Efficacy of Plant-Derived Fungicides at Inhibiting Batrachochytrium salamandrivorans Growth
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungicides
2.2. Bsal Culturing and Zoospore Harvesting
2.3. Minimum Inhibitory Concentration (MIC) of Fungicides against Bsal
2.4. Minimum Fungicidal Concentration (MFC) of Fungicides against Bsal
2.5. Statistical Analyses
2.5.1. MIC Estimation
- Treatment + Plate order + Plate date,
- Treatment * Plate date,
- Treatment * Plate order,
- Treatment * Plate date + Plate order,
- Treatment * Plate order + Plate date, and
- Treatment * Plate date * Plate order.
2.5.2. MFC Estimation
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging fungal threats to animal, plant and ecosystem health. Nature 2012, 484, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Turner, G.G.; Reeder, D.; Coleman, J.T. A Five-year Assessment of Mortality and Geographic Spread of White-Nose Syndrome in North American Bats, with a Look at the Future. Update of White-Nose Syndrome in Bats. Bat Res. News 2011, 52, 13. [Google Scholar]
- Kim, K.; Harvell, C.D. The rise and fall of a six-year coral-fungal epizootic. Am. Nat. 2004, 164, S52–S63. [Google Scholar] [CrossRef] [PubMed]
- Cameron, S.A.; Lozier, J.D.; Strange, J.P.; Koch, J.B.; Cordes, N.; Solter, L.F.; Griswold, T.L. Patterns of widespread decline in North American bumble bees. Proc. Natl. Acad. Sci. USA 2011, 108, 662–667. [Google Scholar] [CrossRef]
- Lorch, J.M.; Knowles, S.; Lankton, J.S.; Michell, K.; Edwards, J.L.; Kapfer, J.M.; Staffen, R.A.; Wild, E.R.; Schmidt, K.Z.; Ballmann, A.E. Snake fungal disease: An emerging threat to wild snakes. Philos. Trans. R. Soc. B: Biol. Sci. 2016, 371, 20150457. [Google Scholar] [CrossRef] [PubMed]
- Martel, A.; Blooi, M.; Adriaensen, C.; Van Rooij, P.; Beukema, W.; Fisher, M.C.; Farrer, R.A.; Schmidt, B.R.; Tobler, U.; Goka, K.; et al. Recent introduction of a chytrid fungus endangers Western Palearctic salamanders. Science 2014, 346, 630–631. [Google Scholar] [CrossRef]
- Van Rooij, P.; Martel, A.; Haesebrouck, F.; Pasmans, F. Amphibian chytridiomycosis: A review with focus on fungus-host interactions. Vet. Res. 2015, 46, 137. [Google Scholar] [CrossRef] [PubMed]
- Foley, J.; Clifford, D.; Castle, K.; Cryan, P.; Ostfeld, R.S. Investigating and managing the rapid emergence of white-nose syndrome, a novel, fatal, infectious disease of hibernating bats. Conserv. Biol. 2011, 25, 223–231. [Google Scholar] [CrossRef]
- Heard, G.W.; Scroggie, M.P.; Ramsey, D.S.; Clemann, N.; Hodgson, J.A.; Thomas, C.D. Can habitat management mitigate disease impacts on threatened amphibians? Conserv. Lett. 2018, 11, e12375. [Google Scholar] [CrossRef]
- Scheele, B.C.; Pasmans, F.; Skerratt, L.F.; Berger, L.; Martel, A.; Beukema, W.; Acevedo, A.A.; Burrowes, P.A.; Carvalho, T.; Catenazzi, A. Amphibian fungal panzootic causes catastrophic and ongoing loss of biodiversity. Science 2019, 363, 1459–1463. [Google Scholar] [CrossRef]
- Martel, A.; Vila-Escale, M.; Fernández-Giberteau, D.; Martinez-Silvestre, A.; Canessa, S.; Van Praet, S.; Pannon, P.; Chiers, K.; Ferran, A.; Kelly, M.; et al. Integral chain management of wildlife diseases. Conserv. Lett. 2020, 13, e12707. [Google Scholar] [CrossRef]
- Yap, T.A.; Nguyen, N.T.; Serr, M.; Shepack, A.; Vredenburg, V.T. Batrachochytrium salamandrivorans and the risk of a second amphibian pandemic. Ecohealth 2017, 14, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Can, Ö.E.; D’Cruze, N.; Macdonald, D.W. Dealing in deadly pathogens: Taking stock of the legal trade in live wildlife and potential risks to human health. Glob. Ecol. Conserv. 2019, 17, e00515. [Google Scholar] [CrossRef] [PubMed]
- Grant, E.H.C.; Muths, E.; Katz, R.A.; Canessa, S.; Adams, M.J.; Ballard, J.R.; Berger, L.; Briggs, C.J.; Coleman, J.T.; Gray, M.J. Using decision analysis to support proactive management of emerging infectious wildlife diseases. Front. Ecol. Environ. 2017, 15, 214–221. [Google Scholar] [CrossRef]
- Carter, E.D.; Miller, D.L.; Peterson, A.C.; Sutton, W.B.; Cusaac, J.P.W.; Spatz, J.A.; Rollins-Smith, L.; Reinert, L.; Bohanon, M.; Williams, L.A.; et al. Conservation risk of Batrachochytrium salamandrivorans to endemic lungless salamanders. Conserv. Lett. 2020, 13, e12675. [Google Scholar] [CrossRef]
- Garner, T.W.; Schmidt, B.R.; Martel, A.; Pasmans, F.; Muths, E.; Cunningham, A.A.; Weldon, C.; Fisher, M.C.; Bosch, J. Mitigating amphibian chytridiomycoses in nature. Philos. Trans. R. Soc. B: Biol. Sci. 2016, 371, 20160207. [Google Scholar] [CrossRef]
- Drawert, B.; Griesemer, M.; Petzold, L.R.; Briggs, C.J. Using stochastic epidemiological models to evaluate conservation strategies for endangered amphibians. J. R. Soc. Interface 2017, 14, 20170480. [Google Scholar] [CrossRef]
- Tompros, A.; Dean, A.D.; Fenton, A.; Wilber, M.Q.; Carter, E.D.; Gray, M.J. Frequency-dependent transmission of Batrachochytrium salamandrivorans in eastern newts. Transbound. Emerg. Dis. 2021, 69, 731–741. [Google Scholar] [CrossRef]
- Silva, S.; Matz, L.; Elmassry, M.M.; San Francisco, M.J. Characteristics of monolayer formation in vitro by the chytrid Batrachochytrium dendrobatidis. Biofilm 2019, 1, 100009. [Google Scholar] [CrossRef]
- Stegen, G.; Pasmans, F.; Schmidt, B.R.; Rouffaer, L.O.; Van Praet, S.; Schaub, M.; Canessa, S.; Laudelout, A.; Kinet, T.; Adriaensen, C.; et al. Drivers of salamander extirpation mediated by Batrachochytrium salamandrivorans. Nature 2017, 544, 353–356. [Google Scholar] [CrossRef]
- Alitonou, G.A.; Noudogbessi, J.-P.; Sessou, P.; Tonouhewa, A.; Avlessi, F.; Menut, C.; Sohounhloue, D. Chemical composition and biological activities of essential oils of Pimenta racemosa (Mill.) JW Moore. from Benin. Int. J. Biosci. 2012, 2, 1–12. [Google Scholar]
- Moghadamtousi, S.Z.; Kadir, H.A.; Hassandarvish, P.; Tajik, H.; Abubakar, S.; Zandi, K. A review on antibacterial, antiviral, and antifungal activity of curcumin.(Report). BioMed Res. Int. 2014, 2014, 186864. [Google Scholar] [CrossRef] [PubMed]
- Marchese, A.; Barbieri, R.; Sanches-Silva, A.; Daglia, M.; Nabavi, S.F.; Jafari, N.J.; Izadi, M.; Ajami, M.; Nabavi, S.M. Antifungal and antibacterial activities of allicin: A review. Trends Food Sci. Technol. 2016, 52, 49–56. [Google Scholar] [CrossRef]
- Marchese, A.; Orhan, I.E.; Daglia, M.; Barbieri, R.; Di Lorenzo, A.; Nabavi, S.F.; Gortzi, O.; Izadi, M.; Nabavi, S.M. Antibacterial and antifungal activities of thymol: A brief review of the literature. Food Chem. 2016, 210, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, Y.G.; Choi, P.; Ham, J.; Park, J.G.; Lee, J. Antibiofilm and Antivirulence Activities of 6-Gingerol and 6-Shogaol Against Candida albicans Due to Hyphal Inhibition. Front. Cell. Infect. Microbioly 2018, 8, 299. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, A.; Kumari, A.; Singh, M.; Kumar, S.; Kumar, S.; Dabla, A.; Chaturvedi, S.; Yadav, V.; Chattopadhyay, D.; Prakash Dwivedi, V. [6]-Gingerol exhibits potent anti-mycobacterial and immunomodulatory activity against tuberculosis. Int. Immunopharmacol. 2020, 87, 106809. [Google Scholar] [CrossRef]
- Alagawany, M.; Farag, M.R.; Abdelnour, S.A.; Dawood, M.A.; Elnesr, S.S.; Dhama, K. Curcumin and its different forms: A review on fish nutrition. Aquaculture 2020, 532, 736030. [Google Scholar] [CrossRef]
- Ezzat Abd El-Hack, M.; Alagawany, M.; Ragab Farag, M.; Tiwari, R.; Karthik, K.; Dhama, K.; Zorriehzahra, J.; Adel, M. Beneficial impacts of thymol essential oil on health and production of animals, fish and poultry: A review. J. Essent. Oil Res. 2016, 28, 365–382. [Google Scholar] [CrossRef]
- Gold, K.K.; Reed, P.D.; Bemis, D.A.; Miller, D.L.; Gray, M.J.; Souza, M.J. Efficacy of common disinfectants and terbinafine in inactivating the growth of Batrachochytrium dendrobatidis in culture. Dis. Aquatic. Org. 2013, 107, 77–81. [Google Scholar] [CrossRef]
- Van Rooij, P.; Pasmans, F.; Coen, Y.; Martel, A. Efficacy of chemical disinfectants for the containment of the salamander chytrid fungus Batrachochytrium salamandrivorans. PLoS ONE 2017, 12, e0186269. [Google Scholar] [CrossRef]
- Kerr, L.A.; Secor, D.H. Bioenergetic trajectories underlying partial migration in Patuxent River (Chesapeake Bay) white perch (Morone americana). Can. J. Fish. Aquat. Sci. 2009, 66, 602–612. [Google Scholar] [CrossRef]
- Shinn, A.P.; Mühlhölzl, A.P.; Coates, C.J.; Metochis, C.; Freeman, M.A. Zoothamnium duplicatum infestation of cultured horseshoe crabs (Limulus polyphemus). J. Invertebr. Pathol. 2015, 125, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Schelkle, B.; Snellgrove, D.; Jones, L.L.; Cable, J. Efficacy of commercially available products against Gyrodactylus turnbulli infections on guppies Poecilia reticulata. Dis. Aquat. Org. 2015, 115, 129–137. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, Q.; Cooper, D. Conservation breeding of Shortfin eels Anguilla australis and Giant kokopu Galaxias argenteus at Mahurangi Technical Institute using aquarium and aquaculture techniques. Int. Zoo Yearb. 2013, 47, 120–128. [Google Scholar] [CrossRef]
- Andrews, J.M. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother. 2001, 48, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Jha, A. Chapter 7—Drug Development Strategies. In Anticandidal Agents; Kumar, A., Jha, A., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 63–71. [Google Scholar]
- Martel, A.; Spitzen-van der Sluijs, A.; Blooi, M.; Bert, W.; Ducatelle, R.; Fisher, M.C.; Woeltjes, A.; Bosman, W.; Chiers, K.; Bossuyt, F. Batrachochytrium salamandrivorans sp. nov. causes lethal chytridiomycosis in amphibians. Proc. Natl. Acad. Sci. USA 2013, 110, 15325–15329. [Google Scholar] [CrossRef]
- Robinson, K.A.; Pereira, K.E.; Bletz, M.C.; Carter, E.D.; Gray, M.J.; Piovia-Scott, J.; Romansic, J.M.; Woodhams, D.C.; Fritz-Laylin, L. Isolation and maintenance of Batrachochytrium salamandrivorans cultures. Dis. Aquat. Org. 2020, 140, 1–11. [Google Scholar] [CrossRef]
- Martel, A.; Van Rooij, P.; Vercauteren, G.; Baert, K.; Van Waeyenberghe, L.; Debacker, P.; Garner, T.W.; Woeltjes, T.; Ducatelle, R.; Haesebrouck, F. Developing a safe antifungal treatment protocol to eliminate Batrachochytrium dendrobatidis from amphibians. Med. Mycol. 2011, 49, 143–149. [Google Scholar] [CrossRef]
- Sanguinetti, M.; Posteraro, B. Susceptibility Testing of Fungi to Antifungal Drugs. J. Fungi 2018, 4, 110. [Google Scholar] [CrossRef]
- Blooi, M.; Pasmans, F.; Rouffaer, L.; Haesebrouck, F.; Vercammen, F.; Martel, A. Successful treatment of Batrachochytrium salamandrivorans infections in salamanders requires synergy between voriconazole, polymyxin E and temperature. Sci. Rep. 2015, 5, 11788. [Google Scholar] [CrossRef]
- Lindauer, A.; May, T.; Rios-Sotelo, G.; Sheets, C.; Voyles, J. Quantifying Batrachochytrium dendrobatidis and Batrachochytrium salamandrivorans Viability. EcoHealth 2019, 16, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Peterson, D.A.; Kimura, H.; Schubert, D. Mechanism of cellular 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) reduction. J. Neurochem. 1997, 69, 581–593. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Use of MTT colorimetric assay to measure cell activation. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Malekinejad, H.; Bazargani-Gilani, B.; Tukmechi, A.; Ebrahimi, H. A cytotoxicity and comparative antibacterial study on the effect of Zataria multiflora Boiss, Trachyspermum copticum essential oils, and Enrofloxacin on Aeromonas hydrophila. Avicenna J. Phytomedicine 2012, 2, 188. [Google Scholar]
- Queen, J.P.; Quinn, G.P.; Keough, M.J. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Multimodel inference: Understanding AIC and BIC in model selection. Sociol. Methods Res. 2004, 33, 261–304. [Google Scholar] [CrossRef]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; Team, R.C. nlme: Linear and nonlinear mixed effects models. R Package Version 3.1–147. 2020. Available online: https://cran.r-project.org/web/packages/nlme/index.html (accessed on 19 August 2022).
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Kosmidis, I. brglm: Bias Reduction in Binary-Response Generalized LinearModels, R Package Version 0.7.2: 2020; Department of Statistical Science, University College London: London, UK, 2011. [Google Scholar]
- Zainal, M.; Mohamad Zain, N.; Mohd Amin, I.; Ahmad, V.N. The antimicrobial and antibiofilm properties of allicin against Candida albicans and Staphylococcus aureus—A therapeutic potential for denture stomatitis. Saudi Dent. J. 2021, 33, 105–111. [Google Scholar] [CrossRef]
- Neelofar, K.; Shreaz, S.; Rimple, B.; Muralidhar, S.; Nikhat, M.; Khan, L.A. Curcumin as a promising anticandidal of clinical interest. Can. J. Microbiol. 2011, 57, 204–210. [Google Scholar] [CrossRef]
- Ficker, C.; Smith, M.; Akpagana, K.; Gbeassor, M.; Zhang, J.; Durst, T.; Assabgui, R.; Arnason, J. Bioassay-guided isolation and identification of antifungal compounds from ginger. Phytother. Res. An. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2003, 17, 897–902. [Google Scholar] [CrossRef]
- De Castro, R.D.; de Souza, T.M.P.A.; Bezerra, L.M.D.; Ferreira, G.L.S.; de Brito Costa, E.M.M.; Cavalcanti, A.L. Antifungal activity and mode of action of thymol and its synergism with nystatin against Candida species involved with infections in the oral cavity: An in vitro study. BMC Complement. Altern. Med. 2015, 15, 1–7. [Google Scholar] [CrossRef]
- Abbaszadeh, S.; Sharifzadeh, A.; Shokri, H.; Khosravi, A.; Abbaszadeh, A. Antifungal efficacy of thymol, carvacrol, eugenol and menthol as alternative agents to control the growth of food-relevant fungi. J. De Mycol. Med. 2014, 24, e51–e56. [Google Scholar] [CrossRef]
- Sadeghi-Ghadi, Z.; Vaezi, A.; Ahangarkani, F.; Ilkit, M.; Ebrahimnejad, P.; Badali, H. Potent in vitro activity of curcumin and quercetin co-encapsulated in nanovesicles without hyaluronan against Aspergillus and Candida isolates. J. De Mycol. Med. 2020, 30, 101014. [Google Scholar] [CrossRef] [PubMed]
- Shadkchan, Y.; Shemesh, E.; Mirelman, D.; Miron, T.; Rabinkov, A.; Wilchek, M.; Osherov, N. Efficacy of allicin, the reactive molecule of garlic, in inhibiting Aspergillus spp. in vitro, and in a murine model of disseminated aspergillosis. J. Antimicrob. Chemother. 2004, 53, 832–836. [Google Scholar] [CrossRef] [PubMed]
- Al-Gendy, A.; Moharram, F.; Zarka, M. Chemical composition, antioxidant, cytotoxic and antimicrobial activities of Pimenta racemosa (Mill.) JW Moore flower essential oil. J. Pharmacogn. Phytochem. 2017, 6, 312–319. [Google Scholar]
- Piotrowski, J.S.; Annis, S.L.; Longcore, J.E. Physiology of Batrachochytrium dendrobatidis, a chytrid pathogen of amphibians. Mycologia 2004, 96, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Spitzen-van der Sluijs, A.; Spikmans, F.; Bosman, W.; de Zeeuw, M.; van der Meij, T.; Goverse, E.; Kik, M.; Pasmans, F.; Martel, A. Rapid enigmatic decline drives the fire salamander (Salamandra salamandra) to the edge of extinction in the Netherlands. Amphib. -Reptil. 2013, 34, 233–239. [Google Scholar] [CrossRef]
- Leontiev, R.; Hohaus, N.; Jacob, C.; Gruhlke, M.C.; Slusarenko, A.J. A comparison of the antibacterial and antifungal activities of thiosulfinate analogues of allicin. Sci. Rep. 2018, 8, 1–19. [Google Scholar] [CrossRef]
- Sandur, S.K.; Pandey, M.K.; Sung, B.; Ahn, K.S.; Murakami, A.; Sethi, G.; Limtrakul, P.; Badmaev, V.; Aggarwal, B.B. Curcumin, demethoxycurcumin, bisdemethoxycurcumin, tetrahydrocurcumin and turmerones differentially regulate anti-inflammatory and anti-proliferative responses through a ROS-independent mechanism. Carcinogenesis 2007, 28, 1765–1773. [Google Scholar] [CrossRef]
- Contreras-Moreno, B.Z.; Velasco, J.J.; Rojas, J.d.C.; Méndez, L.d.C.; Celis, M.T. Antimicrobial activity of essential oil of Pimenta racemosa var. racemosa (Myrtaceae) leaves. J. Pharm. Pharmacogn. Res. 2016, 4, 224–230. [Google Scholar]
- Deberdt, P.; Davezies, I.; Coranson-Beaudu, R.; Jestin, A. Efficacy of Leaf Oil from Pimenta racemosa var. racemosa in Controlling Bacterial Wilt of Tomato. Plant Dis. 2018, 102, 124–131. [Google Scholar] [CrossRef]
- Mansingh, D.P.; Sunanda, O.J.; Sali, V.K.; Vasanthi, H.R. [6]-Gingerol-induced cell cycle arrest, reactive oxygen species generation, and disruption of mitochondrial membrane potential are associated with apoptosis in human gastric cancer (AGS) cells. J. Biochem. Mol. Toxicol. 2018, 32, e22206. [Google Scholar] [CrossRef]
- Elumalai, P.; Kurian, A.; Lakshmi, S.; Faggio, C.; Esteban, M.A.; Ringø, E. Herbal immunomodulators in aquaculture. Rev. Fish. Sci. Aquac. 2020, 29, 33–57. [Google Scholar] [CrossRef]
- Firmino, J.P.; Vallejos-Vidal, E.; Sarasquete, C.; Ortiz-Delgado, J.B.; Balasch, J.C.; Tort, L.; Estevez, A.; Reyes-López, F.E.; Gisbert, E. Unveiling the effect of dietary essential oils supplementation in Sparus aurata gills and its efficiency against the infestation by Sparicotyle chrysophrii. Sci. Rep. 2020, 10, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Souza, C.d.F.; Baldissera, M.D.; Baldisserotto, B.; Heinzmann, B.M.; Martos-Sitcha, J.A.; Mancera, J.M. Essential Oils as Stress-Reducing Agents for Fish Aquaculture: A Review. Front. Physiol. 2019, 10, 785. [Google Scholar] [CrossRef] [PubMed]
- Sutili, F.J.; Gatlin III, D.M.; Heinzmann, B.M.; Baldisserotto, B. Plant essential oils as fish diet additives: Benefits on fish health and stability in feed. Rev. Aquac. 2018, 10, 716–726. [Google Scholar] [CrossRef]
- Tavares-Dias, M. Current knowledge on use of essential oils as alternative treatment against fish parasites. Aquat. Living Resour. 2018, 31, 13. [Google Scholar] [CrossRef]
- Bairwa, M.K.; Jakhar, J.K.; Satyanarayana, Y.; Reddy, A.D. Animal and plant originated immunostimulants used in aquaculture. J. Nat. Prod. Plant Resour. 2012, 2, 397–400. [Google Scholar]
- Dong, J.; Zhang, L.; Liu, Y.; Xu, N.; Zhou, S.; Yang, Q.; Yang, Y.; Ai, X. Thymol Protects Channel Catfish from Aeromonas hydrophila Infection by Inhibiting Aerolysin Expression and Biofilm Formation. Microorganisms 2020, 8, 636. [Google Scholar] [CrossRef]
- Nya, E.J.; Dawood, Z.; Austin, B. The garlic component, allicin, prevents disease caused by Aeromonas hydrophila in rainbow trout, Oncorhynchus mykiss (Walbaum). J. Fish Dis. 2010, 33, 293–300. [Google Scholar] [CrossRef]
- Smith, N.C.; Christian, S.L.; Taylor, R.G.; Santander, J.; Rise, M.L. Immune modulatory properties of 6-gingerol and resveratrol in Atlantic salmon macrophages. Mol. Immunol. 2018, 95, 10–19. [Google Scholar] [CrossRef]
- Hanlon, S.M.; Kerby, J.L.; Parris, M.J. Unlikely remedy: Fungicide clears infection from pathogenic fungus in larval southern leopard frogs (Lithobates sphenocephalus). PLoS ONE 2012, 7, e43573. [Google Scholar] [CrossRef]
- Hanlon, S.M.; Lynch, K.J.; Kerby, J.L.; Parris, M.J. The effects of a fungicide and chytrid fungus on anuran larvae in aquatic mesocosms. Environ. Sci. Pollut. Res. 2015, 22, 12929–12940. [Google Scholar] [CrossRef] [PubMed]
- Rebollar, E.A.; Martínez-Ugalde, E.; Orta, A.H. The Amphibian Skin Microbiome and Its Protective Role Against Chytridiomycosis. Herpetologica 2020, 76, 111, 167–177. [Google Scholar] [CrossRef]
- Carrasco, J.; Navarro, M.J.; Santos, M.; Gea, F.J. Effect of five fungicides with different modes of action on cobweb disease (Cladobotryum mycophilum) and mushroom yield. Ann. Appl. Biol. 2017, 171, 62–69. [Google Scholar] [CrossRef]
- Bosch, J.; Sanchez-Tomé, E.; Fernández-Loras, A.; Oliver, J.A.; Fisher, M.C.; Garner, T.W. Successful elimination of a lethal wildlife infectious disease in nature. Biol. Lett. 2015, 11, 20150874. [Google Scholar] [CrossRef] [PubMed]
- Brauer, V.S.; Rezende, C.P.; Pessoni, A.M.; De Paula, R.G.; Rangappa, K.S.; Nayaka, S.C.; Gupta, V.K.; Almeida, F. Antifungal Agents in Agriculture: Friends and Foes of Public Health. Biomolecules 2019, 9, 521. [Google Scholar] [CrossRef]
- Yoon, M.-Y.; Cha, B.; Kim, J.-C. Recent trends in studies on botanical fungicides in agriculture. Plant Pathol. J. 2013, 29, 1–9. [Google Scholar] [CrossRef] [PubMed]
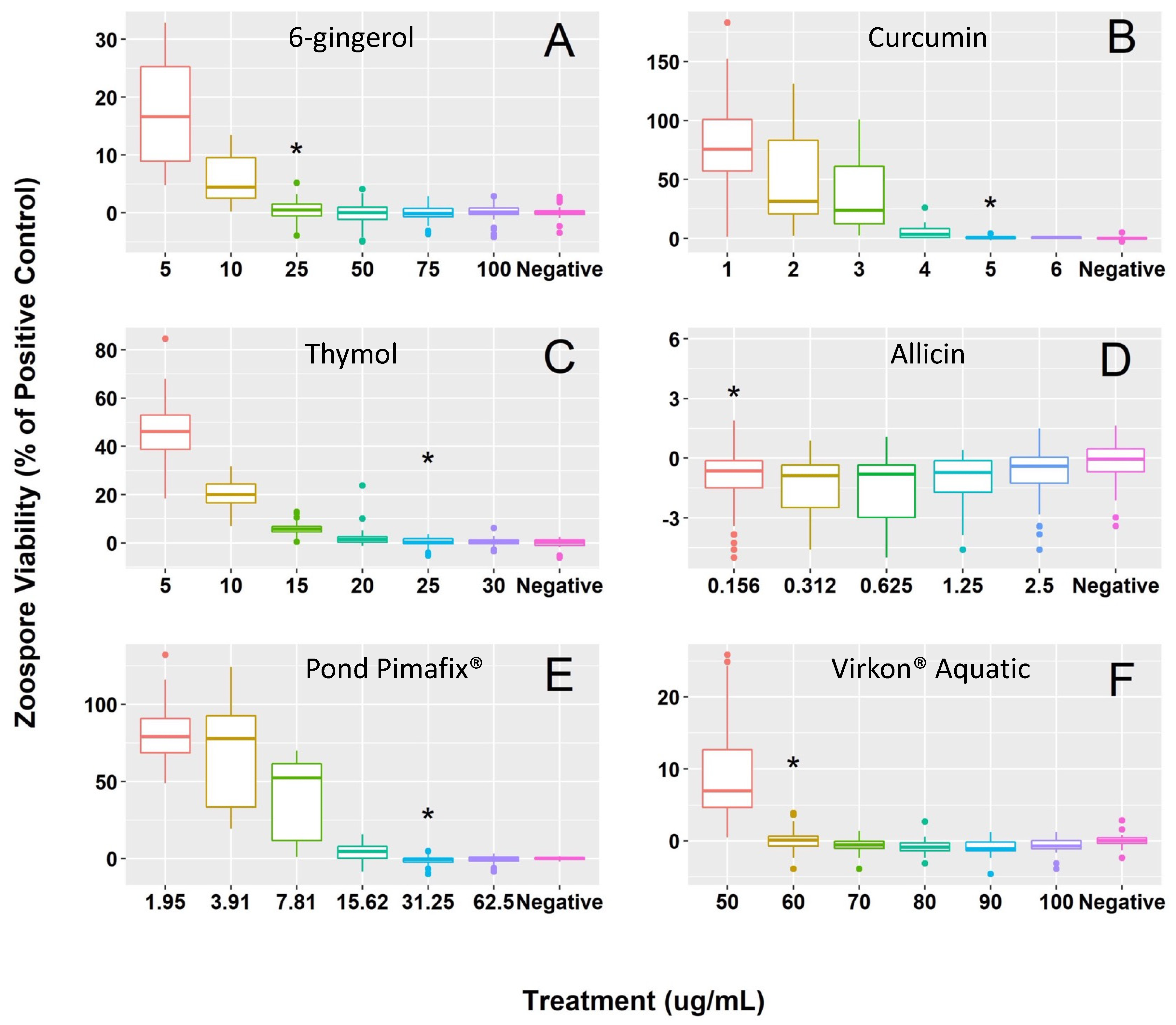
| Fungicide | Concentration Range | MIC | MFC |
|---|---|---|---|
| Thymol | 30, 25, 20, 15, 10, 5 μg/mL | 25 μg/mL | 35 μg/mL |
| Curcumin | 6, 5, 4, 3, 2, 1 μg/mL | 5 μg/mL | 5 μg/mL |
| 6-gingerol | 100, 75, 50, 25, 10, 5 μg/mL | 25 μg/mL | 50 μg/mL |
| Allicin | 2.5, 1.25, 0.625, 0.313, 0.156 μg/mL | 0.156 μg/mL | 0.156 μg/mL |
| Pond Pimafix® | 62.5, 31.25, 15.62, 7.81, 3.91, 1.95 μg/mL | 31.25 μg/mL | 31.25 μg/mL |
| Virkon® Aquatic | 100, 90, 80, 70, 60, 50 μg/mL | 60 μg/mL | 60 μg/mL |
| Fungicide | Best Fit Model | ΔAIC 1 | MIC 2 |
|---|---|---|---|
| Thymol | Treatment + Plate order + Plate date | 0 | 25 μg/mL No interaction model 3 |
| Treatment * Plate date | 3.126 | ||
| Treatment * Plate order | 30.174 | ||
| Treatment * Plate date + Plate order | 2.867 | ||
| Treatment * Plate order + Plate date | 17.330 | ||
| Treatment * Plate date * Plate order | 51.655 | ||
| Curcumin | Treatment + Plate order + Plate date | 286.771 | 5 μg/mL 5/9 plate order*date models 3 2/3 plate date models |
| Treatment * Plate date | 26.616 | ||
| Treatment * Plate order | 305.615 | ||
| Treatment * Plate date + Plate order | 23.766 | ||
| Treatment * Plate order + Plate date | 294.345 | ||
| Treatment * Plate date * Plate order | 0 | ||
| 6-gingerol | Treatment + Plate order + Plate date | 277.191 | 25 μg/mL 6/9 plate order*date models 3/3 plate date models |
| Treatment * Plate date | 146.085 | ||
| Treatment * Plate order | 325.236 | ||
| Treatment * Plate date + Plate order | 116.422 | ||
| Treatment * Plate order + Plate date | 291.269 | ||
| Treatment * Plate date * Plate order | 0 | ||
| Allicin | Treatment + Plate order + Plate date | 0 | 0.156 μg/mL No interaction model 3 |
| Treatment * Plate date | 0.488 | ||
| Treatment * Plate order | 23.047 | ||
| Treatment * Plate date + Plate order | 3.271 | ||
| Treatment * Plate order + Plate date | 3.271 | ||
| Treatment * Plate date * Plate order | 3.271 | ||
| Pond Pimafix® | Treatment + Plate order + Plate date | 358.097 | 31.25 μg/mL 5/9 plate order*date models 2/3 plate date models |
| Treatment * Plate date | 180.598 | ||
| Treatment * Plate order | 318.613 | ||
| Treatment * Plate date + Plate order | 167.370 | ||
| Treatment * Plate order + Plate date | 312.451 | ||
| Treatment * Plate date * Plate order | 0 | ||
| Virkon® Aquatic | Treatment + Plate order + Plate date | 78.395 | 60 μg/mL 7/9 plate order*date models 3/3 plate date models |
| Treatment * Plate date | 92.567 | ||
| Treatment * Plate order | 84.146 | ||
| Treatment * Plate date + Plate order | 80.262 | ||
| Treatment * Plate order + Plate date | 69.128 | ||
| Treatment * Plate date * Plate order | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tompros, A.; Wilber, M.Q.; Fenton, A.; Carter, E.D.; Gray, M.J. Efficacy of Plant-Derived Fungicides at Inhibiting Batrachochytrium salamandrivorans Growth. J. Fungi 2022, 8, 1025. https://doi.org/10.3390/jof8101025
Tompros A, Wilber MQ, Fenton A, Carter ED, Gray MJ. Efficacy of Plant-Derived Fungicides at Inhibiting Batrachochytrium salamandrivorans Growth. Journal of Fungi. 2022; 8(10):1025. https://doi.org/10.3390/jof8101025
Chicago/Turabian StyleTompros, Adrianna, Mark Q. Wilber, Andy Fenton, Edward Davis Carter, and Matthew J. Gray. 2022. "Efficacy of Plant-Derived Fungicides at Inhibiting Batrachochytrium salamandrivorans Growth" Journal of Fungi 8, no. 10: 1025. https://doi.org/10.3390/jof8101025
APA StyleTompros, A., Wilber, M. Q., Fenton, A., Carter, E. D., & Gray, M. J. (2022). Efficacy of Plant-Derived Fungicides at Inhibiting Batrachochytrium salamandrivorans Growth. Journal of Fungi, 8(10), 1025. https://doi.org/10.3390/jof8101025





