718. [Google Scholar] [CrossRef]
- Holt, G.A.; Mcintyre, G.; Flagg, D.; Bayer, E.; Wanjura, J.D.; Pelletier, M.G. Fungal mycelium and cotton plant materials in the manufacture of biodegradable molded packaging material: Evaluation study of select blends of cotton byproducts. J. Biobased Mater. Bioenergy 2012, 6, 431–439. [Google Scholar] [CrossRef]
- Jones, M.; Bhat, T.; Huynh, T.; Kandare, E.; Yuen, R.; Wang, C.H.; John, S. Waste-derived low-cost mycelium composite construction materials with improved fire safety. Fire Mater. 2018, 42, 816–825. [Google Scholar] [CrossRef]
- Borsoi, C.; Scienza, L.C.; Zattera, A.J. Characterization of composites based on recycled expanded polystyrene reinforced with curaua fibers. J. Appl. Polym. Sci. 2013, 128, 653–659. [Google Scholar] [CrossRef]
- Olatunji, O.O.; Akinlabi, S.A.; Mashinini, M.P.; Fatoba, S.O.; Ajayi, O.O. Thermo-gravimetric characterization of biomass properties: A review. In Proceedings of the 3rd Annual Applied Science and Engineering Conference (AASEC 2018), Bandung, Indonesia, 18 April 2018; IOP Conference Series: Materials Science and Engineering. p. 012175. [Google Scholar]
- Jose, J.; Uvais, K.N.; Sreenadh, T.S.; Deepak, A.V.; Rejeesh, C.R. Investigations into the development of a mycelium bio-composite to substitute polystyrene in packaging applications. Arab. J. Sci. Eng. 2021, 46, 2975–2984. [Google Scholar] [CrossRef]
- Yeasmin, M.S.; Mondal, M.I.H. Synthesis of highly substituted carboxymethyl cellulose depending on cellulose particle size. Int. J. Biol. Macromol. 2015, 80, 725–731. [Google Scholar] [CrossRef]
- Ali, N.; Tabi, A.N.M.; Zakil, F.A.; Fauzai, W.N.F.M.; Hassan, O. Yield performance and biological efficiency of empty fruit bunch (EFB) and palm pressed fibre (PPF) as substrates for the cultivation of Pleurotus ostreatus. J. Teknol. 2013, 64, 93–99. [Google Scholar] [CrossRef]
- Hung, K.C.; Yeh, H.; Yang, T.C.; Wu, T.L.; Xu, J.W.; Wu, J.H. Characterization of wood-plastic composites made with different lignocellulosic materials that vary in their morphology, chemical composition and thermal stability. Polymers 2017, 9, 726. [Google Scholar] [CrossRef] [PubMed]
- Angelova, G.; Brazkova, M.; Stefanova, P.; Blazheva, D.; Vladev, V.; Petkova, N.; Slavov, A.; Denev, P.; Karashanova, D.; Zaharieva, R.; et al. Waste rose flower and lavender straw biomass—An innovative lignocellulose feedstock for mycelium bio-materials development using newly isolated Ganoderma resinaceum GA1M. J. Fungi 2021, 7, 866. [Google Scholar] [CrossRef] [PubMed]
- Ghazvinian, A.; Farrokhsiar, P.; Vieira, F.; Pecchia, J.; Gursoy, B. Mycelium-based bio-composites for architecture: Assessing the effects of cultivation factors on compressive strength. Mater. Res. Innov. 2019, 2, 505–514. [Google Scholar]
- Mapook, A.; Hyde, K.D.; Hassan, K.; Kemkuignou, B.M.; Čmoková, A.; Surup, F.; Kuhnert, E.; Paomephan, P.; Cheng, T.; De Hoog, S.; et al. Ten decadal advances in fungal biology leading towards human well-being. Fungal Divers. 2022, 116, 547–614. [Google Scholar] [CrossRef]
- Pegler, D.N. Hyphal analysis of basidiomata. Mycol. Res. 1996, 100, 129–142. [Google Scholar] [CrossRef]
- Webster, J.; Weber, R. Structure and morphogenesis of basidiocarps. In Introduction to Fungi; Cambridge University Press: Cambridge, UK, 2007; 841p. [Google Scholar]
- Mortimer, P.E.; Xu, J.; Karunarathna, S.C.; Hyde, K.D. (Eds.) Lentinus sajor-caju (Polyporales). In Mushrooms for Trees and People: A Field Guide to Useful Mushrooms of the Mekong Region; World Agroforestry Centre (ICRAF): Kunming, China, 2014; 125p. [Google Scholar]
- Luangharn, T.; Karunarathna, S.C.; Dutta, A.K.; Paloi, S.; Promputtha, I.; Hyde, K.D.; Xu, J.; Mortimer, P.E. Ganoderma (Ganodermataceae, Basidiomycota) species from the Greater Mekong Subregion. J. Fungi 2021, 7, 819. [Google Scholar] [CrossRef]
- Gore, V.U.; Mali, P.V. Survey of wood-decaying fungi from Vaijapur Taluka, Aurangabad (M.S.) India. Int. J. Curr. Microbiol. App. Sci. 2021, 10, 59–67. [Google Scholar]
- Liu, R.; Li, X.; Long, L.; Sheng, Y.; Xu, J.; Wang, Y. Improvement of mechanical properties of mycelium/cotton stalk composites by water immersion. Compos. Interfaces 2020, 27, 953–966. [Google Scholar] [CrossRef]
- Abidin, N.M.Z.; Sultan, M.T.H.; Shah, A.U.M.; Safri, S.N.A. Charpy and Izod impact properties of natural fibre composites. In Proceedings of the 6th International Conference on Applications and Design in Mechanical Engineering, Penang Island, Malaysia, 26–27 August 2019; IOP Conference Series: Materials Science and Engineering. p. 012031. [Google Scholar]
- Sivakumar, D.; Kathiravan, S.; Ng, L.F.; Ali, M.B.; Selamat, M.Z.; Sivaraos, S.; Bapokutty, O. Experimental investigation on charpy impact response of kenaf bast fibre reinforced metal laminate system. ARPN J. Eng. Appl. Sci. 2018, 13, 822–827. [Google Scholar]
- Hwang, S.G.; Li, Y.Y.; Lin, H.L. The use of sawdust mixed with ground branches pruned from wax apple or Indian jujube as substrate for cultivation of king oyster mushroom (Pleurotus eryngii). HortScience 2015, 50, 1230–1233. [Google Scholar] [CrossRef]
- Suwannarach, N.; Kumla, J.; Zhao, Y.; Kakumyan, P. Impact of cultivation substrate and microbial community on improving mushroom productivity: A review. Biology 2022, 11, 569. [Google Scholar] [CrossRef]
- Guo, M.; Chorover, J.; Rosario, R.; Fox, R.H. Leachate chemistry of field-weathered spent mushroom substrate. J. Environ. Qual. 2001, 30, 1699–1709. [Google Scholar] [CrossRef]
- Sales-Campos, C.; Ferreira da Eira, A.; de Almeida Minhoni, M.T.; Nogueira de Andrade, M.C. Mineral composition of raw material, substrate and fruiting bodies of Pleurotus ostreatus in culture. Interciencia 2009, 34, 432–436. [Google Scholar]



| Substrates | Fungal Species | Parameters * | ||
|---|---|---|---|---|
| Moisture Content (%) | Density (kg/m3) | Shrinkage (%) | ||
| Sawdust | Ganoderma fornicatum | 62.01 ± 0.64 b | 337.21 ± 13.36 ab | 8.86 ± 1.17 b |
| Ganoderma williamsianum | 61.34 ± 0.73 b | 331.44 ± 9.39 a | 8.40 ± 1.28 b | |
| Lentinus sajor-caju | 61.23 ± 0.53 b | 340.31 ± 16.41 a | 8.10 ± 1.89 b | |
| Schizophyllum commune | 64.23 ± 0.55 a | 318.59 ± 8.14 b | 10.83 ± 1.39 a | |
| Corn husk | Ganoderma fornicatum | 66.98 ± 0.51 b | 232.11 ± 11.52 ab | 12.64 ± 2.70 b |
| Ganoderma williamsianum | 65.11 ± 0.59 c | 239.54 ± 8.65 a | 12.32 ± 1.36 b | |
| Lentinus sajor-caju | 64.94 ± 0.62 c | 240.99 ± 15.61 a | 11.91 ± 1.92 b | |
| Schizophyllum commune | 70.22 ± 0.32 a | 220.74 ± 11.22 b | 15.27 ± 1.45 a | |
| Rice straw | Ganoderma fornicatum | 70.13 ± 0.65 bc | 219.46 ± 8.29 a | 14.26 ± 2.26 b |
| Ganoderma williamsianum | 69.55 ± 0.48 c | 221.05 ± 15.01 a | 13.95 ± 0.80 b | |
| Lentinus sajor-caju | 70.48 ± 0.56 b | 222.76 ± 2.81 a | 13.26 ± 1.03 b | |
| Schizophyllum commune | 74.51 ± 0.73 a | 198.84 ± 10.17 b | 16.31 ± 1.00 a |
| Properties | MBCs | Products * | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Synthetic Foams | Wood-Based Composites | |||||||||
| This Study | Previous Studies | PI | PS | PU | PFR | PP | PW | PB | IB | |
| D (kg/m3) | 198.84–340.31 | 25–954 | 50–400 | 11–50 | 30–100 | 35–120 | 895–920 | 400–800 | 600–800 | 170–430 |
| AS (%) | 8.1–16.31 | 6.2–15.0 | 0.2–1.2 | 0.2–0.6 | - | - | 1.0–2.5 | 1–25 | 0.3–10 | 18.18–30.28 |
| WP (%) | 105.07–208.82 | 24.45–560 | 1.0–3.0 | 0.03–9 | 0.01–72 | 1–15 | 0.01–0.03 | 5–49 | 30.1–200 | 55–380 |
| TD (°C) | 200–325 | 225–375 | 474.1–546.8 | 318–440 | 278–379 | 270–475 | 360–460 | 250–380 | 310–350 | 150–270 |
| CS (MPa) | 0.25–1.87 | 0.03–4.44 | 0.6–1.4 | 0.03–0.69 | 0.002–48 | 0.2–0.55 | 31.19–48.29 | 8–25 | 1.8–3.4 | 0.1–1.21 |
| TS (MPa) | 0.20–0.87 | 0.01–1.55 | 0.44–0.96 | 0.15–0.7 | 0.08–103 | 0.19–0.46 | 31–41.4 | 10–44 | 10–100 | 0.35–1.38 |
| FS (MPa) | 0.06–0.32 | 0.05–4.40 | 0.59–1.36 | 0.07–0.70 | 0.21–57 | 0.38–0.78 | 22–23.2 | 35–78 | 1.5–7 | 2–2.5 |
| IS (kJ/m2) | 0.21–2.70 | - | 0.06–0.12 | 0.01–0.15 | 1.0–1.2 | 0.26–1.63 | 0.02–1 | - | - | - |
| Substrates | Fungal Species | Parameters * | |||
|---|---|---|---|---|---|
| Compression Strength (MPa) | Tensile Strength (MPa) | Flexural Strength (MPa) | Impact Strength (kJ/m2) | ||
| Sawdust | Ganoderma fornicatum | 1.71 ± 0.03 b | 0.34 ± 0.02 b | 0.07 ± 0.00 bc | 0.24 ± 0.00 b |
| Ganoderma williamsianum | 1.85 ± 0.01 a | 0.42 ± 0.01 a | 0.09 ± 0.02 ab | 0.28 ± 0.02 a | |
| Lentinus sajor-caju | 1.87 ± 0.03 a | 0.44 ± 0.03 a | 0.11 ± 0.02 a | 0.30 ± 0.02 a | |
| Schizophyllum commune | 1.59 ± 0.02 c | 0.20 ± 0.01 c | 0.06 ± 0.01 c | 0.21 ± 0.02 b | |
| Corn husk | Ganoderma fornicatum | 0.59 ± 0.01 b | 0.67 ± 0.04 bc | 0.19 ± 0.01 b | 2.05 ± 0.05 c |
| Ganoderma williamsianum | 0.62 ± 0.01 a | 0.75 ± 0.06 b | 0.28 ± 0.03 a | 2.38 ± 0.12 b | |
| Lentinus sajor-caju | 0.62 ± 0.02 a | 0.87 ± 0.06 a | 0.32 ± 0.02 a | 2.70 ± 0.90 a | |
| Schizophyllum commune | 0.58 ± 0.02 b | 0.63 ± 0.06 c | 0.18 ± 0.04 b | 1.49 ± 0.08 d | |
| Rice straw | Ganoderma fornicatum | 0.33 ± 0.01 a | 0.37 ± 0.04 b | 0.10 ± 0.02 b | 0.97 ± 0.10 a |
| Ganoderma williamsianum | 0.36 ± 0.02 a | 0.46 ± 0.03 a | 0.15 ± 0.03 a | 0.99 ± 0.07 a | |
| Lentinus sajor-caju | 0.33 ± 0.04 a | 0.45 ± 0.02 a | 0.16 ± 0.02 a | 1.04 ± 0.08 a | |
| Schizophyllum commune | 0.25 ± 0.03 b | 0.35 ± 0.01 b | 0.07 ± 0.01 b | 0.68 ± 0.09 b | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aiduang, W.; Kumla, J.; Srinuanpan, S.; Thamjaree, W.; Lumyong, S.; Suwannarach, N. Mechanical, Physical, and Chemical Properties of Mycelium-Based Composites Produced from Various Lignocellulosic Residues and Fungal Species. J. Fungi 2022, 8, 1125. https://doi.org/10.3390/jof8111125
Aiduang W, Kumla J, Srinuanpan S, Thamjaree W, Lumyong S, Suwannarach N. Mechanical, Physical, and Chemical Properties of Mycelium-Based Composites Produced from Various Lignocellulosic Residues and Fungal Species. Journal of Fungi. 2022; 8(11):1125. https://doi.org/10.3390/jof8111125
Chicago/Turabian StyleAiduang, Worawoot, Jaturong Kumla, Sirasit Srinuanpan, Wandee Thamjaree, Saisamorn Lumyong, and Nakarin Suwannarach. 2022. "Mechanical, Physical, and Chemical Properties of Mycelium-Based Composites Produced from Various Lignocellulosic Residues and Fungal Species" Journal of Fungi 8, no. 11: 1125. https://doi.org/10.3390/jof8111125
APA StyleAiduang, W., Kumla, J., Srinuanpan, S., Thamjaree, W., Lumyong, S., & Suwannarach, N. (2022). Mechanical, Physical, and Chemical Properties of Mycelium-Based Composites Produced from Various Lignocellulosic Residues and Fungal Species. Journal of Fungi, 8(11), 1125. https://doi.org/10.3390/jof8111125










